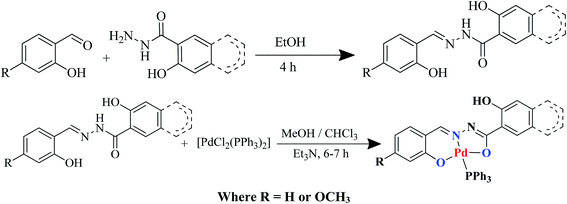
ONO pincer type Pd(II) complexes: synthesis, crystal structure and catalytic activity towards C-2 arylation of quinoline scaffolds†
Vignesh Arumugam a,
Werner Kaminskyb and
Dharmaraj Nallasamy*a
a,
Werner Kaminskyb and
Dharmaraj Nallasamy*a
aInorganic & Nanomaterials Research Laboratory, Department of Chemistry, Bharathiar University, Coimbatore 641 046, India. E-mail: dharmaraj@buc.edu.in; Fax: +91 4222422387; Tel: +91 4222428319 Web: http://ndharmaraj.wix.com/inrl
bDepartment of Chemistry, University of Washington, Seattle, Washington 98195, USA
First published on 4th September 2015
Abstract
Four new palladium(II) complexes featuring ONO pincer type hydrazone ligands were synthesized and characterized by spectroscopic and single-crystal XRD analysis. These complexes showed excellent catalytic activity towards the Suzuki–Miyaura cross coupling reaction of 2-chloroquinoline derivatives with various aryl boronic acids. The main advantages over previous methodologies include low catalyst loading, less problematic reaction media (H2O–DMF (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20%)) and a lower reaction temperature of 60 °C for optimal performance.
20%)) and a lower reaction temperature of 60 °C for optimal performance.
Introduction
Nitrogen based heterocycles are important constituents in a large number of biologically active compounds.1 Among which quinoline scaffolds play a vital role. Several quinolines and their derivatives are currently in use as antibacterial, antifungal, analgesics, antituberculosis, antimalarial, anti-inflammatory, anticancer, antibiotic and anti-HIV drugs.2 In addition, quinoline analogues are valuable synthons for the preparation of nano and mesostructures with enhanced electronic and photonic properties.3 Despite the numerous pharmacological activities and related synthetic methods,4 the development of new, facile and eco-friendly synthetic approaches to 2-substituted quinolines using mild conditions are desirable and subject of ongoing research. 2-Arylated quinoline and their derivatives have proven significant in vivo activities against Leishmania donovani and are in preclinical development.5 Likewise, a few other 2-arylquinolines display significant antiviral activity in HIV-infected cells (Fig. 1).6Within the efforts to realize 2-substituted quinolines, most established routines require elevated reaction temperatures, prolonged reaction times, excess amounts of various reagents, including toxic compounds and harsh reaction conditions7 which are unsuitable for sensitive substrates and thus constitute a challenge in targeted syntheses. Palladium catalyzed Suzuki–Miyaura cross-coupling (SMC) reactions were tried as an alternative, but unfortunately, nitrogen containing heterocycles are problematic substrates for SMC reactions8 because of a tendency to bind with the metal ion and thus deactivates the catalyst.
However, catalysts like [PdCl2(PPh3)2] and Pd(OAc)2/DtBPF (DtBPF = 1,1′-bis(di-tert-butylphosphino)ferrocene) were reported to offer improved results compared to the Pd–L catalysts with electron-rich, bulky, monodentate phosphine ligands such as PtBu3 and PCy2-(biphenyl), but requiring a high catalyst loading (5 mol%).8c,9
Currently, our group is actively pursuing research on the synthesis and biological applications of hydrazone based transition metal complexes with pincer type geometry.10 More recently, we turned our attention to evaluate hydrazone based pincer type palladium complexes as catalysts for the SMC reaction.11 Synthesis of transition metal pincer type complexes with high stability and catalytic activity has been a most productive area of homogeneous catalysis and coordination chemistry.12,13 The remarkable equilibrium between stability and reactivity of the pincer complexes can be controlled by precise ligand design and subsequent co-operative construction of metal complexes.13 As a result, research on the development of pincer ligands possessing CCC, CNC, PNP, SNS, NNN, NCN and SCS donor atoms got much attention.14 However, studies on ONO pincer ligands and corresponding complexes are scanty in the literature. A recent review on Ru, Ir, W, Ta, Re, Pb, Fe and Sb complexes containing ONO pincer ligand moiety highlighted their importance as catalysts in various organic reactions.13a,15 In our previous study,11 we reported for the first time, the catalytic potential of ONO pincer analogous palladium complexes for SMC of challenging aryl chlorides with various aryl boronic acids bearing activating as well as deactivating substituents.
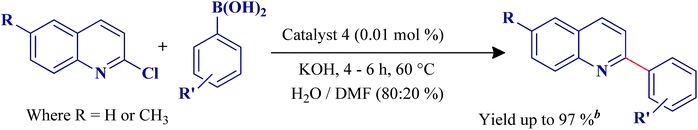
In this manuscript, we describe the synthesis of four new palladium(II) complexes (1–4) bearing ONO pincer type ligands and their catalytic activity towards SMC reaction of 2-chlroroquinoline derivatives with substituted aryl boronic acids. To the best of our insight, this is the first study on the utility of the titled complexes as catalysts in SMC reactions of 2-chlroroquinoline derivatives with substituted aryl boronic acids in H2O–DMF system to synthesis a library of 2-arylquinolines.
Results and discussions
Direct reactions of palladium precursor [PdCl2(PPh3)2] with the pincer type ligands H3L1–H3L4 (where, H3L is pincer type ligand derived from the condensation of salicylaldehyde with salicylic hydrazide (H3L1) or 3-hydroxynaphthoic acid hydrazide(H3L2) and 4-methoxysalicylaldehyde with salicylic hydrazide (H3L3) or 3-hydroxynaphthoic acid hydrazide (H3L4)) yielded complexes 1–4 of the type [Pd(HL)(PPh3)] as depicted in Scheme 1.IR spectra of the four complexes were compared with corresponding free ligands. The absorption due to free OH groups of the ligands observed respectively, at 3719, 3614, 3610 and 3640 cm−1. A strong band observed at 3185, 3260, 3181 and 3249 cm−1 indicated the presence of a N–H functional group in the free ligands. Intense C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O absorption bands were found at 1618, 1641, 1624 and 1623 cm−1 in the spectrum of the free ligands. All four ligands exhibited a very sharp band at 1555, 1572, 1556 and 1556 attributed to the C
O absorption bands were found at 1618, 1641, 1624 and 1623 cm−1 in the spectrum of the free ligands. All four ligands exhibited a very sharp band at 1555, 1572, 1556 and 1556 attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N group. In the IR spectra of the complexes discussed here, non-existence of a strong band from free OH indicated the deprotonation of phenolic oxygen and its coordination to the palladium(II) ion. The N–H and C
N group. In the IR spectra of the complexes discussed here, non-existence of a strong band from free OH indicated the deprotonation of phenolic oxygen and its coordination to the palladium(II) ion. The N–H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations were missing thus proving that the ligands underwent enolisation followed by deprotonation prior to coordination with the palladium(II) ion.16a In addition, a new band occurred due to the C
O stretching vibrations were missing thus proving that the ligands underwent enolisation followed by deprotonation prior to coordination with the palladium(II) ion.16a In addition, a new band occurred due to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–N
N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C group at 1494, 1472, 1488 and 1473 cm−1, respectively. The absorption related to a C–O group was identified as a new sharp band at 1255, 1299, 1222 and 1303 cm−1 in for complexes 1–4. The IR spectral features unambiguously prove that the pincer type ligands H3L1, H3L2, H3L3 and H3L4 were coordinated to palladium(II) ion via the phenolate oxygen, the azomethine nitrogen and the imidolate oxygen in complexes 1–4.16b
C group at 1494, 1472, 1488 and 1473 cm−1, respectively. The absorption related to a C–O group was identified as a new sharp band at 1255, 1299, 1222 and 1303 cm−1 in for complexes 1–4. The IR spectral features unambiguously prove that the pincer type ligands H3L1, H3L2, H3L3 and H3L4 were coordinated to palladium(II) ion via the phenolate oxygen, the azomethine nitrogen and the imidolate oxygen in complexes 1–4.16b
1H NMR spectra of them were recorded in order to study the exact coordination mode of the pincer type ligand in the palladium complexes 1–4. None displayed any signal due to a N–H proton and thus indicated that the oxygen is coordinated to the palladium ion in the imidolate form. Sharp singlet observed in the spectra of complexes 1–4 at 8.61, 9.15, 8.20 and 9.39 ppm, respectively were assigned to the azomethine proton of the pincer ligand. The non-involvement of OH groups, despite their presence in the phenyl/naphthyl ring of the corresponding pincer type ligands in coordination with palladium ion is confirmed by the singlet resonances at 6.38, 5.77, 5.36 and 5.40 ppm, respectively. However, in the spectra of complexes 3 and 4 a sharp singlet at 3.81 and 3.42 ppm, respectively confirmed the presence of a methoxy group. Aromatic protons present in complexes 1–4 were identified in the region of 8.34 to 6.98 ppm.17 Similarly, the 13C NMR resonance of the complexes showed downfield signals at 159.2, 159.3, 159.2 and 157.8 ppm, respectively, owing to the imidolate carbon (N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–O) involved in coordination. The azomethine carbon of the complexes 1–4 gave signals at 151.5, 151.6, 151.5 and 148.5, respectively. Signals featuring in the region of 143.8–105.1 and at 55.2/51.4 ppm have been assigned to aromatic and methoxy carbons present in complexes 1–4.17
C–O) involved in coordination. The azomethine carbon of the complexes 1–4 gave signals at 151.5, 151.6, 151.5 and 148.5, respectively. Signals featuring in the region of 143.8–105.1 and at 55.2/51.4 ppm have been assigned to aromatic and methoxy carbons present in complexes 1–4.17
The molecular structure of complexes 1–4 were determined by single-crystal X-ray diffraction to study the exact coordination mode of the pincer type ligands with palladium ion. Selected bond angles and bond distances are gathered in Table 1 in the ESI.† The ORTEP diagrams provided in Fig. 2–5 shows pincer type ligands coordinated to the palladium ion via the phenolate oxygen, azomethine nitrogen and deprotonated imidole oxygen. Complex 1 crystallized in the triclinic system, but the other three were primitive monoclinic with space groups P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , P21/a, P21/c and P21/c, respectively. The Pd(II) ion adopts a distorted square-planar geometry in all four structures by accommodating the ligand as a binegative tridentate ONO donor and the fourth site is occupied by a triphenylphosphine. The bite angles of O(1)–Pd(1)–O(2) in 1–4 are 174.29(8), 174.30(13), 174.37(6) and 173.74(15), respectively. Similarly, angles N(1)–Pd(1)–P(1) are 174.77(7), 173.09(9), 179.17(5) and 172.74(13). Unit cell packing diagrams of complexes 1–4 are provided in ESI.† The observed bond lengths and bite angles are very similar to those observed in other palladium(II) complexes.18 Relevant data concerning data collection and details of the structure refinements are summarized Table 2 in the ESI.† Based on 1H, 13C NMR and single-crystal XRD data, the pincer type hydrazone ligand is coordinated to the palladium ion in a tridentate fashion by replacing both chloride ions and the triphenylphosphine from the starting precursor.
, P21/a, P21/c and P21/c, respectively. The Pd(II) ion adopts a distorted square-planar geometry in all four structures by accommodating the ligand as a binegative tridentate ONO donor and the fourth site is occupied by a triphenylphosphine. The bite angles of O(1)–Pd(1)–O(2) in 1–4 are 174.29(8), 174.30(13), 174.37(6) and 173.74(15), respectively. Similarly, angles N(1)–Pd(1)–P(1) are 174.77(7), 173.09(9), 179.17(5) and 172.74(13). Unit cell packing diagrams of complexes 1–4 are provided in ESI.† The observed bond lengths and bite angles are very similar to those observed in other palladium(II) complexes.18 Relevant data concerning data collection and details of the structure refinements are summarized Table 2 in the ESI.† Based on 1H, 13C NMR and single-crystal XRD data, the pincer type hydrazone ligand is coordinated to the palladium ion in a tridentate fashion by replacing both chloride ions and the triphenylphosphine from the starting precursor.
| Entry | Catalyst | Base | Solvent | Yield (%) |
|---|---|---|---|---|
| 1 | Catalyst 2 | No base | H2O | No reaction |
| 2 | Catalyst 2 | No base | H2O/DMF | 19 |
| 3 | Catalyst 2 | KOH | H2O/DMF | 90 |
| 4 | Catalyst 2 | K2CO3 | H2O/DMF | 85 |
| 5 | Catalyst 2 | NaOH | H2O/DMF | 84 |
| 6 | Catalyst 2 | Na2CO3 | H2O/DMF | 79 |
| 7 | Catalyst 2 | CH3COONa | H2O/DMF | 52 |
| 8 | Catalyst 2 | Et3N | H2O/DMF | 45 |
| 9 | Catalyst 2 | Pyridine | H2O/DMF | 39 |
| 10 | Catalyst 2 | KOH | H2O/DCM | 40 |
| 11 | Catalyst 2 | KOH | H2O/toluene | 41 |
| 12 | Catalyst 2 | KOH | H2O/CHCl3 | 43 |
| 13 | Catalyst 2 | KOH | H2O/DMF | 90 |
| 14 | Catalyst 2 | KOH | H2O/CH3CN | 72 |
| 15 | Catalyst 2 | KOH | H2O/THF | 61 |
| 16 | Catalyst 2 | KOH | H2O/DMSO | 82 |
| 17 | Catalyst 2 | KOH | H2O/EtOAc | 55 |
| 18 | Catalyst 2 | KOH | H2O/benzene | 53 |
| 19 | Catalyst 2 | KOH | H2O/DMF (10%) | 80 |
| 20 | Catalyst 2 | KOH | H2O/DMF (20%) | 90 |
| 21 | Catalyst 2 | KOH | H2O/DMF (30%) | 83 |
| 22 | Catalyst 2 | KOH | H2O/DMF (40%) | 76 |
| 23 | Catalyst 2 | KOH | H2O/DMF (50%) | 70 |
| 24 | Catalyst 1 | KOH | H2O/DMF (20%) | 85 |
| 25 | Catalyst 2 | KOH | H2O/DMF (20%) | 90 |
| 26 | Catalyst 3 | KOH | H2O/DMF (20%) | 79 |
| 27 | Catalyst 4 | KOH | H2O/DMF (20%) | 96 |
| a Reaction conditions: 2-chloroquinoline or 6-methyl-2-chloroquinoline (3.0 mmol), aryl boronic acid (4.0 mmol), 0.01 mol% of complex 4, KOH (5.0 mmol), H2O (4 mL), and DMF (1 mL) stirred for 4–6 h at 60 °C.b Isolated yield after column chromatography, TON = turnover number = ratio of moles of product formed to moles of catalyst used, TOF = turnover frequency = TON/h. |
|---|
 |
Catalysis
Catalytic activity of the complexes 1–4 were investigated towards SMC reactions of 2-chloroquinoline derivatives with different substituted aryl boronic acids. Reaction conditions were optimized by utilizing 2-chloroquinoline and phenylboronic acid as model substrates in aqueous-organic media and the results are summarized in Table 1.Initially, the catalytic reaction was studied with various bases such as KOH, NaOH, K2CO3, NaHCO3, Na2CO3, CH3COONa, Et3N and pyridine, where KOH provided a coupled product of 90% isolated yield. No reaction was observed in neat water, because of catalyst insolubility (Table 1, entry 1). This problem was circumvented by adding organic solvents to help dissolve the catalyst in the reaction media. A 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20% mixture of H2O–DMF was found to be most effective for the titled coupling reaction. A comparative catalytic study was carried out involving all the four catalysts towards the chosen SMC reaction. Catalysts 2 and 4 performed better than the other two which may be related to a naphthyl unit in the coordinated pincer ligand. In particular catalyst 4 with a methoxy substituent in the salicylaldehyde part, is more effective than catalyst 2, which is composed of non-substituted salicylaldehyde in its molecular architecture. The role of triphenylphosphine ligand in the catalysts 1–4 is to help in activating C–Cl bonds in quinolines.19
20% mixture of H2O–DMF was found to be most effective for the titled coupling reaction. A comparative catalytic study was carried out involving all the four catalysts towards the chosen SMC reaction. Catalysts 2 and 4 performed better than the other two which may be related to a naphthyl unit in the coordinated pincer ligand. In particular catalyst 4 with a methoxy substituent in the salicylaldehyde part, is more effective than catalyst 2, which is composed of non-substituted salicylaldehyde in its molecular architecture. The role of triphenylphosphine ligand in the catalysts 1–4 is to help in activating C–Cl bonds in quinolines.19
In addition, the effect of temperature on this coupling reaction was investigated by using 2-chloroquinoline (3 mmol), phenylboronic acid (4 mmol), KOH (5 mmol), catalyst 4 (0.01 mol%) in H2O–DMF (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20%) and the isolated yield of coupled products were illustrated in Fig. 6. A distinct optimum was reached at 60 °C.
20%) and the isolated yield of coupled products were illustrated in Fig. 6. A distinct optimum was reached at 60 °C.
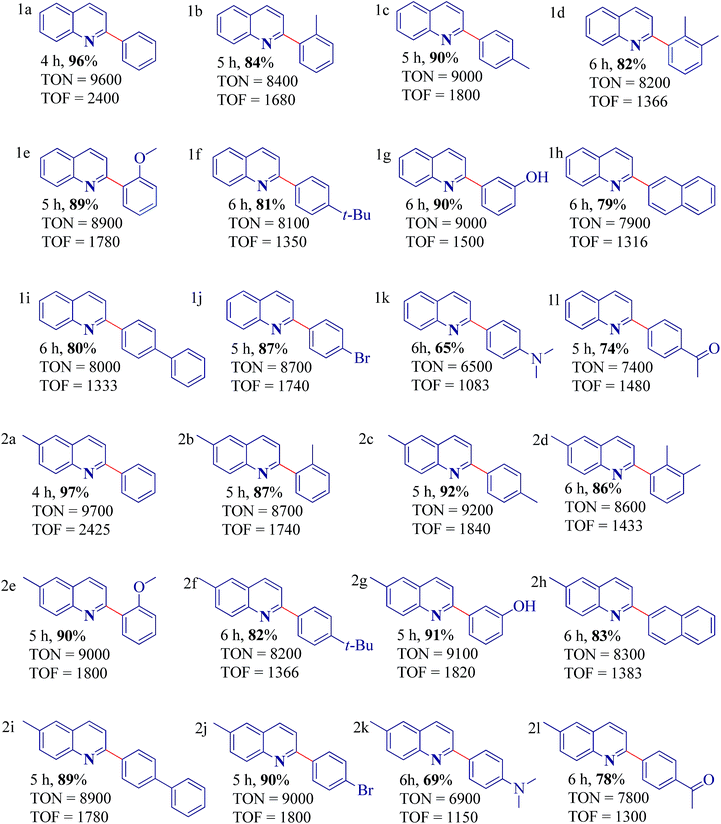
To further explore the scope and application of the reaction, we studied the reaction of various aryl boronic acids with 2-chloroquinoline derivatives under the optimized conditions mentioned above. Aryl boronic acids featuring an electron-donating group such as –CH3, –OCH3, t–(CH3)3C–, –OH and –NR2 furnished the reaction smoothly and afforded 65–92% of the coupled products over a period of 4–6 h (Table 2 entries 1c, 1f, 1k, 2c, 2f and 2k). Interestingly, sterically hindered aryl boronic acids afforded a 82–90% yield (Table 2 entries 1b, 1d, 1e, 1g, 1h, 1i, 2b, 2d, 2e, 2g, 2h and 2i). Similarly, boronic acids bearing moderate electron with drawing groups such as Br and –COCH3 efficiently underwent the coupling reaction, to afford the products in 74–90% (Table 2 entries 1j, 1l, 2j and 2l). Not even a trace of neither homocoupling of aryl boronic acids nor coupling with –OH and –Br groups of the substrates were observed (Table 2, entry 1g, 1j, 2g and 2j). This offers an opportunity to conduct further cross-coupling reactions by exploiting the halogenated positions available on selective boronic acids (Table 2, entry 1j and 2j).
Catalyst loading tests of complex 4 revealed best performance for 0.01 mol%. Reducing catalyst stacking from 0.01–0.0001 mol% under optimized conditions resulted in low yield as reported earlier.20 On the other hand, use of 0.0001 mol% catalyst resulted in high turnover number (Table 3).
| Entry | Mol% | Isolated yield% | TON |
|---|---|---|---|
| 1 | 0.01 | 96 | 9600 |
| 2 | 0.001 | 92 | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
| 3 | 0.0001 | 85 | 850![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
Reusability of the selected catalyst 4, 0.01 mol% was screened and the trend of the isolated yield is summarized in Fig. 7. A 20 mL round bottom flask was charged with 2-chloroquinoline (3.0 mmol) and phenylboronic acid (4 mmol), KOH (5 mmol) in H2O–DMF (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20%). To this reaction mixture was added catalyst 4 (0.01 mol%) and heated to 60 °C. After completion of the reaction, the mixture was extracted with ethyl acetate and the organic layer was subjected to column chromatography to separate the catalyst and product using silica gel and petroleum ether/ethyl acetate (90/10%) as an eluent. The recovered catalyst was washed with distilled water, dried and utilized for the next cycle under same reaction conditions. The identity of the recovered catalyst was confirmed by Rf value (0.54) and melting point of the original catalyst 219–221 °C. The first cycle afforded 96% of the corresponding coupled product, but yield decreased steadily in subsequent cycles (Fig. 7).
20%). To this reaction mixture was added catalyst 4 (0.01 mol%) and heated to 60 °C. After completion of the reaction, the mixture was extracted with ethyl acetate and the organic layer was subjected to column chromatography to separate the catalyst and product using silica gel and petroleum ether/ethyl acetate (90/10%) as an eluent. The recovered catalyst was washed with distilled water, dried and utilized for the next cycle under same reaction conditions. The identity of the recovered catalyst was confirmed by Rf value (0.54) and melting point of the original catalyst 219–221 °C. The first cycle afforded 96% of the corresponding coupled product, but yield decreased steadily in subsequent cycles (Fig. 7).
Very recently, P. T. Perumal et al.21 reported a SnCl2·2H2O mediated synthesis of 2-substituted quinolines in ethanol via A3-coupling followed by reductive cyclization in 89% yield. Advantageously, we herein demonstrated that SMC of aryl boronic acids with 2-chloroquinolines catalyzed by new ONO pincer type palladium(II) complex led to similar 2-arylquinolines in an excellent yield up to 97% in H2O–DMF over a period of 4–6 h with low catalyst loading (0.01 mol%).
Conclusions
Herein, we portrayed the synthesis of four new palladium complexes bearing ONO pincer type ligands along with thorough characterization using IR, 1H and 13C NMR and single-crystal XRD techniques. These complexes showed strong performance in the Suzuki–Miyaura coupling reaction between 2-chloroquinoline scaffolds and aryl boronic acids with low catalyst loading in H2O–DMF (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20%) at moderate temperature. We feel that the present palladium catalyzed methodology offers a simple and straight forward route to synthesize a series of biologically important 2-arylquinolines possessing both activating and deactivating groups in excellent yields.
20%) at moderate temperature. We feel that the present palladium catalyzed methodology offers a simple and straight forward route to synthesize a series of biologically important 2-arylquinolines possessing both activating and deactivating groups in excellent yields.
Experimental section
All chemicals utilized for the synthesis of ligands and their complexes were obtained from Sigma-Aldrich, India. Solvents were purified and dried according to standard procedures.22 Elemental analysis (C, H and N) was performed on a Vario EL III Elemental analyzer instrument. IR spectra (4000–400 cm−1) were recorded on a Nicolet Avatar Model FT-IR spectrophotometer. Melting points were determined with a Lab India instrument.1H and 13C NMR spectra were recorded in deuterated CHCl3 as solvent on BRUKER 400 and 100 MHZ instruments, respectively.Synthesis of ligands
Pincer type ligands H3L were synthesized by condensing equimolar amounts of salicylaldheyde or 4-methoxy salicylaldheyde with salicyl hydrazide (H3L1 and H3L3) or 3-hydroxy-2-naphthoic acid hydrazide (H3L2 and H3L34) in ethanol according to literature method (Scheme 1). The reaction mixture was then refluxed on a water-bath for 4 h and poured into crushed ice. The corresponding solid pincer type ligand formed was filtered, washed several times with distilled water and recrystallized from ethanol in 80–90% yield. The purity of the ligands were checked by various analytical techniques and is in accordance with literature reports.23General method for the synthesis of the palladium complexes
To a warm methanolic solution (20–30 mL) of appropriate ligands (1 equiv.) were added a chloroform solution of [PdCl2(PPh3)2] (1 equiv.) followed by two drops of triethylamine. The reaction mixture was refluxed 6–7 h and kept at room temperature for crystallization. Needle like reddish brown crystals suitable for X-ray studies were obtained on slow evaporation over 45–60 days.Single crystal X-ray diffraction studies
Data collection: single-crystal X-ray diffraction data of complexes 1–4 were collected on Bruker APEX II single crystal X-ray diffractometer. The data was integrated and scaled using SAINT/SADABS from within the APEX2 software package by Bruker (Version 2.1-4). Indexing and unit cell refinement indicated complex 1 is triclinic and all other complexes were primitive monoclinic, with space groups P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , P21/a, P21/c and P21/c, respectively. Solution by direct methods (SHELXS, SIR97)24 produced complete heavy atom phasing models consistent with the proposed structures. The structures were completed by difference Fourier synthesis with SHELXL97.25,26 Scattering factors are from Waasmair and Kirfel.27 Hydrogen atoms were placed in geometrically idealized positions and constrained to ride on their parent atoms with C–H distances in the range 0.95–1.00 Angstrom. Isotropic thermal parameters Ueq were fixed such that they were 1.2 Ueq of their parent atom Ueq for CH's and 1.5 Ueq of their parent atom Ueq in case of methyl groups. All non-hydrogen atoms were refined anisotropically by full-matrix least-squares.
, P21/a, P21/c and P21/c, respectively. Solution by direct methods (SHELXS, SIR97)24 produced complete heavy atom phasing models consistent with the proposed structures. The structures were completed by difference Fourier synthesis with SHELXL97.25,26 Scattering factors are from Waasmair and Kirfel.27 Hydrogen atoms were placed in geometrically idealized positions and constrained to ride on their parent atoms with C–H distances in the range 0.95–1.00 Angstrom. Isotropic thermal parameters Ueq were fixed such that they were 1.2 Ueq of their parent atom Ueq for CH's and 1.5 Ueq of their parent atom Ueq in case of methyl groups. All non-hydrogen atoms were refined anisotropically by full-matrix least-squares.
Catalysis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20%). Then, the catalyst 4 (0.01 mol%) was added to this mixture and stirred at 60 °C. The progress of the SMC reaction was monitored by thin layer chromatography (petroleum ether/ethyl acetate (90–10%)) and the Rf value of the product was measured to be 0.61. After completion of the reaction, the mixture was solvent diluted with ethyl acetate and dried over anhydrous Na2SO4. Solvent was evaporated under vacuo and the residue was purified by column chromatography using petroleum ether/EtOAc as eluent to afford 2-phenylquinoline as a pale yellow solid. The product was washed with distilled water to remove inorganic base. The identity of the product was confirmed by 1H and 13C NMR data.
20%). Then, the catalyst 4 (0.01 mol%) was added to this mixture and stirred at 60 °C. The progress of the SMC reaction was monitored by thin layer chromatography (petroleum ether/ethyl acetate (90–10%)) and the Rf value of the product was measured to be 0.61. After completion of the reaction, the mixture was solvent diluted with ethyl acetate and dried over anhydrous Na2SO4. Solvent was evaporated under vacuo and the residue was purified by column chromatography using petroleum ether/EtOAc as eluent to afford 2-phenylquinoline as a pale yellow solid. The product was washed with distilled water to remove inorganic base. The identity of the product was confirmed by 1H and 13C NMR data.Entry 1a. 2-Phenylquinoline: elemental analysis (%) calculated for C15H11N; C, 87.77; H, 5.40; N, 6.82. Found (%); C, 87.72; H, 5.38; N, 6.81. 1H NMR δ (ppm): 7.96 (d, J = 8 Hz, 2H), 7.85–7.91 (m, 3H), 7.74 (d, J = 7.6 Hz, 1H), 7.32–7.37 (m, 1H), 7.29 (d, J = 7.6 Hz, 2H), 7.26 (d, J = 9.6 Hz, 1H) 7.16–7.18 (m, 1H). 13C NMR: δ (ppm): 157.8, 149.1, 143.1, 140.9, 139.3, 133.7, 130.4, 129.5, 128.3, 128.1, 127.7.
Entry 1b. 2-o-Tolylquinoline: elemental analysis (%) calculated for C16H13N; C, 87.64; H, 5.98; N, 6.39. Found (%); C, 87.61; H, 5.95; N, 6.38. 1H NMR δ (ppm): 7.93 (d, J = 8 Hz, 1H), 7.81–7.86 (m, 1H), 7.63 (d, J = 7.2 Hz, 2H), 7.34–7.37 (m, 1H), 7.29–7.33 (m, 2H), 7.19 (d, J = 8.2 Hz, 2H), 7.13 (d, J = 7.4 Hz, 1H), 2.38 (s, 3H). 13C NMR: δ (ppm): 156.7, 148.9, 143.2, 141.0, 139.5, 137.9, 130.3, 129.6, 129.0, 128.8, 128.5, 126.8, 126.6, 125.3, 125.2, 21.3.
Entry 1c. 2-p-Tolylquinoline: elemental analysis (%) calculated for C16H13N; C, 87.64; H, 5.98; N, 6.39. Found (%); C, 87.62; H, 5.96; N, 6.37. 1H NMR δ (ppm): 7.92 (d, J = 8.4 Hz, 2H), 7.84 (d, J = 8.2 Hz, 1H), 7.80 (d, J = 7.6 Hz, 1H), 7.69 (d, J = 8 Hz, 1H), 7.25–7.38 (m, 1H), 7.10–7.23 (m, 2H), 6.91 (d, J = 7.6 Hz, 1H), 6.80 (s, 1H), 2.43 (s, 3H). 13C NMR: δ (ppm): 158.2, 150.8, 144.2, 140.1, 138.8, 133.3, 130.2, 129.3, 127.9, 127.7, 125.4, 125.1, 124.9, 120.7, 21.6.
Entry 1d. 2-(2,3-Dimethyl-phenyl)-quinoline: elemental analysis (%) calculated for C17H15N; C, 87.52; H, 6.48; N, 6.00. Found (%); C, 87.50; H, 6.46; N, 5.98. 1H NMR δ (ppm): 8.05 (d, J = 9.2 Hz, 1H), 7.91 (q, J = 8.6 Hz, 2H), 7.68 (d, J = 8 Hz, 1H), 7.40–7.48 (m, 1H), 7.35–7.39 (m, 1H), 7.27–7.34 (m, 1H), 7.19–7.23 (m, 2H), 2.93 (s, 3H), 2.76 (s, 3H). 13C NMR: δ (ppm): 156.8, 150.4, 142.0, 140.8, 139.1, 138.8, 133.7, 133.4, 130.3, 129.8, 128.9, 128.7, 128.2, 127.7, 125.5, 125.2, 24.4, 21.1.
Entry 1e. 2-(2-Methoxy-phenyl)-quinoline: elemental analysis (%) calculated for C16H13NO; C, 81.68; H, 5.57; N, 5.95. Found (%); C, 81.67; H, 5.55; N, 5.93. 1H NMR δ (ppm): 7.97 (d, J = 8.4 Hz, 1H), 7.82–7.88 (m, 2H), 7.58 (d, J = 7.8 Hz, 2H), 7.49 (d, J = 6.2 Hz, 1H), 7.47 (d, J = 7.5 Hz, 2H), 7.30–7.38 (m, 1H), 7.10–7.19 (m, 1H), 2.90 (s, 3H). 13C NMR: δ (ppm): 158.3, 150.9, 142.2, 140.1, 139.6, 128.8, 133.5, 131.8, 130.1, 129.2, 128.9, 128.5, 127.8, 127.7, 127.3, 51.4.
Entry 1f. 2-(4-tert-Butyl-phenyl)-quinoline: elemental analysis (%) calculated for C19H19N; C, 87.31; H, 7.33; N, 5.36. Found (%); C, 87.30; H, 7.32; N, 5.34. 1H NMR δ (ppm): 7.91 (s, 2H), 7.81 (d, J = 9.2 Hz, 1H), 7.68 (d, J = 9.6 Hz, 1H), 7.31–7.21 (m, 2H), 7.0 (m, 1H), 1.55 (s, 9H). 13C NMR: δ (ppm): 157.0, 148.8, 143.1, 141.4, 140.3, 137.4, 135.4, 129.5, 128.7, 128.2, 127.5, 126.8, 126.3, 125.9, 125.3, 124.1, 34.6, 29.6.
Entry 1g. 3-Quinolin-2-yl-phenol: elemental analysis (%) calculated for C15H11NO; C, 81.43; H, 5.01; N, 6.33. Found (%); C, 81.42; H, 4.99; N, 6.29. 1H NMR δ (ppm): 7.89–7.98 (m, 1H), 7.87 (s, 2H), 7.71 (d, J = 7.6 Hz, 1H), 7.49 (dd, J = 2.4, 2.4 Hz, 1H), 7.34 (t, J = 8 Hz, 3H), 7.28 (d, J = 7.2 Hz, 1H), 7.10–7.21 (m, 1H), 7.08 (s, 1H). 13C NMR: δ (ppm): 161.6, 159.1, 148.7, 143.9, 141.3, 140.5, 138.2, 134.7, 134.1, 129.7, 129.6, 128.3, 128.2, 127.0, 126.9, 125.1, 124.5.
Entry 1h. 2-Naphthalen-2-yl-quinoline: elemental analysis (%) calculated for C19H13N; C, 89.38; H, 5.13; N, 5.49. Found (%); C, 89.35; H, 5.11; N, 5.45. 1H NMR δ (ppm): 7.83–7.90 (m, 3H), 7.65 (d, J = 11.2 Hz, 1H), 7.50 (d, J = 2 Hz, 1H), 7.34 (dd, J = 1.6, 8 Hz, 1H), 7.25–7.30 (m, 1H), 7.24 (d, J = 1.6 Hz, 2H), 7.20 (t, J = 1.4 Hz, 3H), 7.10–7.19 (m, 1H). 13C NMR: δ (ppm): 155.0, 148.8, 141.1, 140.4, 140.2, 139.5, 136.3, 133.8, 130.3, 129.9, 128.8, 128.6, 128.5, 127.7, 126.5, 125.4, 124.5, 124.0, 119.8, 118.9.
Entry 1i. 2-Biphenyl-4-yl-quinoline: elemental analysis (%) calculated for C21H15N; C, 89.65; H, 5.37; N, 4.98. Found (%); C, 89.62; H, 5.34; N, 4.95. 1H NMR δ (ppm): 7.91–7.94 (m, 2H), 7.88 (d, J = 8.4 Hz, 2H), 7.69 (d, J = 12 Hz, 1H), 7.31–7.37 (m, 3H) 7.28 (d, J = 2.8 Hz, 2H), 7.23 (d, J = 7.2 Hz, 3H), 6.99 (d, J = 8 Hz, 2H). 13C NMR: δ (ppm): 155.0, 147.9, 143.0, 142.7, 139.6, 137.1, 137.0, 129.3, 129.0, 127.9, 126.8, 125.8, 125.1, 119.5, 116.7.
Entry 1j. 2-(4-Bromo-phenyl)-quinoline: elemental analysis (%) calculated for C15H10BrN; C, 63.40; H, 3.55; N, 4.93. Found (%); C, 63.38; H, 3.54; N, 4.91. 1H NMR δ (ppm): 7.48 (d, J = 7.6 Hz, 1H), 7.42 (d, J = 10.4 Hz, 1H), 7.32 (d, J = 6.8 Hz, 2H), 7.20–7.23 (m, 2H) 7.07–7.19 (m, 2H), 6.94 (s, 2H). 13C NMR: δ (ppm): 157.0, 147.6, 142.6, 140.2, 139.2, 137.4, 135.9, 130.3, 128.5, 127.5, 126.1, 125.6, 124.3, 119.4, 116.5, 89.1.
Entry 1k. Dimethyl-(4-quinolin-2-yl-phenyl)-amine: elemental analysis (%) calculated for C17H16N2; C, 82.22; H, 6.49; N, 11.28. Found (%); C, 82.20; H, 6.47; N, 11.25. 1H NMR δ (ppm): 7.96 (d, J = 8 Hz, 1H), 7.83–7.87 (m, 2H), 7.73 (d, J = 7.6 Hz, 1H), 7.32–7.38 (m, 3H), 7.20–7.27 (m, 1H) 7.14 (d, J = 8.8 Hz, 2H), 2.83 (s, 6H). 13C NMR: δ (ppm): 156.5, 149.1, 143.4, 141.5, 141.3, 138.2, 133.5, 131.0, 130.2, 128.4, 128.1, 127.5, 127.1, 126.6, 125.5, 125.3, 124.3, 122.6, 37.8.
Entry 1l. 1-(4-Quinolin-2-yl-phenyl)-ethanone: elemental analysis (%) calculated for C17H13NO; C, 82.57; H, 5.30; N, 5.66. Found (%); C, 82.56; H, 5.27; N, 5.65. 1H NMR δ (ppm): 7.91 (d, J = 8.4 Hz, 2H), 7.85 (d, J = 7.6 Hz, 2H), 7.63 (d, J = 8 Hz, 1H), 7.30–7.37 (m, 1H), 7.25 (d, J = 5.6 Hz, 1H), 7.14–7.20 (m, 1H), 6.95 (d, J = 12.4 Hz, 2H), 2.92 (s, 3H). 13C NMR: δ (ppm): 169.6, 159.3, 143.9, 143.5, 141.7, 141.1, 138.0, 134.1, 134.0, 132.8, 130.3, 128.5, 128.3, 127.7, 127.5, 126.1, 29.0.
Entry 2a. 6-Methyl-2-phenyl-quinoline: elemental analysis (%) calculated for C16H13N; C, 87.64; H, 5.98; N, 6.39. Found (%); C, 87.61; H, 5.95; N, 6.37. 1H NMR δ (ppm): 7.82 (s, 1H), 7.80 (d, J = 7.4 Hz, 2H), 7.59 (d, J = 2 Hz, 2H), 7.57 (t, J = 2 Hz, 1H), 7.49–7.55 (m, 3H), 7.37–7.48 (m, 1H), 2.49 (s, 3H). 13C NMR: δ (ppm): 161.2, 148.1, 147.2, 135.7, 135.3, 129.4, 128.3, 128.1, 127.1, 126.4, 123.1, 122.0, 29.0.
Entry 2b. 6-Methyl-2-o-tolyl-quinoline: elemental analysis (%) calculated for C17H15N; C, 87.52; H, 6.48; N, 6.00. Found (%); C, 87.50; H, 6.45; N, 5.98. 1H NMR δ (ppm): 8.50 (s, 1H), 7.93–8.10 (m, 5H), 7.66 (d, J = 2 Hz, 2H), 7.50–7.57 (m, 1H), 3.35 (s, 3H) 2.51 (s, 3H). 13C NMR: δ (ppm): 160.3, 147.7, 147.4, 135.5, 134.9, 131.9, 128.3, 127.9, 126.9, 122.7, 121.9, 120.9, 120.1, 26.9, 19.8.
Entry 2c. 6-Methyl-2-p-tolyl-quinoline: elemental analysis (%) calculated for C17H15N; C, 87.52; H, 6.48; N, 6.00. Found (%); C, 87.50; H, 6.45; N, 5.97. 1H NMR δ (ppm): 8.17 (s, 1H), 7.85 (d, J = 7.6 Hz, 2H), 7.79 (d, J = 7.6 Hz, 2H), 7.54 (t, J = 7.6 Hz, 1H), 7.44 (t, J = 7.2 Hz, 1H), 7.00 (d, J = 2.4 Hz, 2H), 3.66 (s, 3H), 2.56 (s, 3H). 13C NMR: δ (ppm): 160.2, 147.9, 147.7, 136.0, 135.2, 133.1, 129.2, 128.6, 128.1, 127.0, 124.2, 121.8, 121.2, 121.1, 112.8, 21.9, 19.4.
Entry 2d. 2-(2,3-Dimethyl-phenyl)-6-methyl-quinoline: elemental analysis (%) calculated for C18H17N; C, 87.41; H, 6.93; N, 5.66. Found (%); C, 87.39; H, 6.91; N, 5.62. 1H NMR δ (ppm): 8.20 (s, 1H), 8.03 (d, J = 8.4 Hz, 1H), 7.84 (d, J = 7.6 Hz, 2H), 7.74 (d, J = 2.4 Hz, 1H), 7.55 (t, J = 7.8 Hz, 2H), 7.43–7.46 (m, 2H), 3.96 (s, 3H), 3.91 (s, 3H), 2.50 (s, 3H). 13C NMR: δ (ppm): 160.4, 147.9, 135.6, 134.2, 128.3, 128.0, 127.4, 127.0, 122.9, 122.0, 119.5, 119.4, 111.8, 106.3, 26.8, 19.5, 16.2.
Entry 2e. 2-(2-Methoxy-phenyl)-6-methyl-quinoline: elemental analysis (%) calculated for C17H15NO; C, 81.90; H, 6.06; N, 5.62. Found (%); C, 81.88; H, 6.04; N, 5.59. 1H NMR δ (ppm): 8.71 (s, 1H), 8.19 (d, J = 7.6 Hz, 2H), 7.81 (d, J = 7.6 Hz, 2H), 7.68 (d, J = 8 Hz, 1H), 7.53 (t, J = 7.8 Hz, 1H), 7.74–7.46 (m, 1H), 7.20–7.26 (m, 1H) 2.71 (s, 3H), 1.83 (s, 3H). 13C NMR: δ (ppm): 160.4, 149.5, 146.4, 135.8, 134.4, 131.1, 129.4, 129.1, 128.8, 128.4, 128.2, 127.9, 127.4, 125.8, 12.9, 120.8, 119.5, 119.4, 111.1, 53.5, 25.1.
Entry 2f. 2-(4-tert-Butyl-phenyl)-6-methyl-quinoline: elemental analysis (%) calculated for C20H21N; C, 87.23; H, 7.69; N, 5.09. Found (%); C, 87.21; H, 7.66; N, 5.07. 1H NMR δ (ppm): 8.24 (s, 1H), 7.88 (d, J = 7.6 Hz, 1H), 7.75 (d, J = 6.8 Hz, 1H), 7.39 (d, J = 3.6 Hz, 2H), 7.61 (t, J = 6.4 Hz, 1H), 7.40–7.49 (m, 2H), 7.26–7.38 (m, 1H), 2.35 (s, 3H), 1.49 (s, 9H). 13C NMR: δ (ppm): 161.4, 147.4, 143.9, 139.7, 137.7, 136.5, 131.6, 130.8, 129.3, 129.1, 128.6, 128.3, 127.9, 127.7, 127.5, 126.8, 126.7, 125.3, 123.1, 120.8, 120.4, 119.1, 110.7, 106.5, 31.6, 32.4, 23.4.
Entry 2g. 3-(6-Methyl-quinolin-2-yl)-phenol: elemental analysis (%) calculated for C16H13NO; C, 81.68; H, 5.57; N, 5.95. Found (%); C, 81.65; H, 5.54 N, 5.92. 1H NMR δ (ppm): 8.26 (s, 1H), 8.06 (d, J = 8 Hz, 1H), 7.88 (d, J = 7.2 Hz, 1H), 7.68 (t, J = 12.4 Hz, 1H), 7.58–7.62 (m, 1H), 7.50–7.56 (m, 1H), 7.45 (t, J = 7.6 Hz, 1H), 7.42 (d, J = 11.6 Hz, 1H), 7.37 (s, 1H), 5.31 (s, 1H), 2.50 (s, 3H). 13C NMR: δ (ppm): 158.9, 149.5, 147.1, 138.1, 135.3, 130.1, 129.6, 126.6, 126.3, 123.6, 123.1, 120.7, 119.4, 118.5, 112.8, 25.8.
Entry 2h. 6-Methyl-2-naphthalen-2-yl-quinoline: elemental analysis (%) calculated for C20H15N; C, 89.19; H, 5.61; N, 5.20. Found (%); C, 89.17; H, 5.59; N, 5.18. 1H NMR δ (ppm): 8.20 (s, 1H), 7.95 (d, J = 6.4 Hz, 4H), 7.59 (d, J = 6 Hz, 1H), 7.50 (d, J = 2 Hz, 1H), 7.46 (t, J = 6 Hz, 2H), 7.42–7.46 (m, 1H), 7.30–7.40 (m, 1H), 2.64 (s, 3H). 13C NMR: δ (ppm): 160.4, 147.8, 147.6, 136.3, 135.9, 133.6, 129.3, 127.9, 127.7, 127.0, 125.7, 121.6, 121.3, 121.0, 111.9, 23.1.
Entry 2i. 2-Biphenyl-4-yl-6-methyl-quinoline: elemental analysis (%) calculated for C22H17N; C, 89.46; H, 5.80; N, 4.74. Found (%); C, 89.45; H, 5.78; N, 4.71. 1H NMR δ (ppm): 8.07 (s, 2H), 8.05 (d, J = 7.2 Hz, 2H), 7.89 (d, J = 7.2 Hz, 2H), 7.76 (t, J = 9 Hz, 1H), 7.68 (d, J = 6.4 Hz, 3H), 7.64 (d, J = 5.6 Hz, 1H), 7.43 (t, J = 9 Hz, 2H), 7.21–7.37 (m, 1H), 2.50 (s, 3H). 13C NMR: δ (ppm): 160.9, 147.9, 146.8, 135.2, 133.9, 128.6, 127.8, 127.3, 126.4, 123.2, 122.0, 120.2, 119.4, 117.4, 109.9, 108.3, 31.1.
Entry 2j. 2-(4-Bromo-phenyl)-6-methyl-quinoline: elemental analysis (%) calculated for C16H12BrN; C, 64.45; H, 4.06; N, 4.70. Found (%); C, 64.41; H, 4.03; N, 4.68. 1H NMR δ (ppm): 7.71 (d, J = 7.6 Hz, 2H), 7.60 (s, 1H), 7.41–7.52 (m, 2H), 7.38 (t, J = 3 Hz, 1H), 7.31 (s, 2H), 7.21–7.28 (m, 1H), 2.35 (s, 3H). 13C NMR: δ (ppm): 152.7, 141.1, 136.8, 130.9, 128.5, 124.8, 124.1, 123.2, 122.4, 119.1, 117.0, 116.4, 26.4.
Entry 2k. Dimethyl-[4-(6-methyl-quinolin-2-yl)-phenyl]-amine: elemental analysis (%) calculated for C18H18N2; C, 82.41; H, 6.92; N, 10.68. Found (%); C, 82.39; H, 6.89; N, 10.66. 1H NMR δ (ppm): 7.79 (d, J = 12.8 Hz, 2H), 7.51 (t, J = 12.2 Hz, 2H), 7.44 (t, J = 7.2 Hz, 1H), 7.36 (t, J = 6.8 Hz, 1H), 7.29–7.34 (m, 2H), 6.92–7.15 (m, 1H), 2.96 m (s, 6H), 2.34 (s, 3H). 13C NMR: δ (ppm): 158.3, 141.9, 137.0, 133.5, 130.8, 128.2, 123.2, 122.4, 120.8, 119.8, 118.7, 115.4, 36.7, 29.4.
Entry 2l. 1-[4-(6-Methyl-quinolin-2-yl)-phenyl]-ethanone: elemental analysis (%) calculated for C18H15NO; C, 82.73; H, 5.79; N, 5.36. Found (%); C, 82.71; H, 5.76; N, 5.32. 1H NMR δ (ppm): 8.01 (d, J = 11.2 Hz, 2H), 7.96 (d, J = 12.2 Hz, 2H), 7.74 (d, J = 14.4 Hz, 2H), 7.52 (t, J = 10.4 Hz, 2H), 7.30–7.43 (m, 1H), 3.31 (s, 3H), 2.84 (s, 3H). 13C NMR: δ (ppm): 170.9, 158.3, 140.5, 136.7, 135.4, 133.1, 129.0, 128.8, 125.6, 124.6, 124.1, 123.7, 121.0, 114.8, 29.3, 24.2.
Acknowledgements
Mr Vignesh Arumugam thanks the University Grants Commission (UGC), New Delhi, India, for the award of Junior Research Fellowship (JRF) under UGC-BSR RFSMS.References
- M. S. Butler, J. Nat. Prod., 2004, 67, 2141 CrossRef CAS PubMed
.
-
(a) M. D. Ferretti, A. T. Neto, A. F. Morel, T. S. Kaufman and L. Enrique, Eur. J. Med. Chem., 2014, 81, 253 CrossRef CAS PubMed
; (b) S. Vandekerckhove, S. V. Herreweghe, J. Willems, B. Danneels, T. Desmet, C. Kock, P. J. Smith, K. Chibale and M. Dhooghe, Eur. J. Med. Chem., 2015, 92, 91 CrossRef CAS PubMed
; (c) A. H. Abadi, G. H. Hegazy and A. A. El-Zaher, Bioorg. Med. Chem., 2005, 13, 5759 CrossRef CAS PubMed
; (d) G. C. Muscia, G. Y. Buldain and S. E. Asis, Eur. J. Med. Chem., 2014, 73, 243 CrossRef CAS PubMed
; (e) A. P. Gorka, A. Dios and P. D. Roepe, J. Med. Chem., 2013, 56, 5231 CrossRef CAS PubMed
; (f) S. Mukherjee and M. Pal, Drug Discovery Today, 2013, 18, 389 CrossRef CAS PubMed
; (g) J. A. Makawana, C. B. Sangani, L. Lin and H. L. Zhu, Bioorg. Med. Chem., 2014, 24, 1734 CrossRef CAS PubMed
; (h) N. C. Desai, G. M. Kotadiya and A. R. Trivedi, Bioorg. Med. Chem., 2014, 24, 3126 CrossRef CAS PubMed
; (i) A. Fournet, R. Hocquemiller, F. Roblot, A. Cave, P. Richomme and J. Bruneton, J. Nat. Prod., 1993, 56, 1547 CrossRef CAS
.
-
(a) A. K. Aggarwal and S. A. Jenekhe, Macromolecules, 1991, 24, 6806 CrossRef
; (b) X. Zhang, A. S. Shetty and S. A. Jenekhe, Macromolecules, 1999, 32, 7422 CrossRef CAS
; (c) S. A. Jenekhe, L. Lu and M. M. Alam, Macromolecules, 2001, 34, 7315 CrossRef CAS
.
-
(a) P. Selig and W. Raven, Org. Lett., 2014, 16, 5192 CrossRef CAS PubMed
; (b) L. Kong, Y. Zhou, H. Huang, Y. Yang, Y. Liu and Y. Li, J. Org. Chem., 2015, 80, 1275 CrossRef CAS PubMed
; (c) C. E. Meyet and C. H. Larsen, J. Org. Chem., 2014, 79, 9835 CrossRef CAS PubMed
; (d) N. He, Y. Huo, J. Liu, Y. Huang, S. Zhang and Q. Cai, Org. Lett., 2015, 17, 374 CrossRef CAS PubMed
.
-
(a) V. S. Gopinath, J. Pinjari, T. D. Ravindra, V. Aditya, V. Preeti, S. Rahul, M. Manjunath, P. S. K. Goud, R. Vikram, P. Bose, M. V. S. Rao, G. Suman, S. K. Puri, D. Launay and D. Martin, Eur. J. Med. Chem., 2013, 69, 527 CrossRef CAS PubMed
; (b) K. C. Fang, Y. L. Chen, J. Y. Sheu, T. C. Wang and C. C. Tzeng, J. Med. Chem., 2000, 43, 3809 CrossRef CAS PubMed
; (c) J. Chevalier, S. Atifi, A. Eyraud, A. Mahamoud and J. M. Barbe, J. Med. Chem., 2001, 44, 4023 CrossRef CAS PubMed
; (d) L. T. Phan, T. Jian, Z. Chen, Y. L. Qiu, Z. Wang, T. Beach, A. Polemeropoulos and Y. S. Or, J. Med. Chem., 2004, 47, 2965 CrossRef CAS PubMed
.
- J. Polanski, F. Zouhiri, L. Jeanson, D. Desmaele, J. Angelo, J. Mouscadet, R. Gieleciak, J. Gasteiger and M. Le Bret, J. Med. Chem., 2002, 45, 4647 CrossRef CAS PubMed
.
-
(a) S. Fan, J. Yang and X. Zhang, Org. Lett., 2011, 13, 4374 CrossRef CAS PubMed
; (b) X. Ren, P. Wen, X. Shi, Y. Wang, J. Li, S. Yang, H. Yan and G. Huang, Org. Lett., 2013, 15, 5194 CrossRef CAS PubMed
; (c) S. Zhu, L. Wu and X. Huang, J. Org. Chem., 2013, 78, 9120 CrossRef CAS PubMed
; (d) O. V. Larionov, D. Stephens, A. Mfuh and G. Chavez, Org. Lett., 2014, 16, 864 CrossRef CAS PubMed
; (e) L. Kong, Y. Zhou, H. Huang, Y. Yang, Y. Liu and Y. Li, J. Org. Chem., 2015, 80, 1275 CrossRef CAS PubMed
.
-
(a) M. Feuerstein, H. Doucet and M. Santelli, Tetrahedron Lett., 2005, 46, 1717 CrossRef CAS PubMed
; (b) M. M. Manas, R. Pleixats and A. S. Muns, Synlett, 2006, 18, 3001 CrossRef
; (c) T. Itoh and T. Mase, Tetrahedron Lett., 2005, 46, 3573 CrossRef CAS PubMed
.
- A. E. Thompson, G. Hughes, A. S. Batsanov, M. R. Bryce, P. R. Parry and B. Tarbit, J. Org. Chem., 2005, 70, 388 CrossRef CAS PubMed
.
-
(a) P. Krishnamoorthy, P. Sathyadevi, R. R. Butorac, A. H. Cowley, N. S. P. Bhuvanesh and N. Dharmaraj, Dalton Trans., 2012, 41, 6842 RSC
; (b) P. Sathyadevi, P. Krishnamoorthy, N. S. P. Bhuvanesh, P. Kalaiselvi, V. Vijaya Padma and N. Dharmaraj, Eur. J. Med. Chem., 2012, 55, 420 CrossRef CAS PubMed
; (c) P. Sathyadevi, P. Krishnamoorthy, E. Jayanthi, R. R. Butorac, A. H. Cowley and N. Dharmaraj, Inorg. Chim. Acta, 2012, 384, 83 CrossRef CAS PubMed
; (d) P. Sathyadevi, P. Krishnamoorthy, K. Thanigaimani, P. T. Muthiah and N. Dharmaraj, Polyhedron, 2012, 31, 294 CrossRef CAS PubMed
; (e) P. Krishnamoorthy, P. Sathyadevi, K. Senthilkumar, P. T. Muthiah, R. Ramesh and N. Dharmaraj, Inorg. Chem. Commun., 2011, 14, 1318 CrossRef CAS PubMed
; (f) P. Krishnamoorthy, P. Sathyadevi, R. R. Butorac, A. H. Cowley and N. Dharmaraj, Eur. J. Med. Chem., 2011, 46, 3376 CrossRef CAS PubMed
.
- A. Vignesh, W. Kaminsky, N. S. P. Bhuvanesh and N. Dharmaraj, RSC Adv., 2015, 5, 59428 RSC
.
-
(a) C. J. Moulton and B. L. Shaw, J. Chem. Soc., Dalton Trans., 1976, 1020 RSC
; (b) C. Crocker, R. J. Errington, W. S. McDonald, K. J. Odell, B. L. Shaw and R. J. Goodfellow, J. Chem. Soc., Chem. Commun., 1979, 498 RSC
.
-
(a) M. E. Reilly and A. S. Veige, Chem. Soc. Rev., 2014, 43, 6325 RSC
; (b) J. T. Singleton, Tetrahedron, 2003, 59, 1837 CrossRef CAS
; (c) E. V. Peris and R. H. Crabtree, Coord. Chem. Rev., 2004, 248, 2239 CrossRef CAS PubMed
; (d) M. E. Boom and D. Milstein, Chem. Rev., 2003, 103, 1759 CrossRef PubMed
.
-
(a) T. O. Howell, A. J. Huckaba and T. K. Hollis, Org. Lett., 2014, 16, 2570 CrossRef CAS PubMed
; (b) A. M. Prokhorov, T. Hofbeck, R. Czerwieniec, A. F. Suleymanova, D. N. Kozhevnikov and H. Yersin, J. Am. Chem. Soc., 2014, 136, 9637 CrossRef CAS PubMed
; (c) A. J. Holmes, P. J. Rayner, M. J. Cowley, G. G. R. Green, A. C. Whitwood and S. B. Duckett, Dalton Trans., 2015, 44, 1077 RSC
; (d) Y. Komiyama, J. Kuwabara and T. Kanbara, Organometallics, 2014, 33, 885 CrossRef CAS
; (e) P. García, P. Ren, R. Scopelliti and X. Hu, ACS Catal., 2015, 5, 1164 CrossRef
; (f) Q. Deng, R. L. Melen and L. H. Gade, Acc. Chem. Res., 2014, 47, 3162 CrossRef CAS PubMed
; (g) G. Mancano, M. Bhadbhade and B. A. Messerle, Inorg. Chem., 2014, 53, 10159 CrossRef CAS PubMed
; (h) S. M. Peterson, M. L. Helm and A. M. Appel, Dalton Trans., 2015, 44, 747 RSC
.
- Y. Zhang, X. Li and S. H. Honga, Adv. Synth. Catal., 2010, 352, 1779 CrossRef CAS PubMed
.
-
(a) A. R. B. Rao and S. Pal, J. Organomet. Chem., 2013, 731, 67 CrossRef CAS PubMed
; (b) R. N. Prabhu and R. Ramesh, J. Organomet. Chem., 2012, 718, 43 CrossRef CAS PubMed
.
- M. M. Tamizh and R. Karvembu, Inorg. Chem. Commun., 2012, 25, 30 CrossRef CAS PubMed
.
-
(a) M. M. Tamizh, B. F.T. Cooper, C. L. B. Macdonald and R. Karvembu, Inorg. Chim. Acta, 2013, 394, 391 CrossRef PubMed
; (b) A. R. B. Rao and S. Pal, J. Organomet. Chem., 2014, 762, 58 CrossRef PubMed
.
- L. Botella and C. Najera, Angew. Chem., Int. Ed., 2002, 41, 179 CrossRef CAS
.
-
(a) M. T. Reetz and J. G. Vries, Chem. Commun., 2004, 1559 RSC
; (b) A. V. Gaikwad, A. Holuigue, M. B. Thathagar, J. E. Elshof and G. Rothenberg, Chem.–Eur. J., 2007, 13, 6908 CrossRef CAS PubMed
.
- N. Sudhapriya, A. Nandakumar and P. T. Perumal, RSC Adv., 2014, 4, 58476 RSC
.
- A. I. Vogel, Text Book of Practical Organic Chemistry, Longman, London, 5th edn, 1989 Search PubMed
.
- A. Syamal and D. Kumar, Transition Met. Chem., 1982, 7, 118 CrossRef CAS
.
- A. Altomare, C. Burla, M. Camalli, G. L. Cascarano, C. Giacovazzo, A. Guagliardi, A. G. G. Moliterni, G. Polidori and R. Spagna, J. Appl. Crystallogr., 1999, 32, 115 CrossRef CAS
.
- A. Altomare, G. L. Cascarano, C. Giacovazzo and A. Guagliardi, J. Appl. Crystallogr., 1993, 26, 343 CrossRef
.
- S. Mackay, C. Edwards, A. Henderson, C. Gilmore, N. Stewart, K. Shankland and D. A. MaXus, A computer program for the solution and refinement of crystal structures from diffraction data, University of Glasgow, Scotland, 1997 Search PubMed
.
- D. Waasmaier and A. Kirfel, Acta Crystallogr., Sect. A: Found. Crystallogr., 1995, 51, 416 CrossRef
.
-
(a) Y. Rulong, L. Xingxing, P. Congming, Z. Xiaoqiang, L. Xiaoni, K. Xing and H. Guosheng, Org. Lett., 2013, 15, 4876 CrossRef PubMed
; (b) C. A. Fleckenstein and H. Plenio, Green C hem., 2007, 9, 1287 RSC
; (c) E. M. Courtney and H. L. Catharine, J. Org. C hem., 2014, 79, 9835 CrossRef PubMed
; (d) X. Fuhong, C. Wen, L. Yunfeng and D. Guo-Jun, Org. Biomol. Chem., 2012, 10, 8593 RSC
; (e) T. P. Nitin and S. R. Vivek, J. Org. Chem., 2010, 75, 6961 CrossRef PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1047924–1047927. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra15342e |
| This journal is © The Royal Society of Chemistry 2015 |