Methods to delay deactivation of zeolites on furan acylation: continuous liquid-phase technology and solvent effects†
Abstract
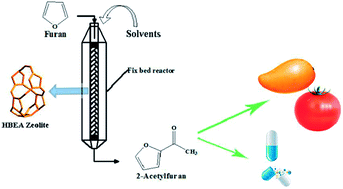
The continuous liquid-phase Friedel–Crafts acylation of furan with acetic anhydride was first carried out in a fixed-bed reactor over an H-beta zeolite. A three-level, four-variable Box-Behnken design combined with response surface methodology was employed to determine the optimal acylation conditions, and the accuracy of the model was verified by validation experiments. In this study, it was found that lower liquid velocity and stronger polarity of the solvent were conducive to enhancing the catalytic activity and stability of the H-beta zeolite. The reaction was limited by the acid sites being poisoned due to the adsorption of the by-product and pore blockage. Moreover, the carbon deposit was proved to be the main reason for the deactivation of the H-beta zeolite by using GC-MS, 13C NMR MAS, 27Al NMR MAS, TG/DTG and NH3-TPD.


 Please wait while we load your content...
Please wait while we load your content...