Hydrothermal carbonization of bamboo in an oxalic acid solution: effects of acid concentration and retention time on the characteristics of products†
Xun Li,
Ming-Fei Li*,
Jing Bian,
Bo Wang,
Ji-Kun Xu and
Run-Cang Sun*
Beijing Key Laboratory of Lignocellulosic Chemistry, Beijing Forestry University, Beijing 100083, China. E-mail: limingfei@bjfu.edu.cn; rcsunn3@bjfu.edu.cn; Fax: +86 10 62336972; Tel: +86 10 62337223, +86 10 62336972
First published on 28th August 2015
Abstract
Bamboo was hydrothermally carbonized in a batch reactor using an oxalic acid solution at 190 °C for the production of hydrochar and aqueous products. The influences of oxalic acid concentration and retention time were examined, and the products obtained were characterized in terms of energy yield, chemical components and structural properties. It was found that the energy densification values at a high acid concentration of 0.8 mol L−1 were comparable to those of most torrefied lignocelluloses. No hemicelluloses were detected in the hydrochar obtained under the conditions of highest severity (0.8 mol L−1 acid concentration and 20 min), indicating that the lignocellulose hemicelluloses were completely removed. In addition, cellulose also showed some degradation; the highest degradation rate of cellulose was 56.69% at the highest carbonization severity. The lignin content showed an increasing trend in the hydrochar with increasing carbonization severity. The thermal stability of the hydrochar increased with the carbonization severity. The aqueous solution obtained had a high antioxidant capacity with a inhibition rate of 63.5% for 2,2-diphenyl-1-picrylhydrazyl, higher than that of a commercial antioxidant, butylated hydroxytoluene (46.4%). The present study indicated that the hydrothermal carbonization of bamboo produced a solid hydrochar with high potential for fuel applications and degraded chemicals with high potential as antioxidants.
1. Introduction
Lignocelluloses are clean and renewable natural resources, which can be transformed into various forms of energy by thermochemical processes. The production of energy from lignocellulose can reduce our dependence on foreign oil, improve air quality and support the sustainable development of society. However, lignocelluloses have many drawbacks such as their high oxygen content, hygroscopic nature and low bulk density. Consequently, the utilization efficiency of lignocelluloses is low and they are not suitable for further processing. Therefore, pretreatment technologies are needed to address these disadvantages for the preparation of a solid fuel, which provides a more suitable feedstock for further conversion such as combustion and gasification. Among the many protocols, carbonization attracts considerable attention because it improves not only the energy content of lignocellulose,1 but also its handling, transportation, and storage properties.2In general, there are two processes to increase energy for the carbonization of lignocellulose: torrefaction and hydrothermal carbonization. Torrefaction is a pyrolysis process at relatively low temperatures ranging from 200 to 300 °C, in an inert gas atmosphere. Numerous studies have been conducted to analyze the products of the torrefaction of various feedstocks under different conditions.3–6 During this process, 80% to 95% of the energy and 70% to 90% of the mass of lignocelluloses are preserved in solid chars.7 On the other hand, considerable attention has been recently paid to the preparation of hydrochar by the hydrothermal carbonization of lignocelluloses for a range of uses. Hydrothermal carbonization refers to the preparation of a solid residue (hydrochar) at temperatures ranging from 180 to 250 °C by applying suspended lignocelluloses and water under a saturated pressure. Three products, solid fuel, aqueous chemicals, and gases, are produced during this endothermic process. A study of loblolly pine carbonized by hot water from 200 to 260 °C indicated that hydrothermal carbonization achieved higher energy densification at the same mass yield compared to dry torrefaction in N2 from 250 to 300 °C.8 Hydrothermal carbonization can be conducted at temperatures 100 °C lower than those used for torrefaction under gas, when achieving the same calorific value. In general, the energy density of lignocellulose is enhanced, and the higher heating value (HHV) is increased by about 3% to 47% after hydrothermal carbonization.9 In addition, the hydrochar obtained showed hydrophobic properties after carbonization, indicating better storage characteristics.10 The hydrothermal process is associated with a series of hydrolysis, condensation, decarboxylation, and dehydration reactions.11 During the hydrothermal carbonization process, acids are also produced and dissolved in the aqueous solution, thus promoting the reactions of lignocelluloses. For instance, it was reported that 0.025 g acetic acid was produced from per gram of loblolly pine by hydrothermal carbonization at 230 °C for 5 min. Formic acid and lactic acid have also been reported to be formed during hydrothermal carbonization.8 Because the major reactions during hydrothermal torrefaction are caused by H+ from water as well as the released acids, it is hoped that the addition of acid could promote the reaction, thus reducing the reaction temperature.
Bamboo has recently received special attention due to its easy propagation, fast growth and regeneration, and high productivity as well as its rapid maturity. The plantation of bamboo achieves efficient utilization of land and yields more biomass per unit area than other plants.12 Bamboo is an important natural resource that is traditionally used to produce furniture, veneers and flooring. A significant amount of bamboo is unsuitable for manufacturing products and is available at the processing sites. Thus, the utilization of bamboo for bioenergy production is worth exploring. Since the potential of products from bamboo hydrothermally carbonized under an acid solution has not been investigated, a comprehensive study is required to achieve this value-added utilization of bamboo. In this study, an oxalic acid solution was used as the reaction medium. Oxalic acid was selected because it is more environmentally friendly than mineral acids such as H2SO4 and it is a natural secondary metabolite product in some plants. The influences of oxalic acid concentration and retention time during bamboo carbonization were examined. The results for the hydrochar in terms of energy yield, chemical components and structural properties are presented. In addition, the liquid products obtained after hydrothermal carbonization were also investigated with respect to their antioxidant capacities.
2. Materials and methods
2.1. Materials
Bamboo (Phyllostachys acuta) was obtained from a firm in Yunnan province (China). The bamboo culms were chopped and smashed to obtain particles with sizes of 20 to 80 mesh. The powder was de-waxed with methylbenzene/ethanol (2/1, v/v) in a Soxhlet extractor for 8 h and then treated with water at 80 °C for 6 h to remove starch before air-drying. The chemical components of the bamboo sample were 46.03% glucan, 25.27% xylan, 0.90% arabinan, 0.32% galactan, 24.19% Klason lignin and 1.67% acid soluble lignin; these were determined according to a method reported previously.132.2. Carbonization of bamboo
The bamboo powder was hydrothermally carbonized in a cylindrical reactor with a Teflon liner with varied acid concentrations and retention times, as shown in Table 1. The selection of acid concentrations and retention times was conducted according to the preliminary investigations. The initial studies indicated that torrefaction with acid concentrations of 0.4–0.8 mol L−1 and retention times of 0–20 min resulted in marked changes in mass yield and product characteristics. In a typical run (R4), 3 g of bamboo sample (S0) was mixed with a 30 mL of 0.6 mol L−1 oxalic acid solution. The mixture was added to a 100 mL cylindrical reactor, which was manufactured with chemically modified Teflon and lined with an ultra-strong aerospace composite fiber material. The pressure of the reactor was monitored by a piezoelectric crystal pressure sensor (0 to 10 ± 0.01 MPa) and the temperature was controlled by a platinum resistance control system (0 to 300 ± 1 °C). After being sealed by screwing, the reactor was heated in a microwave oven (MDS-6G, Sineo Microwave Chemistry Technology Co., Ltd). The main parameters were frequency (2500 MHz) and maximum output power (500 W). The sample was heated by microwave irradiation at 190 °C for 0 min, and then the heating was stopped before cooling to temperatures below 50 °C. The temperature profile was obtained (Fig. 1) and the severity was determined according to previous studies.14,15 Briefly, severity (So) was calculated by the equation: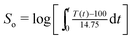 , where t is time and T(t) is the temperature during the process. The mixture was separated using a filter paper. The filtrate was recovered and stored at 4 °C for further analysis. The filtered residue was further washed with water to neutralize the pH and dried at 105 °C for 12 h before analysis.
, where t is time and T(t) is the temperature during the process. The mixture was separated using a filter paper. The filtrate was recovered and stored at 4 °C for further analysis. The filtered residue was further washed with water to neutralize the pH and dried at 105 °C for 12 h before analysis.
| Run | Temperature (°C) | Oxalic acid concentration (mol L−1) | Retention time (min) | Severity | Hydrochar | Liquid phase |
|---|---|---|---|---|---|---|
| R1 | 190 | 0.4 | 0 | 3.33 | S1 | L1 |
| R2 | 190 | 0.4 | 10 | 3.71 | S2 | L2 |
| R3 | 190 | 0.4 | 20 | 4.02 | S3 | L3 |
| R4 | 190 | 0.6 | 0 | 3.33 | S4 | L4 |
| R5 | 190 | 0.6 | 10 | 3.71 | S5 | L5 |
| R6 | 190 | 0.6 | 20 | 4.02 | S6 | L6 |
| R7 | 190 | 0.8 | 0 | 3.33 | S7 | L7 |
| R8 | 190 | 0.8 | 10 | 3.71 | S8 | L8 |
| R9 | 190 | 0.8 | 20 | 4.02 | S9 | L9 |
2.3. Characterization of the products
The chemical component was analyzed according to the National Renewable Energy Laboratory (NREL) standard protocol.13 The sugar and lignin components were assayed with a two-step hydrolysis process using H2SO4. The solid residue after the hydrolysis represents Klason lignin, and the hydrolysate was used for the analysis of acid soluble lignin and sugars. Acid soluble lignin was determined using an UV spectrometer (UV 2300, Shanghai Tianmei Science and Technology Corporation, China) and the sugars were quantified on a high performance anion exchange chromatography system (Dionex ICS 3000, USA) according to a previous study.16 The sugars, including L-arabinose, D-xylose, D-glucose, and D-galactose, were separated with isocratic 5 mM NaOH for 20 min followed by a 0–75 mM NaAc gradient in 5 mM NaOH for 15 min under a nitrogen atmosphere. The contents of glucan, xylan, galactan, and arabinan in the samples were calculated according to stoichiometry during the hydrolysis of polysaccharides into monosaccharides. Cellulose was measured as glucan, hemicellulose was quantified by the sum of xylan, arabinan and galactan, and lignin was calculated as the sum of Klason lignin and acid soluble lignin.Elemental (C, H, O, N, and S) contents were estimated using an elemental analyzer Vario EL III (Elementar, Germany). Higher heating value (HHV) was estimated according to the following formula: HHV (MJ kg−1) = 0.3383ZC + 1.422(ZH − ZO/8), where ZC, ZH, and ZO are the weight percentages of C, H, and O, respectively (since no N and S were detected), after correction for ash and moisture.17 The mass yield of hydrochar (MY), energy densification (ED) and energy yield (EY) are calculated as follows: MY (%) = masschar/massbamboo × 100; ED = HHVchar/HHV bamboo; EY = ED × MY.
FTIR spectra were obtained using an FTIR spectroscope (Thermo Nicolet Corporation, Madison, US) with a MCT detector cooled by liquid nitrogen, according to a previous study.18 The data were obtained in the wavenumber ranging from 4000 to 850 cm−1. XRD analysis of the sample was conducted by scanning the sample on an XRD-6000 instrument (Shimidzu, Japan). The samples were placed on a glass holder, and the X-ray diffractograms were obtained from 2θ = 5° to 40° at a scanning speed of 5° min−1. Thermogravimetric analysis (TGA) was performed using a simultaneous thermal analyzer (SDT Q600, TA instrument). Samples of 5–10 mg were placed in alumina crucibles and heated from 30 °C to 750 °C at a heating rate of 10 °C min−1 in the presence of nitrogen (50 mL min−1).
The aqueous phases were subjected to antioxidant ability analysis against 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical according to the method developed by Brand-Williams et al.19 The aqueous phases were diluted in water at a concentration of 3 g L−1, and 0.1 mL of the solution was mixed with 3.9 mL of 25 mg L−1 DPPH in a methanol solution. The absorbance at 518 nm of the mixture was recorded at 30 min. A solution of the commercial antioxidant butylated hydroxytoluene (BHT) was also used for comparison. The DPPH radical-scavenging activity was calculated using the following formula: DPPH radical scavenging activity (%) = (A0 − A1)/A0 × 100, where A0 is the absorbance of the control and A1 is the absorbance of the lignin sample. In the abovementioned experiments, the analyses were performed in triplicate and the errors were reported. The significant difference was estimated with a two-sided t test, in which p value < 0.05 represents statistical significance.20
3. Results and discussion
3.1. Mass yield and fuel characteristics of hydrochars
The mass yield of hydrochar was significantly influenced by the reaction conditions; the mass yield decreased with increased acid concentration and retention time (Fig. 2). The highest yield was obtained at a retention time of 0 min and an acid concentration of 0.4 mol L−1. The mass yield in the present study was compared to the char previously obtained by torrefaction under an inert gas as well as the hydrochar from grape pomace obtained by hydrothermal carbonization at 200–300 °C.21,22 It was apparent that hemicelluloses significantly degraded due to carbonization, starting from the sample carbonization at the lowest severity, wherein the hemicellulose content was reduced from 35.18% in S0 to 1.09% in S1 (p < 0.05). Galactan was the most unstable hemicellulose and was degraded even at the lowest severity. No hemicelluloses were detected for the hydrochar obtained at the highest severity (S9), indicating that hemicelluloses were completely removed. In addition, cellulose also showed some degradation; the highest degradation rate of cellulose was 56.69% at the highest carbonization severity. The Klason lignin content, representing the acid insoluble components (including modified lignin as well as condensed products from cellulose and hemicellulose degradation), showed an increasing trend in the hydrochars with increasing carbonization severity. The carbonized samples showed an increase in lignin content. This was due to the structural changes of lignin (mainly cleavage of the ether bonds), together with the poly-condensation reaction between the carbohydrate degradation products that formed pseudo-lignin during the acid carbonation process, which resulted in an overestimation of lignin content.6 | ||
| Fig. 2 (a) Mass yield, (b) carbohydrates, and (c) lignin content of hydrochars from bamboo carbonization. | ||
3.2. Energy characteristics
The energy characteristics of the hydrochars obtained from different retention times and oxalic acid concentrations are illustrated in Fig. 3. The contour graph was drawn with Originpro 2015 (OriginLab Corporation, USA), in which the color represents the HHV, energy yield, and energy densification resulting from variations in the concentration of acid and the retention time. The values corresponding to the colors are indicated in the legends on the right part of the graph. As expected, the higher heating value of bamboo improved after carbonization, whereas the HHV values of the hydrochars were enhanced as the retention time and oxalic acid concentration increased. The lowest HHV value was achieved from the carbonization with 0.4 mol L−1 oxalic acid for a retention time of 0 min (18.37 MJ kg−1). The values obtained at an oxalic acid concentration of 0.6 mol L−1 for retention times of 0–20 min were in the range of 19.51–20.00 MJ kg−1, and the values obtained at an oxalic acid concentration of 0.8 mol L−1 for retention times of 0–20 min were in the range of 19.59–20.47 MJ kg−1. These values were comparable to the hydrochars obtained from bedding material.23 | ||
| Fig. 3 (a) Energy yield, (b) HHV, and (c) energy densification of hydrochars from bamboo carbonization at different acid concentrations and retention times. | ||
Energy yield, a means to evaluate the energy retained in hydrochars compared to that retained in the original bamboo, is calculated as the energy densification ratio times the mass yield of the hydrochar. As can be seen, the energy yields were significantly affected by the acid concentration and retention time. The energy yields were in the ranges of 59.81–60.81%, 58.89–62.67%, and 55.26–59.74% for carbonization at acid concentrations of 0.4, 0.6 and 0.8 mol L−1, respectively. The energy intensified as the solid mass decreased with increasing acid concentration and retention time. This was due to the dehydration and decarboxylation reactions, as evidenced by the increase in the carbon content as well as the decrease in the oxygen and hydrogen contents. The energy densification after carbonization was associated with both retention time and acid concentration; these were in the ranges of 1.07–1.12, 1.14–1.17 and 1.14–1.19 for the carbonizations at acid concentrations of 0.4, 0.6 and 0.8 mol L−1, respectively. The energy densification values at high acid concentrations were comparable to those of most torrefied lignocelluloses (such as rich husk, empty fruit bunches, and eucalyptus) obtained at high temperatures (200–300 °C) under an inert gas.2 At the same acid concentration, torrefaction in oxalic acid solution at 190 °C for 20 min (severity 4.02) produced hydrochar with an HHV, mass yield, and energy yield of 19.21 MJ kg−1, 55.06%, and 59.81%, as compared to 21.56 MJ kg−1, 37.46%, and 47.07%, respectively for the hydrochar obtained from torrefaction in an HCl solution at 180 °C for 15 min (severity 3.61).24 It was concluded that torrefaction with oxalic acid solution resulted in relatively higher mass and energy yields but lower HHV values compared to torrefaction in an HCl solution.
The elemental content of the hydrochars after carbonization is shown in Fig. 4. The ternary diagram (plotted by Originpro 2015) presents the variation of carbon–hydrogen–oxygen (C–H–O) for the samples (from S0 to S9), in which the total contents of carbon, hydrogen and oxygen for a given sample are equal to 100% and the amount of each component can be read in the corresponding scale. This diagram shows an increase in carbon content but a decrease in oxygen content with both increasing acid concentration and retention time. The figure shows that with increasing carbonization severity, dehydration and decarboxylation reactions occurred. The highest carbon content of 55.44% and the lowest oxygen content of 38.55% were achieved at S9 compared to those of the raw material (bamboo showed a carbon content of 49.67% and an oxygen content of 44.53%) and other hydrochars.
3.3. Morphological and structural characterization of hydrochars
To gain more insight into the impact of the carbonization conditions on the structure of bamboo, SEM images of bamboo before and after torrefaction are illustrated in Fig. S1 in the ESI.† The images observed by SEM are amplified by factors of 10 K to demonstrate the microstructures (left column), and camera images are also presented (right column). It was expected that due to the presence of the main components of lignocellulose, the SEM images of the hydrochar would more closely resemble the raw material; however, the SEM images of the hydrochar showed a very clear difference, suggesting the rupturing of the lignocellulose structure after the carbonization process. The raw material was light yellow, whereas hydrochars S3 and S6 were brown in color, consistent with partial carbonization; hydrochar S9 was deep brown in color. The image of bamboo shows the cellular structure of lignocellulosic materials. The SEM image of hydrochar S3 indicates some breakdown of the lignocellulose material. Numerous sphere-like micro-particles were observed on the surface of the hydrochars in which S9 showed the biggest particles. These micro-particles were generated by the degradation of carbohydrates during carbonization, followed by precipitation and growth to form spheres.25 The hydrochar samples became crustier than the raw material. This is mainly because the lignin component underwent partial degradation due to its greater thermal stability. In an earlier study by Lynam et al., it was explained that among all lignocellulose components, lignin is the most plentiful in the cell wall; thus, the presence of more lignin or the absence of carbohydrates results in a crusty hydrochar.26The effects of acid concentration and retention time on the degradation of cellulose and hemicelluloses were also supported by the FTIR spectra obtained from the hydrochar and raw material (Fig. S2 in the ESI†). The absorption bands in the spectra were assigned as follows: (a) the peaks around 2900 cm−1 were attributed to the C–H stretching and deforming vibration in CH2 and CH3; (b) the peak at 1730 cm−1 was attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration, mainly from the carbonyl and ester groups of hemicelluloses; (c) the peaks around 1610–1460 cm−1 were attributed to the C
O stretching vibration, mainly from the carbonyl and ester groups of hemicelluloses; (c) the peaks around 1610–1460 cm−1 were attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching from the aromatic rings of lignin; and (d) the peak at 1160 cm−1 was attributed to the C–O–C vibrations from cellulose and hemicelluloses. As can be seen, the presence of peaks around 2900 cm−1 in both the raw material and hydrochar suggests the existence of CH2 and CH3 groups. The peak at 1730 cm−1 appeared in the spectrum of the raw material but was not observed in the spectra of the hydrochar samples, indicating the degradation of hemicelluloses during the carbonization process. The signals of the peaks at 1460, 1510 and 1600 cm−1 attributed to lignin became stronger with increased carbonization severity, which is consistent with the enrichment of lignin content after carbonization. This implied that some fragments and intermediate structures from lignin remained in the hydrochars, i.e. lignin was not completely decomposed and dissolved in the aqueous solution under the carbonization conditions.
C stretching from the aromatic rings of lignin; and (d) the peak at 1160 cm−1 was attributed to the C–O–C vibrations from cellulose and hemicelluloses. As can be seen, the presence of peaks around 2900 cm−1 in both the raw material and hydrochar suggests the existence of CH2 and CH3 groups. The peak at 1730 cm−1 appeared in the spectrum of the raw material but was not observed in the spectra of the hydrochar samples, indicating the degradation of hemicelluloses during the carbonization process. The signals of the peaks at 1460, 1510 and 1600 cm−1 attributed to lignin became stronger with increased carbonization severity, which is consistent with the enrichment of lignin content after carbonization. This implied that some fragments and intermediate structures from lignin remained in the hydrochars, i.e. lignin was not completely decomposed and dissolved in the aqueous solution under the carbonization conditions.
The XRD patterns for the raw material and hydrochars were also compared (Fig. S3 in the ESI†). A distinct and sharp cellulose peak (2θ = 22.7°) was present in all the curves of the samples. The patterns of all the hydrochars were similar to that of the raw material, suggesting that the microcrystalline structure of cellulose was preserved after carbonization under the experimental conditions. The calculated CrI values of the hydrochars were 35.2%, 33.9%, and 35.5% for the samples carbonized with retention times of 0, 10, and 20 min in 0.4 mol L−1 acid, 36.4%, 32.2%, and 31.0% for the samples carbonized with retention times of 0, 10, and 20 min in 0.6 mol L−1 acid, and 37.9%, 32.4%, and 29.9% for the samples carbonized with retention times of 0, 10, and 20 min in 0.8 mol L−1 acid. Overall, higher concentrations of acid and longer retention times resulted in lower CrI values. The decrease in the CrI values of the samples can be explained by the fact that crystalline cellulose was partially decomposed in the carbonization process.
3.4. Thermal behavior of hydrochars
The thermal degradation behaviors of the hydrochars and the raw material were examined by thermogravimetry. Fig. S4 in the ESI† shows the TG/DTG curves of the samples. There was a slight mass loss in the initial stage below 120 °C, attributed to the removal of moisture. Hemicellulose degradation occurred at 200 °C and achieved a maximum at 270 °C. The degradation of hemicellulose also overlapped with that of cellulose, and the peak at 320 °C is assigned to cellulose degradation. The peak at 500 °C corresponds to lignin degradation. From the curves, it can be noted that the thermal degradation behavior of the sample was modified after carbonization. For samples S3 and S9, degradation occurred in a narrow range, and the peak at 349–354 °C was indicative of the highest degradation of cellulose and lignin. No notable degradation peak was observed at 500 °C for the curves of S3 and S9, indicating that the structure of lignin in the hydrochar was modified; this was mainly due to the re-polymerization of the primary degradation products. It was also noted that the residual masses of the residues after the thermal degradation at 750 °C were 7.55 ± 1.1% and 13.8 ± 1.5% for S3 and S9, respectively, as compared to 0% for S0. Overall, the thermal stability of the hydrochar increased significantly after torrefaction (p < 0.05).3.5. Antioxidant activity of an aqueous solution from carbonization
The waste aqueous phase from hydrothermal carbonization is a drawback of the process. To overcome this problem, the waste liquid should be treated to reduce the amount of organics prior to discharge. The recovery and utilization of the chemicals dissolved in the solution can improve the economics of the entire carbonization process. Bamboo contains lignin, and its degraded products are rich in phenolics, which are water soluble. Under carbonization at 0.8 mol L−1 acid for a retention time of 0 min, the maximum organic material amount of 44.98 mg mL−1 was recovered. Based on the previous study, the antioxidant activity of the aqueous solution obtained from carbonization was investigated. As can be seen from Fig. 5, the increase in acid concentration and retention time notably enhanced the antioxidant activity. Under the experimental conditions (concentration, 3 mg mL−1 and test time, 30 min), the carbonization with a retention time of 10 min produced aqueous solutions with inhibition rates of 39.1–47.1%, comparable to that of commercial antioxidant BHT (46.4%). The highest antioxidant capacity with an inhibition rate of 63.5% was obtained for the aqueous solution obtained from carbonization at the highest severity. This behavior was mainly due to the enhancement of the degradation of lignin into small, water-soluble fragments when carbonization was conducted with highly concentrated acid for a long retention time. This was in good agreement with previous results from the subcritical water treatment of plants.27 It was concluded that bamboo can be converted into degraded chemicals after the hydrothermal carbonization process.4. Conclusion
Bamboo was carbonized in an oxalic acid solution for the production of hydrochar and aqueous products. The energy densification values at a high acid concentration of 0.8 mol L−1 were comparable to those of most torrefied lignocelluloses. The increase of the HHV was due to the dehydration and decarboxylation reactions, as evidenced by the increase in the carbon content as well as the decrease in the oxygen and hydrogen contents. Hemicelluloses were significantly degraded due to carbonization, whereas the lignin content showed an increasing trend in the hydrochars with increasing carbonization severity. The thermal stability of the hydrochars increased with the carbonization severity. The aqueous solution obtained under the optimized conditions had a higher antioxidant capacity than that of commercial antioxidant BHT.Acknowledgements
The authors wish to express their gratitude for the financial support from the Fundamental Research Funds for the Central Universities (No. YX2015-01), the Beijing Natural Science Foundation (6154031), the Program of International S &T Cooperation of China (2015DFG31860), and the National Natural Science Foundation of China (31400508, 31110103902).References
- S. M. Kang, X. H. Li, J. Fan and J. Chang, Ind. Eng. Chem. Res., 2012, 51, 9023–9031 CrossRef CAS.
- W.-H. Chen, J. Peng and X. T. Bi, Renewable Sustainable Energy Rev., 2015, 44, 847–866 CrossRef CAS PubMed.
- G. Almeida, J. O. Brito and P. Perre, Bioresour. Technol., 2010, 101, 9778–9784 CrossRef CAS PubMed.
- D. Medic, M. Darr, A. Shah, B. Potter and J. Zimmerman, Fuel, 2012, 91, 147–154 CrossRef CAS PubMed.
- J.-W. Lee, Y.-H. Kim, S.-M. Lee and H.-W. Lee, Bioresour. Technol., 2012, 116, 471–476 CrossRef CAS PubMed.
- S. Neupane, S. Adhikari, Z. Wang, A. J. Ragauskas and Y. Pu, Green Chem., 2015, 17, 2406–2417 RSC.
- R. B. Bates and A. F. Ghoniem, Bioresour. Technol., 2012, 124, 460–469 CrossRef CAS PubMed.
- W. Yan, T. C. Acharjee, C. J. Coronella and V. R. Vasquez, Environ. Prog. Sustainable Energy, 2009, 28, 435–440 CrossRef CAS PubMed.
- J. G. Lynam, C. J. Coronella, W. Yan, M. T. Reza and V. R. Vasquez, Bioresour. Technol., 2011, 102, 6192–6199 CrossRef CAS PubMed.
- T. C. Acharjee, C. J. Coronella and V. R. Vasquez, Bioresour. Technol., 2011, 102, 4849–4854 CrossRef CAS PubMed.
- X. Lu, B. Jordan and N. D. Berge, Waste Manage., 2012, 32, 1353–1365 CrossRef CAS PubMed.
- L. Fryda, C. Daza, J. Pels, A. Janssen and R. Zwart, Biomass Bioenergy, 2014, 65, 28–41 CrossRef CAS PubMed.
- A. Sluiter, B. Hames, R. Ruiz, C. Scarlata, J. Sluiter, D. Templeton and D. Crocker, in Laboratory Analytical Procedure, 2008 Search PubMed.
- S. M. Lee and J.-W. Lee, Bioresour. Technol., 2014, 172, 438–443 CrossRef CAS PubMed.
- T. A. Lloyd and C. E. Wyman, Bioresour. Technol., 2005, 96, 1967–1977 CrossRef CAS PubMed.
- M. F. Li, Y. M. Fan, F. Xu, R. C. Sun and X. L. Zhang, Ind. Crops Prod., 2010, 32, 551–559 CrossRef CAS PubMed.
- X. Z. Yuan, J. Y. Tong, G. M. Zeng, H. Li and W. Xie, Energy Fuels, 2009, 23, 3262–3267 CrossRef CAS.
- C.-Z. Chen, M.-F. Li, Y.-Y. Wu and R.-C. Sun, Ind. Eng. Chem. Res., 2014, 53, 12935–12942 CrossRef CAS.
- W. Brand-Williams, M. E. Cuvelier and C. Berset, LWT--Food Sci. Technol., 1995, 28, 25–30 CrossRef CAS.
- S.-P. Nie and M.-Y. Xie, Food Hydrocolloids, 2011, 25, 144–149 CrossRef CAS PubMed.
- J. J. Chew and V. Doshi, Renewable Sustainable Energy Rev., 2011, 15, 4212–4222 CrossRef PubMed.
- M. Pala, I. C. Kantarli, H. B. Buyukisik and J. Yanik, Bioresour. Technol., 2014, 161, 255–262 CrossRef CAS PubMed.
- I. Oliveira, D. Blohse and H. G. Ramke, Bioresour. Technol., 2013, 142, 138–146 CrossRef CAS PubMed.
- M.-F. Li, Y. Shen, J.-K. Sun, J. Bian, C.-Z. Chen and R. Sun, ACS Sustainable Chem. Eng., 2015, 3, 2022–2029 CrossRef CAS.
- A. B. Fuertes, M. C. Arbestain, M. Sevilla, J. A. Macia-Agullo, S. Fiol, R. Lopez, R. J. Smernik, W. P. Aitkenhead, F. Arce and F. Macias, Aust. J. Soil Res., 2010, 48, 618–626 CrossRef CAS.
- J. Lynam, M. T. Reza, W. Yan, V. Vásquez and C. Coronella, Biomass Convers. Biorefin., 2014, 1–9 Search PubMed.
- B. Aliakbarian, A. Fathi, P. Perego and F. Dehghani, J. Supercrit. Fluids, 2012, 65, 18–24 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra15063a |
| This journal is © The Royal Society of Chemistry 2015 |



