Production of a natural red pigment derived from Opuntia spp. using a novel high pressure CO2 assisted-process†
A. N. Nunesab,
C. Saldanha do Carmoab and
Catarina M. M. Duarte*ab
aInstituto de Tecnologia Química e Biológica António Xavier, Universidade Nova de Lisboa, Av. República, 2780-157 Oeiras, Portugal. E-mail: cduarte@itqb.unl.pt
biBET, Instituto de Biologia Experimental e Tecnológica, Apartado 12, 2781-901 Oeiras, Portugal
First published on 24th September 2015
Abstract
Cactus pears (Opuntia spp.) have been identified to be an excellent source of betalain pigments which can be used as a red food colourant. In this work, pigments from cactus pear fruits were effectively extracted by high pressure carbon dioxide assisted-acidified water extraction after a pre-treatment with CO2 at 375 bar, 55 °C and 60 minutes. Different process conditions, namely pressure, temperature and volume ratio of (solid–liquid mixture)/(pressurized CO2), were tested in order to model the extraction of betalains from prickly pears via the response surface methodology. The best response was achieved at 100 bar, 40 °C and 20% volume ratio of (solid–liquid mixture)/(pressurized CO2). Under these conditions, the betalains yield was 89 ± 0.7 mg per 100 g of dried fruit, 83% of the maximum extractable pigments from Opuntia spp. fruits. Furthermore, Opuntia spp. extracts presented a vivid red colour with high antioxidant activity.
Introduction
In recent years, there is a growing interest in the development of natural colourants due to the health promoting effects of natural substances.1 Furthermore, the safety of synthetic food colourants has been related to high levels of toxicity, allergic reactions, and carcinogenic potential.2–7 Although synthetic dyes have lower production costs and greater stability, the European Union and the United States have restricted their use as food additives.6,8 In this field, betalain pigments provide a natural alternative to synthetic red dyes.Betalains are water-soluble vacuolar nitrogen-containing pigments, which are synthesized from the amino acid tyrosine into two structural groups, namely betacyanins with colour differences from purple to violet and betaxanthins with a range of colour from yellow to orange.3,9–11 The added value of these pigments is increased owing to their double function as colourant and as antioxidant.12–18
The major commercial forms of betalains are produced from red beetroot juices (Beta vulgaris L.), available as either juice concentrates or powders, containing from 0.3% to 1% of pigment. This natural food colourant is classified as additive E-162 (EU) and 73.40 (FDA, USA).2,19–23 However, red beet present some drawbacks including the poor colour spectrum and earthy-like flavour caused by geosmin, as well as high nitrate concentrations associated with the formation of carcinogenic nitrosamines.11,19,21,24
Cactus pears fruit have been identified to be a promising alternative betalainic crop covering a wide coloured spectrum from yellow to purple pigments. Cactus pear (Opuntia spp.) is a tropical or subtropical fruit tree, native to America, which grows in arid and semiarid regions.5,25 The largest genus of the Cactaceae family is mainly used for fruit production. It enables rapid growth, good adaptation to poor soils and low requirement for water. Its fruit, cactus pear fruit or prickly pears, is a fleshy berry, varying in shape, size and colour has very tasty pulp full of seeds.5,22,23,25,26 In contrast to red beetroot, cactus fruits do not contain geosmin and pyrazines that are responsible for the unpleasant pettiness of the former, represents lower risk for microbiological contamination, are highly flavoured, show adequate nutritional properties and contains interesting functional compounds.19,20,22,23 Furthermore, the commercial exploitation of this fruit as an alternative source of food colourants may contribute to the sustainable development of the underdeveloped semi-arid regions.2
Betalains, which are synthesized in the cytoplasm and stored in vacuoles, are mainly extracted through conventional extractions.5,22,27,28 These methods have several drawbacks, such as long extraction time, evaporation of a huge amount of solvent, stability problems, batch-to-batch variations, low selectivity and relative low yields.6,8,29–34 Therefore, it is a key focus to develop novel extraction methods with faster extraction rates and higher betalain extraction yields. An efficient extraction should maximize betalain recovery with minimal degradation using environmentally friendly technologies.
Xu et al., 2010 and Santos & Meireles, 2011 have demonstrated that the explosive effect of High Pressure Carbon Dioxide (HPCD) besides inactivating microorganisms and enzymes could also strengthened the anthocyanin extraction from red cabbage and jabuticaba by its superior abilities in cell membrane modification, intracellular pH decrease, disordering of the intracellular electrolyte balance and removal of vital constituents from cells and cell membranes.6,32 Up to date, there is no study on HPCD assisted-water extraction of betalains. However, Liu et al., 2008 showed that HPCD treatment of a red beet extract proved to be effective in inactivating the enzymes responsible for phenolic and betalains degradation. This treatment resulted in no significant change in the colour shade of that extract.35
The aim of this work was to obtain betalain-rich extracts from Opuntia spp. fruits. Within the present study, it was investigated for the first time, the feasibility of using a two-step process, HPCD pre-treatment followed by HPCD assisted-water extraction. Process variables were optimized, such as, pressure, temperature and volume ratio of (solid–liquid mixture)/(pressurized CO2) (RS–L/CO2 (%)) for the maximum recovery of betalains using acidified water. The yield obtained was compared with pressurized water extraction (PWE) and water extraction (WE). The red pigment was compared with commercial red beet extract regarding colour characteristics.
Experimental
Materials
Chemicals used for different extractions methodologies were: carbon dioxide 99.95% from Air Liquide (Lisbon, Portugal), methanol absolute 99.99% from Fisher Scientific (Waltham, MA, USA) and citric acid from Sigma-Aldrich (St Quentin Fallavier, France).For phytochemical characterization: sodium carbonate (Na2CO3) was purchased from Sigma-Aldrich (St Quentin Fallavier, France), Folin–Ciocalteau reagent was acquired from Panreac (Barcelona, Spain) and gallic acid was purchased from Fluka (Germany).
Chemicals used for antioxidant activity assays were: 2′,2′-azobis(2-amidinopropane)dihydrochloride (AAPH), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), caffeic acid (C9H8O4), cobalt fluoride tetrahydrate (CoF2), hydrogen peroxide (H2O2) and picolinic acid (C6H5NO2) from Sigma-Aldrich (St Quentin Fallavier, France) and iron chloride (FeCl3) from Riedel-de-Haën (Seelze, Germany). Disodium fluorescein (FL) was from TCI Europe (Antwerp, Belgium).
Reagents used for phosphate buffer solution (PBS) and sodium phosphate buffer solution (SPB) preparation included sodium chloride (NaCl), potassium chloride (KCl), monopotassium phosphate (KH2PO4) and sodium phosphate monobasic monohydrate (NaH2PO4·H2O) from Sigma-Aldrich (St Quentin Fallavier, France) and sodium phosphate dibasic dehydrate (Na2HPO4·2H2O) from Riedel-de-Haën (Seelze, Germany).
Opuntia spp. fruits preparation
Wild Opuntia spp. fruits were harvested by hand during October 2013. Fruits were collected from a plant growing in the South of Portugal (Algarve – Quarteira – N37°04.400, W008°06.100). All prickly pears fruits were harvested at comparable ripening stages (physiological maturity). For sample preparation, spikes were removed with a brush and Opuntia spp. fruits were freeze dried at −100 °C, in the absence of light, during 72 hours (FreeZone Plus 4.5 L Cascade Freezer Dry System, LABCONCO®, Kansas City, United States of America). The resulting dehydrated fruits were kept in a cold, dry and dark environment until further analyses.Opuntia spp. fruits extraction procedures
From the literature6,32 and some preliminary experimental results (data not shown), process variables and their ranges extraction pressure (100–250 bar), extraction temperature (40–70 °C) and volume ratio of (solid–liquid mixture)/(pressurized CO2) (20–80%) were chosen (Table 1). After selection of independent variables and their ranges, experiments were established based on a CCFC design and each independent variable was coded at three levels between −1, 0 and +1. Taking into account the determined factor range to the extraction pressure (100–250 bar), the values 100 bar (level −1), 175 bar (level 0) and 250 bar (level +1) were selected. The same levels were defined for the temperature and RS–L/CO2 according to Table 1. A total of 17 assays including three replicates of the center points were generated (Table 2).
| Variable, factors, unit | Levels | ||
|---|---|---|---|
| −1 | 0 | +1 | |
| Temperature, X1 (°C) | 40 | 55 | 70 |
| Pressure, X2 (bar) | 100 | 175 | 250 |
| RS–L/CO2, X3 (%) | 20 | 50 | 80 |
| Experiment number | Temperature, X1 (°C) | Pressure, X2 (bar) | RS–L/CO2, X3 (%) | |||
|---|---|---|---|---|---|---|
| 1 | 40 | (−1) | 100 | (−1) | 20 | (−1) |
| 2 | 40 | (−1) | 100 | (−1) | 80 | (+1) |
| 3 | 40 | (−1) | 250 | (+1) | 20 | (−1) |
| 4 | 40 | (−1) | 250 | (+1) | 80 | (+1) |
| 5 | 70 | (+1) | 100 | (−1) | 20 | (−1) |
| 6 | 70 | (+1) | 100 | (−1) | 80 | (+1) |
| 7 | 70 | (+1) | 250 | (+1) | 20 | (−1) |
| 8 | 70 | (+1) | 250 | (+1) | 80 | (+1) |
| 9 | 70 | (+1) | 175 | (0) | 50 | (0) |
| 10 | 70 | (+1) | 175 | (0) | 50 | (0) |
| 11 | 55 | (0) | 100 | (−1) | 50 | (0) |
| 12 | 55 | (0) | 250 | (+1) | 50 | (0) |
| 13 | 55 | (0) | 175 | (0) | 20 | (−1) |
| 14 | 55 | (0) | 175 | (0) | 80 | (+1) |
| 15 (C) | 55 | (0) | 175 | (0) | 50 | (0) |
| 16 (C) | 55 | (0) | 175 | (0) | 50 | (0) |
| 17 (C) | 55 | (0) | 175 | (0) | 50 | (0) |
The extractions were carried out in a supercritical fluid extractor (Thar Technology, Pittsburgh, PA, USA, model SFE-500F-2-C50). Before each extraction, a High Pressure Carbon Dioxide (HPCD) pre-treatment of dried Opuntia spp. fruits was performed. A given weight (4.2–11.1 g) of dried Opuntia spp. fruits were placed in the extraction vessel, which was pre-heated to 55 °C and then pressurized by CO2 until 375 bar for 60 minutes. These conditions were chosen accordingly to Liu et al., 2008.35
At the end of each pre-treatment, pressure was slowly release and a specific volume (84–222 mL) of acidified water pH 5.0 by citric acid (ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, w/v) was added to the extraction vessel which was pre-heated to the required temperature (40–70 °C). Liquid CO2 was fed in the extraction vessel using a TharSFC P-50 high pressure pump (Thar Technology, Pittsburgh, PA, USA). The vessel was pressurized with carbon dioxide until reaching the required pressure (100–250 bar) and held for 30 minutes. After this extraction time, the depressurization was performed by releasing CO2 into the atmosphere. The resulting extracts were kept in a cold, dry and dark environment until further analyses.
20, w/v) was added to the extraction vessel which was pre-heated to the required temperature (40–70 °C). Liquid CO2 was fed in the extraction vessel using a TharSFC P-50 high pressure pump (Thar Technology, Pittsburgh, PA, USA). The vessel was pressurized with carbon dioxide until reaching the required pressure (100–250 bar) and held for 30 minutes. After this extraction time, the depressurization was performed by releasing CO2 into the atmosphere. The resulting extracts were kept in a cold, dry and dark environment until further analyses.
The extraction vessel was filled with about 10 g of dried Opuntia spp. fruits and laboratory glass beads were placed on both endings of the cell, in order to achieve a uniform distribution of the solvent flow. Acidified distilled water pH 5.0 by citric acid was delivered to the extraction vessel using a TharSFC P-50 high pressure pump (Thar Technology, Pittsburgh, PA, USA) until the desired pressure 100 bar. The solvent was preheated on a heat exchanger to a temperature of 40 °C. The pressure on the extraction vessel was maintained by an automated back pressure regulator (TharSFC ABPR, Thar Technology, Pittsburgh, PA, USA), which was located between the extraction vessel and the first fraction collector, with a total solvent flow rate of 10 g min−1. The resulting extract was kept in a cold, dry and dark environment until further analyses.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, w/v), for 30 minutes at 40 °C. The resulting extracts were kept in a cold, dry and dark environment until further analyses.
20, w/v), for 30 minutes at 40 °C. The resulting extracts were kept in a cold, dry and dark environment until further analyses.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) for 30 minutes at room temperature.36 The extract was centrifuged at 5000 RCF (relative centrifugal force) for 10 minutes (MIKRO 220 R, Hettich Zentrifugen, Tuttlingen, Germany). The supernatant was recovered after centrifugation and the residue was extracted again with aqueous methanol until absence of colour was obtained. All extracts were combined and kept in a cold, dry and dark environment until further analyses.
1, v/v) for 30 minutes at room temperature.36 The extract was centrifuged at 5000 RCF (relative centrifugal force) for 10 minutes (MIKRO 220 R, Hettich Zentrifugen, Tuttlingen, Germany). The supernatant was recovered after centrifugation and the residue was extracted again with aqueous methanol until absence of colour was obtained. All extracts were combined and kept in a cold, dry and dark environment until further analyses.Opuntia spp. fruit extracts characterization
| % Betacyanins = ([a/1129] × DF × 100), |
| % Betaxanthins = ([y/750] × DF × 100), |
The total betalain content was expressed as the sum of betacyanin and betaxanthins content. The results were presented as mg of betalains per 100 g of dried fruit, expressed as a mean of triplicates.
Antioxidant activity
The colour strength of all extracts was determined as the absorbance units at the maximum absorption wavelength of a 1% (v/v) solution.19
The colour of Opuntia spp. extract were assessed by CIElab method using a Minolta Colorimeter CR-200 (Osaka, Japan) described using 3 attributes or specific qualities of visual sensation: tonality, luminosity and chromatism.
CIELab colour or space system is based on a sequential or continuous Cartesian representation of 3 orthogonal axes: L*, a* and b*. Coordinate L* represents clarity (L* = 0 black and L* = 100 colourless), a* green/red colour component (a* > 0 red, a* < 0 green) and b* blue/yellow colour component (b* > 0 yellow, b* < 0 blue).
C* is the chroma or colour purity and h° refers to the hue angle of tone and indicates the sample's colour (0° or 360° = red, 90° = yellow, 180° = green, and 270° = blue). C* was determined according to the expression C* = [(a*) + (b*)2]1/2 and h° according to the expression h° = arctan(b*/a*).
The colour parameters were expressed as a mean of triplicates. These values were then converted to RGB (Red, Green, and Blue colour values), using the software OpenRGB (Logicol).
Results and discussion
Aiming at producing a red natural colourant from Opuntia spp. fruits a two-step process was developed. The first step consisted in a High Pressure Carbon Dioxide (HPCD) pre-treatment of dried prickly pears at 375 bar, 55 °C during 60 min. These conditions were chosen accordingly to Liu et al., 2008 for avoiding enzymatic degradation.35 Moreover, this HPCD pre-treatment was applied in order to disrupt the cell vacuoles upon the depressurization of CO2, making betalains more available. In a second step High pressure carbon dioxide assisted-water extraction was explored. RSM was applied aiming at maximizing the extraction yield and stability of betalains. In order to define the extraction media, some previous studies on betalains extraction from Opuntia spp. were taken into account. Castellar et al., 2003 and Sanchez-Gonzalez et al., 2013 concluded that maximum stability of the Opuntia spp. pigments was achieved at pH 5.9,34 Consequently, the water used in all extractions experiments was acidified until pH 5 using citric acid. The resulting extracts were evaluated in terms of betalains yield, phytochemical composition, antioxidant capacity and colour analyses.Modelling of betalain extraction through HPCD assisted-water extraction
The HPCD assisted-water extraction experiments were carried out according to the CCFC previously described. The obtained results, i.e. the betalains yield, total phenolic content, antioxidant activity (ORAC, HORAC and HOSC), colour strength and L* value are shown in Table 3. These results were used to estimate both, linear and quadratic effects of the variables and also their linear interactions. For total phenolic content, ORAC, HOSC, colour strength and L*, a lack of fit of the polynomial models exhibited by low values of R2 and Radj2 was observed.| Experiment number | HPCD assisted-water extraction | Phytochemical composition | Antioxidant activity | Colour properties | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T, °C | P, bar | RS–L/CO2, % | TBC | TPC, mg/100 g dried extract | ORAC, μmol TE/100 g dried extract | HORAC, μmol CAE/100 g dried extract | HOSC, μmol TE/100 g dried extract | Colour strength | L* | a* | b* | C* | h° | RGB colour | ||
| mg/100 g dried Opuntia | mg/100 g dried extract | |||||||||||||||
| a HPCD, High Pressure Carbon Dioxide; HPCDAWE, High Pressure Carbon Dioxide Assisted Water Extraction; PWE, Pressurized Water Extraction; WE, Water Extraction; TBC, Total Betalain Content; TPC, Total Phenolic Content; ORAC, Oxygen radical absorbance capacity; HORAC, Hydroxyl radical adverting capacity; HOSC, Hydroxyl radical scavenging capacity. | ||||||||||||||||
| With HPCD pre-treatment (375 bar, 55 °C, 60 min) | ||||||||||||||||
| 1 | 40 | 100 | 20 | 88.7 ± 0.7 | 190.6 ± 4.4 | 929 ± 22 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 104 ± 886 104 ± 886 |
9423 ± 995 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 828 ± 865 828 ± 865 |
5.03 ± 0.20 | 28.28 ± 0.05 | 62.41 ± 0.09 | 24.61 ± 0.01 | 67.08 | 0.38 |  |
| 2 | 40 | 100 | 80 | 85.0 ± 0.7 | 184.8 ± 4.2 | 930 ± 4 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 697 ± 790 697 ± 790 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 417 ± 751 417 ± 751 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 762 ± 901 762 ± 901 |
3.95 ± 0.08 | 30.26 ± 0.02 | 64.36 ± 0.06 | 20.51 ± 0.04 | 67.57 | 0.31 |  |
| 3 | 40 | 250 | 20 | 79.8 ± 0.6 | 180.2 ± 4.1 | 890 ± 11 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 109 ± 844 109 ± 844 |
9666 ± 810 | 9605 ± 994 | 4.29 ± 0.13 | 32.76 ± 0.08 | 66.23 ± 0.10 | 21.34 ± 0.08 | 69.58 | 0.31 |  |
| 4 | 40 | 250 | 80 | 74.6 ± 0.6 | 179.3 ± 4.1 | 934 ± 8 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 585 ± 1762 585 ± 1762 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 244 ± 670 244 ± 670 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 542 ± 1236 542 ± 1236 |
4.18 ± 0.05 | 31.01 ± 0.04 | 64.57 ± 0.06 | 19.35 ± 0.04 | 67.41 | 0.29 |  |
| 5 | 70 | 100 | 20 | 73.9 ± 0.6 | 175.9 ± 4.0 | 901 ± 39 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 828 ± 1548 828 ± 1548 |
9327 ± 839 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 613 ± 1450 613 ± 1450 |
4.79 ± 0.03 | 30.39 ± 0.03 | 63.22 ± 0.06 | 21.73 ± 0.01 | 66.85 | 0.33 |  |
| 6 | 70 | 100 | 80 | 72.8 ± 0.6 | 175.1 ± 4.0 | 864 ± 4 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 271 ± 1643 271 ± 1643 |
9416 ± 1077 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 250 ± 864 250 ± 864 |
4.38 ± 0.05 | 33.59 ± 0.00 | 67.30 ± 0.10 | 24.20 ± 0.04 | 71.55 | 0.35 |  |
| 7 | 70 | 250 | 20 | 76.2 ± 0.6 | 173.9 ± 4.0 | 962 ± 43 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 653 ± 1325 653 ± 1325 |
9084 ± 1168 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 857 ± 940 857 ± 940 |
4.28 ± 0.13 | 30.32 ± 0.03 | 63.80 ± 0.02 | 26.88 ± 0.03 | 69.24 | 0.40 |  |
| 8 | 70 | 250 | 80 | 81.6 ± 0.7 | 177.5 ± 4.1 | 929 ± 9 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 849 ± 1479 849 ± 1479 |
9558 ± 725 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 372 ± 1027 372 ± 1027 |
4.42 ± 0.06 | 33.96 ± 0.01 | 66.01 ± 0.01 | 23.61 ± 0.01 | 70.11 | 0.34 |  |
| 9 | 40 | 175 | 50 | 79.7 ± 0.6 | 170.5 ± 3.9 | 890 ± 10 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 642 ± 831 642 ± 831 |
8499 ± 897 | 8659 ± 1092 | 4.99 ± 0.10 | 34.76 ± 0.03 | 67.44 ± 0.08 | 19.56 ± 0.02 | 70.21 | 0.28 |  |
| 10 | 70 | 175 | 50 | 77.2 ± 0.6 | 179.3 ± 3.9 | 960 ± 37 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 809 ± 842 809 ± 842 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 003 ± 841 003 ± 841 |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 292 ± 1119 292 ± 1119 |
4.34 ± 0.06 | 33.16 ± 0.03 | 66.57 ± 0.01 | 25.24 ± 0.03 | 71.19 | 0.36 |  |
| 11 | 55 | 100 | 50 | 75.4 ± 0.6 | 167.0 ± 3.8 | 969 ± 49 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 829 ± 926 829 ± 926 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 330 ± 1150 330 ± 1150 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 245 ± 934 245 ± 934 |
4.62 ± 0.05 | 33.72 ± 0.03 | 67.64 ± 0.01 | 20.99 ± 0.04 | 70.82 | 0.30 |  |
| 12 | 55 | 250 | 50 | 76.0 ± 0.6 | 174.8 ± 4.0 | 937 ± 13 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 428 ± 1151 428 ± 1151 |
8576 ± 1062 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 163 ± 807 163 ± 807 |
4.59 ± 0.04 | 30.22 ± 0.04 | 64.93 ± 0.01 | 23.48 ± 0.04 | 69.05 | 0.35 |  |
| 13 | 55 | 175 | 20 | 76.2 ± 0.6 | 173.6 ± 4.0 | 934 ± 39 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 182 ± 839 182 ± 839 |
9108 ± 452 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 401 ± 635 401 ± 635 |
4.85 ± 0.23 | 28.69 ± 0.04 | 62.38 ± 0.04 | 24.75 ± 0.02 | 67.11 | 0.38 |  |
| 14 | 55 | 175 | 80 | 74.8 ± 0.6 | 171.8 ± 3.9 | 872 ± 33 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 862 ± 1054 862 ± 1054 |
8640 ± 688 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 575 ± 1570 575 ± 1570 |
4.28 ± 0.07 | 31.97 ± 0.02 | 64.60 ± 0.10 | 19.99 ± 0.00 | 67.62 | 0.30 |  |
| 15 (C) | 55 | 175 | 50 | 81.1 ± 0.6 | 181.3 ± 4.1 | 933 ± 30 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 220 ± 1212 220 ± 1212 |
7182 ± 814 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 174 ± 882 174 ± 882 |
4.07 ± 0.09 | 32.78 ± 0.02 | 67.35 ± 0.01 | 21.79 ± 0.02 | 70.79 | 0.31 |  |
| 16 (C) | 55 | 175 | 50 | 86.8 ± 0.7 | 187.8 ± 4.3 | 982 ± 23 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 476 ± 1460 476 ± 1460 |
8203 ± 616 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 689 ± 848 689 ± 848 |
3.71 ± 0.11 | 30.81 ± 0.04 | 65.17 ± 0.08 | 22.82 ± 0.08 | 69.04 | 0.34 |  |
| 17 (C) | 55 | 175 | 50 | 85.1 ± 0.7 | 184.6 ± 4.2 | 929 ± 31 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 209 ± 1466 209 ± 1466 |
7766 ± 873 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 248 ± 1440 248 ± 1440 |
4.51 ± 0.12 | 30.52 ± 0.02 | 64.93 ± 0.04 | 24.55 ± 0.04 | 69.41 | 0.36 |  |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Without HPCD pre-treatment | ||||||||||||||||
| HPCDAWE | 40 | 100 | 20 | 64.6 ± 0.3 | 175.6 ± 0.8 | 742 ± 14 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 447 ± 1323 447 ± 1323 |
8159 ± 838 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 848 ± 1019 848 ± 1019 |
3.77 ± 0.03 | 53.30 ± 0.04 | 80.55 ± 0.02 | 10.00 ± 0.06 | 81.16 | 0.12 |  |
| PWE | 40 | 100 | 30.4 ± 1.2 | 164.2 ± 6.3 | 787 ± 22 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 995 ± 1417 995 ± 1417 |
6834 ± 751 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 164 ± 1078 164 ± 1078 |
1.67 ± 0.07 | 41.67 ± 0.02 | 75.74 ± 0.06 | 5.29 ± 0.05 | 75.92 | 0.07 |  |
|
| WE | 40 | 1 | 45.4 ± 2.0 | 146.6 ± 4.9 | 818 ± 3 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 205 ± 1050 205 ± 1050 |
7611 ± 709 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 042 ± 1306 042 ± 1306 |
2.06 ± 0.03 | 36.33 ± 0.04 | 59.37 ± 0.02 | 15.25 ± 0.05 | 61.29 | 0.25 |  |
|
The response surfaces (Fig. 1) fitted to betalains yield can be described by second-order polynomial models as a function of pressure, temperature and RS–L/CO2 (Table 4). In these response surface models, the significant effects p < 0.05 and those having confidence range smaller than the value of the effect, or smaller than the standard deviation (data not shown), were included in the model equations of these surfaces. It is better to accept factor with values higher than 0.05 rather than to take the chance of missing an important factor.44 The good values for both R2 and Radj2 of these models (Table 4) suggest a close agreement between the experimental data and the theoretical values predicted by the model. About 82% or 74% of the observed results concerning the betalains yield and HORAC are explained by the present model (see ESI†). However, no optimum conditions were observed in the response surface for the betalains extraction. Therefore, only the identification of the region corresponding to the best response can be achieved.
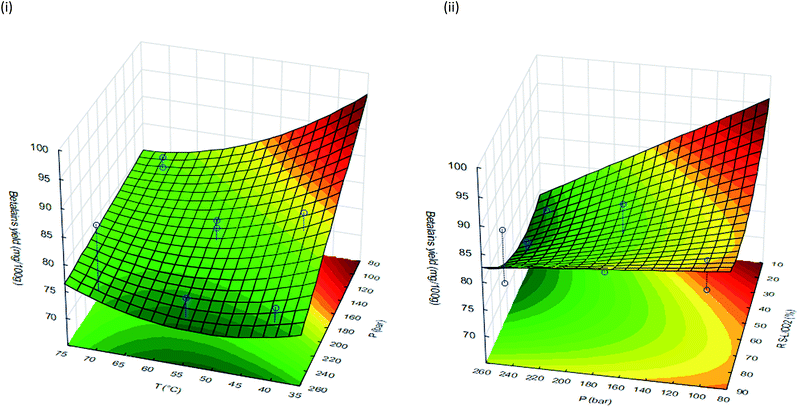 | ||
| Fig. 1 Response surfaces fitted to the betalains yield as a function of (i) pressure and temperature and (ii) pressure and RS–L/CO2. | ||
| Polynomial model equations | R2 | Radj2 |
|---|---|---|
| BY = 163.6 − 0.237A − 1.701B + 0.009B2 − 0.634C + 0.003C2 + 0.003AB + 0.001AC + 0.003BC | 0.817 | 0.581 |
| HORAC = 8964 + 0.306B2 − 40.57C − 0.891C2 − 0.204AB + 0.232AC | 0.736 | 0.617 |
The analysis of the data showed that the recovery of betalains was negative affected (p < 0.05) by the extraction pressure within the tested range (100–250 bar). Accordingly, the lower pressure tested (100 bar) led to an increase in betalains yield. This effect was more pronounced at lower temperatures (40 °C) and lower RS–L/CO2 (20%).
In addition, the recovery of betalains was significant affect (p < 0.05) by some interactions between factors, pressure with temperature and pressure with RS–L/CO2 (Fig. 1). With simultaneous increase of pressure and temperature values, the betalains yield decreased. This result may be explained by the negative impact of higher pressure and temperature on the pigments stability during the extraction process. This effect was also reported by Santos & Meireles, 2011 for other pigment.6 When the pressure and RS–L/CO2 values decreased the recovery of betalains increased. This effect can be explained by the increase of the volume of pressurized CO2 (lower RS–L/CO2), which possibly plays a crucial role during the betalains extraction.
The best response can be achieved at 100 bar, 40 °C and 20% volume ratio of (solid–liquid mixture)/(pressurized CO2). Under these conditions, the betalains yield was 89 ± 0.7 mg per 100 g of dried fruit.
The repeatability (coefficient of variation) of the extraction process through HPCD assisted-water extraction was 4%, taking into account three experiments of the design (center points).
Available literature on Opuntia spp. fruits illustrates a high variation of betalains content. The total pigment content depends on the respective species and clone investigated and may range from 5–110 mg/100 g.9,14,34 Our results agree with those findings reported in literature (17.8 mg/100 g fresh fruit).
The effect of high pressure carbon dioxide on the two-step extraction process
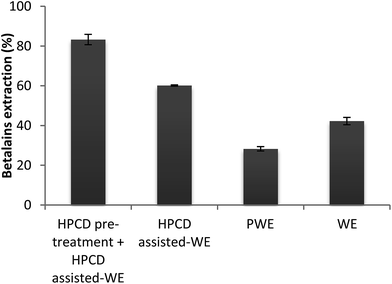
The effect of HPCD on betalains extraction was also studied throughout the HPCD pre-treatment and HPCD assisted-water extraction steps. A comparison of extraction percentage of betalains obtained with two-step extraction process, HPCD assisted-water extraction without pre-treatment, pressurized water extraction (PWE) and water extraction (WE) with respect to the maximum extractable pigment (107 ± 2.12 mg of betalains/100 g dried fruit) is shown in Fig. 2.The greatest extraction percentage of betalains was obtained with two-step extraction process, with extraction percentage of 83% of the maximum extractable pigments from Opuntia spp. fruits (Fig. 2). The amount of pigments extracted by HPCD assisted-water extraction, PWE and WE were lower (28–60%). The promising effect of the use of pressurized CO2 for betalains extraction seems irrefutable when the results obtained using this technique were compared to that obtained using other extraction methods. Comparing WE with PWE and HPCD assisted-water extraction at same extraction conditions (temperature, solvent and ratio S–L) it was possible to conclude that the last was more efficient in extracting betalains from Opuntia spp. fruits. In addition, the HPCD pre-treatment before the HPCD assisted-water extraction has also enhanced the extraction efficiency.
According to Santos & Meireles, 2011 and Xu et al., 2010, there are five possible forms associated with CO2 in a HPCD assisted-water extraction, including supercritical CO2, carbonic acid and its dissociated products (H+, HCO3− and CO32−). These different forms might play different roles in the HPCD assisted-water extraction of betalains. Firstly, supercritical CO2 combines high diffusivity of gas with solvent strength of liquids with non-polar and lipophilic properties to dissolve phospholipid layer of cell membranes, improving the penetration of water into the cellular matrix and efflux of betalains from cell vacuoles to the outside of the cell. Secondly, the generation of in situ carbonic acid, when the CO2 is added into HPCD assisted-water extraction system, decrease pH. This leads to a positive impact on betalains extraction and stability from Opuntia spp. fruits. Finally, the explosive effect during the rapid CO2 depressurization causes disruption of cell vacuoles, making betalains more available, thus enhancing extraction efficiency.6,32
In addition, the effect of pressurized carbon dioxide has been reported to inactivate microorganisms and inhibit enzyme activity (polyphenoloxidase and peroxidase), which are responsible for betalains degradation.45 Furthermore, the absence of oxygen in the extraction system is other advantage of this methodology because the presence of oxygen is one of the factors that affect betalains stability.6,32 Overall, these advantages have contributed for the higher yields of betalains using HPCD.
Opuntia spp. fruit extracts characterization
Regarding the total phenolic content, the values varied between 864 ± 4.13 and 982 ± 23.0 mg GAE/100 g dried extract. Among all extracts the ORAC, HORAC and HOSC varied between 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 209 ± 926 and 12
209 ± 926 and 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 862 ± 1212 μmol TE/100 g dried extract, 7182 ± 688 and 11
862 ± 1212 μmol TE/100 g dried extract, 7182 ± 688 and 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 417 ± 873 μmol CAE/100 g dried extract, and 8659 ± 901 and 15
417 ± 873 μmol CAE/100 g dried extract, and 8659 ± 901 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 762 ± 1440 μmol TE/100 g dried extract.
762 ± 1440 μmol TE/100 g dried extract.
The antioxidant activity results are in line with that has been obtained by other authors. Several works have demonstrated the potent antiradical scavenging activity of betalains in vitro.13,15,26,46 It was shown that betacyanins i.e. betanin acts as a scavenger of reactive oxygen species (DPPH-, galvinoxyl-, superoxide- and hydroxyl radicals).16 Furthermore, other authors have proved that betalains prevent active oxygen-induced and free radical-mediated oxidation of biological molecules.12,15,17,18
Firstly, a spectrophotometric study for all extracts was performed by normalizing to an absorbance of 0.70 ± 0.05 at 535 nm, the betanin λmax value (see ESI†). It can be seen that Opuntia spp. fruit extract showed single maximum wavelengths at 535 nm and presented a symmetrical spectrum. These results indicated that Opuntia spp. fruit extract spectrum is characteristic of a single-composition (betanin/isobetanin).19
Opuntia spp. fruits only contain the red pigments betanin and isobetanin, which are the compounds present in the additive, red beet (E-162). These characteristics make cactus pear a promising source of betacyanin pigments, which would be suitable for food applications.
The colour parameters of the Opuntia spp. extracts was measured using CIELab method. The results are presented in Table 3. From the results, lightness ranged from the L* = 28.7 (best response) to the L* = 34.8. All the samples had positive a* values, as expected from their red colour ranging from a* = 62.4 to a* = 67.6. It was observed positive values of b* parameter (blueness–yellowness) ranging from b* = 19.3 to b* = 26.9. This means that all samples had a more yellow colour than blue. Chroma, which expresses the brilliance or purity of a colour, was similar in all extracts. The hue angle, which indicates the tonality, ranged between 0.28 and 0.40, as predictable from their red colour.
Finally, colour properties of the selected Opuntia spp. fruit extract were compared with those of a commercial red food colourant (Table 5).
| Colour strength | Colour parameters | |||||||
|---|---|---|---|---|---|---|---|---|
| L* | a* | b* | C* | h° | ΔE* | RGB colour | ||
| Opuntia spp. fruit extract | 5.0 ± 0.2 | 28.7 ± 0.0 | 62.4 ± 0.1 | 24.6 ± 0.0 | 67.1 | 0.38 | 0.0 |  |
| Red beet concentrate19 | 5.2 ± 0.1 | 69.5 ± 0.1 | 57.9 ± 0.2 | −1.0 ± 0.0 | 57.9 | 359 | 8.4 |  |
Red beet pigment has been extensively commercialized as a food colourant. E-162 and 73.40 are the commercial codes for red beet pigment in Europe and U.S.A., respectively.
According to the results, the Opuntia spp. extract presented high colour strength like the commercial form. The total colour difference value (ΔE*) was 8.4, more than 5 units of difference indicate that human eye is capable of distinguishing Opuntia spp. fruit extract from the red beet concentrate. Opuntia spp. fruit extract had the lowest value of lightness. Both had positive a* values due to their red colour. Greater dispersion was observed in the b* parameter, the Opuntia spp. fruit extract presented more yellowness colour than blueness. The brilliance or purity of a colour was highest in Opuntia spp. fruit extract and, according to its hue value, was the reddest colourant. Therefore, Opuntia spp. fruit extract, with its pleasant flavour, could be considered as a promising natural betalain food colourant.
Conclusions
A novel high pressure CO2-assisted process was proposed in this work to promote red pigment recovery from Opuntia spp. fruits. The best operating conditions to obtained betalains-rich extracts were achieved at 100 bar, 40 °C and 20% RS–L/CO2, after high pressure CO2 pre-treatment at 375 bar, 55 °C and 60 min. Under these optimized conditions, the betalain extraction yield was 2-fold increased when comparing to conventional water extraction. This process provided betalain-rich extract with a vivid red colour and high colour strength and significant antioxidant activity (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 862 ± 1212 μmol TE/100 g, 11
862 ± 1212 μmol TE/100 g, 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 417 ± 873 μmol CAE/100 g and 15
417 ± 873 μmol CAE/100 g and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 762 ± 1440 μmol TE/100 g dried extract, for ORAC, HORAC and HOSC, respectively). Consequently, the Opuntia spp. fruit extract described in this study represents a natural red pigment alternative to red beet extracts with intensive reddish colour and improved sensory properties, without geosmin and pyrazines.
762 ± 1440 μmol TE/100 g dried extract, for ORAC, HORAC and HOSC, respectively). Consequently, the Opuntia spp. fruit extract described in this study represents a natural red pigment alternative to red beet extracts with intensive reddish colour and improved sensory properties, without geosmin and pyrazines.
Acknowledgements
The authors acknowledge the financial support received from Portuguese Fundação para a Ciência e Tecnologia (FCT) through PTDC/AGR-AAM/099645/2008 project, PEst-OE/EQB/LA0004/2011 grant and for the doctoral (SFRH/BDE/51856/2012) fellowship. The authors are grateful to Eng. Armando Ferreira for the use of Minolta Colorimeter CR-200 at INIAV (Instituto Nacional de Investigação Agrária e Veterinária).References
- C. Saldanha do Carmo, A. N. Nunes, A. T. Serra, S. Ferreira-Dias, I. Nogueira and C. M. M. Duarte, Green Chem., 2015, 17, 1510–1518 RSC.
- H. M. C. Azeredo, Int. J. Food Sci. Technol., 2009, 44, 2365–2376 CrossRef CAS PubMed.
- H.-O. Boo, S.-J. Hwang, C.-S. Bae, S.-H. Park, B.-G. Heo and S. Gorinstein, Ind. Crops Prod., 2012, 40, 129–135 CrossRef CAS PubMed.
- D. M. Jiménez-Aguilar, A. E. Ortega-Regules, J. D. Lozada-Ramírez, M. C. I. Pérez-Pérez, E. J. Vernon-Carter and J. Welti-Chanes, J. Food Compos. Anal., 2011, 24, 889–894 CrossRef PubMed.
- J. Prakash Maran, S. Manikandan and V. Mekala, Ind. Crops Prod., 2013, 49, 304–311 CrossRef CAS PubMed.
- D. T. Santos and M. A. A. Meireles, Innovative Food Sci. Emerging Technol., 2011, 12, 398–406 CrossRef CAS PubMed.
- M. Shahid, S. U. Islam and F. Mohammad, J. Cleaner Prod., 2013, 53, 310–331 CrossRef CAS PubMed.
- M. E. Borges, R. L. Tejera, L. Díaz, P. Esparza and E. Ibáñez, Food Chem., 2012, 132, 1855–1860 CrossRef CAS PubMed.
- R. Castellar, J. M. Obón, M. Alacid and J. A. Fernández-López, J. Agric. Food Chem., 2003, 51, 2772–2776 CrossRef CAS PubMed.
- F. Delgado-Vargas, A. R. Jiménez and O. Paredes-López, Natural pigments: carotenoids, anthocyanins, and betalains–characteristics, biosynthesis, processing, and stability, 2000, vol. 40 Search PubMed.
- D. A. Moreno, C. García-Viguera, J. I. Gil and A. Gil-Izquierdo, Phytochem. Rev., 2008, 7, 261–280 CrossRef CAS.
- M. Allegra, P. G. Furtmüller, W. Jantschko, M. Zederbauer, L. Tesoriere, M. a. Livrea and C. Obinger, Biochem. Biophys. Res. Commun., 2005, 332, 837–844 CrossRef CAS PubMed.
- Y. Cai, M. Sun and H. Corke, J. Agric. Food Chem., 2003, 51, 2288–2294 CrossRef CAS PubMed.
- F. C. Stintzing and R. Carle, Mol. Nutr. Food Res., 2005, 49, 175–194 CAS.
- L. Tesoriere, D. Butera, M. Allegra, M. Fazzari and M. A. Livrea, J. Agric. Food Chem., 2005, 53, 1266–1270 CrossRef CAS PubMed.
- T. Esatbeyoglu, A. E. Wagner, R. Motafakkerazad, Y. Nakajima, S. Matsugo and G. Rimbach, Food Chem. Toxicol., 2014, 73, 119–126 CrossRef CAS PubMed.
- J. Kanner, S. Harel and R. Granit, J. Agric. Food Chem., 2001, 49, 5178–5185 CrossRef CAS PubMed.
- L. Tesoriere, D. Butera, D. D'Arpa, F. Di Gaudio, M. Allegra, C. Gentile and M. A. Livrea, Free Radical Res., 2003, 37, 689–696 CrossRef CAS PubMed.
- R. Castellar, J. M. Obón and J. A. Fernández-López, J. Sci. Food Agric., 2006, 86, 122–128 CrossRef PubMed.
- F. Gandía-Herrero, J. Cabanes, J. Escribano, F. García-Carmona and M. Jiménez-Atiénzar, J. Agric. Food Chem., 2013, 61, 4294–4302 CrossRef PubMed.
- F. Gandía-Herrero, M. Jiménez-Atiénzar, J. Cabanes, F. García-Carmona and J. Escribano, J. Agric. Food Chem., 2010, 58, 10646–10652 CrossRef PubMed.
- J. P. Maran and S. Manikandan, Dyes Pigm., 2012, 95, 465–472 CrossRef PubMed.
- J. M. Obón, M. R. Castellar, M. Alacid and J. A. Fernández-López, J. Food Eng., 2009, 90, 471–479 CrossRef PubMed.
- C. Vergara, J. Saavedra, C. Sáenz, P. García and P. Robert, Food Chem., 2014, 157, 246–251 CrossRef CAS PubMed.
- C. Saénz, S. Tapia, J. Chávez and P. Robert, Food Chem., 2009, 114, 616–622 CrossRef PubMed.
- D. Butera, L. Tesoriere, F. Di Gaudio, A. Bongiorno, M. Allegra, A. M. Pintaudi, R. Kohen and M. A. Livrea, J. Agric. Food Chem., 2002, 50, 6895–6901 CrossRef CAS PubMed.
- H. M. C. de Azeredo, A. C. Pereira, A. C. R. de Souza, S. T. Gouveia and K. C. B. Mendes, Int. J. Food Sci. Technol., 2009, 44, 2464–2469 CrossRef PubMed.
- G. J. Swamy, A. Sangamithra and V. Chandrasekar, Dyes Pigm., 2014, 111, 64–74 CrossRef CAS PubMed.
- J. Azmir, I. S. M. Zaidul, M. M. Rahman, K. M. Sharif, A. Mohamed, F. Sahena, M. H. A. Jahurul, K. Ghafoor, N. A. N. Norulaini and A. K. M. Omar, J. Food Eng., 2013, 117, 426–436 CrossRef CAS PubMed.
- M. Herrero, A. Cifuentes and E. Ibañez, Food Chem., 2006, 98, 136–148 CrossRef CAS PubMed.
- H. W. Huang, C. P. Hsu, B. B. Yang and C. Y. Wang, Trends Food Sci. Technol., 2013, 33, 54–62 CrossRef CAS PubMed.
- Z. Xu, J. Wu, Y. Zhang, X. Hu, X. Liao and Z. Wang, Bioresour. Technol., 2010, 101, 7151–7157 CrossRef CAS PubMed.
- K. J. Gupta, H. Rolletschek, B. Neelwarne, N. P. Shetty, K. Jørgensen and H. J. L. Jørgensen, Red Beet Biotechnology: Food and Pharmaceutical Applications, 2012, pp. 91–104 Search PubMed.
- N. Sanchez-Gonzalez, M. R. Jaime-Fonseca, E. San Martin-Martinez and L. G. Zepeda, J. Agric. Food Chem., 2013, 61, 11995–12004 CrossRef CAS PubMed.
- X. Liu, Y. Gao, X. Peng, B. Yang, H. Xu and J. Zhao, Innovative Food Sci. Emerging Technol., 2008, 9, 24–31 CrossRef CAS PubMed.
- S. H. Guzmán-Maldonado, A. L. Morales-Montelongo, C. Mondragón-Jacobo, G. Herrera-Hernández, F. Guevara-Lara and R. Reynoso-Camacho, J. Food Sci., 2010, 75, 485–492 CrossRef PubMed.
- V. L. Singleton and J. A. Rossi, Am. J. Enol. Vitic., 1965, 16, 144–158 CAS.
- A. T. Serra, A. A. Matias, A. V. M. Nunes, M. C. Leitão, D. Brito, R. Bronze, S. Silva, A. Pires, M. T. Crespo, M. V. San Romão and C. M. Duarte, Innovative Food Sci. Emerging Technol., 2008, 9, 311–319 CrossRef CAS PubMed.
- D. Huang, B. Ou, M. Hampsch-Woodill, J. A. Flanagan and R. L. Prior, J. Agric. Food Chem., 2002, 50, 4437–4444 CrossRef CAS PubMed.
- R. P. Feliciano, M. N. Bravo, M. M. Pires, A. T. Serra, C. M. Duarte, L. V. Boas and M. R. Bronze, Food Anal. Methods, 2009, 2, 149–161 CrossRef.
- B. W. Bolling, Y. Y. Chen, A. G. Kamil and C. Y. O. Chen, J. Food Sci., 2012, 77, 69–75 CrossRef PubMed.
- B. Ou, M. Hampsch-Woodill, J. Flanagan, E. K. Deemer, R. L. Prior and D. Huang, J. Agric. Food Chem., 2002, 50, 2772–2777 CrossRef CAS PubMed.
- J. Moore, J. J. Yin and L. Yu, J. Agric. Food Chem., 2006, 54, 617–626 CrossRef CAS PubMed.
- P. D. Haaland, Experimental design in biotechnology, CRC Press, 1989 Search PubMed.
- X. Liu, Y. Gao, H. Xu, Q. Hao, G. Liu and Q. Wang, Food Chem., 2010, 119, 108–113 CrossRef CAS PubMed.
- F. C. Stintzing, K. M. Herbach, M. R. Mosshammer, R. Carle, W. Yi, S. Sellappan, C. C. Akoh, R. Bunch and P. Felker, J. Agric. Food Chem., 2005, 53, 442–451 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra14998c |
| This journal is © The Royal Society of Chemistry 2015 |