Microwave-assisted organic syntheses: microwave effect on intramolecular reactions – the Claisen rearrangement of allylphenyl ether and 1-allyloxy-4-methoxybenzene†
Satoshi Horikoshi*a,
Tomoki Watanabea,
Momoko Kamataa,
Yumiko Suzukia and
Nick Serponeb
aDepartment of Materials and Life Sciences, Faculty of Science and Technology, Sophia University, 7-1 Kioicho, Chiyodaku, Tokyo 102-8554, Japan. E-mail: horikosi@sophia.ac.jp; Fax: +81-3-3238-3361; Tel: +81-3-3238-4662
bPhotoGreen Laboratory, Dipartimento di Chimica, Universita di Pavia, via Taramelli 12, Pavia 27100, Italy
First published on 15th October 2015
Abstract
This article examined how and the possible effect microwaves may have on intramolecular reactions such as those of the Claisen-type rearrangement carried out in dimethyl sulfoxide (DMSO) solvent and in solvent-free, microwave irradiation conditions. For comparison, the reaction was also performed by conventional heating using an oil bath. 2-Allylphenol was synthesized from allylphenyl ether in DMSO solvent under stirring conditions as a model intramolecular reaction taking place via the Claisen rearrangement using a commercial microwave chemical apparatus together with conventional heating; no enhancement of the reaction occurred. To further examine the influence of microwave irradiation on Claisen rearrangement reactions, we also investigated the transformation of 1-allyloxy-4-methoxybenzene to 2-allyl-4-methoxyphenol under both solvent-free conditions (no stirring) and in DMSO medium; here also no reaction enhancement was observed. This notwithstanding, microwaves did impact the formation of a by-product formed in the latter reaction, which was identified by GC and GC/MS as 4-methoxyphenol, the yield of which was nearly fourfold greater (ca. 6%) under microwave irradiation than under oil-bath heating (ca. 1.5%). The latter suggests that under solvent-free conditions a microwave non-thermal effect influenced the formation of this by-product during the Claisen rearrangement process, contrary to the case where the reaction was performed in DMSO medium for which the yields were identical (ca. 2.5%), regardless of whether the reactant was microwave or oil-bath heated.
1. Introduction
Microwave radiation has been used extensively as a heat source in many organic syntheses since 2000,1 as a means of significantly accelerating processes, in general, and organic reactions, in particular.2 However, in spite of this advantageous feature of microwaves, a direct comparison with conventional heating is neither straightforward nor simple because factors such as the distribution of temperature3 and reactor types, among others, in microwave heating are in most cases different from conventional heating. A significant advantage of microwaves is that substrates can be heated directly in a short time thereby avoiding the use of (organic) solvents. From the point of view of industrial manufacturing and Green Chemistry, the use of a technology that can deliver high yields of products in relatively short times is not only ecofriendly but financially attractive as well.Our studies in microwave-assisted chemistry have consistently focused on how and what particular features of microwave radiation impact syntheses, in general, and organic syntheses involving intermolecular reactions, in particular.4–19 In this regard, in a previous study we investigated specific (non-thermal) microwave effects that might have an impact on organic syntheses by examining the synthesis of monoglycerylcetyldimethylammonium chloride from 3-chloro-1,2-propanediol and N,N-dimethylhexadecylamine in 2-propanol solvent, and under solvent-free conditions.18 No effects were evident in homogeneous 2-propanol media under temperature conditions identical to conventional heating; however, heterogeneous solvent-free conditions brought out non-insignificant specific microwave effects as evidenced by variant product yields: 62% by microwave irradiation versus 47% by conventional heating (typically using an oil bath). This variance was attributed to thermal conduction and localized hot spots formed under microwave irradiation. The model proposed for the solvent-free synthesis considered hydrophilic 3-chloro-1,2-propanediol molecules forming preferentially H-bonded domains (size, 2–20 μm) heated by microwaves and dispersed in a sea of hydrophobic N,N-dimethylhexadecylamine molecules. Moreover, the ionic liquid 1-butyl-3-methylimidazolium tetrafluoroborate, produced under solvent-free conditions, was obtained in high yields by microwave irradiation: 87% under 5.8 GHz microwave heating conditions versus 21% under conventional heating.20 Evidently, the condition needed to observe microwave specific phenomena in organic syntheses is that the starting substrates have different large microwave absorption features. That is, the microscopic dissimilar distribution of temperature should enhance the collision frequency between two different molecules.
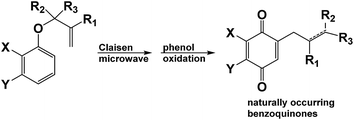
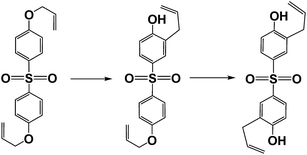
As part of our systematic studies then, the present study focusses on the how microwave radiation might affect intramolecular reactions of the Claisen-type rearrangement. In this regard, studies of microwave-assisted syntheses of naturally occurring 1,4-benzoquinones and of bis-(3-allyl-4-hydroxyphenyl)sulfone by the Claisen rearrangement process have been reported by Davis et al. (reaction (1))21 and Yamamoto and coworkers (reaction (2)),22 respectively, among several other studies.23–34 In nearly all these studies, however, the interest focused on the formation of the major product from the Claisen rearrangement, as minor by-products are often not reported or otherwise noted.
 | (1) |
 | (2) |
To enhance the heat effect of the microwaves in Claisen rearrangements, a strong microwave absorbing solvent (e.g., water, alcohols) is typically used35 so that any direct effect between reacting substrates and microwaves could not be delineated. Accordingly, to assess the how and what effect microwaves may have, if any, on intramolecular reactions, we examined the synthesis of 2-allylphenol from the Claisen rearrangement of the corresponding allylphenyl ether in dimethyl sulfoxide (DMSO) solvent, a strong microwave absorber solvent, and using commercially available microwave chemical apparatuses. Further, the Claisen rearrangement of 1-allyloxy-4-methoxybenzene to 2-allyl-4-methoxyphenol was investigated under solvent-free, non-stirring conditions, as well as in the presence of DMSO using frequency-precise (2.450000 GHz) microwaves emitted from a semiconductor generator combined with a single-mode cavity. The latter process also yielded 4-methoxyphenol in non-insignificant yields.
2. Experimental setup
2.1 Microwave equipment with single-mode applicator and semiconductor generator
The microwave irradiation setup with a single-mode TE103 cavity (transverse electric 103 mode), schematically illustrated in Fig. 1a, included a short plunger, an iris, a three-stub tuner, a power monitor and an isolator. Continuous microwave radiation was generated from a 2.450000 GHz microwave semiconductor generator (Fuji Electronic Industrial Co. Ltd.; GNU-201AA; maximal power, 200 W). The resonance of the microwaves was adjusted with the iris and the plunger at 1.5 cycles. Heating of the reactor contents was achieved by positioning the quartz tube reactor in the single-mode microwave apparatus of Fig. 1a and b at the position of maximal electric field density (E field; Fig. 1a inset) within the waveguide. Temperatures of the solutions were measured at 3 s intervals with an optical fiber thermometer (FL-2000, Anritsu Meter Co. Ltd.). The wavelength of propagation of the microwaves in the TE103 mode within the waveguide was 14.78 cm (estimated from eqn (3)):36
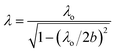 | (3) |
 | ||
| Fig. 1 (a) Details of the experimental setup and positioning of the samples in the single-mode microwave resonator; maximal position of the electric field (E field) density. (b) Photograph of the single-mode microwave resonator and the 2.45 GHz semiconductor microwave generator. Reproduced from ref. 36; Copyright 2012 by the International Microwave Power Institute. | ||
The synthesis of 2-allyl-4-methoxyphenol by the Claisen rearrangement process was examined by positioning the reactor containing the initial substrate 1-allyloxy-4-methoxybenzene at the maximal microwaves' E-field density (Fig. 1a inset). The microwave input power was ca. 170 W (without DMSO) and 46 W with DMSO present as the latter is a strong microwave absorber. An electric field monitor (Fuji Electronic Industrial Co. Ltd.) was used to maintain the sample tube at the maximal position of the E-field density, as the reproducibility of such experiments is often diminished if such operations are neglected. Under our conditions, no significant positional changes of the electric field were needed on heating.
2.2 Analytical methods and simulations
The yields of product(s) formed were determined by gas chromatographic analyses using a Shimadzu model 2014 FID-GC (Restek Rtx-5 capillary column; helium was the carrier gas; column temperature, 100–280 °C varied at 15 °C min−1 intervals). Potential by-products that may form were identified by gas chromatography/mass spectrometry techniques (GC/MS; Shimadzu model GCMS-QP2010 Ultra; Restek Rxi-5sil capillary column; helium was the carrier gas). Dielectric constant and dielectric loss of the pure chemicals subjected to the 2.45 GHz microwaves were measured with an Agilent Technologies 8720C Network Analyzer using a slim probe system. Unless otherwise noted, proton NMR spectra were recorded in CD3Cl solvent with a 400 MHz Bruker Avance DPX 400 NMR spectrometer; chemical shifts are quoted in parts per million (ppm).The RF model in the COMSOL multiphysics software version 4.3a, based on a finite element method, was used for the calculation and 3D simulation of the electromagnetic distribution, power density, and the temperature distribution under microwave irradiation. The geometric model of the waveguide reflected the experimental setup used. The power density, or power dissipated per unit volume, was calculated from the electric field strength using eqn (4):
P = 2πfε0ε′′|![[E with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0045_20d1.gif) |2 |2
| (4) |
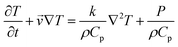 | (5) |
![[v with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0076_20d1.gif) is the velocity vector (m s−1), and P is the volumetric heat (W m−3).
is the velocity vector (m s−1), and P is the volumetric heat (W m−3).
2.3 Microwave-assisted synthesis of 2-allylphenol in DMSO with commercial microwave equipment
The initial substrate allylphenyl ether (0.134 g; Wako Pure Chem. Co) and DMSO solvent (5 mL) was placed in the reactor. The thermally driven reaction was then performed with microwave heating from commercial microwave equipment or heating from an oil bath at 180 ± 1 °C for reaction times of 30, 60, and 120 min. The resulting product 2-allylphenol formed from the Claisen rearrangement process was subsequently washed twice with toluene (5 mL), and its yield determined by gas chromatography in comparison with a standard 2-allylphenol sample from Wako Pure Chem. Co. The microwave apparatuses with multimode applicators were the Milestone StartSYNTH and Shikoku Instrumentation Co. Ltd. μReactor systems; in both cases the reactor consisted of a two-neck quartz tube connected to a reflux condenser. An Anton-Paar Monowave300 microwave apparatus was also used albeit with a standard Pyrex closed reactor.2.4 Synthesis of the precursor ether 1-allyloxy-4-methoxybenzene
The 1-allyloxy-4-methoxybenzene substrate was synthesized by addition of 4-methoxyphenol (4 g; standard sample; purity > 99.0%; Tokyo Chemical Industry Co., Ltd.), potassium carbonate (4 g), and allyl bromide (4 g) to an acetone (30 mL) solution that was subsequently refluxed with oil-bath heating for 3 days. Completion of the reaction was confirmed by thin layer chromatographic techniques (TLC) using hexane as the developing solvent. Potassium carbonate was removed by filtration, after which acetone was removed using a rotary evaporator. The resulting residue was added to various water/ethyl acetate aliquots (each 20 mL) in a separating funnel to permit extraction of the organic layer. At this stage, any 4-methoxyphenol left was removed by adding aqueous sodium hydroxide (4 wt%), following which the small amount of water was removed by addition of sodium sulfate (5 g). The mixture was then filtered and the ethyl acetate removed from the filtrate using a rotary evaporator. The residual sample was then treated by flash chromatography on silica gel (230–400 mesh) in hexane. The end point of product separation was confirmed by TLC on aluminum-backed silica plates pre-coated with silica (0.2 mm); the plates were then developed using UV fluorescence and iodine. The synthesis yield of 1-allyloxy-4-methoxybenzene was 19%. A proton NMR spectrum of this product (see Fig. S-1 in ESI†) in CD3Cl revealed peaks at δ (ppm) = 3.775 (s, CH3, 3H), 4.48–4.52 (m, CH2, 2H), 5.29–5.50 (m, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2, 2H), 6.03–6.16 (m, C
CH2, 2H), 6.03–6.16 (m, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH, 1H), 6.855–6.895 (m, ArH, 4H). These results accord with those reported by Lin and coworkers.25
CH, 1H), 6.855–6.895 (m, ArH, 4H). These results accord with those reported by Lin and coworkers.25
2.5 Microwave-assisted synthesis of 2-allyl-4-methoxyphenol in a single-mode microwave system
The initial ethereal substrate 1-allyloxy-4-methoxybenzene (0.2 g) was placed in a quartz tube reactor (length: 20.0 cm; internal diameter: 0.5 cm) and then heated either by microwave dielectric heating or by oil-bath heating at 170 °C under solvent-free and non-stirring conditions for reaction times of 60, 120, and 240 min. The resulting product was washed twice with toluene (1 mL), and its yield also determined by gas chromatography. The major product resulting from the Claisen rearrangement, 2-allyl-4-methoxyphenol, was subsequently purified by vacuum distillation, its purity being evidenced by gas chromatographic analyses. The proton NMR spectrum (see Fig. S-2 in ESI†) of 2-allyl-4-methoxyphenol in CD3Cl solvent showed peaks at δ (ppm) = 3.38 (d, J = 6.4 Hz, CH2, 2H), 3.76 (s, CH3, 3H), 4.62 (s, OH, 1H), 5.13–5.20 (m, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2, 2H), 5.95–6.01 (m, C
CH2, 2H), 5.95–6.01 (m, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH, 1H), 6.66–6.79 (m, ArH, 3H).
CH, 1H), 6.66–6.79 (m, ArH, 3H).
The microwave-assisted synthesis of 2-allyl-4-methoxyphenol from the 1-allyloxy-4-methoxybenzene (0.2 g) precursor was also investigated in DMSO solvent (5 mL) in a quartz reactor (length: 20.0 cm; internal diameter: 2.0 cm) in a single-mode microwave system with the reactants heated to 170 °C; the solution was stirred using a micro-stirring bar. Subsequent to the work-up of the reaction, the product was analyzed by the same techniques as above.
3. Results and discussion
3.1 Microwave-assisted synthesis of 2-allylphenol in DMSO solvent
As a first step in our query on the effect of microwaves on intramolecular reactions we carried out the synthesis of 2-allylphenol via the Claisen rearrangement of allylphenyl ether in DMSO solvent using three commercial microwave chemical apparatuses. The chemical yields of the resulting 2-allylphenol were 8.3% under microwave irradiation for 120 min using the commercial Monowave300 and StartSYNTH systems (Fig. 1), whereas the yields were 7.2% when using oil-bath heating and the commercial μReactor system with microwave heating (Fig. 2). Although the synthesis was performed at the same microwave frequency (2.45 GHz; magnetron generator), the small difference in yields is likely due to the different types of reactors used. For instance, while a standard closed reactor was used in the Monowave300 apparatus, the reactor used for the StartSYNTH, μReactor and oil-bath heating consisted of a two-neck quartz tube connected to a reflux condenser. In all cases the reaction temperature was 180 ± 1 °C. No significant enhancement, if any, was observed when the reaction was carried out under microwave heating relative to oil-bath heating.To further explore the possible enhancing effect of microwaves in an intramolecular reaction, we then performed the synthesis of 2-allylphenol under solvent-free conditions using the single-mode microwave apparatus of Fig. 1 in which the microwaves irradiated directly the precursor allylphenyl ether undergoing the Claisen rearrangement. The microwave absorption capacity of allylphenyl ether is fairly small (dielectric loss: ε′′r = 0.363) relative to DMSO (ε′′r = 15.98) solvent, a strong microwave absorber, so that microwave heating the initial substrate in the absence of DMSO achieved temperatures no greater than 150 °C. In DMSO media, however, the microwaves heated DMSO preferentially which, subsequent to heat transfer to allylphenyl ether, ultimately led to the formation of 2-allylphenol. The microwaves were thus used simply as a heat source in this process.
3.2 Microwave-assisted synthesis of 2-allyl-4-methoxyphenol under solvent-free conditions
To improve the microwave absorption capacity of the starting substrate, the precursor 1-allyloxy-4-methoxybenzene was synthesized and subsequently employed in the Claisen process in its conversion to 2-allyl-4-methoxyphenol utilizing the single-mode apparatus of Fig. 1 as the quantity obtained in the synthesis of this precursor was rather small. The use of commercial reactors and microwave apparatuses proved inadequate in this case for such a small quantity and also because of difficulties in measuring temperatures accurately. Solvent-free conditions precluded stirring during the reaction. Accordingly, the temperature distribution in neat 1-allyloxy-4-methoxybenzene was the first step to examine under microwave heating.The temperature distribution in the quartz reactor was determined by monitoring the temperature with an optical fiber thermometer at the upper, at the center and at the lower portions of the reactor that contained the starting substrate (0.2 g; liquid level, 15 mm; Fig. 3). At the lower part, the temperature was 170 °C (usual measuring position), and 185 °C and 178 °C at the upper part and at the center, respectively. The 15 °C temperature variation between the upper and lower parts of the reactor was rather unexpected for such a little sample volume. By comparison, under oil-bath heating the temperature distribution was 170 °C throughout the sample. Accordingly, oil-bath heating of the sample was also performed at the highest temperature reached on microwave heating (185 °C).
 | ||
| Fig. 3 Temperature distribution of neat 1-allyloxy-4-methoxybenzene and neat DMSO solvent under microwave heating. | ||
The temperature distribution observed upon microwave heating a sample of neat 1-allyloxy-4-methoxybenzene required 170 W microwaves because microwave absorption by this substrate was rather low (dielectric loss: ε′′r = 0.827). Accordingly, the temperature distribution of the strong microwave absorber dimethyl sulfoxide (DMSO; ε′′r = 15.98) solvent was also examined in neat form (no other substrate present) with the same microwave system; 42 W microwaves were needed to bring pure DMSO solvent (alone) to 170 °C at the lower part of the reactor. From the data reported in Fig. 3, it is clear that the temperature distribution inside the reactor showed a similar tendency, though the extent of absorption of microwave radiation for the sample changed. This infers that the temperature distribution of the sample under microwave heating reflected the distribution of the microwaves' electric field (see below).
The chemical yield of the Claisen rearrangement product 2-allyl-4-methoxyphenol formed with oil-bath heating after 4 h under solvent-free conditions was 24% at 170 °C (empty triangles; Fig. 4), whereas the yield was 36% with microwave irradiation at 170 °C (full circles). The yield of product by the oil-bath heating method at 185 °C (full triangles, Fig. 4) was more than twice the chemical yield observed at 170 °C (empty triangles). Interestingly, averaging the yields at 185 °C and 170 °C from oil-bath heating gave yields (empty circles) nearly identical to those from microwave heating (full circles). Evidently, the synthesis of 2-allyl-4-methoxyphenol by the Claisen reaction is not enhanced when using the microwave method vis-à-vis the conventional method indicating that the microwave effect on the reaction is strictly a thermal one, unlike intermolecular reactions wherein non-thermal effects have been ascertained.4,6,9
The distribution of the electric field inside the waveguide was simulated by an analysis of the electromagnetic field using the RF model in the COMSOL multiphysics software version 4.3a. The 1-allyloxy-4-methoxybenzene substrate was located at the maximal electric field position inside the waveguide, and the microwave loss assessed. Results of the simulation are presented in Fig. 5, which shows the microwave electric field is concentrated at the surface between the 1-allyloxy-4-methoxybenzene sample and air. Therefore, the microwaves (wavelength, ca. 14.78 cm) were concentrated mostly at the microvolume sample, resulting in a distribution of temperature within this microvolume.
Fig. 6 displays a photograph of various samples of 1-allyloxy-4-methoxybenzene that were subsequently heated by the microwaves (170–185 °C) and conventionally by an oil bath (185 °C) for 60, 120 and 180 min {note that because of difficulties in detecting differences in color for such small samples by photography, the samples were dissolved in 2 mL of toluene solvent for better delineation}. With oil-bath heating, the color of the solutions changed from nearly colorless to a light brownish tinge, whereas under microwave heating the samples turned from pale pink (at 60 min) to a more pronounced reddish tinge after 180 min. Gas chromatographic patterns showed peaks with retention times at 8.095 min (1-allyloxy-4-methoxybenzene), 8.967 min (2-allyl-4-methoxyphenol), and a peak of an unknown substrate at 6.860 min at the completion of the reaction. The latter peak increased with reaction time, and was more intense under microwave heating than oil-bath heating. An analysis of this unknown substrate by GC/MS techniques revealed a mass signal at m/z = 124.135, which a library search of the GC/MS equipment identified it as pertaining to 4-methoxyphenol that was subsequently further confirmed by a GC/MS spectrum of a standard sample of 4-methoxyphenol whose GC signal in toluene solvent also occurred at a retention time of 6.860 min. The discoloration of the solution was likely the result of air oxidation of the by-product 4-methoxyphenol toward hydroquinone/benzoquinone-type systems.
The chemical yields of 4-methoxyphenol produced as a by-product in the synthesis of 2-allyl-4-methoxyphenol are summarized in Fig. 7. Under microwave heating the yield was 5.9% after 4 h of irradiation at a temperature of 170–185 °C. By contrast, the yield was 1.9% upon oil-bath heating at 185 °C and 1.5% at 170 °C; the yield at 200 °C was 2.8%. Evidently, microwave heating enhanced the formation of 4-methoxyphenol by nearly a factor of 3.5 compared to the average yield (data of 185 °C and 170 °C) obtained from oil-bath heating.
The relationship between reaction temperature under microwave heating and oil-bath heating with the percent yields of 2-allyl-4-methoxyphenol and 4-methoxyphenol is reported in Fig. 8a and b, respectively, for a reaction time of 3 h. There were almost no changes in the yields of 2-allyl-4-methoxyphenol in the temperature range 152–178 °C under microwave and oil-bath heating, indicating that microwaves had no effect on the Claisen intramolecular reaction other than providing the necessary thermal energy as provided by the more conventional oil-bath heating method. Curiously, however, under identical temperature conditions the yields of 4-methoxyphenol were significantly greater (threefold) under microwave irradiation than under oil-bath heating. Formation of 4-methoxyphenol by the microwave method seems to follow the formation of 2-allyl-4-methoxyphenol, which infers that 4-methoxyphenol may have been generated from a reaction involving the formation of 2-allyl-4-methoxyphenol, a point discussed further below.
3.3 Microwave-assisted synthesis of 2-allyl-4-methoxyphenol in DMSO solvent
To assess the influence of a solvent on the chemical yields of 2-allyl-4-methoxyphenol via the Claisen rearrangement of 1-allyloxy-4-methoxyphenol, the reaction was also performed in the presence of the strong microwave absorber DMSO solvent under vigorous stirring conditions (mini-bar magnet). The variance in the distribution of temperature was about 3 °C under these conditions. The solution was brought to a temperature of 170 °C using 46 W microwaves.The chemical yield data of 2-allyl-4-methoxyphenol produced in DMSO media are summarized in Fig. 9a. There was little variation – in fact negligible within experimental error – between the yields from microwave heating and oil-bath heating under identical temperature conditions. The yields of the by-product 4-methoxyphenol are reported in Fig. 9b. After 4 h of heating the yields at 170 °C were 2.4% (microwave) and 2.3% (oil bath), while under non-stirring conditions and microwave irradiation the corresponding yield was unexpectedly 2.5% for a temperature distribution of 183 °C at the upper part of the reactor and 170 °C at the lower part. The small variance in temperature had no influence on the yields.
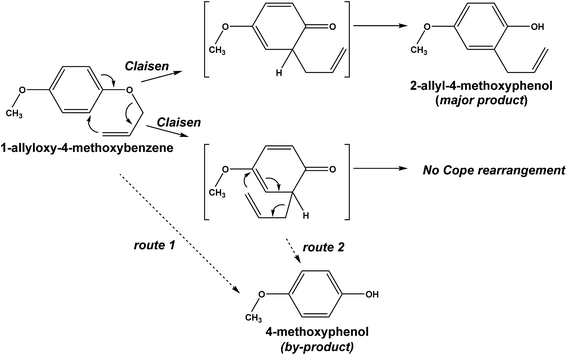
3.4 The Claisen rearrangement and formation of 4-methoxyphenol
The thermal [3,3] sigmatropic reaction involving the Claisen rearrangement of a substituted allyl aryl ether typically yields an ortho-dienone species, which can then enolize readily to give an ortho-allyphenol system (Scheme 1).25 However, when the rearrangement process occurs at an ortho position that bears a substituent, a concomitant Cope rearrangement process can take place that when followed by enolization leads to formation of a para-allyphenol species. Such an arrangement is usually irreversible in forming phenolic compounds (under thermodynamic control) so that the ortho-Claisen species is typically the major product.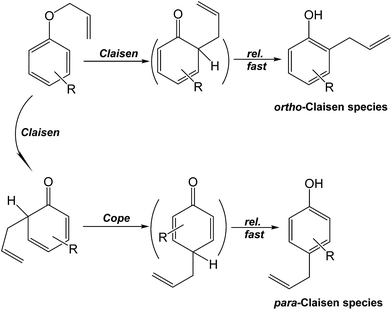 | ||
| Scheme 1 Claisen rearrangement processes for ally aryl ethers. After ref. 25. | ||
Bagnell and co-workers reported that formation of phenol, a by-product obtained subsequent to the microwave-assisted synthesis of 2-allylphenol from allylphenyl ether via the Claisen rearrangement, occurred on subjecting the allylphenol in aqueous media to high temperature conditions (200 °C).37 Although a small quantity of water was used in the present study in several processes in the conversion of the 1-allyloxy-4-methoxybenzene to 2-allyl-4-methoxyphenol, it is doubtful that such a small quantity of water would have had a significant influence on the overall Claisen process under the conditions used. To test this inference, the 1-allyloxy-4-methoxybenzene (0.2 g) was added to 0.1 mL of ion-exchanged water and then heated by microwave irradiation for 4 h. The chemical yield of 4-methoxyphenol that formed under these conditions decreased to 1.8%, indicating that the presence of water was not at the origin of 4-methoxyphenol by the microwave heating method. To test the latter assertion, 0.1 mL of acetic acid was added to 0.2 g of 1-allyloxy-4-methoxybenzene in lieu of water. The resulting solution was then exposed to microwave irradiation for 4 h; no increase in the yield of formation of 4-methoxyphenol was observed.
Our presently available results permit proposing a possible reaction pathway (Scheme 2) for the intramolecular rearrangement of 1-allyloxy-4-methoxybenzene by the Claisen process, which leads to formation of the major product 2-allyl-4-methoxyphenol. However, unlike the events displayed in Scheme 1, no Cope rearrangement is possible as the preferential para-position for rearrangement is blocked by the 4-methoxy group. On the other hand, formation of 4-methoxyphenol could, in principle, occur via two possible routes.
Route 1 might generate the by-product directly from 1-allyloxy-4-methoxybenzene in line with reports that route 1 involving a catalyzed reaction38 or where the 1-allyloxy-4-methoxybenzene ether is exposed to UV radiation (radical reactions39) leads to formation of 4-methoxyphenol during the Claisen process (photo-Claisen process39). However, neither of these processes were relevant under our experimental conditions. The alternative route of forming 4-methoxyphenol might be during the attempted occurrence of the Cope rearrangement (route 2).
To probe whether the 4-methoxyphenol occurred through route 1 or route 2, we examined the synthesis of 2-allylphenol using the corresponding allylphenyl ether by subjecting it to both microwave and oil-bath heating. No formation of phenol occurred under both heating methods (Scheme 3) either through the occurrence of the Cope rearrangement process (similar to route 2), or through the direct involvement of the allylphenyl ether (similar to route 1), especially under microwave heating. Thus we conclude that formation of 4-methoxyphenol occurred by neither routes postulated in Scheme 2.
 | ||
| Scheme 3 Proposed mechanism of the synthesis of phenol by the Claisen and Cope rearrangement of allylphenyl ether. | ||
Nonetheless, to the extent that the precursor product to the Claisen rearrangement examined herein, namely 1-allyloxy-4-methoxybenzene, was formed by reacting 4-methoxyphenol with allyl bromide,21 it is not inconceivable that under microwave heating (and to some extent oil-bath heating) a different pathway involving the precursor might be taking place to yield 4-methoxyphenol concomitant with and/or subsequent to the Claisen rearrangement process. Accordingly, the question as to how 4-methoxyphenol formed will have to await further studies.
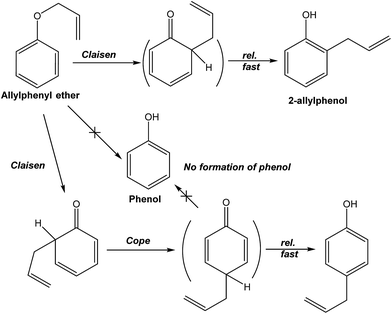
4. Summary
The question posed in this study was the how and what possible effect(s), if any, did microwaves have on intramolecular reactions such as those of the Claisen-type rearrangement carried out in DMSO solvent or solvent-free, microwave irradiation conditions, and with or without stirring. For the intramolecular reaction occurring in the strong microwave absorbing solvent (DMSO), the microwaves heated the solvent first that was subsequently followed by the transfer of thermal energy from the solvent to the starting substrate (reaction (6)). Microwave radiation was used as the heat source for this process. However, even if the microwaves had heated the starting substrate directly, there was no enhancement of the reaction (reaction (7)) demonstrating that only microwave thermal effects were effective. Unlike intermolecular reactions, no microwave non-thermal effects could be inferred in the intramolecular Claisen rearrangement process.
 | (6) |
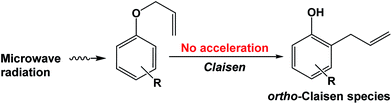 | (7) |
The above inferences notwithstanding, microwaves did have an impact on the yields of the by-product identified as 4-methoxyphenol. At 170 °C, the yield of this by-product was nearly fourfold greater (ca. 6%) under microwave irradiation than under oil-bath heating (ca. 1.5%), which strongly suggests that, under solvent-free conditions, microwave non-thermal effect(s) were influential during the various mechanistic steps in the Claisen rearrangement of the initial substrate, at least in those steps that led to the by-product. By contrast, the yields of 4-methoxyphenol in DMSO solvent media were identical (ca. 2.5%) regardless of whether the precursor substrate was microwave or oil-bath heated.
Acknowledgements
We are grateful to the Japan Society for the Promotion of Science (JSPS; Grant-in-aid for Scientific Research No. C-25420820) for financial support. We also thank Sophia University for a grant from the Sophia University-wide Collaborative Research Fund to S. H. One of us (N. S.) is grateful to Prof. Albini of the University of Pavia (Italy) for his continued hospitality during the many winter semesters in his laboratory.References
- See e.g., Microwaves in Organic Synthesis, ed. A. de la Hoz and A. Loupy, Wiley-VCH Verlag, GmbH, Weinheim, Germany, 3rd edn, 2012, vol. 1 and 2 Search PubMed.
- See e.g., G. Majetich and R. Hicks, J. Microwave Power, 1995, 30, 27 Search PubMed.
- C. O. Kappe, Angew. Chem., Int. Ed., 2004, 43, 6250 CrossRef CAS PubMed.
- S. Horikoshi, M. Kamata, S. Sakamoto, T. Mitani and N. Serpone, Ind. Eng. Chem. Res., 2014, 53, 14941 CrossRef CAS.
- S. Horikoshi and N. Serpone, Catal. Sci. Technol., 2014, 4, 1197 CAS.
- S. Horikoshi, S. Matsuzaki, S. Sakamoto and N. Serpone, Radiat. Phys. Chem., 2014, 97, 48 CrossRef CAS.
- S. Horikoshi and N. Serpone, Catal. Today, 2014, 224, 225 CrossRef CAS.
- S. Horikoshi, T. Sumi and N. Serpone, Chemical Engineering and Processing: Process Intensification, 2013, 72, 59 CrossRef.
- S. Horikoshi, A. Osawa, S. Sakamoto and N. Serpone, Appl. Catal., A, 2013, 460–461, 52 CrossRef CAS.
- S. Horikoshi, T. Sumi and N. Serpone, J. Microwave Power, 2012, 46, 215 Search PubMed.
- S. Horikoshi, S. Matsuzaki, T. Mitani and N. Serpone, Radiat. Phys. Chem., 2012, 81, 1885 CrossRef CAS.
- S. Horikoshi, Y. Suttisawat, A. Osawa, C. Takayama, X. Chen, S. Yang, H. Sakai and N. Serpone, J. Catal., 2012, 289, 266 CrossRef CAS.
- S. Horikoshi, A. Osawa, M. Abe and N. Serpone, J. Phys. Chem. C, 2011, 115, 23030 CAS.
- S. Horikoshi, S. Sato, M. Abe and N. Serpone, Ultrason. Sonochem., 2011, 18, 938 CrossRef CAS PubMed.
- S. Horikoshi and N. Serpone, Mini-Rev. Org. Chem., 2011, 8, 299 CrossRef CAS.
- S. Horikoshi, M. Muratani, K. Miyabe, K. Ohmura, T. Hirowatari, N. Serpone and M. Abe, J. Oleo Sci., 2011, 60, 375 CrossRef CAS PubMed.
- S. Horikoshi, A. Osawa, Y. Suttisawat, M. Abe and N. Serpone, Org. Process Res. Dev., 2010, 14, 1461 CrossRef.
- S. Horikoshi, M. Fukui, K. Tsuchiya, M. Abe and N. Serpone, Chem. Phys. Lett., 2010, 491, 244 CrossRef CAS.
- S. Horikoshi, M. Abe, M. Kajitani and N. Serpone, Utilization of 5.8 GHz microwaves in organic syntheses, Proc. GCMEA 2008 MAJIC, 1st Global Congress on Microwave Energy Applications The New Flame for Humanity, Lake Biwa, Otsu, Japan, August 4–8 2008 Search PubMed.
- S. Horikoshi, T. Hamamura, M. Kajitani, M. Yoshizawa-Fujita and N. Serpone, Org. Process Res. Dev., 2008, 12, 1089 CrossRef CAS.
- C. J. Davis, T. E. Hurst, A. M. Jacob and C. J. Moody, J. Org. Chem., 2005, 70, 4414 CrossRef CAS PubMed.
- T. Yamamoto, Y. Wada, H. Enokida, M. Fujimoto, K. Nakamura and S. Yanagida, Green Chem., 2003, 5, 690 RSC.
- S. Kotha, K. Mandal, A. Chandra Deb and S. Banerjee, Tetrahedron Lett., 2004, 45, 9603 CrossRef CAS.
- D. K. Deodhar, A. S. Tipnis and S. D. Samant, Indian J. Chem., 2010, 49B, 1552 CAS.
- Y. Lin, J. Cheng and Y.-H. Chu, Tetrahedron, 2007, 63, 10949 CrossRef CAS.
- I. A. Sayed, V. V. Thakur, M. D. Nikalje, G. K. Dewkar, S. P. Kotkar and A. Sudalai, Tetrahedron, 2005, 61, 2831 CrossRef.
- A. M. Jacob and C. J. Moody, Tetrahedron Lett., 2005, 46, 8823 CrossRef CAS.
- M. Mansha, Y. Abbas and N. Ullah, Synth. Commun., 2015, 45, 599 CrossRef.
- D. Craig, F. Paina and S. C. Smith, Chem. Commun., 2008, 3408 RSC.
- E. Gajdošíková, M. Martinková, J. Gonda and P. Čonka, Molecules, 2008, 13, 2837 CrossRef PubMed.
- H. Valizadeh and A. Shockravi, J. Heterocycl. Chem., 2006, 43, 763 CrossRef CAS.
- D. O. Tymoshenko, Mini-Rev. Org. Chem., 2015, 5, 85 CrossRef.
- X. Li, X. Zhang, Z. Yu, X. Liu, Q. You and Q. Guo, J. Chem. Res., 2011, 35, 630 CrossRef CAS.
- S. M. M. Lopes, B. S. Santos, F. Palacios and T. M. V. D. Pinho e Melo, Microwave-assisted reactions of allenic esters: [3 + 2] annulations and allenoate-Claisen rearrangement (RG-4087UP), ARKIVOC, 2010, 70–81 CAS , Part (v).
- P.-K. Chen, M. R. Rosana, G. B. Dudley and A. E. Stiegman, J. Org. Chem., 2014, 79, 7425 CrossRef CAS PubMed.
- S. Horikoshi, T. Sumi and N. Serpone, J. Microwave Power, 2012, 46, 215 Search PubMed.
- L. Bagnell, T. Cablewski, C. R. Strauss and W. Trainor, J. Org. Chem., 1996, 61, 7355 CrossRef CAS PubMed.
- M. Chouhan, K. Kumar, R. Sharma, V. Grover and V. A. Nair, Tetrahedron Lett., 2013, 54, 4540 CrossRef CAS.
- I. Iwakura, A. Yabushita, J. Liu, K. Okamura and T. Kobayashi, Phys. Chem. Chem. Phys., 2012, 14, 9696 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra18039b |
| This journal is © The Royal Society of Chemistry 2015 |