Fabrication of ZnIn2S4–g-C3N4 sheet-on-sheet nanocomposites for efficient visible-light photocatalytic H2-evolution and degradation of organic pollutants
Hong Liu*,
Zhitong Jin,
Zhengzheng Xu,
Zhe Zhang and
Dan Ao
Department of Chemical Engineering, School of Environmental and Chemical Engineering, Shanghai University, 99 Shangda Road, Shanghai 200444, P. R. China. E-mail: liuhong@shu.edu.cn; Fax: +86-21-66137725; Tel: +86-21-66137487
First published on 9th November 2015
Abstract
ZnIn2S4–g-C3N4 sheet-on-sheet nanocomposites with different g-C3N4 contents were synthesized by a facile hydrothermal method and characterized by thermogravimetric analysis (TGA), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), Fourier transform infrared spectroscopy (FT-IR), transmission electron microscopy (TEM), high-resolution transmission electron microcopy (HRTEM), N2 adsorption–desorption, ultraviolet-visible diffuse reflection spectroscopy (DRS), photoluminescence (PL) spectroscopy and photoelectrochemical (PEC) experiments. The photocatalytic activities of these samples were evaluated by the photocatalytic H2-production and degradation of organic pollutants (methyl orange and phenol) under visible-light illumination (λ > 420 nm). The results showed that the ZnIn2S4–g-C3N4 composite photocatalysts displayed higher photocatalytic activity than the pristine g-C3N4 and ZnIn2S4 both for H2-evolution and degradation of pollutants. The optimal g-C3N4 content was determined to be 40 wt%, and the corresponding H2-production rate was 953.5 μmol h−1 g−1, which was about 1.91 times higher than that of pure ZnIn2S4. The enhanced photocatalytic activity of ZnIn2S4–g-C3N4 composites should be attributed to the well-matched band structure and intimate contact interfaces between ZnIn2S4 and g-C3N4, which led to the effective transfer and separation of the photogenerated charge carriers. Moreover, the ZnIn2S4–g-C3N4 composites showed excellent stability during the photocatalytic reactions under visible light. A possible mechanism of the enhanced photocatalytic activity of ZnIn2S4–g-C3N4 composites was proposed and supported by the PL and PEC results.
1. Introduction
Photocatalysis, as a green technology, is attracting considerable interests due to its great potential in solving global environmental and energy problems.1–3 TiO2 is the most widely studied photocatalyst due to its unique characteristics such as high photocatalytic activity, low cost, stability and nontoxicity. However, TiO2 only works in the ultraviolet region due to its wide band gap (∼3.2 eV), which restricts its practical applications.4,5 Therefore, exploring new photocatalytic materials with high photocatalytic activity under visible-light (∼43% of the solar spectrum) is indispensable in this field.As a typical metal-free polymeric semiconductor, graphitic carbon nitride (g-C3N4) is a very promising material for solar energy utilization because of its high thermal and chemical stability, nontoxicity, easy to prepare and desirable band gap of 2.7 eV.6–12 The potential applications of g-C3N4 in photocatalytic hydrogen evolution, CO2 reduction, degradation of pollutants, and photocatalytic organic synthesis have been reported.6,7,9–12 However, the photocatalytic efficiency of pure g-C3N4 is limited by the high recombination rate of photogenerated electron–hole pairs and low surface area.6,9 To enhance its photocatalytic performance, a number of methods have been exploited, including preparing novel nanostructures (mesoporous structure13,14 and nanosheets7,11), doping with metal or nonmetal elements,15–18 deposition of noble metal,19 protonating by strong acids20 and construction of heterojunction composite.21–23 Among them, designing heterojunction composite is regarded as one of the best methods to promote the separation of photogenerated electrons and holes, which can effectively improve the photocatalytic efficiency.
ZnIn2S4, as a typical member of the AB2X4 family of semiconductors, is a good candidate for a photocatalyst under the visible-light irradiation due to its suitable bandgap (2.34–2.48 eV).24–26 ZnIn2S4 exhibits two distinct polymorphs based on cubic and hexagonal lattices. Previous studies revealed that both polymorphs of ZnIn2S4 are active for photocatalytic hydrogen generation and pollutants degradation under visible-light irradiations and show considerable chemical stability.27–30 However, the photocatalytic efficiency over pure ZnIn2S4 is low due to the short lifetimes of the photo-generated electron–hole pairs. To enhance the photocatalytic performance of ZnIn2S4, numerous effects have been made. For example, by controlling morphologies of ZnIn2S4,26,31–34 doping with metals,35,36 and incorporation of multiwalled carbon nanotubes,37 reduced graphene oxide28,30 or other semiconductors (such as CdS,38 MoS2,29,39 CdIn2S4 (ref. 40) and TiO2 (ref. 41)), the photocatalytic performance of ZnIn2S4 have been enhanced to a certain degree. However, it should be noted that the photocatalytic activity of ZnIn2S4 still requires further improvement.
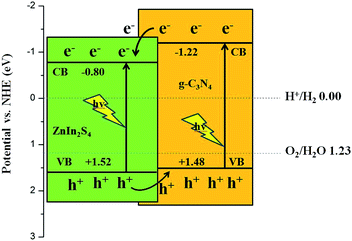
Similar to g-C3N4, hexagonal ZnIn2S4 has an intrinsic layered structure based on a stacking of packets of S–Zn–S–In–S–In–S layers.29 It is expected that it is facile to grow layer ZnIn2S4 on g-C3N4 surface due to their analogous layered structures which can minimize the lattice mismatch. Besides this, the position of the conduction band (CB) of g-C3N4 (−1.12 eV vs. NHE42) is more negative than that of ZnIn2S4 (−0.68 eV vs. NHE39) and thus provides possibility for a directional transfer of the photo-generated electrons from g-C3N4 to ZnIn2S4. Moreover, due to the valence band (VB) of ZnIn2S4 (+1.62 eV vs. NHE39) are more positive than that of g-C3N4 (+1.57 eV vs. NHE42), the holes on VB of ZnIn2S4 will migrate to that of g-C3N4, achieving the separation of charge carriers in composites. Therefore, it can be supposed that combining ZnIn2S4 with g-C3N4 may result in an improved photocatalytic efficiency compared with individual g-C3N4 and ZnIn2S4. However, to the best of our knowledge, no work related to ZnIn2S4–g-C3N4 photocatalyst has been reported so far.
In this study, ZnIn2S4–g-C3N4 sheet-on-sheet nanocomposites with different g-C3N4 contents were synthesized by a facile hydrothermal method. The as-prepared ZnIn2S4–g-C3N4 composites showed a significantly enhanced photocatalytic performance for hydrogen evolution and degradation of organic pollutants under visible light irradiation. A possible enhancement mechanism for the improved photocatalytic activity in the ZnIn2S4–g-C3N4 composites was also proposed.
2. Experimental
2.1 Materials preparation
All the chemicals were of analytical grade from the Sinopharm Chemical Reagent Co., Ltd.2.2 Characterization
The crystal structure of the samples was identified by an X-ray diffractometer (XRD, D-MAX-2550, λ = 0.15418 nm) using Cu Kα radiation. The morphologies and microstructures of the photocatalysts were evaluated by transmission electron microscopy (TEM FEI TECNAI G20) and high-resolution transmission electron microscopy (HRTEM FEI TECNAI G2F20). The surface elemental component and the chemical state of the sample were analyzed by X-ray photoelectron spectroscopy (XPS, ESCALAB 250Xi). FT-IR spectrum was performed on a FT-IR spectrophotometer (AVATAR370) using KBr disks at room temperature. Ultraviolet-visible (UV-vis) diffuse reflectance spectra (DRS) of the samples were obtained on an UV-vis spectrophotometer (Hitachi U-3010) using BaSO4 as a reference. The photoluminescence (PL) spectra were measured using a fluorescence spectrophotometer (Hitachi RF-5301) at room temperature. The Brunauer–Emmett–Teller (BET) specific surface areas of the samples were analyzed by nitrogen adsorption using a Micromeritics ASAP 2020 nitrogen adsorption apparatus. Thermogravimetric analysis (TGA) was performed on a Netzsch 409 PC thermal analyzer under nitrogen atmosphere with a heating rate of 10 °C min−1.2.3 Photocatalytic measurement
The photocatalytic hydrogen-production experiment was conducted in a closed gas circulation and evacuation system fitted with a top window Pyrex cell. A 300 W xenon lamp (PLS-SXE300C, Beijing Perfectlight Co. Ltd., China) coupled with a UV cut-off filter (λ > 420 nm) was used to provide the visible light. In each run, 0.05 g photocatalyst was dispersed in a 100 mL aqueous solution containing 10 mL trolamine, which served as a sacrificial agent. Before the irradiation with visible light, the suspension was degassed with N2 for 1 h to drive away the O2 in the system. The reaction cell was kept at room temperature with cooling water. The produced H2 was detected using an online gas chromatography (GC7900, N2 carrier, 5A molecular sieve column, TCD detector).Photocatalytic degradation activity of the samples was estimated by monitoring the degradation of methyl orange (MO) and phenol in an aqueous solution under visible light irradiation. The reaction was performed in a photochemical reactor (BL-GHX-V, Shanghai Bilon Instruments Co., Ltd, China) equipped with a 500 W Xe lamp combined with a 420 nm cut-off filter as a light source. All experiments were conducted at room temperature in air. In a typical photocatalytic experiment, 0.02 g photocatalyst was added into 50 mL pollutant solutions (10 mg L−1) in a reaction cell with a Pyrex jacket. Prior to irradiation, the suspension was magnetically stirred in the dark for 30 min to reach an adsorption–desorption equilibrium. Then, the suspension was exposed to visible light irradiation under magnetic stirring. At given time intervals, about 5 mL suspensions were collected and centrifuged (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 6 min) to remove the photocatalyst particles. Then, the pollutant concentration of the obtained solution was analyzed by a UV-vis spectrophotometer (Agilent 8453) by checking the absorbance at 506 nm and 270 nm for MO and phenol, respectively. The total organic carbon (TOC) assays were carried out using a TOC analyzer (Multi N/C 2100). To probe the oxidizing species in the photocatalytic degradation, the disodium ethylenediamine tetraacetate (EDTA-2Na), isopropyl alcohol (IPA), or benzoquinone (BQ), was added in the solution. The concentrations of the quenching chemicals were 2 mM.
000 rpm, 6 min) to remove the photocatalyst particles. Then, the pollutant concentration of the obtained solution was analyzed by a UV-vis spectrophotometer (Agilent 8453) by checking the absorbance at 506 nm and 270 nm for MO and phenol, respectively. The total organic carbon (TOC) assays were carried out using a TOC analyzer (Multi N/C 2100). To probe the oxidizing species in the photocatalytic degradation, the disodium ethylenediamine tetraacetate (EDTA-2Na), isopropyl alcohol (IPA), or benzoquinone (BQ), was added in the solution. The concentrations of the quenching chemicals were 2 mM.
2.4 Photoelectrochemical measurements
Photocurrent measurements were performed on a CHI 660E electrochemical workstation (Chenhua Instrument, Shanghai, China) in a conventional three electrode configuration with a Pt foil as the counter electrode and a Ag–AgCl (saturated KCl) as the reference electrode. A 300 W Xe arc lamp severed as a light source. A 0.5 M Na2SO4 aqueous solution was used as the electrolyte. The working electrodes were prepared as follows: 10 mg of the prepared photocatalyst was ground with 20 μL of a poly(3,4-ethylenedioxythiophene)/poly(styrenesulfonate) (PEDO/TPSS, 1.3–1.7%) aqueous solution and 100 μL of distilled water to make a slurry. The slurry was then spread on a 1.5 cm × 1.0 cm indium-tin oxide (ITO) glass substrate with an active area of about 0.3 cm2 by the doctor-blade method, using adhesive tape as the space. The film was dried in air and annealed at 150 °C for 30 min in a flowing N2 atmosphere. The photoresponses of the samples as light on and off were measured at 0.0 V. Electrochemical impedance spectroscopy (EIS) measurements were determined at an AC voltage magnitude of 5 mV with the frequency range of 106 to 0.01 Hz with the initial potential (0 V) in 0.01 M Na2SO4.3. Results and discussion
3.1 Structure and properties characterization
The contents of ZnIn2S4–g-C3N4 composites were determined by thermogravimetric analysis (TGA) under nitrogen atmosphere from room temperature to 800 °C with a heating rate of 10 °C min−1. The decomposition of the g-C3N4 nanosheets start at 560 °C and is complete at ∼760 °C which is attributed to the burning of g-C3N4 as shown in Fig. 1. In all ZnIn2S4–g-C3N4 composites, this weight loss region can also be observed and the g-C3N4 content can be easily determined from the corresponding weight loss in the thermogram. As shown in Table 1, the real g-C3N4 contents are nearly consistent with the theoretical values.| Samples | g-C3N4 content (wt%) | SBET (m2 g−1) |
|---|---|---|
| ZnIn2S4 | 0 | 66.9 |
| ZISCN20 | 20.5 | 63.9 |
| ZISCN30 | 27.4 | 42.3 |
| ZISCN40 | 38.6 | 35.2 |
| ZISCN50 | 52.9 | 29.8 |
| g-C3N4 nanosheet | 100 | 10.4 |
Fig. 2 shows the XRD patterns of g-C3N4 nanosheets, ZnIn2S4 and ZnIn2S4–g-C3N4 composites with different g-C3N4 contents. The XRD pattern of the ZnIn2S4 can be assigned to a hexagonal phase of ZnIn2S4 (JCPDS no. 03-065-2023) and the characteristic peaks at 21.4°, 27.8° and 47.3° are attributed to (006), (102) and (110) crystal planes of ZnIn2S4.29 Two distinct peaks around 13.1° and 27.7° are observed in the XRD pattern of g-C3N4 nanosheets, corresponding to the (100) and (002) diffraction planes, respectively.6–12 For all ZnIn2S4–g-C3N4 composites, only broadened diffraction peaks for ZnIn2S4 have been observed due to the overlapping of the tow closely peaks for ZnIn2S4 (27.8°) and g-C3N4 (27.7°).
The morphologies and microstructures of g-C3N4 nanosheets, ZnIn2S4 and ZISCN40 were characterized by TEM and HRTEM. It can be observed from Fig. 3a that pure g-C3N4 shows a two-dimensional nanosheet structure with a wrinkled surfaces. The pristine ZnIn2S4 sample (Fig. 3b) exhibits microsphere morphology with a diameter of about 1.5 μm. Further observation show that individual ZnIn2S4 microsphere are built by numerous nanosheets with a thickness of about 10 nm (Fig. 3c). After hybridization with g-C3N4, the ZnIn2S4 microspheres are unfolded and distributed uniformly as nanosheets on the surface of g-C3N4 nanosheets to form a 2D sheet-on-sheet structure (Fig. 3d). This phenomenon may be ascribed to the important effect of surface groups of g-C3N4 in the crystal growth and self-assembly process of ZnIn2S4 materials. The 2D sheet-on-sheet composite structure offers the maximum contact area between two components (ZnIn2S4 and g-C3N4 nanosheets), which will facilitate the transfer of photogenerated carriers and improve the photocatalytic performance. The HRTEM image of ZISCN40 (Fig. 3e) reveals that a lattice spacing of ZnIn2S4 crystallites is ∼0.324 nm, corresponding to the (102) plane of ZnIn2S4.30 The EDS spectrum (Fig. 3f) of the composites clearly confirms the presence of Zn, In, S, C and N elements (other peaks originate from the substrate).
 | ||
| Fig. 3 TEM images of (a) g-C3N4 nanosheets, (b and c) ZnIn2S4 and (d) ZISCN40, (e) HRTEM image and (f) EDS spectrum of ZISCN40. | ||
Fig. 4 shows the FT-IR spectra of g-C3N4 nanosheets, ZnIn2S4 and the ZnIn2S4–g-C3N4 (40%) sample. In the FT-IR spectra of g-C3N4, the broad peak at 3250 cm−1 can be attributed to stretching mode of the N–H bond.43 And the peaks located at about 1240, 1320, 1410, 1460, 1575 and 1640 cm−1 are ascribed to aromatic C–N stretching vibration modes and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching vibration modes.44,45 The peak at 812 cm−1 is originated from characteristic breathing vibration of triazine ring.44 For ZnIn2S4, the broad band centered at 3430 cm−1 and 1620 cm−1 are contributed to the surface absorbed water molecules and hydroxyl groups.46 In the case of the ZnIn2S4–g-C3N4 composite, the IR bands characteristic of g-C3N4 and ZnIn2S4 are also identified, confirming that the composite photocatalyst is composed of g-C3N4 and ZnIn2S4.
N stretching vibration modes.44,45 The peak at 812 cm−1 is originated from characteristic breathing vibration of triazine ring.44 For ZnIn2S4, the broad band centered at 3430 cm−1 and 1620 cm−1 are contributed to the surface absorbed water molecules and hydroxyl groups.46 In the case of the ZnIn2S4–g-C3N4 composite, the IR bands characteristic of g-C3N4 and ZnIn2S4 are also identified, confirming that the composite photocatalyst is composed of g-C3N4 and ZnIn2S4.
The X-ray photoelectron spectra (XPS) were further employed to elucidate the surface elemental composition of g-C3N4, ZnIn2S4 and ZnIn2S4–g-C3N4 and to further study the interaction of ZnIn2S4 with the g-C3N4 support. The XPS survey spectrum of ZISCN40 (Fig. 5a) confirms that the composite is mainly composed of C, N, Zn, In and S. The C 1s spectrum of pure g-C3N4 (Fig. 5b) can be deconvoluted into three peaks at 284.7, 286.4 and 288.3 eV. The C peak at 288.3 eV is identified as sp2-bonded carbon (N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), the C peak at 284.7 eV is corresponded to graphitic carbon which was usually observed on the XPS characterization for carbon nitrides.44,45 The weak peak at 286.4 eV can be assigned to sp3-coordinated carbon bonds from the defects on g-C3N4 surfaces.44,45 The N 1s spectrum of pure g-C3N4 (Fig. 5c) can be deconvoluted into three peaks at 398.1, 399.3 and 400.7 eV, respectively, corresponding to the nitrogen atoms in the aromatic rings (C
N), the C peak at 284.7 eV is corresponded to graphitic carbon which was usually observed on the XPS characterization for carbon nitrides.44,45 The weak peak at 286.4 eV can be assigned to sp3-coordinated carbon bonds from the defects on g-C3N4 surfaces.44,45 The N 1s spectrum of pure g-C3N4 (Fig. 5c) can be deconvoluted into three peaks at 398.1, 399.3 and 400.7 eV, respectively, corresponding to the nitrogen atoms in the aromatic rings (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–C), tertiary nitrogen (N–(C)3) and C–N–H.47,48 In comparison with pure g-C3N4, the negative shift of the C 1s peaks and positive shift of N 1s peaks are observed for the ZISCN40 sample, indicating the g-C3N4 structure has changed after interaction with ZnIn2S4 to form the heterojunction. The main binding energy peaks located at 1044.5 eV (Zn 2p1/2), 1021.5 eV (Zn 2p3/2), 452.4 eV (In 3d3/2), 444.9 eV (In 3d5/2), 161.5 eV (S 2p3/2) and 162.7 eV (S 2p1/2) can be observed from the high resolution spectra of the pristine ZnIn2S4 in Fig. 5d–f, respectively. The binding energy values are very close to the reported ones,29,30,38,39 indicating that the valence states of Zn, In and S are +2, +3 and −2, respectively. After modification with g-C3N4, slight shifts toward lower binding energies can be observed from the high resolution spectra of ZISCN40 in Fig. 5d–f, implying that the binding energies of the core level electrons of those metal and sulfide ions are affected due to the possible chemical bonding actions among the composite components. The XPS results clearly indicate the presence of chemical bonds between g-C3N4 and ZnIn2S4, rather than a simple physical mixture.
N–C), tertiary nitrogen (N–(C)3) and C–N–H.47,48 In comparison with pure g-C3N4, the negative shift of the C 1s peaks and positive shift of N 1s peaks are observed for the ZISCN40 sample, indicating the g-C3N4 structure has changed after interaction with ZnIn2S4 to form the heterojunction. The main binding energy peaks located at 1044.5 eV (Zn 2p1/2), 1021.5 eV (Zn 2p3/2), 452.4 eV (In 3d3/2), 444.9 eV (In 3d5/2), 161.5 eV (S 2p3/2) and 162.7 eV (S 2p1/2) can be observed from the high resolution spectra of the pristine ZnIn2S4 in Fig. 5d–f, respectively. The binding energy values are very close to the reported ones,29,30,38,39 indicating that the valence states of Zn, In and S are +2, +3 and −2, respectively. After modification with g-C3N4, slight shifts toward lower binding energies can be observed from the high resolution spectra of ZISCN40 in Fig. 5d–f, implying that the binding energies of the core level electrons of those metal and sulfide ions are affected due to the possible chemical bonding actions among the composite components. The XPS results clearly indicate the presence of chemical bonds between g-C3N4 and ZnIn2S4, rather than a simple physical mixture.
 | ||
| Fig. 5 XPS spectra of g-C3N4 nanosheets, ZnIn2S4 and ZISCN40: (a) survey spectrum of ZISCN40, (b) C 1s, (c) N 1s, (d) Zn 2p, (e) In 3d, (f) S 2p. | ||
The Brunauer–Emmett–Teller specific surface areas (SBET) of pure g-C3N4 nanosheets, ZnIn2S4, and the as-prepared ZnIn2S4–g-C3N4 composites were measured by nitrogen adsorption and summarized in Table 1. As listed in Table 1, the BET specific surface area of the pristine ZnIn2S4 is about 66.9 m2 g−1. For the ZnIn2S4–g-C3N4 composites, the surface area decreases with the enhancement of g-C3N4 mass ratio because the surface area of g-C3N4 nanosheets is only 10.4 m2 g−1.
The UV-vis diffuse reflectance spectra of the ZnIn2S4–g-C3N4 composites with different weight ratios of g-C3N4, as well as g-C3N4 nanosheets and ZnIn2S4 are compiled in Fig. 6a. Pure g-C3N4 sample shows absorption wavelengths from the UV to the visible range up to 460 nm. For ZnIn2S4, the absorption edge is about 600 nm, exhibiting the broad absorption region of visible-light. The ZnIn2S4–g-C3N4 composites have absorption edges to longer wavelengths in comparison with the pure g-C3N4. The observations are attributed to the interaction between g-C3N4 and ZnIn2S4 in the composites.48,49 The interaction probably plays a significant role in improving the separation of the photogenerated electron–hole pairs to enhance the photocatalytic activity. The band gap energy of a semiconductor can be calculated by the following equation: αhν = A(hν − Eg)n/2, in which α, h, ν, Eg, and A are the absorption coefficient, Planck constant, the light frequency, the band gap, and a constant, respectively. The value of n depends on the type of optical transition of the semiconductor (n = 1 for direct transition and n = 4 for indirect transition). For g-C3N4 and ZnIn2S4, the n value of 1 is used.50,51 Therefore, as can be seen from Fig. 6b, the corresponding band gap values of ZnIn2S4, ZISCN20, ZISCN30, ZISCN40, ZISCN50 and g-C3N4 nanosheets are found to be 2.32, 2.34, 2.35, 2.38, 2.40 and 2.70 eV, respectively. The valence band edge potential and the conduction band edge potential of a semiconductor material can be determined by using the following equation: EVB = X − Ee + 0.5Eg, where EVB represents valence band edge potential, X is the electronegativity of the semiconductor estimated by the geometric mean of the electronegativity of the constituent atoms. Ee is the energy of free electrons on the hydrogen scale (∼4.5 eV), Eg is the band gap energy of the semiconductor. The CB edge potential ECB can be determined by ECB = EVB − Eg. The X value of pure ZnIn2S4 and g-C3N4 is about 4.86 and 4.63 eV. The EVB of bare ZnIn2S4 and g-C3N4 nanosheets can be assigned to be +1.52 and +1.48 eV, respectively, and the corresponding ECB of ZnIn2S4 and g-C3N4 can be estimated to be −0.80 and −1.22 eV, respectively.
3.2 Photocatalytic activity
Photocatalytic hydrogen evolution activities of as-prepared samples were evaluated under visible light (λ > 420 nm) irradiation using trolamine as the sacrificial reagent to quench photoinduced holes. As shown in Fig. 7, the photocatalytic H2-production rate is negligible when g-C3N4 alone is used as photocatalyst due to fast recombination of photocatalytic electron–hole pairs, indicating that pure g-C3N4 nanosheets is not active for photocatalytic hydrogen production. Pristine ZnIn2S4 without g-C3N4 shows a low photocatalytic activity with the H2 evolution rate of 500.0 μmol h−1 g−1. After introduction of g-C3N4, the H2-production activity of ZnIn2S4 is significant enhanced. The rate of H2 evolution over ZnIn2S4–g-C3N4 composites increases with increasing g-C3N4 content, achieving a maximum of 953.5 μmol h−1 g−1 at the g-C3N4 content of 40 wt%, which is about 1.91 times higher than that of pure ZnIn2S4. A further increase in the content of g-C3N4 leads to a reduction in the photocatalytic activity. This decrease is probably due to the fact that excessive amount of g-C3N4 may cover the active sites on the surface of ZnIn2S4.45,52 Therefore, an appropriate g-C3N4 loading amount is crucial to achieve the optimized photocatalytic activity of ZnIn2S4 photocatalyst.The photocatalytic degradation performances of as-prepared samples were also evaluated by using MO and phenol as the model pollutant under visible light irradiation (Fig. 8). The blank experiments (without any photocatalyst) show that direct photolysis of MO and phenol under visible light can be neglected. Fig. 8a shows that the pure g-C3N4 nanosheets has a poor activity, in which only 5.7% of MO is decomposed after irradiation for 120 min. Moreover, only 72.6% of MO can be removed by ZnIn2S4 for the same irradiation time. After coupling with g-C3N4, the photodegradation activity of ZnIn2S4 is remarkably enhanced. It can be observed that the optimum g-C3N4 mass ratio is 40 wt%. The degradation ratio of MO is up to 95.3% in the presence of ZISCN40 after 120 min irradiation. Fig. 8b displays the photodegradation activity of phenol over the as-prepared samples. Both pure g-C3N4 and ZnIn2S4 samples show low activities, in which only 21.4% and 52.5% of phenol are degraded after reacting for 240 min, respectively. In contrast, all the ZnIn2S4–g-C3N4 samples display much improved activities. The trend of activity change of these ZnIn2S4–g-C3N4 samples for the photodegradation of phenol is the same as that of MO degradation. The degradation ratio of phenol on the optimal photocatalyst (ZISCN40) reaches to 72.3% after 240 min irradiation. In addition, after 120 min or 240 min irradiation the mineralization rate of MO and phenol over ZISCN40 reaches to 70.0% and 58.0%, respectively (Fig. 9).
 | ||
| Fig. 8 Photocatalytic activities of g-C3N4 nanosheets, ZnIn2S4 and ZnIn2S4–g-C3N4 composites on the degradation of the target organic pollutions under visible-light irradiation: (a) MO, (b) phenol. | ||
 | ||
| Fig. 9 TOC removal during (a) MO and (b) phenol degradation over ZISCN40 under visible light illumination. | ||
To understand the photocatalytic mechanism, the main active oxidants in the photocatalytic degradation process were identified. It is well-known that the oxidants generated in the photocatalytic process can be measured through trapping by IPA (a scavenger of hydroxyl radicals), EDTA-2Na (a scavenger of holes) or BQ (a scavenger of ˙O2−). It can be clearly seen from Fig. 10 that the addition of EDTA-2Na greatly reduces the photodegradation rate of MO and phenol in the ZISCN40 suspension, whereas the addition of BQ has little effect on the photodegradation rate of MO and phenol. In addition, the addition of IPA as hydroxyl radicals' scavenger causes a minor change in the photocatalytic degradation of the above pollutants. It indicates that both the holes and hydroxyl radicals are the active species in the photocatalytic degradation of MO and phenol, while ˙O2− can be negligible in the reaction.
 | ||
| Fig. 10 Photocatalytic degradation of MO (a) and phenol (b) with the addition of hole and radical scavenger. | ||
To reveal the stability of as-synthesized photocatalysts, the cycling hydrogen evolution and MO degradation experiments were performed over ZISCN40 under the same conditions. As shown in Fig. 11, there is no significant decrease in H2-production rate and photodegradation rate after four consecutive cycles, indicating the good stability of the ZnIn2S4–g-C3N4 composite during photocatalytic reactions.
 | ||
| Fig. 11 Cycling runs for (a) the photocatalytic hydrogen evolution and (b) MO degradation in the presence of ZISCN40 under visible light illumination. | ||
3.3 Mechanism for the enhanced photocatalytic activity of ZnIn2S4–g-C3N4 composites
It has been widely accepted that the separation efficiency of electrons and holes played a crucial role in the photocatalytic reaction. Photoluminescence (PL) analysis was applied to reveal the efficiency of charge carrier trapping and separation of the photoinduced electrons and holes in semiconductors. Fig. 12a shows the PL spectra of pure g-C3N4 nanosheets, ZnIn2S4 and ZISCN40 sample with an excitation wavelength of 385 nm. It can be seen that the pure g-C3N4 has a strong emission peak at around 460 nm, which corresponds to band gap recombination of electron–hole pairs. The PL intensity of bare ZnIn2S4 is weak probably because the amount of photogenerated electron–hole pairs is less under the same irradiation conditions.53,54 Similar results were also observed on the BiOBr/g-C3N4,54 Ag3PO4/g-C3N4,55 Fe2O3/g-C3N4,56 ZnFe2O4/g-C3N4,57 ZnS/g-C3N4,51 BiVO4/g-C3N4 (ref. 58) and MnFe2O4/g-C3N4 (ref. 59) heterojunction systems by other groups. The ZnIn2S4–g-C3N4 composite has a lower peak than that of g-C3N4, which indicates that the composite has a lower electrons and holes recombination rate. Fig. 12b shows the photocurrent transient response for the electrodes of bare g-C3N4 nanosheets, ZnIn2S4 and ZISCN40 under visible light irradiation. As it can be seen, with the light switched-on and -off cycles, the ZISCN40 sample exhibits the highest photocurrent transient response under visible light irradiation, which is greatly larger than that for bare g-C3N4 and ZnIn2S4. It suggests the remarkably enhanced carrier separation ratio over ZISCN40. To further confirm the above results, electrochemical impedance spectroscopy (EIS), a useful measurement to characterize charge carrier transportation, was also performed. As can be seen from Fig. 12c, the diameter of the Nyquist semicircle for the ZISCN40 sample is smaller than that of ZnIn2S4 and g-C3N4, which indicates that the hybrid composite has a lower resistance than that of ZnIn2S4 and g-C3N4. The PL and photoelectrochemical results confirmed the superior charge transfer and recombination inhibition in the ZnIn2S4–g-C3N4 composite photocatalyst, which is responsible for the enhanced photocatalytic activity. | ||
| Fig. 12 (a) PL spectra, (b) transient photocurrent responses and (c) electrochemical impedance spectroscopy of g-C3N4 nanosheets, ZnIn2S4 and ZISCN40 under visible light irradiation. | ||
The high separation efficiency of photo-generated carriers should be ascribed to the suitable band potentials of g-C3N4 nanosheets and ZnIn2S4. The possible mechanism of separation and transportation of electron–hole pairs at the interface of ZnIn2S4–g-C3N4 composite under visible light is proposed and illustrated in Scheme 1. Under the visible light illumination, both g-C3N4 and ZnIn2S4 can be excited and produce photogenerated electron–hole pairs. Since the CB edge potential of g-C3N4 nanosheets (∼−1.22 eV vs. NHE) is more negative than that of ZnIn2S4 (∼−0.80 eV vs. NHE), the photoinduced electrons on the CB of g-C3N4 can directly transfer to the CB of ZnIn2S4. While the corresponding VB edge potential of ZnIn2S4 (∼+1.52 eV vs. NHE) is more positive than that of g-C3N4 nanosheets (∼+1.48 eV vs. NHE), the photogenerated holes on the VB of ZnIn2S4 can immigrate to the VB of g-C3N4. Thus, the photogenerated electrons and holes move in opposite directions, which can effectively reduce the recombination probability and enhance the charge separation efficiency, leading to a remarkable enhancement of the photocatalytic activity.
4. Conclusions
Novel ZnIn2S4–g-C3N4 sheet-on-sheet nanocomposites were prepared by a facile hydrothermal method. Under visible light irradiation, the obtained ZnIn2S4–g-C3N4 composites exhibited a significantly enhanced photocatalytic activity for hydrogen evolution and degradation of pollutants than pure ZnIn2S4 and g-C3N4. The optimal content of g-C3N4 was found to be 40 wt%. The enhanced photocatalytic performance of the composites was ascribed to the efficient charge separation and transfer on the interface between ZnIn2S4 and g-C3N4. More importantly, the as-prepared nanocomposites possessed high reusability. Therefore, they can be used as a promising photocatalyst for practical applications in environmental purification and clean hydrogen energy production from water splitting.Acknowledgements
The authors gratefully acknowledge the National Natural Science Foundation of China (11472164) and Innovative Research Team (IRT13078) for financial support. The authors also thank Lab for Microstructure, Instrumental Analysis and Research Center, Shanghai University, for materials characterizations.Notes and references
- H. Tong, S. X. Ouyang, Y. P. Bi, N. Umezawa, M. Oshikiri and J. H. Ye, Adv. Mater., 2012, 24, 229–251 CrossRef CAS PubMed.
- M. R. Hoffmann, S. T. Martin, W. Y. Choi and D. W. Bahnemann, Chem. Rev., 1995, 95, 69–96 CrossRef CAS.
- X. B. Chen, S. H. Shen, L. J. Guo and A. S. Mao, Chem. Rev., 2010, 110, 6503–6570 CrossRef CAS PubMed.
- H. Y. Li, H. W. Yu, L. Sun, J. L. Zhai and X. R. Han, Nanoscale, 2015, 7, 1610–1615 RSC.
- Z. D. Meng, L. Zhu, J. G. Choi, M. L. Chen and W. C. Oh, J. Mater. Chem., 2011, 21, 7596–7603 RSC.
- X. C. Wang, K. Maeda, A. Thomas, K. Takanabe, G. Xin, J. M. Carlsson, K. Domen and M. Antonietti, Nat. Mater., 2009, 8, 76–80 CrossRef CAS PubMed.
- S. B. Yang, Y. J. Gong, J. S. Zhang, L. Zhan, L. L. Ma, Z. Y. Fang, R. Vajtai, X. C. Wang and P. M. Ajayan, Adv. Mater., 2013, 25, 2452–2456 CrossRef CAS PubMed.
- W. J. Wang, J. C. Yu, D. H. Xia, P. K. Wong and Y. C. Li, Environ. Sci. Technol., 2013, 47, 8724–8732 CAS.
- S. W. Cao and J. G. Yu, J. Phys. Chem. Lett., 2014, 5, 2101–2107 CrossRef CAS PubMed.
- J. Mao, T. Y. Peng, X. H. Zhang, K. Li, L. Q. Ye and L. Zan, Catal. Sci. Technol., 2013, 3, 1253–1260 CAS.
- H. X. Zhao, H. T. Yu, X. Quan, S. Chen, H. M. Zhao and H. Wang, RSC Adv., 2014, 4, 624–628 RSC.
- F. Z. Su, S. C. Mathew, G. Lipner, X. Z. Fu, M. Antonietti, S. Blechert and X. C. Wang, J. Am. Chem. Soc., 2010, 132, 16299–16301 CrossRef CAS PubMed.
- J. S. Zhang, F. S. Guo and X. C. Wang, Adv. Funct. Mater., 2013, 23, 3008–3014 CrossRef CAS.
- J. Xu, Y. J. Wang and Y. F. Zhu, Langmuir, 2013, 29, 10566–10572 CrossRef CAS PubMed.
- X. F. Chen, J. S. Zhang, X. Z. Fu, M. Antonietti and X. C. Wang, J. Am. Chem. Soc., 2009, 131, 11658–11659 CrossRef CAS PubMed.
- X. C. Wang, X. F. Chen, A. Thomas, X. Z. Fu and M. Antonietti, Adv. Mater., 2009, 21, 1609–1612 CrossRef CAS.
- Y. Wang, J. S. Zhang, X. C. Wang, M. Antonietti and H. R. Li, Angew. Chem., Int. Ed., 2010, 49, 3356–3359 CrossRef CAS PubMed.
- G. Liu, P. Niu, C. H. Sun, S. C. Smith, Z. G. Chen, G. Q. Lu and H. M. Cheng, J. Am. Chem. Soc., 2010, 132, 11642–11648 CrossRef CAS PubMed.
- L. Ge, C. C. Han, J. Liu and Y. F. Li, Appl. Catal., A, 2011, 409–410, 215–222 CrossRef CAS.
- Y. J. Zhang, A. Thomas, M. Antonietti and X. C. Wang, J. Am. Chem. Soc., 2009, 131, 50–51 CrossRef CAS PubMed.
- Y. Hou, Z. H. Wen, S. M. Cui, X. R. Guo and J. H. Chen, Adv. Mater., 2013, 25, 6291–6297 CrossRef CAS PubMed.
- Q. Li, N. Zhang, Y. Yang, G. Z. Wang and D. H. L. Ng, Langmuir, 2014, 30, 8965–8972 CrossRef CAS PubMed.
- K. Katsumata, R. Motoyoshi, N. Matsushita and K. Okada, J. Hazard. Mater., 2013, 260, 475–482 CrossRef CAS PubMed.
- J. Shen, J. T. Zai, Y. P. Yuan and X. F. Qian, Int. J. Hydrogen Energy, 2012, 37, 16986–16993 CrossRef.
- L. Shang, C. Zhou, T. Bian, H. J. Yu, L. Z. Wu, C. H. Tung and T. R. Zhang, J. Mater. Chem. A, 2013, 1, 4552–4558 CAS.
- B. Chai, T. Y. Peng, P. Zeng, X. H. Zhang and X. J. Liu, J. Phys. Chem. C, 2011, 115, 6149–6155 CAS.
- Z. B. Lei, W. S. You, M. Y. Liu, G. H. Zhou, T. Takata, M. Hara, K. Domen and C. Li, Chem. Commun., 2003, 17, 2142–2143 RSC.
- Y. J. Chen, H. Ge, L. Wei, Z. H. Li, R. S. Yuan, P. Liu and X. Z. Fu, Catal. Sci. Technol., 2013, 3, 1712–1717 CAS.
- L. Wei, Y. J. Chen, Y. P. Lin, H. S. Wu, R. S. Yuan and Z. H. Li, Appl. Catal., B, 2014, 144, 521–527 CrossRef CAS.
- L. Ye, J. L. Fu, Z. Xu, R. S. Yuan and Z. H. Li, ACS Appl. Mater. Interfaces, 2014, 6, 3483–3490 CAS.
- X. L. Gou, F. Y. Cheng, Y. H. Shi, L. Zhang, S. J. Peng, J. Chen and P. W. Shen, J. Am. Chem. Soc., 2006, 128, 7222–7229 CrossRef CAS PubMed.
- N. S. Chaudhari, A. P. Bhirud, R. S. Sonawane, L. K. Nikam, S. S. Warule, V. H. Rane and B. B. Kale, Green Chem., 2011, 13, 2500–2506 RSC.
- F. Fang, L. Chen, Y. B. Chen and L. M. Wu, J. Phys. Chem. C, 2010, 114, 2393–2397 CAS.
- X. F. Bai and J. S. Li, Mater. Res. Bull., 2011, 46, 1028–1034 CrossRef CAS.
- S. H. Shen, J. Chen, X. X. Wang, L. Zhao and L. J. Guo, J. Power Sources, 2011, 196, 10112–10119 CrossRef CAS.
- F. Li, G. P. Chen, J. H. Luo, Q. L. Huang, Y. H. Luo, Q. B. Meng and D. M. Li, Catal. Sci. Technol., 2013, 3, 1993–1999 CAS.
- B. Chai, T. Y. Peng, P. Zeng and X. H. Zhang, Dalton Trans., 2012, 41, 1179–1186 RSC.
- J. G. Hou, C. Yang, H. J. Cheng, Z. Wang, S. Q. Jiao and H. M. Zhu, Phys. Chem. Chem. Phys., 2013, 15, 15660–15668 RSC.
- G. H. Tian, Y. J. Chen, Z. Y. Ren, C. G. Tian, K. Pan, W. Zhou, J. Q. Wang and H. G. Fu, Chem.–Asian J., 2014, 9, 1291–1297 CrossRef CAS.
- Y. G. Yu, G. Chen, G. Wang and Z. S. Lv, Int. J. Hydrogen Energy, 2013, 38, 1278–1285 CrossRef CAS.
- S. Yang, L. Li, W. H. Yuan and Z. L. Xia, Dalton Trans., 2015, 44, 6374–6383 RSC.
- H. Katsumata, Y. Tachi, T. Suzukib and S. Kaneco, RSC Adv., 2014, 4, 21405–21409 RSC.
- Y. L. Chen, J. H. Li, Z. H. Hong, B. Shen, B. Z. Lin and B. F. Gao, Phys. Chem. Chem. Phys., 2014, 16, 8106–8113 RSC.
- S. C. Yan, Z. S. Li and Z. G. Zou, Langmuir, 2009, 25, 10397–10401 CrossRef CAS PubMed.
- Q. J. Xiang, J. G. Yu and M. Jaroniec, J. Phys. Chem. C, 2011, 115, 7355–7363 CAS.
- H. F. Li, H. T. Yu, S. Chen, H. M. Zhao, Y. B. Zhang and X. Quan, Dalton Trans., 2014, 43, 2888–2894 RSC.
- L. Q. Ye, J. Y. Liu, Z. Jiang, T. Y. Peng and L. Zan, Appl. Catal., B, 2013, 142–143, 1–7 CAS.
- L. Ge, F. Zuo, J. K. Liu, Q. Ma, C. Wang, D. Z. Sun, L. Bartels and P. Y. Feng, J. Phys. Chem. C, 2012, 116, 13708–13714 CAS.
- X. S. Zhou, Z. H. Luo, P. F. Tao, B. Jin, Z. J. Wu and Y. S. Huang, Mater. Chem. Phys., 2014, 143, 1462–1468 CrossRef CAS.
- J. S. Chen, F. Xin, X. H. Yin, T. Y. Xiang and Y. W. Wang, RSC Adv., 2015, 5, 3833–3839 RSC.
- Y. Q. Shi, S. H. Jiang, K. Q. Zhou, B. B. Wang, B. Wang, Z. Gui, Y. Hu and R. K. K. Yuen, RSC Adv., 2014, 4, 2609–2613 RSC.
- J. G. Yu, S. H. Wang, B. Cheng, Z. Lin and F. Huang, Catal. Sci. Technol., 2013, 3, 1782–1789 CAS.
- Q. Li, H. Meng, J. G. Yu, W. Xiao, Y. Q. Zheng and J. Wang, Chem.–Eur. J., 2014, 20, 1176–1185 CrossRef CAS PubMed.
- Y. J. Sun, W. D. Zhang, T. Xiong, Z. W. Zhao, F. Dong, R. Q. Wang and W. K. Ho, J. Colloid Interface Sci., 2014, 418, 317–323 CrossRef CAS.
- H. Katsumata, T. Sakai, T. Suzuki and S. Kaneco, Ind. Eng. Chem. Res., 2014, 53, 8018–8025 CrossRef CAS.
- Y. Liu, Y. X. Yu and W. D. Zhang, Int. J. Hydrogen Energy, 2014, 39, 9105–9113 CrossRef CAS.
- J. Chen, S. H. Shen, P. H. Guo, P. Wu and L. J. Guo, J. Mater. Chem. A, 2014, 2, 4605–4612 CAS.
- N. Tian, H. W. Huang, Y. He, Y. X. Guo, T. R. Zhang and Y. H. Zhang, Dalton Trans., 2015, 44, 4297–4307 RSC.
- K. Vignesh, A. Suganthi, B. K. Min and M. Kang, J. Mol. Catal. A: Chem., 2014, 395, 373–383 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |