Spinel nickel ferrite nanoparticles strongly cross-linked with multiwalled carbon nanotubes as a bi-efficient electrocatalyst for oxygen reduction and oxygen evolution†
Pengxi Liab,
Ruguang Maacd,
Yao Zhouacd,
Yongfang Chenae,
Qian Liu*acd,
Guihua Pengb,
Zhenhua Liangb and
Jiacheng Wang*acd
aState Key Laboratory of High Performance Ceramics and Superfine Microstructure, Shanghai Institute of Ceramics, Chinese Academy of Sciences, 1295 Dingxi Road, Shanghai 200050, China. E-mail: qianliu@sunm.shcnc.ac.cn; jiacheng.wang@mail.sic.ac.cn
bState Key Laboratory Cultivation Base for the Chemistry and Molecular Engineering of Medicinal Resources, Ministry of Science and Technology of China, School of Chemistry & Pharmaceutical of Guangxi Normal University, Guilin 541004, Guangxi, China
cInnovation Center for Inorganic Materials Genomic Science, Shanghai Institute of Ceramics, Chinese Academy of Sciences, Shanghai 200050, P. R. China
dShanghai Institute of Materials Genome, Shanghai 200444, P. R. China
eUniversity of the Chinese Academy of Sciences, Beijing 100049, P. R. China
First published on 20th August 2015
Abstract
It is of great concern to explore new electrocatalysts for the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER). In this study, spinel NiFe2O4 nanoparticles cross-linked with the outer walls of multiwalled carbon nanotubes (MWCNTs) were successfully prepared by a simple, scalable hydrothermal method. The as-synthesized NiFe2O4/MWCNT nanohybrid shows not only a better ORR catalytic activity than pure NiFe2O4 and MWCNTs, but also a close four-electron reaction pathway. Meanwhile, the NiFe2O4/MWCNT nanohybrid exhibits a much higher OER catalytic activity when compared to NiFe2O4, MWCNTs and commercial Pt/C in terms of the onset potential and current density. Moreover, the NiFe2O4/MWCNT nanohybrid demonstrates the preeminent long-term durability measured by the current–time chronoamperometric test for both the ORR and OER, which evidently outperforms commercial Pt/C. The excellent bi-functional electrocatalytic activities of the NiFe2O4/MWCNT nanohybrid are attributed to the strong coupling between the NiFe2O4 nanoparticles and the MWCNTs as well as the network structure.
Introduction
Nowadays, rechargeable metal–air batteries have attracted more and more immediate attention due to their probable advantages like low-cost, environmental benignity and high energy density.1 Nevertheless, the sluggish kinetics of the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) has been the most critical issue that affects the cycling efficiency, thus impeding the development of metal–air batteries.2 Pt-based materials are usually used as the most reliable electrocatalysts to enhance the kinetics of the ORR and OER.3 However, they are very precious and have insufficient availability for extensive commercial production and application.4 Therefore, it is highly necessary to develop non-noble electrocatalysts as substitutes for the ORR and OER.Recently, researchers have made great efforts to search for non-precious metal catalysts as substitutes for Pt-catalysts, such as metal oxides and metal-free carbon materials.3c,5 Yeo et al. reported that gold-supported Co3O4 exhibited enhanced catalytic activities for the OER.6 Single crystalline NiO nanoflakes have been demonstrated to show significant ORR catalytic activities for metal–air batteries.7 In addition, N-doped carbon nanotubes as efficient and durable metal-free cathodic catalysts for the ORR have been reported by Feng and his co-workers.8 Among these non-precious catalysts, spinel oxides exhibit good catalytic activities for the ORR and OER, have low cost and have environmental friendliness. However, due to their poor electron conductivity, they are usually supported on a conducting carbon matrix aimed at speeding up electron transport. For example, Lou et al. reported the synthesis of NiCo2O4/reduced graphene oxide (rGO) hybrid nanosheets by a simple polyol process followed by thermal annealing treatment in air. The resulting nanocomposite exhibits a remarkable electrocatalytic activity and favorable kinetics for the ORR in alkaline electrolyte.9 The spinel ZnCo2O4/N-doped carbon nanotube composite prepared via a hydrothermal strategy demonstrates an estimable electrocatalytic activity for the ORR, attributed to the synergistic effect of ZnCo2O4 and the N-doped carbon nanotube.10 Dai and his co-workers studied the electrocatalytic performance of Ni–Fe layered double hydroxide on carbon nanotubes for water oxidation.5b Lee et al. have reported mesoporous NiCo2O4 nanoplatelets anchored on graphene as a bi-functional electrocatalyst for the ORR and OER.11 Prabu et al. synthesized CoMn2O4 nanoparticles anchored on nitrogen-doped graphene nanosheets as a bi-functional electrocatalyst for both the ORR and OER.12 Multiwalled carbon nanotubes (MWCNTs) possess many topological defects in the tube walls so that they have more active sites on the surface than other graphite carbon materials.13 To the best of our knowledge, NiFe2O4/MWCNT nanocomposites have been reported previously in the application of gas sensing,14 for the determination of the voltammetric behaviour of dopamine,15 and in magnetic hyperthermia,16 but their electrocatalytic activities for both the ORR and OER have been seldom reported.
In this paper, we exhibit spinel NiFe2O4 nanoparticles cross-linked by multiwalled carbon nanotubes (MWCNTs) via a simple, scalable hydrothermal method. The electrocatalytic activities of the NiFe2O4/MWCNT nanohybrid in alkaline solution were systematically investigated by using a rotating disk electrode (RDE) and rotating ring-disk electrode (RRDE). During the oxygen reduction reaction (ORR) process, the NiFe2O4/MWCNT nanohybrid shows a more positive onset potential and higher current density than pure NiFe2O4 and MWCNTs. For the oxygen evolution reaction (OER), the NiFe2O4/MWCNT nanohybrid exhibits a better catalytic activity than NiFe2O4, MWCNTs and commercial Pt/C, in view of the onset potential and current density. Moreover, the NiFe2O4/MWCNT nanohybrid demonstrates superior durability on the basis of the current–time chronoamperometric test for both the ORR and OER.
Experimental section
Oxidative treatment of MWCNTs
To acquire water-soluble MWCNTs, pristine MWCNTs were subjected to oxidative treatment according to the reported method.17 Briefly, 1 g of MWCNTs was suspended in 80 mL of a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of concentrated H2SO4/HNO3, sonicated for 1 h and then transferred into a Teflon-lined stainless autoclave and heated to 100 °C for 12 h. After cooling, the resulting product was collected by filtration, washed with water and ethanol three times, and then dried at 100 °C overnight.
1 mixture of concentrated H2SO4/HNO3, sonicated for 1 h and then transferred into a Teflon-lined stainless autoclave and heated to 100 °C for 12 h. After cooling, the resulting product was collected by filtration, washed with water and ethanol three times, and then dried at 100 °C overnight.
Preparation of the NiFe2O4/MWCNT nanohybrid
The NiFe2O4/MWCNT nanohybrid was synthesized via a general hydrothermal approach. In a typical synthesis procedure, 0.04 g of MWCNTs was dissolved in a mixture of H2O (40 mL) and EtOH (20 mL) and subjected to ultrasonication for 120 min to form a homogenous solution. Then, 0.582 g of Ni(NO3)2·6H2O, 1.616 g of Fe(NO3)3·9H2O (the molar ratio of Ni/Fe = 1/2) and 0.4 g of polyvinylpyrrolidone (PVP) were dissolved under magnetic stirring for 1 h. Subsequently, 6 mL NH4OH was added drop by drop under drastic stirring for 1 h, which was used as a precipitation reagent. The mixed solution was transferred into a Teflon-lined stainless autoclave and heated to 180 °C for 12 h. After the black product was collected by filtering, washed with ethanol and deionized water, and dried under vacuum, it was then annealed at 400 °C in nitrogen for 4 h to dislodge the residuary PVP molecules in the nanohybrid. The pure NiFe2O4 nanoparticles were prepared using the same procedure in the absence of MWCNTs.Characterization
The crystal structures of the as-synthesized samples were investigated using an X-ray powder diffraction (XRD) diffractometer (D8 ADVANCE) with Cu Kα radiation (40 kV and 200 mA). Transmission electron microscopy (TEM) images were obtained on a JEM-2100F transmission electron microscope. Thermogravimetric (TG) analysis was performed on a thermal analysis instrument (STA449C) in an air flow within a temperature range of 30 to 800 °C at a heating rate of 10 °C min−1, aimed at confirming the amount of MWCNTs in the NiFe2O4/MWCNT nanohybrid. Surface analysis of the samples was performed with an X-ray photoelectron spectroscopy spectrometer (XPS, ESCALAB 250 X-ray photoelectron spectrometer) with Al Kα radiation. Raman spectra were recorded on a DXR Raman Microscope (Thermal Scientific Co., USA) with a 532 nm excitation length.Electrode preparation and electrochemical measurements
The electrochemical activities of the materials were measured on a PINE instrument using a conventional three electrode system with 0.1 M KOH aqueous solution via rotating disk electrode (RDE) voltammetry, rotating ring-disk electrode (RRDE) voltammetry and cyclic voltammetry (CV) for the ORR. The Ag/AgCl electrode and Pt flake electrode were used as the reference and counter electrodes, respectively. The glassy carbon electrode (GCE, diameter = 5 mm) was used as the working electrode. The RRDE electrode consists of a GC disk (0.2475 cm2 of geometric surface area), surrounded by a Pt ring (0.1866 cm2 of geometric surface area). The active sample (5 mg) was dispersed in 1 mL of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) water/ethanol mixed solution containing 30 μL of Nafion solution (5 wt%) by sonication for about 2 h to form a homogeneous ink. Then, 20 μL of the ink was pipetted onto a GCE, yielding a catalyst level of 0.64 mg cm−2. The electrode with the catalyst was dried at 50 °C, and was used as the working electrode for further electrochemical measurements. Commercial 20 wt% Pt/C (Johnson Matthey) was used for comparison.
1 (v/v) water/ethanol mixed solution containing 30 μL of Nafion solution (5 wt%) by sonication for about 2 h to form a homogeneous ink. Then, 20 μL of the ink was pipetted onto a GCE, yielding a catalyst level of 0.64 mg cm−2. The electrode with the catalyst was dried at 50 °C, and was used as the working electrode for further electrochemical measurements. Commercial 20 wt% Pt/C (Johnson Matthey) was used for comparison.
For the ORR test, the electrolyte (0.1 M KOH) was purged with high-purity O2 gas to ensure O2 saturation. The CV measurement was performed by sweeping the potential from 0 V to −0.9 V at a scan rate of 50 mV s−1. The LSV tests were carried out by sweeping the potential from 0 V to −0.9 V at a scan rate of 10 mV s−1, with the electrode rotating at 400, 625, 900, 1225, 1600, and 2025 rpm. The kinetic parameters of the ORR tests could be determined by the Koutecky–Levich equation (eqn (1)):
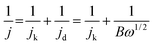 | (1) |
| B = 0.2nFCO2DO22/3υ−1/6 | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), DO2 is the diffusion coefficient of oxygen (DO2 = 1.86 × 10−5 cm2 s−1), ν is the kinetics viscosity of the solution (ν = 0.01 cm−2 s−1), CO2 is the bulk concentration of O2 dissolved in the electrolyte (CO2 = 1.21 × 10−6 mol cm−3).
485 C mol−1), DO2 is the diffusion coefficient of oxygen (DO2 = 1.86 × 10−5 cm2 s−1), ν is the kinetics viscosity of the solution (ν = 0.01 cm−2 s−1), CO2 is the bulk concentration of O2 dissolved in the electrolyte (CO2 = 1.21 × 10−6 mol cm−3).
For the RRDE test, the ring potential was held at 0.2 V (vs. Ag/AgCl) at a scanning rate of 10 mV s−1. The percentage of H2O2 and the transferred electron number during the ORR process can be calculated via the following equations (eqn (3) and (4)):
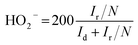 | (3) |
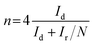 | (4) |
For the OER test, the electrolyte was purged with high-purity N2 gas to spin off the O2 and ensure N2 saturation during the voltammetry testing. The LSV measurements were performed at a scan rate of 10 mV s−1 and a rotating speed of 1600 rpm.
Results and discussion
TG measurements were performed to confirm the content of the MWCNTs in the NiFe2O4/MWCNT nanohybrid. Fig. 1 shows the TG curve of the NiFe2O4/MWCNT powder measured in an air flow from 30 to 800 °C. It can be clearly observed that there is a weight decrease of about 1.4 wt% from 30 to 405 °C, which is ascribed to the desorption of physically absorbed water as well as the decomposition of the hydroxide compounds in the precursor. The weight loss of about 5.4 wt% from 405 to 625 °C is ascribed to the combustion loss of the MWCNTs in the NiFe2O4/MWCNT nanohybrid.The XRD profiles of NiFe2O4 and NiFe2O4/MWCNT are shown in Fig. 2. The characteristic peaks of the NiFe2O4/MWCNT nanohybrid and pure NiFe2O4 can be well indexed as cubic spinel phase (PDF#54-0964) with no collateral peaks. Furthermore, the intense and sharp peaks indicate that both the pure NiFe2O4/MWCNT nanohybrid and pure NiFe2O4 are well crystallized. No additional peaks ascribed to the MWCNTs were observed due to a very low content of the MWCNTs in the nanohybrid.
The morphology and microstructure of the as-synthesized NiFe2O4/MWCNT nanohybrid were investigated using transmission electron microscopy (TEM). By using the one-pot hydrothermal treatment, the NiFe2O4 nanoparticles grew successfully and were homogeneously dispersed on the MWCNTs, as is evidenced by the low-magnification TEM image (Fig. 3a). The large length to diameter ratio of the MWCNTs results in the evident aggregation of the final hybrid. This unique network structure can create more active sites and allows the easy transport of O2 and the electrolyte in the NiFe2O4/MWCNT nanohybrid during the electrocatalysis process. The high-magnification TEM image (Fig. 3b) further reveals that these NiFe2O4 nanoparticles dotted on the outer walls of the MWCNTs have typical sizes of about 10–20 nm. Meanwhile, high-resolution TEM (HR-TEM) can provide more information about the crystal structure of the NiFe2O4 nanoparticles and the contacting interface of the NiFe2O4 nanoparticles and MWCNTs (Fig. 3c and d). The measured d spacing of 0.18 nm is labelled as the lattice spacing of the (400) plane of NiFe2O4 and the lattice spacing about 0.33 nm is consistent with the interlayer distance of graphite (Fig. 3d). It is clear that the NiFe2O4 nanocrystals are closely cross-linked with the MWCNTs, resulting in more disordered crystal planes for NiFe2O4 and the MWCNTs at the interfaces. This disordered structure could lead to the formation of more defects, which could be beneficial for adsorbing and activating oxygen molecules and thus improves the electrocatalytic activity for the ORR and OER.
The surface chemical composition and cation oxidation states of NiFe2O4 and the NiFe2O4/MWCNT nanohybrid were characterized by X-ray photoelectron spectroscopy (XPS). As expected, the XPS survey spectra in Fig. 4a exhibits Ni 2p, Fe 2p, O 1s and C 1s peaks for both pristine NiFe2O4 and the NiFe2O4/MWCNT nanohybrid. The de-convolution of the Ni 2p peak of NiFe2O4 and the NiFe2O4/MWCNT nanohybrid shows four peaks (Fig. 4b). In the pristine NiFe2O4 sample, the peaks at a binding energy of 855.9 and 873.6 eV correspond to Ni 2p3/2 and Ni 2p1/2, respectively.18 The satellite peaks at around 862.5 and 880.7 are two shake-up type peaks of Ni at the high binding energy side of the Ni 2p3/2 and Ni 2p1/2 edge. The Ni 2p3/2 and Ni 2p1/2 main peaks and satellite peaks demonstrate the presence of Ni2+cations in NiFe2O4. Nevertheless, after the hybridization of the NiFe2O4 nanoparticles with MWCNTs via in situ hydrothermal growth, there is an up-shift of ca. 3.5 eV for Ni 2p3/2 and 1.2 eV for Ni 2p1/2 at the binding energy of the peaks for the NiFe2O4/MWCNT nanohybrid compared with those for pristine NiFe2O4. This blue-shift of the binding energies for the Ni 2p peaks reveals the strong coupling between the NiFe2O4 nanoparticles and the MWCNTs in the nanohybrid possibly via Ni–O–C bonds during the hydrothermal treatment. The Fe 2p spectra are shifted into two peaks (Fig. 4c). The peaks at a binding energy of around 711.5 and 725.1 eV are assigned to Fe 2p3/2 and Fe 2p1/2, respectively, suggesting the presence of Fe3+cations in the as-synthesized samples.19 Meanwhile, no shift in the binding energy of the Fe 2p XPS peaks is exhibited after the hybridization of NiFe2O4 and the MWCNTs when comparing the Fe 2p spectra of NiFe2O4 and the NiFe2O4/MWCNT nanohybrid. As shown in Fig. 4d, the O 1s spectrum of NiFe2O4 shows a peak at 530.4 eV,20 corresponding to the lattice oxygen in the Ni/Fe–O framework while a positive shift of about 0.4 eV in the binding energy for the O 1s peak is observed for the NiFe2O4/MWCNT nanohybrid. It also indicates the strong coupling via Ni–O–C bonds between NiFe2O4 and the MWCNTs in the NiFe2O4/MWCNT nanohybrid.
 | ||
| Fig. 4 (a) Full XPS spectra and (b–d) high-resolution Ni 2p (b), Fe 2p (c), and O 1s XPS spectra of NiFe2O4 and the NiFe2O4/MWCNT nanohybrid. | ||
Raman spectroscopy also provides more information about the structural properties of the as-prepared NiFe2O4/MWCNT nanohybrid. As displayed in Fig. 5, the characteristic D and G bands of carbon materials locate at around 1349 and 1582 cm−1, respectively, for pure MWCNTs. The D band is a double-resonance Raman mode, which can be understood as a measurement of structural disorder coming from amorphous carbon and any defects.21 The G band originates from the tangential in-plane stretching vibrations of the carbon–carbon bonds within the graphitic sheets.22 The second order 2D band at 2693 cm−1 is activated independently of defects through a two-phonon double resonance process, which is responsible for its dispersive nature and sensitivity to the structural ordering of the tube walls and the graphitic electronic structure.23 For the NiFe2O4/MWCNT nanohybrid, all of the D, G and 2D bands show a red-shift, compared with pure MWCNTs. It could indicate the existence of the coupling between NiFe2O4 and the MWCNTs, in good agreement with the results of HR-TEM and XPS. Furthermore, the ratio of the intensities of the D and G bands, R = ID/IG, can be used to evaluate the disorder density of carbon materials.24 The ID/IG ratios for the MWCNTs and NiFe2O4/MWCNT are calculated to be 0.16 and 0.26, respectively. The ID/IG ratio increases after the MWCNTs were hybridized with NiFe2O4, which reveals that the disorder density and defect of the graphitic carbon sheets increase accordingly.
The ORR activity of the NiFe2O4/MWCNT nanohybrid as measured with CV and RDE in a conventional three-electrode system is shown in Fig. 6. In order to understand the electrocatalytic performance of NiFe2O4/MWCNT during the ORR process, the ORR activities of pure NiFe2O4 and the MWCNTs are also included for comparison.
Fig. 6a demonstrates the CV curves of the NiFe2O4/MWCNT nanohybrid in N2 and O2-saturated 0.1 M KOH solution. The NiFe2O4/MWCNT nanohybrid shows no redox peak in the potential range from 0 to −0.9 V (vs. Ag/AgCl) in N2-saturated 0.1 M KOH solution. However, a reduction peak corresponding to the ORR at −0.35 V (vs. Ag/AgCl) can be observed in O2-saturated solution, indicating the occurrence of the ORR on the surface of the NiFe2O4/MWCNT nanohybrid. Fig. 6b exhibits the LSVs of the MWCNTs, NiFe2O4 and the NiFe2O4/MWCNT nanohybrid in O2-saturated 0.1 M KOH solution at a rotation rate of 1600 rpm. The NiFe2O4/MWCNT nanohybrid shows a higher diffusion-limiting current density and more positive onset potential than pure NiFe2O4 nanoparticles and MWCNTs. Due to the high electric conductivity of MWCNTs, the hybridization of the NiFe2O4 nanoparticles with MWCNTs can endow the resulting nanohybrid with high electric conductivity, which is favourable for increasing the ORR activity. The ORR activity of the NiFe2O4/MWCNT nanohybrid is enhanced compared to those of pure MWCNTs and NiFe2O4, also suggesting the synergistic effect of the two components in the nanohybrid is possibly ascribed to the strong coupling between NiFe2O4 and the MWCNTs. The corresponding Tafel plots for the samples in the low overpotential region are shown in Fig. 6c. The NiFe2O4/MWCNT nanohybrid shows a smaller Tafel slope (93 mV per dec) than pure NiFe2O4 (115 mV per dec) and the MWCNTs (101 mV per dec), which can indicate the enhanced ORR kinetics after the hybridization of the NiFe2O4 nanoparticles with the MWCNTs. The LSVs for the ORR of the NiFe2O4/MWCNT nanohybrid at different rotation rates are shown in Fig. 6d. As can be seen, the diffusion limiting currents are improved with the increasing rotation rates. Fig. 6e demonstrates the corresponding Koutecky–Levich plots obtained from the inverse current density (j−1) as a function of the inverse of the square root of the rotation rate (ω−1/2) for NiFe2O4/MWCNT at −0.53, −0.56, −0.59 and −0.62 V (vs. Ag/AgCl), respectively. The plots are almost parallel and linear, indicating the first-order dependence of the kinetics of the ORR of the NiFe2O4/MWCNT surface. Fig. 6f shows the transferred electron number n of the MWCNTs, NiFe2O4 and NiFe2O4/MWCNT based on the LSVs (Fig. 6d and S1†) in the potential range of −0.53–−0.62 V (vs. Ag/AgCl). The transferred electron number n of the MWCNTs and NiFe2O4 is in the range of 2.34–3.12 and 2.20–2.62, respectively. For NiFe2O4/MWCNT, the transferred electron number n is in the range of 3.95–4.00. These results indicate that the integration of NiFe2O4 and the MWCNTs could not only significantly improve the electrocatalytic activity of NiFe2O4/MWCNT but also obtain the close 4e− ORR catalytic pathway to obtain the maximum energy capacity.
In order to further confirm the catalytic activities and reaction pathways of the NiFe2O4/MWCNT nanohybrid for the ORR, the LSVs of NiFe2O4/MWCNT and Pt /C (Fig. 7a) were further obtained by the rotating ring-disk electrode (RRDE) in O2-saturated 0.1 M KOH solution at a rotation rate of 1600 rpm. The ring potential was held at 0.2 V (vs. Ag/AgCl). The formation of peroxide species (HO2−) and the electron transferred number n based on the corresponding RRDE data during the ORR procedure are shown in Fig. 7b. The measured HO2− yield of the NiFe2O4/MWCNT nanohybrid and commercial Pt/C is 15.7–19.7% and 7.17–7.36%, respectively, over the potential range from −0.53 to −0.62 V (vs. Ag/AgCl). The calculated electron transferred numbers n for the NiFe2O4/MWCNT nanohybrid and Pt/C are 3.61–3.68 and 3.84–3.85, respectively. It suggests that an intrinsic four-electron pathway for the ORR by the NiFe2O4/MWCNT nanohybrid is the dominant mechanism. This result is consistent with that calculated from the slopes of the K–L plots.
The electrocatalytic activity of the NiFe2O4/MWCNT nanohybrid as well as the control catalysts for the OER was also measured. The anodic LSV curves for the OER by various electrocatalysts collected in N2-saturated 0.1 M KOH solution at a rotation speed of 1600 rpm are shown in Fig. 8a. Note that the onset potential of the NiFe2O4/MWCNT nanohybrid (0.42 V vs. Ag/AgCl) is smaller than those of pure NiFe2O4 (0.56 V), the MWCNTs (0.49 V) and commercial Pt/C (0.54 V). Furthermore, the current density of NiFe2O4/MWCNT reaches 39.5 mA cm−2 at 1.0 V, which is much larger than those of NiFe2O4 (11.7 mA cm−2), the MWCNTs (23.7 mA cm−2) and commercial Pt/C (27.8 mA cm−2). The OER kinetics was investigated via the Tafel plots in the low over-potential region (Fig. 8b). At low over-potentials, the Tafel slope is 93, 148, 112 and 105 mV per dec for the NiFe2O4/MWCNT nanohybrid, NiFe2O4, the MWCNTs and commercial Pt/C, respectively. It implies a much faster OER activity of the NiFe2O4/MWCNT nanohybrid by the Tafel slopes than those for pristine NiFe2O4, the MWCNTs, and commercial Pt/C. These results clearly indicate that the NiFe2O4/MWCNT nanohybrid possesses a much higher OER electrocatalytic activity than NiFe2O4 and the MWCNTs, which is mainly ascribed to the unique network structures of the hybrids and the strong coupling and synergistic effect between NiFe2O4 nanoparticles and the MWCNT matrix. The present NiFe2O4/MWCNT nanohybrid (0.42 V and 24.5 mA cm−2) significantly outperforms the previously reported catalysts (e.g. Co3O4/graphene (0.54 V and 7.3 mA cm−2),25 NiCo2O4/graphene (0.55 V and 19.8 mA cm−2),26 CoFe2O4/graphene (0.54 V and 13.6 mA cm−2),27 and CoFe2O4/biocarbon (0.48 V and 17.7 mA cm−2))19a in terms of the onset potentials and limiting current density at 0.8 V (Table S1†), implying the very high excellence of MWCNTs as a matrix to cross-link transition metal oxide nanoparticles as highly efficient electrocatalysts.
Another main challenge for metal–air battery development is the durability of the catalysts. The stability of NiFe2O4/MWCNT and commercial Pt/C for the ORR and OER was examined by the chronoamperometric method in O2-saturated and N2-statured 0.1 M KOH solution, respectively, at a rotation rate of 1600 rpm. As shown in Fig. 9a, the ORR current density of NiFe2O4/MWCNT decreases by about 13.1% at a constant potential of −0.6 V (vs. Ag/AgCl) over 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s of continuous operation, while the ORR current density of commercial Pt/C decreases by 29.9% after 20
000 s of continuous operation, while the ORR current density of commercial Pt/C decreases by 29.9% after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s. For the OER durability (Fig. 9b), the current density of the NiFe2O4/MWCNT nanohybrid decreases by only 15.9% while commercial Pt/C decreases by about 34.5% at a constant potential of 0.8 V after 20
000 s. For the OER durability (Fig. 9b), the current density of the NiFe2O4/MWCNT nanohybrid decreases by only 15.9% while commercial Pt/C decreases by about 34.5% at a constant potential of 0.8 V after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s. These results indicate that the NiFe2O4/MWCNT nanohybrid has long-time stability for both the ORR and OER. The strong coupling between the NiFe2O4 nanoparticles and the MWCNTs can prevent the detachment and aggregation of the NiFe2O4 nanoparticles during the catalytic process, which enhances the cycling stability of the electrode.
000 s. These results indicate that the NiFe2O4/MWCNT nanohybrid has long-time stability for both the ORR and OER. The strong coupling between the NiFe2O4 nanoparticles and the MWCNTs can prevent the detachment and aggregation of the NiFe2O4 nanoparticles during the catalytic process, which enhances the cycling stability of the electrode.
Conclusions
In summary, NiFe2O4 nanoparticles were successfully cross-linked with the outer walls of multiwalled carbon nanotubes (MWCNTs) via a simple and scalable hydrothermal procedure. The particle size of the NiFe2O4 nanoparticles coupled with the MWCNTs was about 10–20 nm. The final NiFe2O4/MWCNT composite demonstrates a unique network structure, affording more active sites and allowing the easy transport of O2 and electrolyte during the catalytic process. High-resolution TEM, X-ray photoelectron spectroscopy and Raman analysis confirmed the existence of the strong coupling between the NiFe2O4 nanoparticles and the MWCNTs. The results of the electrochemical measurements indicate that NiFe2O4/MWCNT possess promising activity for both the ORR and OER, in terms of the onset potential and current density. The hybridization of NiFe2O4 nanoparticles with MWCNTs also results in a close four-electron reaction pathway for the ORR, implying that the maximum energy efficiency could be obtained. Moreover, the NiFe2O4/MWCNT nanohybrid demonstrates superior durability investigated by the current–time chronoamperometric test for both the ORR and OER when compared to commercial Pt/C. The excellent catalytic activities and stabilities of the NiFe2O4/MWCNT nanohybrid are mainly owing to the strong coupling and synergistic effect between the NiFe2O4 nanoparticles and the MWCNT matrix, as well as the network structure.Acknowledgements
The authors thank the financial support from the Shanghai Institute of Ceramics, the Key Project for Young Researcher of State Key Laboratory of High Performance Ceramics and Superfine Microstructure, the One Hundred Talent Plan of Chinese Academy of Sciences, the National Natural Science Foundation of China (No. 21307145), the Youth Science and Technology Talents “Sail” Program of Shanghai Municipal Science and the Technology Commission (No. 15YF1413800), and the Research Grant (No.14DZ2261200) from the Shanghai Government.Notes and references
- (a) L. Hu, H. Zhong, X. Zheng, Y. Huang, P. Zhang and Q. Chen, Sci. Rep., 2012, 2, 986 Search PubMed; (b) B. Guan, D. Guo, L. Hu, G. Zhang, T. Fu, W. Ren, J. Li and Q. Li, J. Mater. Chem. A, 2014, 2, 16116 RSC; (c) J. Kim, X. Yin, K. C. Tsao, S. Fang and H. Yang, J. Am. Chem. Soc., 2014, 136, 14646 CrossRef CAS PubMed.
- (a) Y. C. Lu, D. G. Kwabi, K. P. C. Yao, J. R. Harding, J. Zhou, L. Zuin and Y. Shao-Horn, Energy Environ. Sci., 2011, 4, 2999 RSC; (b) Y. C. Lu, Z. C. Xu, H. A. Gasteiger, S. Chen, K. Hamad-Schifferli and Y. Shao-Horn, J. Am. Chem. Soc., 2010, 132, 12170 CrossRef CAS PubMed; (c) H. Xia, Y. Wan, W. Assenmacher, W. Mader, G. Yuan and L. Lu, NPG Asia Mater., 2014, 6, e126 CrossRef CAS PubMed.
- (a) L. Ma, S. Sui and Y. Zhai, J. Power Sources, 2008, 177, 470 CrossRef CAS PubMed; (b) D. Yu, Y. Xue and L. Dai, J. Phys. Chem. Lett., 2012, 3, 2863 CrossRef CAS; (c) R. M. P. Li, Y. Zhou, Y. Chen, Z. Zhou, G. Liu, Q. Liu, G. Peng and J. Wang, RSC Adv., 2015, 5, 44476 RSC.
- (a) C. T. Hung, N. Yu, C. T. Chen, P. H. Wu, X. Han, Y. S. Kao, T. C. Liu, Y. Chu, F. Deng, A. Zheng and S.-B. Liu, J. Mater. Chem. A, 2014, 2, 20030 RSC; (b) J. Y. Choi, R. S. Hsu and Z. W. Chen, J. Phys. Chem. C, 2010, 114 CAS; (c) L. Wang, F. Yin and C. Yao, Int. J. Hydrogen Energy, 2014, 39, 15913 CrossRef CAS PubMed.
- (a) X. Han, T. Zhang, J. Du, F. Cheng and J. Chen, Chem. Sci., 2013, 4, 368 RSC; (b) M. Gong, Y. Li, H. Wang, Y. Liang, J. Z. Wu, J. Zhou, J. Wang, T. Regier, F. Wei and H. Dai, J. Am. Chem. Soc., 2013, 135, 219 Search PubMed; (c) J. Wang, R. Ma, Y. Zhou and Q. Liu, J. Mater. Chem. A, 2015, 3, 12836 RSC; (d) J. A. Koza, Z. He, A. S. Miller and J. A. Switzer, Chem. Mater., 2012, 24, 3567 CrossRef CAS.
- B. S. Yeo and A. T. Bell, J. Am. Chem. Soc., 2011, 133, 5587 CrossRef CAS PubMed.
- S. Ci, J. Zou, G. Zeng, Q. Peng, S. Luo and Z. Wen, RSC Adv., 2012, 2, 5185 RSC.
- L. Feng, Y. Yan, Y. Chen and L. Wang, Energy Environ. Sci., 2011, 4, 1892 CAS.
- G. Zhang, B. Y. Xia, X. Wang and X. W. Lou, Adv. Mater., 2013, 26, 2408 CrossRef PubMed.
- Z. Pu, Q. Liu, C. Tang, A. M. Asiri, A. H. Qusti, A. O. Al-Youbi and X. Sun, J. Power Sources, 2014, 257, 170 CrossRef CAS PubMed.
- D. U. Lee, B. J. Kim and Z. Chen, J. Mater. Chem. A, 2013, 1, 4754 CAS.
- M. Prabu, P. Ramakrishnan and S. Shanmugam, Electrochem. Commun., 2014, 41, 59 CrossRef CAS PubMed.
- (a) W. Xiong, F. Du, F. Liu, A. Perez, M. Supp, T. S. Ramakrishnan, L. Dai and L. Jiang, J. Am. Chem. Soc., 2010, 132, 15839 CrossRef CAS PubMed; (b) Y. Tang, B. L. Allen, D. R. Kauffman and A. Star, J. Am. Chem. Soc., 2009, 131, 13200 CrossRef CAS PubMed; (c) W. Zhang, A. U. Shaikh, E. Y. Tsui and T. M. Swager, Chem. Mater., 2009, 21, 3234 CrossRef CAS; (d) Y. Liang, H. Wang, P. Diao, W. Chang, G. Hong, Y. Li, M. Gong, L. Xie, J. Zhou, J. Wang, T. Z. Regier, F. Wei and H. Dai, J. Am. Chem. Soc., 2012, 134, 15849 CrossRef CAS PubMed.
- R. Hajihashemi, A. M. Rashidi, M. Alaie, R. Mohammadzadeh and N. Izadi, Mater. Sci. Eng., C, 2014, 44, 417 CrossRef CAS PubMed.
- A. A. Ensafi, B. Arashpour, B. Rezaei and A. R. Allafchian, Mater. Sci. Eng., C, 2014, 39, 78 CrossRef CAS PubMed.
- K. Stojak Repa, D. Israel, J. Alonso, M. H. Phan, E. M. Palmero, M. Vazquez and H. Srikanth, J. Appl. Phys., 2015, 117, 17C723 CrossRef PubMed.
- J. Zhang, H. Zou, Q. Qing, Y. Yang, Q. Li, Z. Liu, X. Guo and Z. Du, J. Phys. Chem. B, 2003, 107, 3712 CrossRef CAS.
- (a) C. Jin, F. Lu, X. Cao, Z. Yang and R. Yang, J. Mater. Chem. A, 2013, 1, 12170 RSC; (b) M. Fu, Q. Jiao and Y. Zhao, J. Mater. Chem. A, 2013, 1, 5577 RSC.
- (a) S. Liu, W. Bian, Z. Yang, J. Tian, C. Jin, M. Shen, Z. Zhou and R. Yang, J. Mater. Chem. A, 2014, 2, 18012 RSC; (b) W. Bian, Z. Yang, P. Strasser and R. Yang, J. Power Sources, 2014, 250, 196 CrossRef CAS PubMed.
- L. Liu, L. Sun, J. Liu, X. Xiao, Z. Hu, X. Cao, B. Wang and X. Liu, Int. J. Hydrogen Energy, 2014, 39, 11258 CrossRef CAS PubMed.
- J. By A, R. Saito, G. Dresselhaus and M. S. Dresselhaus, Philos. Trans. R. Soc. London, Ser. A, 2004, 362, 2311 CrossRef PubMed.
- M. S. Dresselhaus, G. Dresselhaus, R. Saito and A. Jorio, Phys. Rep., 2005, 409, 47 CrossRef PubMed.
- (a) M. A. Pimenta, G. Dresselhaus, M. S. Dresselhaus, L. G. Cancado, A. Jorio and R. Saito, Phys. Chem. Chem. Phys., 2007, 9, 1276 RSC; (b) P. Delhaes, M. Couzi, M. Trinquecoste, J. Dentzer, H. Hamidou and C. Vix-Guterl, Carbon, 2006, 44, 3005 CrossRef CAS PubMed.
- S. Osswald, M. Havel and Y. Gogotsi, J. Raman Spectrosc., 2007, 38, 728 CrossRef CAS PubMed.
- C. Sun, F. Li, C. Ma, Y. Wang, Y. Ren, W. Yang, Z. Ma, J. Li, Y. Chen, Y. Kim and L. Chen, J. Mater. Chem. A, 2014, 2, 7188 CAS.
- D. U. Lee, B. J. Kim and Z. Chen, J. Mater. Chem. A, 2013, 1, 4754 CAS.
- W. Bian, Z. Yang, P. Strasser and R. Yang, J. Power Sources, 2014, 250, 196 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Additional figures and table mentioned in the text. See DOI: 10.1039/c5ra14713a |
| This journal is © The Royal Society of Chemistry 2015 |








