Bio-polyurethanes from Sapium sebiferum oil reinforced with carbon nanotubes: synthesis, characterization and properties
Guiying Wu,
Yanli Fan,
Xin He and
Yunjun Yan*
Key Laboratory of Molecular Biophysics of the Ministry of Education, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan 430074, China. E-mail: yanyunjun@hust.edu.cn; Fax: +86-27-87792213; Tel: +86-27-87792213
First published on 17th September 2015
Abstract
The physicochemical properties and fatty acid profile of natural Sapium sebiferum oil (SSO) were analyzed and found to have a high iodine value of 186.8 g/100 g. Then, a novel SSO-based polyurethane (PU) was successfully synthesized via an in situ polymerization method, and multi-walled carbon nanotubes (CNTs) were modified by an acid mixture and ethylenediamine (EDA) to further reinforce the PU matrix. The microstructure and the properties of the functionalized CNTs and the PU/CNT composites were characterized by Fourier transform infrared (FTIR), transmission electron microscopy (TEM), thermogravimetric analysis (TGA), scanning electron microscopy (SEM) and differential scanning calorimetry (DSC). The results showed that amino-functionalized CNTs could homogeneously disperse in the PU matrix and significantly improved the thermal and mechanical properties of the composite. Compared with pure PU, the glass transition temperature and initial decomposition temperature of the PU matrix with amino-functionalized CNTs were respectively enhanced by 17.1 and 32.5 °C. Meanwhile, 295% improvement in the tensile strength, 111.3% enhancement in the Young's modulus and 22.4% increment in the elongation at break of the composite were observed. The properties of the PU/CNT composites indicate the great potential applications of SSO in polymer materials.
1. Introduction
Nowadays, polyurethanes (PUs) are one of the most important polymer materials, which have been widely used in construction, packaging, insulation, upholstery, footwear, machinery industries and medical devices due to their versatile properties.1 Depending on the polyols and isocyanates used and the polymerization method, the chemistry of PUs is suitable for different types of synthetic materials such as thermoplastics and thermosets, flexible and rigid foams, as well as interpenetrating polymer networks.2 However, conventional PUs are usually synthesized with unrenewable petroleum-based polyols, which consume much energy and result in big environmental pollution.3 Thus, for the requirements of environment friendly and sustainability in the chemical industry, it is urgent to utilize renewable resources for polymer synthesis.4 In recent years, much attention has been paid to the synthesis of PUs from natural resources such as plant oils because of their competitive cost, global abundance, biological degradation and special built-in functionality (ester functions and insaturations).5 However, plant-oil-based PUs are mostly prepared from food oils, which are severely limited in supply because of their contradiction to the large demand for food by a growing world population.Sapium sebiferum, commonly referred to as Chinese tallow tree, is one of the most economical woody oil plants. It is widely distributed in China, Japan, India and the southern coastal United States, and it can be grown on alkaline, saline, droughty, and acidic soils.6 It grows rapidly, reaching maturity within 3–4 years and it generates economic yields during its productive life span of 70 to 100 years. Its fruit production is heavy, averaging around 4–10 tons of seeds per hectare every year. Furthermore, the fruits can yield a drying oil from the kernels (13–32 wt% per kernel) with high iodine value.7 As the oil is inedible because of some toxic constituents,8 it does not compete with human food and farming land. Therefore, Sapium sebiferum oil (SSO) is one of the most suitable platform feedstock for the production of various polymers such as PUs.
As widely reported in the literature,9–12 carbon nanotubes (CNTs) are commonly used experimentally as reinforcing fillers for composites because of their remarkable low density, excellent mechanical strength, thermal stability and thermal conductivity. However, CNTs are very difficult to disperse uniformly in a polymer matrix, because of insufficient interacts in the interface between the polymer and CNTs, resulting in mobility of the CNTs in the matrix.13 Modification of CNTs is necessary to enable their high potential application in polymer composites. In the present study, we introduced carboxylic acid groups onto the surface of CNTs via an oxidation process. Ethylenediamine (EDA) was allowed to undergo a coupling reaction with carboxylic acid groups to introduce amino groups, which can directly react with the isocyanato of the PUs. Therefore, EDA was used as an excellent cross-linker to prepare the high-quality PU/CNT composites.
Experiments were carried out to use the raw material SSO to prepare PU/CNT composites. The main objectives of the study were (1) to evaluate the physicochemical properties and fatty acids profiles of SSO to determine whether the use of SSO as raw material for PU preparation is feasible, (2) to synthesize the high-quality SSO-based PUs, and (3) to utilize CNTs as reinforcing reagent in the matrix to improve the properties of PUs.
2. Materials and methods
2.1. Materials
SSO was purchased from a refinery factory of Dawu County (Hubei province, China). Multi-walled CNTs (Shenzhen Nanotech Port Co., Ltd) had an average length of 5–15 μm, a diameter of 40–60 nm and a specific surface area of 40–70 m2 g−1. Isophorone diisocyanate (IPDI), N,N′-dicyclohexylcarbodiimide (DCC) and EDA were bought from Aladdin Chemistry Ltd Co. (China). Peracetic acid, acetic acid, methyl alcohol, fluoboric acid, chloroform, toluene, ethanol, acetone, concentrated nitric acid (HNO3, 70%), and concentrated sulfuric acid (H2SO4, 98%) were obtained from Sinopharm Chemical Reagent Ltd Co. (Shanghai, China). Pristine CNTs and polyols prepared from SSO were dried at 60 °C for 4 h under vacuum to remove moisture.2.2. Synthesis methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio by volume). Firstly, 1 g of pristine CNTs was added to 100 mL of the acid mixture in a conical flask. This mixture was then sonicated for 4 h at ambient temperature in an ultrasonic bath to introduce carboxylic acid groups on the CNT surface, and washed with distilled water on polycarbonate filter paper with 0.45 μm pore size until the washings displayed no acidity. The product was dried in a vacuum oven at 80 °C for 4 h. The acid-treated CNTs (CNT-COOH) powders were dispersed in the solvent EDA and refluxed with DCC catalyst at 120 °C for 48 h to introduce amino groups (see Scheme 1).15 The reaction mixture was then diluted with methanol and filtered through size polycarbonate filter paper with 0.45 μm pore, and the residue was washed thoroughly with excess methanol. Finally, the amino-functionalized CNT (CNT-NH2) powders were dried at 80 °C for 4 h in a vacuum oven.
1 ratio by volume). Firstly, 1 g of pristine CNTs was added to 100 mL of the acid mixture in a conical flask. This mixture was then sonicated for 4 h at ambient temperature in an ultrasonic bath to introduce carboxylic acid groups on the CNT surface, and washed with distilled water on polycarbonate filter paper with 0.45 μm pore size until the washings displayed no acidity. The product was dried in a vacuum oven at 80 °C for 4 h. The acid-treated CNTs (CNT-COOH) powders were dispersed in the solvent EDA and refluxed with DCC catalyst at 120 °C for 48 h to introduce amino groups (see Scheme 1).15 The reaction mixture was then diluted with methanol and filtered through size polycarbonate filter paper with 0.45 μm pore, and the residue was washed thoroughly with excess methanol. Finally, the amino-functionalized CNT (CNT-NH2) powders were dried at 80 °C for 4 h in a vacuum oven.
The PU/CNT composites were prepared via the reaction of IPDI and polyol-219 with 2 wt% CNTs using previously reported methods, as shown in Scheme 2.16 CNTs and acetone were adequately mixed by sonication for 30 min. The mixture and IPDI were allowed to react under a magnetic stirring in a N2 atmosphere in a water-cooled condenser at 80 °C for 2 h. The solution was then cooled to 60 °C and polyol-219 was added (1.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mole ratio of –NCO to –OH groups). After reaction for 2 h, the product was degassed under vacuum for 10 min, poured onto a sheet of release paper, and then heated in an oven at 60 °C for 8 h. The PU/CNT composites prepared with pristine CNTs, CNT-COOH, and CNT-NH2 are designated as PU/CNT-P, PU/CNT-C and PU/CNT-N, respectively.
1 mole ratio of –NCO to –OH groups). After reaction for 2 h, the product was degassed under vacuum for 10 min, poured onto a sheet of release paper, and then heated in an oven at 60 °C for 8 h. The PU/CNT composites prepared with pristine CNTs, CNT-COOH, and CNT-NH2 are designated as PU/CNT-P, PU/CNT-C and PU/CNT-N, respectively.
2.3. Testing and measurement
Physicochemical properties of SSO were analyzed through the following methods. The measurement of moisture content, peroxide value, iodine value, acid value, saponification value, phospholipids content, viscosity and specific gravity were conducted according to the National Standards for Testing Vegetable Oil of China (GB/T 9696-2008, 5538-2005, 5532-2008, 5530-2005, 5534-2008, 5537-2008, 265-88, and 1884-2000, respectively).The composition of fatty acids in SSO was analyzed on a GC-9790 gas chromatography equipped with a 19091N-133 innowax capillary column (30 m × 0.25 mm × 0.1 μm, Agilent Technologies Inc., USA). The injector and the detector temperatures were set at 240 and 280 °C, respectively. Methyl esters were prepared by the reaction of about 0.1 mL oil and 1 mL NaOMe (1 M) and then centrifuged (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 10 min). The sample for analysis was gained by mixing 10 μL of the upper layer with 290 μL of n-hexane and 300 μL of standard solution.
000 rpm, 10 min). The sample for analysis was gained by mixing 10 μL of the upper layer with 290 μL of n-hexane and 300 μL of standard solution.
To examine the significant absorption bands of the CNTs, PU and PU/CNT composites, Fourier transform infrared (FTIR) spectra of the samples in the wavenumber range of 4000–400 cm−1 were obtained at a resolution of 4 cm−1 on Bruker Vertex70 FTIR spectrophotometer at room temperature.
The elemental compositions on the surface of the CNTs were measured by X-ray photoelectron spectroscopy (XPS) (AXIS-ULTRA DLD-600W, Japan). A monochromatic Al Kα X-ray was used as the incident radiation.
Morphological studies of the CNTs were carried out by transmission electron microscopy (TEM) (Tecnai G2 20 FEI instrument, Holland). The samples were diluted in ethanol and well dispersed by sonification for 15 min. A drop of the sample solution was then placed on a copper grid by using a micropipette and dried before observation.
To investigate the thermal stability of the CNTs, PU, and PU/CNT composites, thermogravimetric analysis (TGA) was performed with the aid of a Pyris1 TGA instrument. The samples were subjected to 50 to 600 °C at a heating rate of 10 °C min−1 in a nitrogen atmosphere. The weight of the measured samples was about 5 mg.
The morphology of the pure PU and PU/CNT composites was observed by scanning electron microscopy (SEM) (Nova NanoSEM 450). Surfaces of the composites were freeze-fractured in liquid nitrogen, and all specimens were coated with platinum before SEM observation.
The glass transition temperatures (Tgs) of the pure PU and PU/CNT composites were determined by differential scanning calorimetry (Diamond DSC) from −10 to 150 °C at a heating rate of 10 °C min−1 under nitrogen flow.
Mechanical properties of the PU and PU/CNT films (100 mm × 10 mm × 1 mm) were determined on a CMT4104 universal testing machine (Shenzhen SANS Testing Machine Ltd Co., Shenzhen, China) at a speed of 50 mm min−1. For each composite, five specimens were tested, and the average value was obtained.
3. Results and discussion
3.1. Physicochemical properties and fatty acid profile of SSO
Data collected from the study of the physicochemical properties of SSO are shown in Table 1. The oil content (45%–60%) of S. sebiferum fruit is higher than that of linseed, soybean and palm kernel (33.3%, 18.4%, and 44.6%, respectively).17 The experimental result shows that SSO has low acid value (1.36 mg KOH per g) and moisture contents (0.12%), which do not affect the quality of synthetic PUs. The iodine value and peroxide value of the oil are relatively high, reflecting its high content of polyunsaturated fatty acid, a low resistance to oxidation, and short shelf life. Saponification value of the oil is 201.28 mg KOH per g, which indicates that the oil consists of normal triglycerides, and is suitable for the production of liquid soap and shampoo industries. The dynamic viscosity of the oil at 25 °C is 47.87 mPa s, indicating good fluidity. The tested physicochemical properties of SSO are close to those of soybean oil, suggesting that it can be used as a feedstock for polyol production.| Properties | Sapium sebiferum oil | Soybean oil |
|---|---|---|
| Specific gravity (g cm−3, 25 °C) | 0.92 ± 0.03 | 0.86 |
| Moisture content (%) | 0.12 ± 0.01 | 0.14 |
| Viscosity (mPa s, 25 °C) | 47.87 ± 0.37 | — |
| Acid value (mg KOH per g) | 1.36 ± 0.04 | 0.25 |
| Saponification value (mg KOH per g) | 201.28 ± 1.26 | 184.20 |
| Peroxide value (meq kg−1) | 4.17 ± 0.08 | — |
| Iodine value (g/100 g) | 186.75 ± 0.94 | 127.00 |
| Molecular weight | 841.99 ± 5.87 | 914.80 |
The fatty acid profile of SSO (Table 2) was compared with those of other seven oils, such as those of soybean oil, sunflower oil, jatropha oil, linseed oil, castor oil, tung oil and palm oil.7,16,17 The major fatty acids in SSO are palmitic, stearic, oleic, linoleic and linolenic acid. The highest percentage is composed of linolenic acid (42.7%), followed by linoleic acid (29.8%) and oleic acid (14.2%). Thus, SSO can be classified as linolenic–linoleic oil. It has a higher iodine value compared with those of other seven plant oils (see Table 2). The iodine value of the oil is 186.8 g/100 g, reflecting its high unsaturation and polyunsaturated fatty acid content. The number of double bonds per molecule attains 6.6, much higher than those of castor oil (3.0) and soybean oil (4.5). This results in a high hydroxyl value of the synthesized polyols.18 All of these results indicate that SSO, an inedible vegetable oil, is suitable for polymer synthesis.
| Name | Palmitic | Stearic | Oleic | Linoleic | Linolenic | Iodine value (g of I2/100 g) |
|---|---|---|---|---|---|---|
| Soybean oil | 14.0 | 4.0 | 23.3 | 52.2 | 5.6 | 128.7 |
| Sunflower oil | 6.5 | 2.0 | 45.4 | 46.0 | 0.1 | 120.2 |
| Jatropha oil | 13.0 | 8.4 | 42.5 | 33.0 | 3.2 | 103.6 |
| Linseed oil | 5.0 | 4.0 | 22.0 | 17.0 | 52.0 | 180.0 |
| Castor oil | 1.5 | 0.5 | 5.0 | 4.0 | 0.5 | 102.2 |
| Tung oil | 1.3 | 1.3 | 7.0 | 5.5 | 0.3 | 165 |
| Palm oil | 41.8 | 3.4 | 41.9 | 11.0 | — | 43.3 |
| Sapium sebiferum kernel oil | 7.1 | 2.0 | 14.2 | 29.8 | 42.7 | 186.8 |
3.2. Characterization of the modified CNTs
The FTIR spectrum for pristine CNTs is shown in Fig. 1a. The broad band at 3440 cm−1 is attributed to the presence of O–H groups on the surface of pristine CNTs and is believed to result from tight binding of moisture from the ambient atmosphere to the CNTs. In the FTIR spectrum of CNT-COOH (Fig. 1b), the peak at 1726 cm−1 is attributed to acid carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) stretching. The new peaks around 2921 and 2845 cm−1 for CNT-NH2 (Fig. 1c) may be assigned to C–H stretching vibration of methylene in EDA molecules. The characteristic peak of acid carbonyl (C
O) stretching. The new peaks around 2921 and 2845 cm−1 for CNT-NH2 (Fig. 1c) may be assigned to C–H stretching vibration of methylene in EDA molecules. The characteristic peak of acid carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) at 1726 cm−1, which CNT-COOH produced, disappeared, and a new peak at around 1657 cm−1 may be attributed to amide carbonyl (C
O) at 1726 cm−1, which CNT-COOH produced, disappeared, and a new peak at around 1657 cm−1 may be attributed to amide carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) stretching.13 In addition, new bands at 1568 and 1232 cm−1 correspond to N–H in-plane and C–N bond stretching, respectively.19 The above results confirm a successful grafting on the CNTs with EDA.
O) stretching.13 In addition, new bands at 1568 and 1232 cm−1 correspond to N–H in-plane and C–N bond stretching, respectively.19 The above results confirm a successful grafting on the CNTs with EDA.
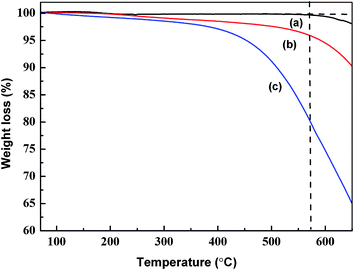
Fig. 2 shows the weight losses of pristine CNTs, CNT-COOH, and CNT-NH2 by TGA, which may be used to estimate the degree of functionalization of the modified CNTs.20 In Fig. 2a, the weight loss for pristine CNTs at 570 °C is very slight (about 0.3 wt%) because of the presence of disordered and amorphous carbons on the CNTs surface.21 This loss indicates good thermal stability below 570 °C. According to Fig. 2b, the weight loss of CNT-COOH (about 4.1 wt%) can be ascribed to the destructive effect of chemical acid treatment on the CNT-COOH surface. In contrast, the curve for CNT-NH2 in Fig. 2c shows a sharp weight loss (19.8 wt%) from 350 to 570 °C. This may be attributed to the large-scale oxidative thermal decomposition of EDA grafted on the CNT-NH2 surface.
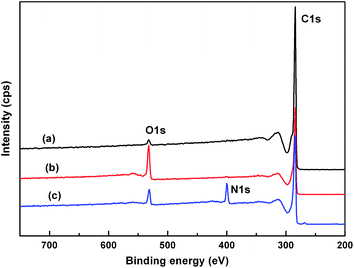
Fig. 3 shows the survey spectra of pristine CNTs, CNT-COOH, and CNT-NH2 by XPS. The peaks at 532, 400, and 284 eV correspond to O1s, N1s, and C1s electrons, respectively. The intensities of the O1s increased significantly after the mixed acid treatment relative to CNT-COOH. Correspondingly, the successful surface amino-functionalized CNTs (CNT-NH2) reduced the relative intensity of O1s with the appearance of N1s peak.
Fig. 4 shows TEM images of the morphology of pristine CNTs and CNT-NH2. As seen in Fig. 4a, there are many black spots and gray fragments in the structure of pristine CNTs, which indicate the present of impurities caused by amorphous carbon, carbon black, and carbon nanoparticles during the preparation of CNTs.22 In Fig. 4b, the ends of CNT-NH2 have already been opened and the wall surface is rather smooth, showing that most of the impurities had been removed by the modification. CNT-NH2 showed less entanglement instead of numerous CNT bundles, suggesting that the grafted EDA could promote the effective dispersion of CNTs.9 In comparison with that of pristine CNTs, the TEM image of CNT-NH2 reveals nanotube bundle adequately covered by a layer of organic matter (Fig. 4c and d). In addition, a clear boundary and flatten packing of the nanotubes is visible. Therefore, the results confirm the chemical functionalization of CNTs with EDA.
3.3. FTIR spectroscopy of pure PU and PU/CNT composites
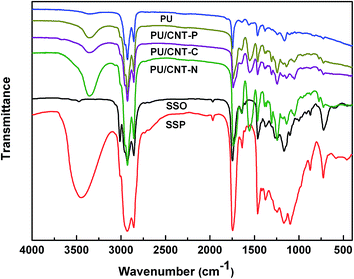
Representative FTIR spectra of the composites are shown in Fig. 5. The characteristic peak of SSO at 3011 cm−1 due to C–H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H stretching disappeared after epoxidation and hydroxylation treatments. In contrast, a new characteristic absorption peak at 3447 cm−1 in the spectrum of Sapium sebiferum polyol (SSP) emerged. As it is due to the –OH group, it implies complete hydroxylation.16 In the spectra of pure PU and PU/CNT composites, the disappearance of the stretching vibration band at 2270 cm−1, which corresponds to –NCO groups of IPDI, suggests that –OH groups of SSP have consumed –NCO groups. Moreover, characteristic bands at 3368 cm−1 (N–H stretching vibration), 1726 cm−1 (carbonyl C
C–H stretching disappeared after epoxidation and hydroxylation treatments. In contrast, a new characteristic absorption peak at 3447 cm−1 in the spectrum of Sapium sebiferum polyol (SSP) emerged. As it is due to the –OH group, it implies complete hydroxylation.16 In the spectra of pure PU and PU/CNT composites, the disappearance of the stretching vibration band at 2270 cm−1, which corresponds to –NCO groups of IPDI, suggests that –OH groups of SSP have consumed –NCO groups. Moreover, characteristic bands at 3368 cm−1 (N–H stretching vibration), 1726 cm−1 (carbonyl C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration), 1549 cm−1 (N–H out-of-plane bending and C–N stretching vibrations), and 1244 cm−1 (ester C–O stretching vibration) confirm the formation of urethane groups (–NH–(C
O stretching vibration), 1549 cm−1 (N–H out-of-plane bending and C–N stretching vibrations), and 1244 cm−1 (ester C–O stretching vibration) confirm the formation of urethane groups (–NH–(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)–O–) after the reaction.23 In addition, by comparison of pure PU with PU/CNT composites, it is very difficult to find any new peak. The results suggest that pure PU and PU/CNT composites were prepared successfully and the segmented structure of PU was not affected by CNTs.
O)–O–) after the reaction.23 In addition, by comparison of pure PU with PU/CNT composites, it is very difficult to find any new peak. The results suggest that pure PU and PU/CNT composites were prepared successfully and the segmented structure of PU was not affected by CNTs.
3.4. The microstructure of pure PU and PU/CNT composites
Fig. 6 shows SEM images of the dispersion and interactions of CNTs in the PU matrix. The image of the composite with pristine CNTs in Fig. 6b shows entangled CNT bundles, which resulted in non-uniform dispersion. Displacement of most of pristine CNTs from the fractured surface demonstrates poor compatibility and adhesion between the CNTs and the matrix.9 In contrast, more dispersed bright dots and fewer bundles in the PU/CNT-C are apparent in Fig. 6c, thus indicating good adhesion of CNT-COOH to the matrix. This adhesion is due to the increased polarity of the CNTs with carboxylic groups in the PU matrix. Fig. 6d features the brightest dots and almost no bundles in the composite with CNT-NH2, which suggest high dispersion of CNT-NH2 in the PU matrix and their tight binding to the matrix. This is attributed to the interaction of –NH2 groups of CNT-NH2 with –NCO groups of the PU matrix.24,25 The presence of CNTs changes the direction of fracture propagation, resulting in more dissipation of external energy, which leads to an increase in strength and modulus of the PU/CNT composites. Therefore, the degree of CNTs dispersion in the PU matrix directly correlates with its efficiency for improving mechanical, thermal, and other properties.233.5. Thermal properties of pure PU and PU/CNT composites
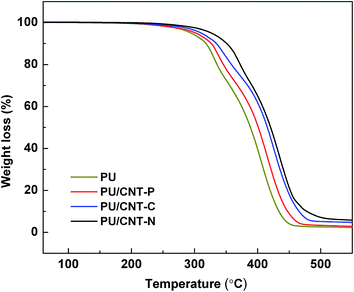
DSC curves of pure PU and PU/CNT composites are presented in Fig. 7. Tg values of all PU/CNT composites are higher than that of pure PU. Tg values of PU/CNT-P, PU/CNT-C, and PU/CNT-N gradually increased, reaching a maximum value of 73.7 °C (PU/CNT-N). The results therefore suggest that CNTs have a positive effect on the crystallization of the polymer, resulting in an increase in Tg. As reported in the literature,24,26 the nanotubes act as nucleating agents, which enhance the crystallization process. Tg represents the mobility of polymer chains in the matrix at the molecular level and an increase in the Tg value indicates a decrease in molecular movement in the polymer.27 Because active –NH2 groups on the surface of CNT-NH2 can react with –NCO groups of IPDI, more chemical interactions occur between CNT-NH2 and the PU matrix, resulting in more networks and higher Tg for PU/CNT-N compared with that for PU/CNT-C. Moreover, the Tg of PU/CNT-C is higher than that of PU/CNT-P because of the greater dispersibility and compatibility of CNT-COOH in the PU matrix, as confirmed by the SEM images (Fig. 6b and c). The increase in Tg values in the PU/CNT composites corresponds to an improvement of mechanical properties such as tensile strength and modulus, due to shifting of Tg toward higher temperatures.Fig. 8 and 9 present TGA and DTG curves for pure PU and PU/CNT composites, respectively. In comparison with that of pure PU, the TGA curves for the composites are obviously shifted toward higher temperatures. Values of the initial decomposition temperature (IDT), which is the temperature at 5% weight loss, are listed in Table 3. The IDTs of PU/CNT-P, PU/CNT-C, and PU/CNT-N are higher than that of pure PU and are delayed by 8.8, 17.6 and 32.5 °C, respectively. Residues of the PU/CNT composites at 550 °C are also shown in Table 3. These results suggest that addition of the CNTs to the PU matrix greatly enhances its thermal stability. Thermal degradations of pure PU and PU/CNT composites take place at two stages, corresponding to the hard and soft segments because of thermodynamic incompatibility of the two segments of the PU matrix.28 The temperature for the maximum rate of degradation (Tmax) for each of the two stages for pure PU and PU/CNT composites is listed in Fig. 9 and Table 3. At each stage, the thermal stability of the PU/CNT composites was significantly enhanced by addition of the CNTs, especially in the case of CNT-NH2. This observation indicates that the incorporation of CNTs into the PU matrix substantially enhanced the thermal stability of the composites because of high thermal stability and high thermal conductivity of the CNTs, which can promote heat dissipation in the matrix.29,30 Tmax values of PU/CNT-C increased to 347.2 and 426.2 °C at two stages, respectively. This improvement may be attributed to the homogeneous dispersion of CNT-COOH in the matrix and the uniform heat distribution throughout the composite.29 Tmax values of PU/CNT-N were enhanced to 370.8 and 436.3 °C, which are the largest improvement in the PU/CNT composites. This improvement is due to the formation of chemical bonds between PU and CNT-NH2, which significantly reduces thermal boundary resistance in PU/CNT-N and thus allows smooth transfer of heat from the PU matrix to the CNTs.30,31 Therefore, the more uniform dispersion and stronger interfacial interaction in the PU/CNT-N composite may contribute to its higher thermal stability compared with those of PU/CNT-P and PU/CNT-C.
| Sample | IDT (°C) | Tmax | Residue at 550 °C (%) | |
|---|---|---|---|---|
| 1st step (°C) | 2nd step (°C) | |||
| PU | 293.1 | 330.8 | 405.7 | 2.7 |
| PU/CNT-P | 301.9 | 337.7 | 414.1 | 2.8 |
| PU/CNT-C | 310.7 | 347.2 | 426.2 | 4.7 |
| PU/CNT-N | 325.6 | 370.8 | 436.3 | 4.9 |
3.6. Mechanical properties of pure PU and PU/CNT composites
The mechanical properties of pure PU and PU/CNT composites are listed in Table 4. Their tensile strength and Young's modulus are higher than those of pure PU. Compared with pure PU, PU/CNT-P, PU/CNT-C, and PU/CNT-N respectively have 66%, 179%, and 295% higher tensile strength and 19.9%, 69.9%, and 111.3% larger Young's modulus. It is demonstrated that adding CNTs to the PU matrix could reinforce and toughen the polymer, because of the effect of the intrinsic mechanical properties, weight fractions and aspect ratios of the nanotubes.25 PU/CNT-C has higher tensile strength and modulus than those of pure PU and PU/CNT-P because of hydrophilic carboxylic groups on the CNTs and better dispersion in this composite.24 PU/CNT-N has the highest tensile strength and modulus among the composites, because the incorporation of CNT-NH2 into the polymer can lead to interaction between CNTs and polymeric chains, which favors stress transfer to CNTs.11,30 Therefore, the strong interaction between CNT-NH2 and the matrix greatly enhances dispersion and interfacial adhesion, thereby strengthening the overall mechanical properties of the composites.32 These findings are consistent with the SEM and DSC results. The elongation at break of the PU/CNT composites increased by 3.9%, 15.5%, and 22.4%, respectively, as compared with that of pure PU (Table 4). Generally, using common fillers for PU materials effectively improved their strength, but usually decreased their elongation at break.33 Strength, modulus and elongation at break in the PU/CNT composites were simultaneously improved, when the CNT content of the composites is 2 wt%. This result may be caused by easier cubic deformation of the CNTs due to their unique cage structure and topology.13 However, the higher CNT content (5 wt%) of the PU matrix can decrease the elongation at break of the composite because of uneven CNT dispersion.| Tensile strength (MPa) | Elongation at break (%) | Young's modulus (MPa) | |
|---|---|---|---|
| PU | 6.2 ± 0.3 | 185.1 ± 13.4 | 18.6 ± 1.1 |
| PU/CNT-P | 10.3 ± 0.5 | 192.3 ± 14.6 | 22.3 ± 1.6 |
| PU/CNT-C | 17.3 ± 0.9 | 213.8 ± 15.7 | 31.6 ± 2.2 |
| PU/CNT-N | 24.5 ± 1.2 | 226.5 ± 16.2 | 39.3 ± 3.1 |
PUs from various types of plant oils show a wide range of properties due to their differences in the reactivity and relative percentages of fatty acids. This variety results in their many applications.16 In Table 5,16,34,35 most properties of SSO-based PU closely match those of the other common plant-oil-based PUs. However its Tg is higher than that of the plant-oil-based PUs probably because of more hydroxyl groups and higher cross-linking density of SSO-based polyols used in PU synthesis.36 Furthermore, adding small amounts (2 wt%) of the CNTs to the PU matrix could significantly improve its Tg, thermal stability and mechanical properties. Therefore, the PU/CNT composites in this work may have promising application in adhesives and decorative coatings.
| Sample | DSC (°C) | TGA | Tensile strength (MPa) | Elongation at break (%) | ||
|---|---|---|---|---|---|---|
| IDT (°C) | Tmax1 (°C) | Tmax2 (°C) | ||||
| Soybean | 20.9 | 242 | — | — | 4.5 ± 0.6 | 329.6 ± 47.2 |
| Sunflower | 41 | — | 284 | 408 | 23.18 ± 0.8 | 750 ± 5 |
| Jatropha | — | 286.7 | 313.9 | 393.1 | 2.4 ± 0.5 | 325.1 ± 46.8 |
| Linseed | 35.9 | 150 | 267 | 312 | 3.8 ± 1 | 256 ± 6 |
| Castor | −40.2 | 281 | 354 | 434 | 6.3 ± 0.3 | 389.1 ± 12.3 |
| Tung | 34.8 | 266.1 | 382.9 | 422.5 | 4.8 ± 0.5 | 14.9 ± 0.5 |
| Palm | 39.7 | 191.9 | 275 | 447 | 1.5 ± 0.3 | — |
| Sapium sebiferum | 56.6 | 293.1 | 330.8 | 405.7 | 6.2 ± 0.3 | 185.1 ± 13.4 |
4. Conclusion
In the study, the physicochemical properties and fatty acid profile of SSO were analyzed and confirmed a feasibility of polymer exploitation. A bio-polyurethane was synthesized from natural SSO-based polyol and IPDI via in situ polymerization and the functionalized CNTs at 2 wt% loading were used to reinforce the PU matrix. CNT-NH2 showed the best CNT dispersion throughout the PU matrix and was the most effective among the composites at improving the thermal and mechanical properties of PU. In comparison with that of pure PU, Tg of the PU/CNT-N composite increased from 56.6 to 73.7 °C, and its IDT increased from 293.1 to 325.6 °C. Meanwhile, the tensile strength, Young's modulus, and elongation at break of PU/CNT-N were also enhanced by 295%, 111.3%, and 22.4%, respectively. The significant improvement of thermal and mechanical properties of the PU/CNT composites is due to the homogeneous dispersion of CNT-NH2 and the presence of strong interfacial adhesion between CNT-NH2 and the PU matrix. The synthetic PU/CNT composites showed promising application in adhesives and decorative coatings.Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (No. 31070089, 31170078 and J1103514), the National High Technology Research and Development Program of China (2011AA02A204, 2013AA065805), the Innovation Foundation of Shenzhen Government (JCYJ20120831111657864) and the Fundamental Research Funds for HUST (No. 2014NY007). Many thanks are indebted to Analytical and Testing Center of HUST for their valuable assistances in FTIR, SEM, TEM, TGA and DSC measurement.References
- A. Lee and Y. Deng, Eur. Polym. J., 2015, 63, 67–73 CrossRef CAS PubMed.
- D. V. Palaskar, A. Boyer, E. Cloutet, C. Alfos and H. Cramail, Biomacromolecules, 2010, 11, 1202–1211 CrossRef CAS PubMed.
- Y. Yan, X. Li, G. Wang, X. Gui, G. Li, F. Su, X. Wang and T. Liu, Appl. Energy, 2014, 113, 1614–1631 CrossRef CAS PubMed.
- M. Desroches, S. Caillol, V. Lapinte, R. Auvergne and B. Boutevin, Macromolecules, 2011, 44, 2489–2500 CrossRef CAS.
- Z. S. Petrovi, Polym. Rev., 2008, 109–155 CrossRef PubMed.
- A. E. Atabani, A. S. Silitonga, H. C. Ong, T. M. I. Mahlia, H. H. Masjuki, I. A. Badruddin and H. Fayaz, Renewable Sustainable Energy Rev., 2013, 18, 211–245 CrossRef CAS PubMed.
- Y. Liu, H. Xin and Y. Yan, Ind. Crops Prod., 2009, 30, 431–436 CrossRef CAS PubMed.
- Q. Li and Y. Yan, Appl. Energy, 2010, 87, 3148–3154 CrossRef CAS PubMed.
- S. Yang, C. M. Ma, C. Teng, Y. Huang, S. Liao, Y. Huang, H. Tien, T. Lee and K. Chiou, Carbon, 2010, 48, 592–603 CrossRef CAS PubMed.
- P. Russo, M. Lavorgna, F. Piscitelli, D. Acierno and L. Di Maio, Eur. Polym. J., 2013, 49, 379–388 CrossRef CAS PubMed.
- M. Raja, S. H. Ryu and A. M. Shanmugharaj, Colloids Surf., A, 2014, 450, 59–66 CrossRef CAS PubMed.
- C. Zhang, D. Vennerberg and M. R. Kessler, J. Appl. Polym. Sci., 2015, 132 DOI:10.1002/app.42515.
- J. Xiong, Z. Zheng, W. Song, D. Zhou and X. Wang, Composites, Part A, 2008, 39, 904–910 CrossRef PubMed.
- J. Xiong, D. Zhou, Z. Zheng, X. Yang and X. Wang, Polymer, 2006, 47, 1763–1766 CrossRef CAS PubMed.
- T. Ramanathan, F. T. Fisher, R. S. Ruoff and L. C. Brinson, Chem. Mater., 2005, 17, 1290–1295 CrossRef CAS.
- G. Wu, X. He, L. Xu, H. Zhang and Y. Yan, RSC Adv., 2015, 5, 27097–27106 RSC.
- E. Akbar, Z. Yaakob, S. K. Kamarudin, M. Ismail and J. Salimon, Eur. J. Appl. Eng. Sci. Res., 2009, 29, 396–403 Search PubMed.
- A. Zlatanić, C. Lava, W. Zhang and Z. S. Petrović, J. Polym. Sci., Part B: Polym. Phys., 2004, 42, 809–819 CrossRef PubMed.
- H. Kuan, C. M. Ma, W. Chang, S. Yuen, H. Wu and T. Lee, Compos. Sci. Technol., 2005, 65, 1703–1710 CrossRef CAS PubMed.
- H. Tahermansouri and E. Biazar, New Carbon Mater., 2013, 28, 199–207 CrossRef CAS.
- S. Yang, C. M. Ma, C. Teng, Y. Huang, S. Liao, Y. Huang, H. Tien, T. Lee and K. Chiou, Carbon, 2010, 48, 592–603 CrossRef CAS PubMed.
- Y. Li, S. Wang, Z. Luan, J. Ding, C. Xu and D. Wu, Carbon, 2003, 41, 1057–1062 CrossRef CAS.
- C. Wang, X. Chen, H. Xie and R. Cheng, Composites, Part A, 2011, 42, 1620–1626 CrossRef PubMed.
- N. G. Sahoo, Y. C. Jung, H. J. Yoo and J. W. Cho, Macromol. Chem. Phys., 2006, 207, 1773–1780 CrossRef CAS PubMed.
- A. K. Barick and D. K. Tripathy, Composites, Part A, 2010, 41, 1471–1482 CrossRef PubMed.
- S. A. R. Hashmi, H. C. Prasad, R. Abishera, H. N. Bhargaw and A. Naik, Mater. Des., 2015, 67, 492–500 CrossRef CAS PubMed.
- H. Xia and M. Song, Soft Matter, 2005, 1, 386–394 RSC.
- A. K. Barick and D. K. Tripathy, Mater. Sci. Eng., B, 2011, 176, 1435–1447 CrossRef CAS PubMed.
- Q. Jing, J. Y. Law, L. P. Tan, V. V. Silberschmidt, L. Li and Z. Dong, Composites, Part A, 2015, 70, 8–15 CrossRef CAS PubMed.
- J. Xiong, Z. Zheng, X. Qin, M. Li, H. Li and X. Wang, Carbon, 2006, 44, 2701–2707 CrossRef CAS PubMed.
- S. S. Mahapatra, S. K. Yadav, H. J. Yoo, J. W. Cho and J. Park, Composites, Part B, 2013, 45, 165–171 CrossRef CAS PubMed.
- W. D. Zhang, L. Shen, I. Y. Phang and T. Liu, Macromolecules, 2004, 37, 256–259 CrossRef CAS.
- J. Xiong, Y. Liu, X. Yang and X. Wang, Polym. Degrad. Stab., 2004, 86, 549–555 CrossRef CAS PubMed.
- X. Yang, S. Li, J. Xia, J. Song, K. Huang and M. Li, Ind. Crops Prod., 2015, 63, 17–25 CrossRef CAS PubMed.
- S. Saalah, L. C. Abdullah, M. M. Aung, M. Z. Salleh, D. R. Awang Biak, M. Basri and E. R. Jusoh, Ind. Crops Prod., 2015, 64, 194–200 CrossRef CAS PubMed.
- X. Kong, G. Liu, H. Qi and J. M. Curtis, Prog. Org. Coat., 2013, 76, 1151–1160 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |