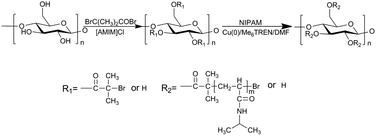
Thermal-sensitive Starch-g-PNIPAM prepared by Cu(0) catalyzed SET-LRP at molecular level
Leli Wang,
Ying Wu,
Yongjun Men,
Jianan Shen and
Zhengping Liu*
Beijing Key Lab of Energy Conversion and Storage Materials, BNU Key Lab of Environmentally Friendly and Functional Polymer Materials, College of Chemistry, Beijing Normal University, Beijing 100875, China. E-mail: lzp@bnu.edu.cn; Fax: +86 10 58802075; Tel: +86 10 58806896
First published on 13th August 2015
Abstract
Single electron transfer living radical polymerization (SET-LRP) was employed to prepare thermal-sensitive starch graft poly(N-isopropylacrylamide) (Starch-g-PNIPAM) with soluble Starch-Br as macro-initiator at molecular level. And Starch-g-PNIAPM with improved graft ratio and controlled length of graft chains were obtained. The effects of molar ratio of monomer to initiator and temperature on polymerization were investigated. The thermal responsibilities investigated by 1H NMR and UV showed that the LCST ranged from 31.5 °C to 23 °C with the increase of the length of PNIPAM chains and the concentrations of copolymers. Starch-g-PNIPAM hydrogels were gained when adding N,N′-methylenebisacrylamide as crosslinking agent and the swelling and deswelling behaviors were also investigated. The obtained hydrogels possess a high equilibrium swelling ratio as high as 1826%, and an extremely rapid shrinking rate as fast as losing more than 85% of water in 5 min.
Introduction
Starch graft poly(N-isopropylacrylamide) (Starch-g-PNIPAM) consisting of both natural starch and synthetic stimuli responsive poly(N-isopropylacrylamide) (PNIPAM) has attracted increasing attention due to its potential applications in controlled drug release, immobilization of enzyme, super water absorbent material and size-selective absorbent. Compared with bulk PNIPAM, Starch-g-PNIPAM has advantages for the improvements of biodegradability and biocompatibility. The conventional method to prepare Starch-g-PNIPAM was grafting PNIPAM from starch by traditional free radical polymerization. In 2000, Zhuo and co-workers prepared temperature-sensitive hydrogels with improved surface property by polymerization of NIPAM with gelated corn starch.1 In 2006, Chen and co-workers synthesized smart carboxymethyl starch graft PNIPAM (CMS-g-PNIPAM) with core–shell structure in supercritical carbon dioxide.2 In 2010, poly(NIPAM-co-DMAAm) grafted starch microspheres were prepared by Fundueanu and co-workers as well.3 In the same year, Wang and co-workers prepared PNIPAM hydrogels with high mechanical strength crosslinked by a starch-based crosslinker.4However, influenced by strong intramolecular and intermolecular hydrogen bonds, natural starch is difficult to be dissolved in traditional solvents, which confined the synthesis of Starch-g-PNIPAM to surface initiation and at granule level, significantly limiting the graft ratio, graft density and further application. Recently, ionic liquid, employed as an environmentally benign green solvent, instead of classical organic ones,5–11 has attracted increasingly attentions in the field of dissolution and modification of natural polymers,12–26 which makes the graft polymerization at molecular level and the improvement of graft ratio possible. Our group27 synthesized Starch-g-PS in ionic liquid [EMIM]Ac using traditional free radical polymerization with potassium persulfate as initiator. The introduction of ionic liquid [EMIM]Ac provided an effective way to dissolve corn starch, making it a homogeneous polymerization at molecular level. However, due to the uncontrollability of traditional free radical polymerization, the graft ratio and the length of graft chains were out of control. In our recent work,28 a starch-based macro-initiator with good solubility was synthesized using [AMIM]Cl as the reaction medium and used for ATRP of styrene and methyl methacrylate at molecular level, which significantly improved the graft ratio and graft density. Single electron transfer living radical polymerization (SET-LRP) is another novel living polymerization approach after the invention of ATRP.29,30 SET-LRP becomes an attractive method in polymer chemistry and material chemistry, due to its good controllability, milder reactive conditions and lighter color products as compared with ATRP.31–34 To the best of our knowledge, modification of cellulose via SET-LRP has been investigated for several years,33,35–39 while its applications in preparation of starch graft copolymers need deep research.
In this study, SET-LRP method was employed to synthesize Starch-g-PNIPAM at molecular level in DMF with soluble Starch-Br as macro-initiator and Cu(0) power/Me6TREN as catalytic system. The graft ratio of PNIPAM was significantly improved compared to previous work. The thermal response of Starch-g-PNIPAM was investigated by 1H NMR and UV. Starch-g-PNIPAM hydrogels with relative high swelling ratio were formed when N,N′-methylenebisacrylamide was added as crosslinking agent.
Experimental section
Materials
Corn starch (from Aladdin, AR) was dried in vacuum at 60 °C for 48 h before usage. Ionic liquid [AMIM]Cl was synthesized according to the literature procedure40 and used after being dried in vacuum at 80 °C for 48 h. N-Isopropyl acrylamide (NIPAM) (from TCI, 98%) was recrystallized from hexane/ethyl acetate. Me6TREN was synthesized according to the literature procedure.39 Other reagents such as 2-bromoisobutyryl bromide (BrBiB), N,N′-methylenebisacrylamide and Cu power were purchased from Aladdin and used as received.Synthesis of Starch-Br
Starch-based macro-initiator (Starch-Br) was synthesized as the description in our previous work:28 corn starch was dissolved in [AMIM]Cl at concentration of 10 wt% with mechanically stirred at 80 °C under N2 flow. After cooling the mixture to room temperature, a certain amount of BrBiB ([AGU]/[BrBiB] = 1/5, molar ratio) was added with N2 flow under an ice-cold bath. The mixture was reacted at room temperature for 6 h with mechanically stirred. The products were precipitated by adding into excessive deionized water. The precipitate was washed thoroughly and filtered, followed by dried in vacuum at 60 °C for 24 h.Synthesis of Starch-g-PNIPAM copolymer and hydrogel via SET-LRP
A desired amount of Starch-Br, NIPAM, Cu power and Me6TREN were completely dissolved in DMF in a 25 mL Schlenk flask. The reaction mixture was degassed by three freeze-pump-thaw cycles and was left to react for a prescribed time period at 30 °C for SET-LRP. The reaction was stopped by exposing the mixture to air. Then the mixture was dialyzed against deionized water for a week and lyophilized.Starch-g-PNIPAM hydrogel was prepared as follow: a desired amount of Starch-Br, NIPAM, Cu power, Me6TREN and N,N′-methylenebisacrylamide were completely dissolved in solvent in a 25 mL Schlenk flask. The reaction mixture was degassed by three freeze-pump-thaw cycles and was left to react at 30 °C for 48 h. The product was immersed in deionized water which was refreshed at an interval of 8 h for a week, and then lyophilized for 24 h.
Monomer conversion and grafting ratio were calculated according to eqn (1) and (2), respectively, where W1 (g) is the dry weight of Starch-Br, W2 (g) is the dry weight of starch graft copolymers, and Wmon (g) is the weight of the monomer at the beginning of the polymerization. Swelling ratio of Starch-g-PNIPAM hydrogels was calculated according to eqn (3), where Ws and Wd are the weight of hydrogel samples at swollen and dry state, respectively.
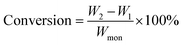 | (1) |
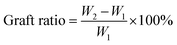 | (2) |
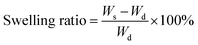 | (3) |
Characterization
Fourier Transform Infrared (FTIR) spectra were recorded on an Omnic Avatar 360 with the pressed KBr discs. Nuclear magnetic resonance (NMR) spectra were obtained in DMSO-d6 or D2O on a Bruker Avance 400 MHz spectrometer.Differential scanning calorimetry (DSC) was performed on a Mettler DSC 1 instrument with a heating rate of 10 °C min−1 from 0 to 200 °C after eliminating thermal history. Thermogravimetric Analysis (TGA) was performed on a METTLER TOLEDO TGA-SDA 851e analyzer with a heating rate of 10 °C min−1 from 25 to 600 °C under nitrogen atmosphere.
The temperature response of Starch-g-PNIPAM was determined by measuring the light transmittance at the wavelength of 500 nm on a UV-Vis spectrophotometer (TU-1901), equipped with a circulating water bath. The samples were equilibrated for 15 min before being measured at each temperature. The LCST was determined as the intersection of the maximal slope tangent and the initial horizontal tangent in UV spectra.
The structures of Starch-g-PNIPAM hydrogels were examined by scan electron microscopy (SEM, Hitachi S-4800, Japan). The samples were observed by SEM after spraying platinum.
Results and discussion
Synthesis of Starch-g-PNIPAM via SET-LRP
Starch-based macroinitiator with DS = 1.20 prepared in ionic liquid [AMIM]Cl was used as initiator of SET-LRP to prepare Starch-g-PNIPAM. Cu(0) power/Me6TREN and DMF were used as catalytic system and solvent, respectively. The reaction process is described in Scheme 1.A series of influencing factors on the polymerization were investigated and the results are summarized in Table 1. The graft polymerization was conducted at molecular level obtaining Starch-g-PNIPAM with high graft ratio. The monomer conversion, graft ratio and graft chain length could be tuned by molar ratio of [NIPAM]/[Starch-Br], temperature and time. The number average molecule weight (Mn) of grafted PNIPAM chains was calculated by 1H NMR spectra rather than GPC, because it was difficult to cleave PNIPAM chains from starch backbone. Similar phenomena were discovered in the previous research.36,41 Seen from Table 1, Mn varied from 600 to 8490 g mol−1 under experimental conditions, while graft ratio ranged from 229% to 2652%. As shown in entries 3, 5 and 7, rate of polymerization, Mn, as well as graft ratio declined with the decrease of molar ratio of [NIPAM]/[Starch-Br]. Monomer conversion decreased from 35.08 to 16.24% with the decrease of molar ratio of [NIPAM]/[Starch-Br] from 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 40
1 to 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, while keeping the other reaction variables constant. A gradual decrease of [NIPAM] also resulted in a decrease in Mn from 3990 to 600 g mol−1. As displayed in entries 1–4, the monomer conversion and Mn increased with time. However, at the beginning of the polymerization, the rate of polymerization was very slow (7.09%, 20 min), which might be ascribed to the influence of an induction period.42 Temperature is another crucial factor for free radical polymerization. Experiments at different temperatures (30 °C and 50 °C) were conducted to investigate the effect of temperature. Comparing entries 3 and 11, polymerizations at 50 °C were remarkably faster than that at 30 °C. When keeping other reaction conditions constant, raising temperature from 30 °C to 50 °C resulted in the increase of monomer conversion from 35.08% to 75.22%, and the rise of Mn from 3990 to 8490 g mol−1.
1, while keeping the other reaction variables constant. A gradual decrease of [NIPAM] also resulted in a decrease in Mn from 3990 to 600 g mol−1. As displayed in entries 1–4, the monomer conversion and Mn increased with time. However, at the beginning of the polymerization, the rate of polymerization was very slow (7.09%, 20 min), which might be ascribed to the influence of an induction period.42 Temperature is another crucial factor for free radical polymerization. Experiments at different temperatures (30 °C and 50 °C) were conducted to investigate the effect of temperature. Comparing entries 3 and 11, polymerizations at 50 °C were remarkably faster than that at 30 °C. When keeping other reaction conditions constant, raising temperature from 30 °C to 50 °C resulted in the increase of monomer conversion from 35.08% to 75.22%, and the rise of Mn from 3990 to 8490 g mol−1.
| Entry | [NIPAM]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [Starch-Br]a [Starch-Br]a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [Cu] [Cu]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [Me6TREN] [Me6TREN] |
Temp. (°C) | Time (min) | Conv.b (%) | Mn (1H NMR)c | Graft ratio (%) |
|---|---|---|---|---|---|---|
a [Starch-Br] = mole of bromine, DS = 1.20, calculated from 1H NMR.b The monomer conversion was determined by weighing the samples.c Calculated by 1H NMR. Ic+f = Ic + If = 6Id + If,  , Mn = 113.16 × DP (DP, the degree of polymerization of PNIPAM chains. Ic, Id, If and Ic+f, the integral areas of peaks c, d, f, and c + f in 1H NMR spectrum, respectively).d 2 wt% of N,N′-methylenebisacrylamide was added.e 5 wt% of N,N′-methylenebisacrylamide was added.f Using [EMIM]DMP as solvent. , Mn = 113.16 × DP (DP, the degree of polymerization of PNIPAM chains. Ic, Id, If and Ic+f, the integral areas of peaks c, d, f, and c + f in 1H NMR spectrum, respectively).d 2 wt% of N,N′-methylenebisacrylamide was added.e 5 wt% of N,N′-methylenebisacrylamide was added.f Using [EMIM]DMP as solvent. |
||||||
| 1 | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 20 | 7.09 | 1260 | 250 |
| 2 | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 40 | 29.78 | 2120 | 1050 |
| 3 | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 60 | 35.08 | 3990 | 1237 |
| 4 | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 180 | 56.74 | 6170 | 2006 |
| 5 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 60 | 25.53 | 1580 | 450 |
| 6 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 90 | 34.04 | 2260 | 600 |
| 7 | 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 60 | 16.24 | 600 | 229 |
| 8 | 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 600 | 81.56 | 3570 | 1150 |
| 9 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 600 | 75.81 | 1940 | 267 |
| 10 | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
50 | 30 | 52.22 | 5660 | 1841 |
| 11 | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
50 | 60 | 75.22 | 8490 | 2652 |
| 12d | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 1440 | 88.74 | — | 3128 |
| 13e | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 1440 | 89.37 | — | 3150 |
| 14d,f | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 1440 | 88.97 | — | 3145 |
| 15e,f | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
30 | 1440 | 90.12 | — | 3186 |
Fig. 1 shows the kinetic plots of graft polymerization of PNIPAM onto starch with molar ratio of [NIPAM]/[Starch-Br] as 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 50
1 and 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, where [M]0 and [M] represent the concentration of monomer before polymerization and at time t. In the first 20 minutes of the polymerization with molar ratio of [NIPAM]/[Starch-Br] as 100
1, where [M]0 and [M] represent the concentration of monomer before polymerization and at time t. In the first 20 minutes of the polymerization with molar ratio of [NIPAM]/[Starch-Br] as 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at 30 °C, polymerization proceeded slowly compared with polymerization after 20 minutes. It is speculated that there is an induction period at the beginning of the polymerization. A similar phenomenon was discovered in the polymerization with molar ratio of 50
1 at 30 °C, polymerization proceeded slowly compared with polymerization after 20 minutes. It is speculated that there is an induction period at the beginning of the polymerization. A similar phenomenon was discovered in the polymerization with molar ratio of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at 30 °C and 100
1 at 30 °C and 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at 50 °C. The induction period may be caused by the oxide layer on the surface of Cu power.42 A rectilinear dependence of ln([M]0/[M]) vs. time as well as linear evolution of Mn vs. concentration were observed in the kinetic plots, confirming the liveness and controllability of polymerization process.
1 at 50 °C. The induction period may be caused by the oxide layer on the surface of Cu power.42 A rectilinear dependence of ln([M]0/[M]) vs. time as well as linear evolution of Mn vs. concentration were observed in the kinetic plots, confirming the liveness and controllability of polymerization process.
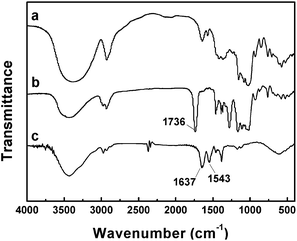
In order to certify that PNIPAM side chains were successfully grafted onto starch backbone, graft copolymers were characterized by FTIR. Fig. 2 shows FTIR spectra of corn starch, Starch-Br and Starch-g-PNIPAM. Compared with the spectrum of corn starch, a typical absorption peak of the O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group at 1736 cm−1 (Fig. 2b) was observed, indicating the successful synthesis of Starch-Br. Vibrations at 1637 cm−1 and 1543 cm−1 in Fig. 2c were assigned to N–C
O group at 1736 cm−1 (Fig. 2b) was observed, indicating the successful synthesis of Starch-Br. Vibrations at 1637 cm−1 and 1543 cm−1 in Fig. 2c were assigned to N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and N–H, respectively, which demonstrated that PNIPAM was grafted onto starch.
O and N–H, respectively, which demonstrated that PNIPAM was grafted onto starch.
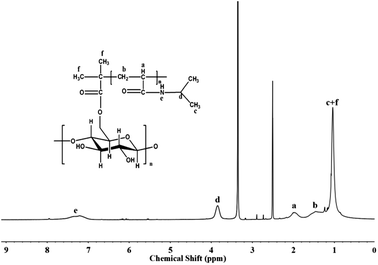
1H NMR spectra showed in Fig. 3 were employed to give a further demonstration of graft polymerization. The chemical shifts at δ = 3.8 ppm and at δ = 1.0 ppm were assigned to –CH– and –CH3 of isopropyl in the side chains, respectively. The signals in the range of δ = 1.3–2.1 ppm should be ascribed to –CH2–CH– in the polymer chains. The signal at δ = 7.2 ppm represented proton of –NH– group. The signals of starch backbone were not strong enough to be observed because the molar ratio of monomer to Starch-Br was relative high.
Temperature response of Starch-g-PNIPAM
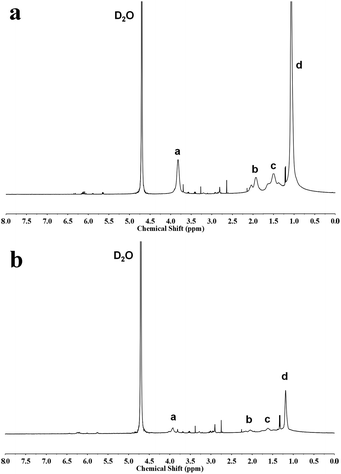
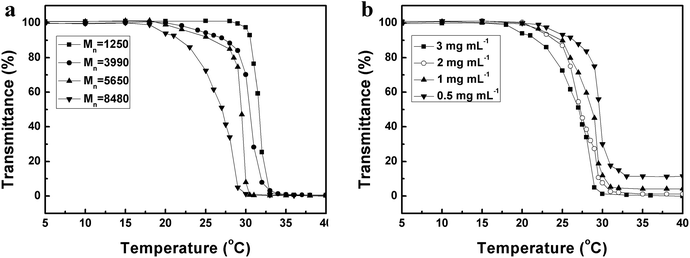
As is well known, PNIPAM is one of the representative temperature-sensitive polymers with responsive temperature of ∼32 °C.43,44 Starch-g-PNIPAM consisted of starch backbone and PNIPAM side chains, was expected to be thermo-responsible. 1H NMR and UV spectra were used to measure the thermosensitivity of Starch-g-PNIPAM. Fig. 4 shows 1H NMR spectra of Starch-g-PNIPAM in D2O at 25 °C and 35 °C, respectively. The intensities of signals a, b, c and d, which were the characteristic peaks of PNIPAM side chains, decreased obviously during temperature changing from 25 °C to 35 °C, indicating that Starch-g-PNIPAM have a good response to temperature.Fig. 5 shows the changes of optical transmittance of Starch-g-PNIPAM at the wavelength of 500 nm obtained in aqueous solutions measured by UV during the heating process. The optical transmittance of solutions exhibited a saltation at lower critical solution temperature (LCST) during the heating process. The LCST ranged from 31.5 °C to 23 °C with the increase of molecular weight of graft chains and concentrations of copolymers. The influence of molecular weight was studied, as indicated in Fig. 5a, the LCST of the solution decreased with the increasing graft chains length. Moreover, keeping the concentrations of copolymers as 3 mg mL−1, the LCST decreased from 31.5 °C to 29 °C, 28 °C and 23 °C when raising molecular weight of graft chains from 1250 to 3990, 5650 and 8480 g mol−1. Fig. 5b depicts the effect of concentration of copolymers on LCST. Keeping the molecular weight of graft chains as 8480 g mol−1, the LCST of the solution with a relative high concentration of 3 mg mL−1 was 23 °C, which is much lower than that of solutions with relative low concentrations of 2 mg mL−1 (24 °C), 1 mg mL−1 (28.5 °C) and 0.5 mg mL−1 (29.5 °C). These phenomena could be explained by the influence of solubility. Starch-g-PNIPAM with longer side chains and at lower concentration dissolved better in water, and the temperature to force them from dissolved state to insoluble state should be higher, resulting in an increase in LCST.
Thermal properties of Starch-g-PNIPAM
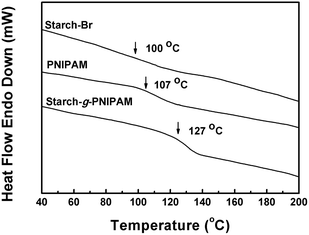
TGA and DSC were employed to measure the thermal properties of Starch-g-PNIPAM. Fig. 6 depicts representative thermograms of Starch-Br, PNIPAM and Starch-g-PNIPAM. Starch-Br showed typical degradation behaviour of starch with a decomposition temperature of about 220 °C, while PNIPAM showed two-step weight loss at 190 °C and 330 °C. All of the distinct onset degradation temperature could be observed in TGA curve of Starch-g-PNIPAM, but the weight loss of starch backbone was much less than that of PNIPAM chains, demonstrating the high graft ratio of Starch-g-PNIPAM. DSC curves of Starch-Br, PNIPAM and Starch-g-PNIPAM are shown in Fig. 7. The glass transition temperature (Tg) of Starch-Br was observed at about 100 °C, and the Tg of bulk PNIPAM appeared at about 107 °C. However, Starch-g-PNIPAM showed Tg at 127 °C, which was even higher than that of bulk PNIPAM. This phenomenon may be caused by the restriction of PNIPAM chains to starch backbone.33,45 After graft polymerization, the mobility of PNIPAM chains was hindered by starch backbone, resulting in an increase in Tg.Swelling behaviour of Starch-g-PNIPAM hydrogels
Starch-g-PNIPAM hydrogels could be prepared by adding a certain amount of N,N′-methylenebisacrylamide at the beginning of polymerization when using DMF as solvent (entries 12 and 13, Table 1). Since ionic liquid is a kind of good solvent for starch, we chose ionic liquid 1-ethyl-3-methylimidazolium dimethylphosphate ([EMIM]DMP) as another solvent to prepare hydrogels (entries 14 and 15, Table 1).The surface morphology of Starch-g-PNIPAM hydrogels prepared in DMF and [EMIM]DMP are displayed in Fig. 8. Macroporous structures were observed in hydrogel prepared in DMF. A large amount of these pores were obstructed. However, in hydrogel prepared in [EMIM]DMP, open channels formed with smaller pores were observed. Fig. 9 depicts the temperature sensitive swelling and deswelling process of Starch-g-PNIPAM hydrogels with different degree of crosslinking prepared in DMF and [EMIM]DMP, respectively. The equilibrium swelling ratio of Starch-g-PNIPAM hydrogels with degree of crosslinking of 2% at 20 °C could reach 1097% and 1479%, respectively, much higher than hydrogels prepared by polymerization of NIPAM from gelated corn starch,1 and slightly higher than hydrogels prepared by polymerization of NIPAM from carboxymethyl starch at pH = 7.2 Compared with hydrogels prepared in DMF, hydrogels prepared in [EMIM]DMP could uptake more water and swell faster. Hydrogels prepared in [EMIM]DMP with degree of crosslinking of 2% and 5% could uptake 92% and 97% of their maximum water-uptake capacity in 120 min, while it took almost 1000 min for those prepared in DMF to uptake 89% and 86% of their maximum water-uptake capacity. This improvement in swelling rate might be caused by the microstructure differences between the hydrogels. Hydrogels prepared in [EMIM]DMP possessing open channels behaved better in swelling process. The degree of crosslinking is another important factor that affected swelling behaviour. Hydrogels with higher degree of crosslinking swelled slower and uptook less water than ones with lower degree of crosslinking. High degree of crosslinking stood for high restriction to hydrophilic PNIPAM, which resulted in a decrease in equilibrium swelling ratio and shrinking rate.
As to deswelling at 40 °C, hydrogels prepared in DMF and [EMIM]DMP both shrinked quickly in 5 min after being warmed up to 40 °C, which was more than 4 times faster than previous work.1,4 To our best knowledge, in the conventional hydrogels, the interaction between PNIPAM and water in the surface region of hydrogels were destroyed firstly and formed a dense layer at the beginning of deswelling process, which would prevent the release of water. However, in our Starch-g-PNIPAM hydrogels, despite of the confine of dense layer, hydrogen bonds between –OH in starch and PNIPAM could provide channels for water to be released, which significantly improved shrinking rate.1,4 In addition, the preparation at molecular level was another explanation for rapid shrinking rate.
Five swelling and deswelling circles were conducted. Results demonstrated that hydrogels had almost the same good performance in swelling process after at least 5 circles, since there was no significant difference in swelling ratio observed (Fig. 10).
 | ||
| Fig. 10 Equilibrium swelling ratio of Starch-g-PNIPAM hydrogels prepared in DMF with degree of crosslinking of 2% for five swelling-deswelling circles. | ||
All hydrogels were hydrolysed in 10% HCl aqueous for different time and then the swelling experiments were conducted. Hydrogels with degree of crosslinking of 2% disintegrated during hydrolysis, while hydrogels with degree of crosslinking of 5% maintained their shapes. The equilibrium swelling ratios of hydrogels after hydrolysis for different time are displayed in Fig. 11. The results showed that the equilibrium swelling ratio increased with the hydrolysis time and reached a highest value of 1826%. This might be ascribed to the hydrolysis of starch backbone during the process. On one hand, starch is a portion that difficult to dissolve and swelling in water, which disappeared gradually during the hydrolysis process, leading to an increase in equilibrium swelling ratio. On the other hand, starch also played a role of crosslinker in hydrogels. When starch was hydrolysed, it had the same effect as a decrease of degree of crosslinking, which resulted in an increase in swelling ratio.
 | ||
| Fig. 11 Equilibrium swelling ratio of Starch-g-PNIPAM hydrogels with different hydrolysis time. The hydrogels was prepared in DMF with degree of crosslinking of 5%. | ||
Conclusions
Cu(0) catalyzed SET-LRP has been used for the first time for the modification of starch. Thermal-sensitive Starch-g-PNIPAM with high graft ratio and controlled chain length was obtained at molecular level, bringing about the improvement of swelling ratio of Starch-g-PNIPAM hydrogels. The polymerization could be tuned by varying molar ratio, temperature and time. Study of thermal response demonstrated that the LCST of Starch-g-PNIPAM copolymer ranged from 31.5 °C to 23 °C with the increase of the length of PNIPAM chains and the concentrations of copolymers. Starch-g-PNIPAM hydrogels obtained at molecular level showed up an excellent performance in swelling behaviour, reaching a highest swelling ratio of 1826%, and a remarkable rapid shrinking rate as fast as losing more than 85% of water in 5 min, indicating the potential application in drug release. Furthermore, the present methodology is expected to be applied to a wide range of stimuli responsive monomers to develop other new functional materials based on natural starch.Acknowledgements
This work is supported by the National Natural Science Foundation of China (No. 51173019) and the Program for Changjiang Scholars and Innovative Research Team in University.Notes and references
- X. Zhang and R. Zhuo, J. Colloid Interface Sci., 2000, 223, 311–313 CrossRef CAS.
- L. Cao, L. Chen, X. Chen, L. Zuo and Z. Li, Polymer, 2006, 47, 4588–4595 CrossRef CAS PubMed.
- G. Fundueanu, M. Constantin, P. Ascenzi and B. C. Simionescu, Biomed. Microdevices, 2010, 12, 693–704 CrossRef CAS.
- Y. Tan, K. Xu, P. Wang, W. Li, S. Sun and L. Dong, Soft Matter, 2010, 6, 1467–1471 RSC.
- C. Fu and Z. Liu, Polymer, 2008, 49, 461–466 CrossRef CAS PubMed.
- J. Wang and Z. Liu, Green Chem., 2012, 14, 3204–3210 RSC.
- J. Wang and Z. Liu, Chin. Sci. Bull., 2013, 58, 1262–1266 CrossRef CAS.
- J. Dou and Z. Liu, Green Chem., 2012, 14, 2305–2313 RSC.
- J. Dou, Z. Liu, K. Mahmood and Y. Zhao, Polym. Int., 2012, 61, 1470–1476 CrossRef CAS PubMed.
- Q. Peng, K. Mahmood, Y. Wu, L. Wang, Y. Liang, J. Shen and Z. Liu, Green Chem., 2014, 16, 2234–2242 RSC.
- A. Sankri, A. Arhaliass, I. Dez, A. C. Gaumont, Y. Grohens, D. Lourdin, I. Pillin, A. Rolland-Sabaté and E. Leroy, Carbohydr. Polym., 2010, 82, 256–263 CrossRef CAS PubMed.
- Q. Liu, M. H. A. Janssen, F. van Rantwijk and R. A. Sheldon, Green Chem., 2005, 7, 39–42 RSC.
- A. Biswas, R. L. Shogren, D. G. Stevenson, J. L. Willett and P. K. Bhowmik, Carbohydr. Polym., 2006, 66, 546–550 CrossRef CAS PubMed.
- H. Zhao, G. A. Baker, Z. Song, O. Olubajo, T. Crittle and D. Peters, Green Chem., 2008, 10, 696–705 RSC.
- A. Lehmann, B. Volkert, M. Hassan-Nejad, T. Greco and H.-P. Fink, Green Chem., 2010, 12, 2164–2171 RSC.
- M. Abe, Y. Fukaya and H. Ohno, Green Chem., 2010, 12, 1274–1280 RSC.
- K. Lappalainen, J. Kärkkäinen and M. Lajunen, Carbohydr. Polym., 2013, 93, 89–94 CrossRef CAS PubMed.
- K. Lappalainen, J. Kärkkäinen, J. Panula-Perälä and M. Lajunen, Starch/Staerke, 2012, 64, 263–271 CrossRef CAS PubMed.
- X. Lu, Z. Luo, S. Yu and X. Fu, J. Agric. Food Chem., 2012, 60, 9273–9279 CrossRef CAS PubMed.
- W. Xie, Y. Zhang and Y. Liu, Carbohydr. Polym., 2011, 85, 792–797 CrossRef CAS PubMed.
- R. L. Shogren and A. Biswas, Carbohydr. Polym., 2010, 81, 149–151 CrossRef CAS PubMed.
- S. Mateyawa, D. F. Xie, R. W. Truss, P. J. Halley, T. M. Nicholson, J. L. Shamshina, R. D. Rogers, M. W. Boehm and T. McNally, Carbohydr. Polym., 2013, 94, 520–530 CrossRef CAS PubMed.
- R. P. Swatloski, S. K. Spear, J. D. Holbrey and R. D. Rogers, J. Am. Chem. Soc., 2002, 124, 4974–4975 CrossRef CAS PubMed.
- A. Xu, J. Wang and H. Wang, Green Chem., 2010, 12, 268–275 RSC.
- H. Zhang, J. Wu, J. Zhang and J. He, Macromolecules, 2005, 38, 8272–8277 CrossRef CAS.
- J. Shen, L. Wang, Y. Men, Y. Wu, Q. Peng, X. Wang, R. Yang, K. Mahmood and Z. Liu, RSC Adv., 2015, 5, 60330–60338 RSC.
- Y. Men, X. Du, J. Shen, L. Wang and Z. Liu, Carbohydr. Polym., 2015, 121, 348–354 CrossRef CAS PubMed.
- L. Wang, J. Shen, Y. Men, Y. Wu, Q. Peng, X. Wang, R. Yang, K. Mahmood and Z. Liu, Polym. Chem., 2015, 6, 3480–3488 RSC.
- V. Percec, T. Guliashvili, J. S. Ladislaw, A. Wistrand, A. Stjerndahl, M. J. Sienkowska, M. J. Monteiro and S. Sahoo, J. Am. Chem. Soc., 2006, 128, 14156–14165 CrossRef CAS PubMed.
- K. Matyjaszewski, N. V. Tsarevsky, W. A. Braunecker, H. Dong, J. Huang, W. Jakubowski, Y. Kwak, R. Nicolay, W. Tang and J. A. Yoon, Macromolecules, 2007, 40, 7795–7806 CrossRef CAS.
- J. A. Syrett, M. W. Jones and D. M. Haddleton, Chem. Commun., 2010, 46, 7181–7183 RSC.
- S. L. Potisek, D. A. Davis, N. R. Sottos, S. R. White and J. S. Moore, J. Am. Chem. Soc., 2007, 129, 13808–13809 CrossRef CAS PubMed.
- J. O. Zoppe, Y. Habibi, O. J. Rojas, R. A. Venditti, L.-S. Johansson, K. Efimenko, M. Osterberg and J. Laine, Biomacromolecules, 2010, 11, 2683–2691 CrossRef CAS PubMed.
- J. Tom, B. Hornby, A. West, S. Harrisson and S. Perrier, Polym. Chem., 2010, 1, 420–422 RSC.
- Q. Yan, J. Yuan, F. Zhang, X. Sui, X. Xie, Y. Yin, S. Wang and Y. Wei, Biomacromolecules, 2009, 10, 2033–2042 CrossRef CAS PubMed.
- X. Sui, J. Yuan, M. Zhou, J. Zhang, H. Yang, W. Yuan, Y. Wei and C. Pan, Biomacromolecules, 2008, 9, 2615–2620 CrossRef CAS PubMed.
- L. Fan, H. Chen, Z. Hao and Z. Tan, J. Polym. Sci., Part A: Polym. Chem., 2013, 51, 457–462 CrossRef CAS PubMed.
- M. S. Hiltunen, J. Raula and S. L. Maunu, Polym. Int., 2011, 60, 1370–1379 CAS.
- J. Voepel, U. Edlund, A.-C. Albertsson and V. Percec, Biomacromolecules, 2011, 12, 253–259 CrossRef CAS PubMed.
- T. Meng, X. Gao, J. Zhang, J. Yuan, Y. Zhang and J. He, Polymer, 2009, 50, 447–454 CrossRef CAS PubMed.
- L. Ma, R. Liu, J. Tan, D. Wang, X. Jin, H. Kang, M. Wu and Y. Huang, Langmuir, 2010, 26, 8697–8703 CrossRef CAS PubMed.
- Q. Zhang, P. Wilson, Z. Li, R. McHale, J. Godfrey, A. Anastasaki, C. Waldron and D. M. Haddleton, J. Am. Chem. Soc., 2013, 135, 7355–7363 CrossRef CAS PubMed.
- J. Virtanen, M. Arotçaréna, B. Heise, S. Ishaya, A. Laschewsky and H. Tenhu, Langmuir, 2002, 18, 5360–5365 CrossRef CAS.
- M. Arotçaréna, B. Heise, S. Ishaya and A. Laschewsky, J. Am. Chem. Soc., 2002, 124, 3787–3793 CrossRef PubMed.
- W. Yin, H. Yanga and R. Cheng, Eur. Phys. J. E: Soft Matter Biol. Phys., 2005, 17, 1–5 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |