Differently ordered carbonaceous structures synthesized by bubbled Ar or He plasmas inside methylene blue solutions with contrasting Escherichia coli growth inhibition effects†
Abstract
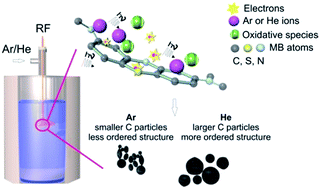
This work is a comparative study on the synthesis of carbonaceous structures with different ordering, aided by an atmospheric pressure plasma in contact with methylene blue aqueous solutions, by utilizing two different inert plasma gases, argon and helium respectively. The study follows the diagnosis of the two plasmas by optical emission spectroscopy, their different degradation effects on methylene blue solutions and the characterization of the carbonaceous structures obtained as a result of plasma treatment with transmission electron microscopy, Raman and X-ray photoelectron spectroscopy. The carbon based structures obtained with the helium plasma have a higher degree of ordering and are larger in size compared to the Ar plasma ones. The bacterial growth inhibition assays show opposite behaviours: one structure inhibits the growth while the other favours it.

 Please wait while we load your content...
Please wait while we load your content...