Synthesis and evaluation of novel quinuclidinone derivatives as potential anti-proliferative agents†
Jigar Y.
Soni
a,
Shridhar
Sanghvi
b,
R. V.
Devkar
b and
Sonal
Thakore
*a
aDepartment of Chemistry, Faculty of Science, The M. S. University of Baroda, Vadodara, 390 002, India. E-mail: chemistry2797@yahoo.com; Tel: +91-0265-2795552
bDepartment of Zoology, Faculty of Science, The M. S. University of Baroda, Vadodara, 390 001, India
First published on 18th September 2015
Abstract
In this study a new series of substituted (Z)-4-(3-oxoquinuclidin-2-ylidene) benzamides and substituted (Z)-4-((3-oxoquinuclidin-2-ylidene)methyl)benzoates have been designed and synthesised as potential anti-cancer agents. This set of compounds were prepared by using a common intermediate (Z)-4-((3-oxoquinuclidin-2-ylidene)methyl)benzoic acid. They were well characterized by various spectroscopic techniques as well as a crystallographic study and screened for anti-cancer activity. A cell viability assay using MTT was performed on A549 & L132 cell lines and IC50 values were determined. Analogues 4c and 5e exhibited the most potent anti-cancer activity among all the analogues synthesized in this present study. A haemolytic assay using normal human erythrocytes was performed to study the blood compatibility of the compounds. Acridine orange/ethidium bromide (AO/EB) staining also showed cell death. To get a better insight into the mechanism of cell death DAPI (4′,6-diamido-2-phenylindol nuclear staining) and DNA fragmentation studies were performed. A Structure Activity Relationship (SAR) was explored to facilitate further development of this new class of compounds.
1 Introduction
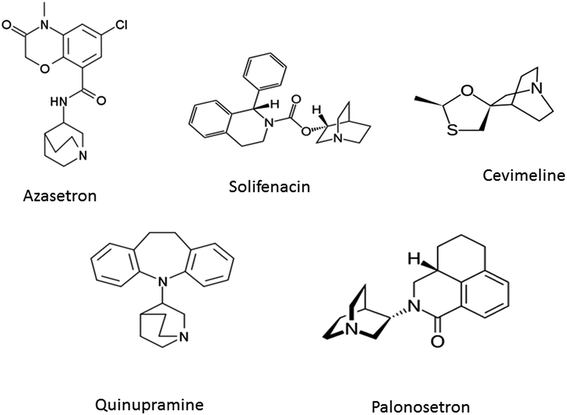
Cancer, a serious health problem, is one of the main causes of mortality in the developing as well as developed countries.1 Different theories have been proposed for the cause of cancer and several strategies have been formulated and examined for combating the disease. Survival rates for five years of some cancers have significantly improved in the past two decades while those of other cancers, such as lung and pancreatic cancer remain low.2 The major form of cancer treatment is chemotherapy, alone or combined with radiation. However, the majority of cancers develop resistance to chemotherapy during treatment. As a result, the design and discovery of non-traditional, efficient and safe classes of chemical agents are the prime targets in contemporary medicinal chemistry.3The molecule 3-quinuclidinone hydrochloride possesses variety of biological spectrum and is a part of many existing drugs such as azasetron, palonosetron, solifenacin, cevimeline, quinupramine (Fig. 1). Literature survey revealed that several derivatives of quinuclidines have been reported to show wide range of biological activity such as Alzheimer's disease4 chronic obstructive pulmonary disease,5 antihistamine-bronchodilating agents,6 α7 and α4β2 nicotinic receptors inhibitory activity.7 According to Malki et al. analogs of quinuclidinone can provide an excellent scaffold for novel anti-cancer agents with improved safety profile.8 They used lung carcinoma cells for study and observed that in more potent derivatives, the carbonyl group of quinuclidinone was intact.8 Further in another study they observed that quinuclidinone derivatives induce apoptosis in human breast cancer cells via reduced expression level of Bcl-2, Bcl-XL and increased mitochondrial apoptotic pathways by activating the release of cytochrome C.9 The derivatives of this molecule may have a selective mode of action as they are structurally unique, and yet have a great deal of known chemistry upon which to prepare analogs. In search of novel more potent anti-cancer compounds with greater affinity for cancer cells than healthy normal cells, we decided to explore the anti-cancer activity of some novel quinuclidinone derivatives. We have recently reported the synthesis of quinuclidinone hydrochloride from isonipecotic acid.10 In the present article we report the synthesis of some quinuclidinone based ester and amide derivatives with cytotoxicity and apoptosis-inducing property in lung cancer cells.
2 Result and discussion
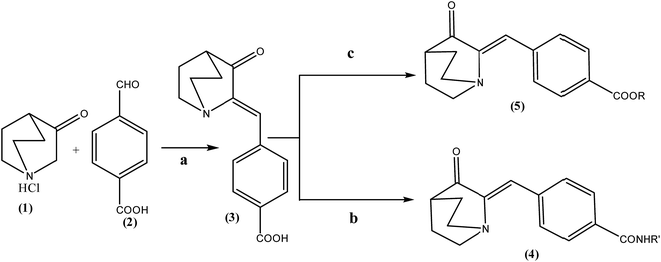
2.1 Synthesis
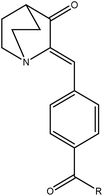
In the first step 3-quinuclidinone hydrochloride (1) was refluxed with 4-formyl benzoic acid (2) in the presence of sodium hydroxide, using absolute ethanol as solvent to give 4-(3-oxo-1-aza-bicyclo[2.2.2]oct-2-ylidenemethyl)-benzoic acid (3) (Scheme 1). Formation of this intermediate acid (3) was confirmed from the broad absorption band at around 3200 cm−1 in the infrared spectrum (IR) and downfield peak at 13.1 δ ppm in 1H NMR. The acid derivative (3) was converted to the acid chloride with thionyl chloride in the presence of triethylamine. The acid chloride was further converted in to the corresponding ester or amide derivative by reaction with appropriate alcohol/amine. The structure of synthesized compounds established on the basis of elemental analysis and spectral data. In case of amide derivatives (4a to 4f), the NH stretching frequency appeared around 3300 to 3450 cm−1. The formation of ester (5a to 5e) was confirmed from the disappearance of the broadband of carboxyl in the IR spectrum and appearance of absorption band in the region 1728–1750 cm−1. 13C NMR downfield peak appeared around 165–169 δ ppm, which further confirms the formation of ester derivatives. Single crystal of compound 4c and 5c (Fig. 6 and 7) were obtained by slow evaporation method using methanol as a solvent. The crystal structures of compound 4c and 5c shows the presence of double bond with Z geometry. This is well in agreement with previous report.11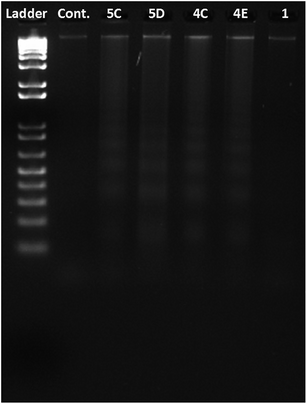 | ||
| Fig. 4 DNA ladder test for apoptosis: ladder formation suggests induction of apoptosis (lane 1: ladder, lane 2: control, lane 3: 5d, lane 4: 5c, lane 5: 4c, lane 6: 4e, lane 7: quinuclidinone HCl 1). | ||
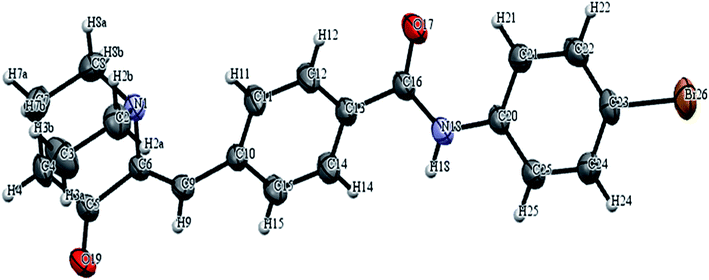 | ||
| Fig. 6 ORTEP diagram of compound 4c with atom numbering scheme. Displacement ellipsoids are drawn at the 50% probability. | ||
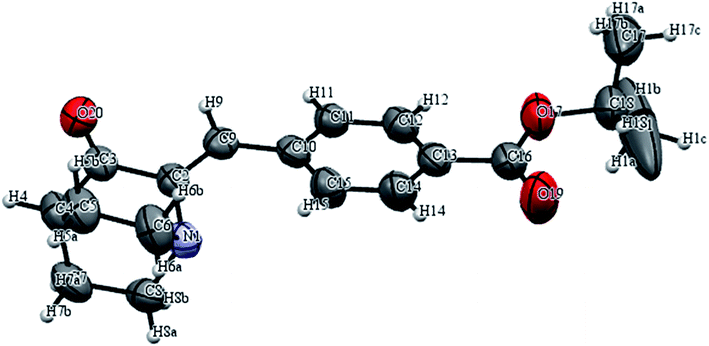 | ||
| Fig. 7 ORTEP diagram of compound 5c with atom numbering scheme. Displacement ellipsoids are drawn at the 50% probability. | ||
2.2 Biological assay and structure activity relationship (SAR)
The synthesized compounds were screened for MTT assay. All compounds were screened at 1, 5, 10 and 20 μM concentrations. The results obtained in a cytotoxicity assay for quinuclidinone and its ester and amide analogues accounted for decreased cell viability in a dose dependent manner. The results also suggest that the synthesized compounds showed specificity for cancer cells over normal lung cells.The half inhibitory concentration IC50 values in μM concentration were determined and presented in Table 1. The results indicate that compared to esters all amide derivative exhibited reasonably good activity. Among amides the derivative having unsubstituted phenyl ring (4a) was found to be less active among all. The introduction of either electron donating group (methyl) or electron withdrawing group (bromo) increases the activity. Amide derivative having parabromophenyl group (4c) was found to be most potent among all aromatic amides. In order to determine if aromatic ring is essential for activity, derivatives with cyclohexyl (4d) and heterocyclic ring such as morpholine (4e) and pyrrolidine (4f) were synthesized. In all these cases an increase in the activity was observed.
In ester series the derivatives in which the oxygen of ester group was attached to a secondary carbon exhibited better activity. Thus esters of secondary alcohols such as isopropyl alcohol, sec-butanol, and cyclopropyl alcohol were found to be more active. The compound (5c) bearing isopropyl group was found to be most active followed by (5d) and (5e) which were moderately active. Lowering of activity was observed when methyl (5a) and ethyl groups (5b) were introduced.
Synthesis of new chemotherapeutic agents with selective cytotoxicity towards cancer cells is always a major challenge for chemists and biotechnologists. Based upon the results obtained in the cytotoxicity assay the derivatives 4c, 4e, 5c and 5d were selected for a detailed scrutiny to assess the mode of cell death.
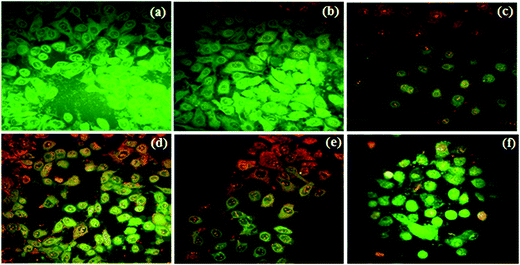
Cancer cells were stained with fluorescent stains to gain a deeper insight into the mechanism of cell death. Induction of apoptosis is a key event and a preferred pathway for induction of cell death by a test compound.12 Hence, a fluorescent probe (AO/EB) was used to gather qualitative evidence on apoptosis. It has been reported that the viable cells show green fluorescence and late apoptotic cells show orange to red fluorescence with condensed chromatin.13 We could observe more orange to red fluorescent cells 4c, 4e and 5c as compared to 5d and quinuclidinone HCl 1 treated cells suggesting induction of apoptosis (Fig. 2).
Nuclear condensation resulting due to any test compound is assessed using DAPI staining.14 In our study, condensation/fragmentation/distortion of nuclei was evident in all treatment groups (Fig. 3). Further confirmation on apoptosis was obtained from DNA ladder assay, wherein apoptosis induced DNA damage is accredited to possible induction of apoptotic pathway.15 In the present study, all the test compounds showed moderate to heavy ladder formation (Fig. 4) suggesting induction of apoptosis.
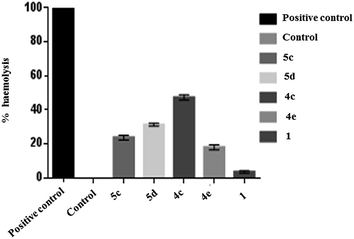
Novel compounds that may be potent in destruction of cancer cells often cause damage to the red blood corpuscles (RBC) and hence, haemolytic assay provides a clue on its merit in not destroying the RBC.16 Hence, haemolytic assay is a popular tool to assess the relative impact of test compounds on RBCs.16 In our study 4e, 5e, 5f, and the quinuclidinoneHCl 1 accounted for moderate haemolysis indicating at their relative safety for in vivo use as a possible therapeutant (Fig. 5).
3 Conclusions
In conclusion a series of novel quinuclidinone based amides and esters (4a to 4f and 5a to 5f) were synthesized. The structures of title compounds were well supported by spectroscopic data and elemental analysis. Test compounds were able to induce apoptosis of A549 lung carcinoma cells with minimal damage to the L132 normal lung cells. The most potent compounds (4c, 4e, 5c and 5d) were subjected to further investigations. DNA fragmentation suggests that the cytotoxic effect of the compound is selectively mediated through the induction of apoptosis. Additional experiments are required to determine the mechanism of action and for better elucidation of structure activity relationships of this class of molecules.4 Experimental
4.1 Materials and methods
Commercial grade solvents and reagents (alcohols and amines) were purchased from Sigma-Aldrich or Alfa Aeser or Spectrochem, Mumbai, India and used without further purification. Quinuclidinone hydrochloride was prepared as described in literature.10 Melting points were measured using a (Buchi B-545) melting point apparatus and were uncorrected. Infrared spectra were recorded on a Perkin-Elmer RX 1 spectrometer. Elemental analyses were recorded on Thermo Finnigan Flash 11-12 series EA. 1H and 13C NMR spectra were recorded on an Advance Bruker (400 MHz) spectrometer in suitable deuterated solvents. 1HNMR data were recorded as follows: chemical shift measured in parts per million (ppm) downfield from TMS multiplicity, observed coupling constant (J) in Hertz (Hz), proton count. Multiplicities are reported as singlet (s), broad singlet (br s), doublet (d), triplet (t), quartet (q) and multiplet (m). 13C NMR chemical shifts are reported in ppm downfield from TMS. Solvents and reagents were purified by literature methods. Mass spectra were determined by LC/MS, using Shimadzu LCMS 2020 and AB Sciex 3200 QTRAP. The reaction progress was monitored by TLC in ultraviolet light as well as with iodine vapor.4.2 Biological assay
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 15 min. 350 μL of the supernatant was again lysed in 106 μL of second lysis buffer (150 mM NaCl, 10 mM Tris–HCl (pH 8.0), 40 mM EDTA, 1% SDS and 0.2 mg mL−1 of proteinase K, at final concentration) for 4 h at 37 °C. The DNA was extracted with phenol/chloroform/isoamyl alcohol (25
000 rpm for 15 min. 350 μL of the supernatant was again lysed in 106 μL of second lysis buffer (150 mM NaCl, 10 mM Tris–HCl (pH 8.0), 40 mM EDTA, 1% SDS and 0.2 mg mL−1 of proteinase K, at final concentration) for 4 h at 37 °C. The DNA was extracted with phenol/chloroform/isoamyl alcohol (25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25
25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v/v), and the pellet thus obtained was washed with ethanol and re-suspended for RNAase digestion in 15 μL of 10 mM Tris, 1 mM of EDTA (pH 8.5), and 50 μg mL−1 of RNAase for 1 h at 37 °C. The fragmented DNA was quantified on 2% agarose gel electrophoresis.19
1 v/v/v), and the pellet thus obtained was washed with ethanol and re-suspended for RNAase digestion in 15 μL of 10 mM Tris, 1 mM of EDTA (pH 8.5), and 50 μg mL−1 of RNAase for 1 h at 37 °C. The fragmented DNA was quantified on 2% agarose gel electrophoresis.19
4.3 Synthesis of (Z)-4-((3-oxoquinuclidin-2-ylidene)methyl)benzoic acid (3)
Compound 1 and formyl benzoic acid were taken in absolute ethanol and the solution was refluxed with catalytic amount of sodium hydroxide for about 3–4 h. The reaction progress was monitored by TLC. After completion the reaction mixture was acidified with glacial acetic acid. Crude product precipitated was filtered and washed with water. It was dried under vacuum and recrystallized using IPA-water.4.4 General procedure for the preparation of compounds amides (4)
The product of step one (1 mmol) was taken in 20 ml dichloromethane and triethyl amine (3 mmol) was added. The resulting mixture was stirred at 0 °C and thionyl chloride (1.1 mmol) was added carefully. It was then warmed to room temperature and refluxed for 30 min. After solvent evaporation the unreacted thionyl chloride was removed under vacuum. The acid chloride thus obtained was taken in 20 ml acetonitrile and appropriate amine (1.12 mol) was added followed by K2CO3 (3 mmol). The reaction mixture was refluxed and the progress of the reaction was monitored by TLC. After completion, the alcohol was evaporated under vacuum. The residue was dissolved in ethyl acetate (50 ml) and washed with water and sat. NaHCO3 solution. The organic layer was dried with sodium sulfate and solvent evaporated to give the ester as solid product.4.5 General procedure for the preparation of esters (5)
To the acid chloride obtained above, appropriate alcohol was added in excess and the mixture was refluxed. The progress of the reaction was monitored by TLC. After completion, the alcohol was evaporated under vacuum. The residue was dissolved in ethyl acetate (50 ml) and washed with water and sat. NaHCO3 solution. The organic layer was dried with sodium sulfate and solvent evaporated to give the ester as solid product.13C NMR (100 MHz, DMSO): δ 25.3, 32.8, 47.3, 48.8, 123.1, 127.7, 131.9, 135.7, 136.4, 146.35, 165.3, 205.4. IR (KBr, cm−1): 3341, 2926, 2853, 1745, 1703, 1653, 1534, 1500, 800. ESI/MS 338.3 [M + 1]+ calculated for C21H26N2O2.Anal. calcd for:: C, 74.53; H, 7.74; N, 8.28; O, 9.45; found C, 74.78; H, 7.51; N, 8.37.
Acknowledgements
The authors are grateful to the Synth Services Pvt. Ltd, Jarod, Vadodara, India, for financial assistance. The authors thank the DST-PURSE program for Single Crystal X-ray Diffraction, DST FIST program for NMR analysis, Dr Vikram Sarabhai Central instrumentation facility for LCMS and confocal microscopy at the Faculty of Science, The Maharaja Sayajirao University of Baroda and Blue cross pathology lab, Vadodara for haemolytic study.References
- Y. Jiao, B.-T. Xin, Y. Zhang, J. Wu, X. Lu, Y. Zheng, W. Tang and X. Zhou, Eur. J. Med. Chem., 2015, 90, 170 CrossRef CAS PubMed.
- A. Malki, R. Laha and S. C. Bergmeier, Bioorg. Med. Chem. Lett., 2014, 24, 1184 CrossRef CAS PubMed.
- N. Inceler, A. Yilmaz and S. N. Baytas, Med. Chem. Res., 2013, 22, 3109 CrossRef CAS.
- M. I. Rodríguez-Franco, I. Dorronsoro, A. Castro, A. Martínez, A. Badía and J. E. Baños, Bioorg. Med. Chem., 2003, 11, 2263 CrossRef.
- J. P. Starck, L. Provins, B. Christophe, M. Gillard, S. Jadot, P. Lo Brutto, L. Quéré, P. Talaga and M. Guyaux, Bioorg. Med. Chem. Lett., 2008, 18, 2675 CrossRef CAS PubMed.
- L. Farnell, C. R. Ganellin, G. J. Durant, E. Parsons, C. R. Ganellin, J. Port, W. G. Richards, R. Ganellin, C. R. Ganellin, W. Black, H. Pullman, J. Port, M. Kakudo, D. H. Aschman and J. W. Black, J. Med. Chem., 1975, 18, 666 CrossRef.
- H. R. Arias, J. J. López, D. Feuerbach, A. Fierro, M. O. Ortells and E. G. Pérez, Int. J. Biochem. Cell Biol., 2013, 45, 2420 CrossRef CAS PubMed.
- A. Malki, A. B. Pulipaka, S. C. Evans and S. C. Bergmeier, Bioorg. Med. Chem. Lett., 2006, 16, 1156 CrossRef CAS PubMed.
- A. Malki and E. S. El Ashry, J. Chemother., 2012, 24, 268 CrossRef CAS PubMed.
- J. Y. Soni, V. Premasagar and S. Thakore, Lett. Org. Chem., 2015, 12, 277 CrossRef CAS.
- N. R. Madadi, S. Parkin and P. A. Crooks, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2012, 68, 0730 Search PubMed.
- S. Thakore, M. Valodkar, J. Y. Soni, K. Vyas, R. N. Jadeja, R. V. Devkar and P. S. Rathore, Bioorg. Chem., 2013, 46, 26 CrossRef CAS PubMed.
- M. Valodkar, R. N. Jadeja, M. C. Thounaojam, R. V. Devkar and S. Thakore, Mater. Chem. Phys., 2011, 128, 83 CrossRef CAS PubMed.
- K. Vyas, R. N. Jadeja, D. Patel, R. V. Devkar and V. K. Gupta, Polyhedron, 2013, 65, 262 CrossRef CAS PubMed.
- R. Singh, R. N. Jadeja, M. C. Thounaojam, T. Patel, R. V. Devkar and D. Chakraborty, Inorg. Chem. Commun., 2012, 23, 78 CrossRef CAS PubMed.
- F. Hayat, E. Moseley, A. Salahuddin, R. L. van Zyl and A. Azam, Eur. J. Med. Chem., 2011, 46, 1897 CrossRef CAS PubMed.
- M. Kutwin, E. Sawosz, S. Jaworski, N. Kurantowicz, B. Strojny and A. Chwalibog, Nanoscale Res. Lett., 2014, 9, 257 CrossRef PubMed.
- P. J. Shiny, A. Mukherjee and N. Chandrasekaran, Bioprocess Biosyst. Eng., 2014, 37, 991 CrossRef CAS PubMed.
- M. Nath, M. Vats and P. Roy, Inorg. Chim. Acta, 2014, 423, 70 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1025491 and 1051487 contains the crystallographic data for the compounds 4c and 5c respectively. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra15127a |
| This journal is © The Royal Society of Chemistry 2015 |