Advanced treatment of acrylic fiber manufacturing wastewater with a combined microbubble-ozonation/ultraviolet irradiation process
Tianlong Zhenga,
Tao Zhangb,
Qunhui Wang*ac,
Yanli Tiana,
Zhining Shid,
Nicholas Smalee and
Banghua Xua
aDepartment of Environmental Engineering, University of Science and Technology Beijing, 30 Xueyuan Road, Haidian District, Beijing 100083, China. E-mail: wangqh59@sina.com; Fax: +86-010-62332778; Tel: +86-010-62332778
bWater Desalination and Reuse Center, King Abdullah University of Science and Technology, Thuwal 4700, Saudi Arabia
cBeijing Key Laboratory of Resource-oriented Treatment of Industrial Pollutants, University of Science and Technology Beijing, 30 Xueyuan Road, Haidian District, Beijing 100083, China
dSchool of Earth and Environmental Sciences, The University of Adelaide, South Australia 5005, Australia
eThe Bionics Institute, Victoria, Australia 3002
First published on 9th September 2015
Abstract
This work investigated the effectiveness of a combination of microbubble-ozonation and ultraviolet (UV) irradiation for the treatment of secondary wastewater effluent of a wet-spun acrylic fiber manufacturing plant. Under reactor condition (ozone dosage of 48 mg L−1, UV fluence rate of 90 mW cm−2, initial pH of 8.0, and reaction time of 120 min), the biodegradability (represented as BOD5/CODcr) of the wastewater improved from 0.18 to 0.47. This improvement in biodegradability is related to the degradation of alkanes, aromatic compounds, and other bio-refractory organic compounds. The combination of microbubble-ozonation and UV irradiation synergistically improved treatment efficiencies by 228%, 29%, and 142% for CODcr, UV254 removal and BOD5/CODcr respectively after 120 min reaction time, as compared with the sum efficiency of microbubble-ozonation alone and UV irradiation alone. Hydroxyl radical production in the microbubble-ozonation/UV process was about 1.8 times higher than the sum production in microbubble-ozonation alone and UV irradiation alone. The ozone decomposition rate in the combined process was about 4.1 times higher than that in microbubble-ozonation alone. The microbubble-ozonation/UV process could be a promising technique for the treatment of bio-refractory organics in the acrylic fiber manufacturing industry.
1. Introduction
Wastewater produced in wet-spun acrylic fiber manufacturing industry contains numerous bio-refractory organic pollutants such as organic nitriles, alkanes, aromatic compounds, esters, phenols, and amides.1 If not properly handled, such wastewater can severely pollute receiving waters. There is still no fully mature biological treatment technique for the comprehensive treatment of wet-spun acrylic fiber manufacturing wastewater to meet effluent discharge standards.The microbubble-ozonation technique has been widely used in the treatment of bio-refractory industrial wastewater.1–3 This technique has overcome some of the limiting factors of the traditional ozonation process such as low ozone dissolution and slow gas–liquid mass transfer rate.4 The generation of hydroxyl free radicals (˙OH) from ozone can improve the oxidation effects as ˙OH possesses a higher oxidation potential than molecular ozone.5 Hydroxyl radical generation can be improved during microbubble ozone intrusion.6 More recently, has been found that microbubble collapse in microbubble-ozonation also promotes hydroxyl radical production.7,8 However, the generation rate of hydroxyl radicals in the microbubble-ozonation process is still not high enough for efficient degradation of refractory organic pollutants especially when the concentration of these pollutants is relatively high. Ultraviolet (UV) irradiation enhances ˙OH production during ozonation treatment because of the photolysis of ozone.9–12 Microbubble and UV irradiation enhanced ozonation has not yet been investigated in the literature, and couple be a promising technology for advanced treatment of bio-refractory wastewater, especially for the wet-spun acrylic fiber manufacturing wastewater (having high concentrations refractory organic pollutants).
In this study, the microbubble-ozonation/UV combined process was applied to treat the secondary effluent of a wet-spun acrylic fiber manufacturing plant. We optimized reactor parameters (i.e., ozone dosage, UV fluence rate, initial pH and reaction time), and then, investigated removal rates of the contaminants and improvement of biodegradability under the optimized treatment condition. This new combined process was assessed by an experimentally determined synergistic effect, quantification of hydroxyl radical production, and ozone decomposition rate.
2. Experimental
2.1. Wastewater
The experimental wastewater was secondary effluent of an acrylic fiber manufacturing wastewater in Northern China. The wastewater was stored at 4 °C before use. Physical and chemical properties of the wastewater are shown in Table 1. This wastewater is characteristic in its complex organic components, high toxicity, and low biodegradability.| Parameter | CODcr (mg L−1) | BOD5 (mg L−1) | TOC (mg L−1) | NH3–N (mg L−1) | Total nitrogen (mg L−1) | UV254 (Abs per cm) | BOD5/CODcr | pH |
|---|---|---|---|---|---|---|---|---|
| a Note: S.D. is the abbreviation of standard deviation. | ||||||||
| Range of values | 275–345 | 40–100 | 90–145 | 50–72 | 75–90 | 0.35–0.50 | 0.08–0.21 | 6.5–8.3 |
| Mean ± S.D. | 322 ± 25 | 59 ± 16 | 113 ± 19 | 65 ± 7 | 82 ± 8 | 0.42 ± 0.05 | 0.18 ± 0.02 | 8.0 ± 0.3 |
2.2. Experimental setup
The experimental apparatus is shown in Fig. 1. The reactor was made of transparent rigid Plexiglas with an inner diameter of 80 mm, a height of 1200 mm, and an effective volume of 6 L. A TCRI Microbubble Generator (Japan) was used to produce microbubbles, with a mean bubble diameter of less than 45 μm under the operational pressure of 0.4 MPa. For comparative purposes, a cylindrical 40 μm micropore titanium plate placed at the bottom of the reactor was used to generate ozone macrobubbles (with a mean bubble diameter of approximately 1 mm) with ozone gas produced by an ozone generator (CF-YG5, Shanmei Shuimei Co. Beijing) at a rate of 5 g O3/h, using dehumidified air as gas source (flow rate of air of 0.5 L min−1). In addition, a low-pressure mercury vapor UV lamp equipped within a quartz well was submerged through the centre of the reactor. Five UV lamps (i.e., 10 W (15 mm diameter × 212 mm height), 14 W (15 mm diameter × 303 mm height), 18 W (15 mm diameter × 356 mm height), 23 W (15 mm diameter × 436 mm height), and 28 W (15 mm diameter × 550 mm height) produced by Cnlight Co., Ltd, Guangdong, China) were used to investigate the influence of irradiation intensity. The UV fluence rate13 of the above described UV lamps at distance of 4 cm from the centre were 50, 70, 90, 114 and 139 mW cm−2, respectively.At the beginning of the experiment, 3 L of secondary acrylic fiber manufacturing wastewater was pumped into the reactor with a peristaltic pump. When the microbubble-ozonation/UV combined process was activated, the macrobubble pathway was closed, and vice versa. In the microbubble-ozonation/UV process, the wastewater was continuously circulated between the microbubble generator and the reactor.
Ozone gas exhausted from the reactor was absorbed with 2% KI solution. The temperature of the reaction solution maintained at 20 °C throughout the experiment. Samples withdrawn at predetermined time intervals were immediately purged with nitrogen gas to remove residual ozone. For gas chromatography-mass spectrometry (GC-MS) analysis, the residual ozone was not purged, but rather was left to decompose over two days.
2.3. Analysis
Most of the wastewater quality parameters were measured according to The Water and Wastewater Monitoring and Analysis Method (4th Edition).14 In addition, CODcr, BOD5, TOC, and UV254 were respectively measured by a COD rapid digestion apparatus (DIS-1A, Shenzhen Changhong Instrument, Co., Ltd, China), oxiTop system (OxiTop, WTW, Germany), vario TOC analyzer (vario TOC, Elementar, Germany) and an UV-visible spectrophotometer (UV-752, METASH, China). The toxicity of samples was analyzed using Modulus Single Tube Luminometer (Turner Biosystems, USA), which is based on the light emission inhibition of luminescent bacteria, the methods of which can be found in Martins et al.15 Further, the toxicity level expressed as EC50, representing samples concentrations causing 50% bacteria inhibition, was acquired using the dilution of each sample. The dissolved oxygen concentration and the pH were determined with a dissolved oxygen meter (HQ30D, Hach, USA) and an automatic potentiometric titrator meter (ZD-2, LEICI, China) respectively at 20 °C. The pH for all solution used in this experiment was adjusted by adding 0.1 mol L−1 of HCl or NaOH solution, controlling it at the desired level. The gaseous ozone concentration was measured with the iodometric method.16 The concentration of dissolved ozone was tested with the indigo colorimetric method (Standard Method 4500-O3 B).17The hydroxyl radical concentration was determined with a three-dimensional excitation-emission matrix fluorescence spectroscopy (3D-EEM) (F2700, Hitachi, Japan) after reaction with disodium salt of terephthalic acid (NaTA) and filtration with a 0.45 μm PVDF membrane. NaTA reacts with hydroxyl radicals to form 2-hydroxyterephthalic acid (HTA) that gives a bright stable fluorescence (λemission = 425 nm, λexcitation = 315 nm).18 TA and NaTA are extensively applied to detect hydroxyl radicals produced in aqueous phase.19,20 In this study, samples (5 mL) collected from the reactor were reacted with 0.5 mM NaTA (5 mL) at the pH of 6.85 in which the buffer was 10 mM mixed non-fluorescent phosphate solution. The samples were analyzed within a few hours of being collected. EEM spectra were scanned from 200 to 450 nm for excitation and 280 to 500 nm for emission.
GC-MS was used for the analysis of the main organic compounds in the wastewater. The wastewater sample (150 mL) was extracted with 50 mL of CH2Cl2 (Chromatogram Pure Grade, Fisher, USA) under acidic (pH 2.0), neutral (pH 7.0), and alkaline (pH 12.0) conditions. The three extracts were mixed together, dehydrated with anhydrous sodium sulfate and dried under the flow of nitrogen gas. The residual was dissolved in 1.0 mL of CH2Cl2 and then 1 μL of the solution was injected into a Shimadzu GCMS-QP2010 plus system (Shimadzu., Japan) and separated with a capillary column (30 m × 0.25 mm i.d., J & W Scientific 122-5032 DB-5, USA). The GC oven temperature was maintained at 50 °C for 1 min, raised at a rate of 10 °C min−1 to 60 °C (held for 2 min), and then further raised at 10 °C min−1 to 250 °C (held for 5 min). Injector port, interface and ion source temperatures were 250, 280 and 300 °C, respectively. MS was operated in electron ionization mode (EI) at 70 eV. Identification of the compounds was based on the NIST 05 mass spectral library database.
CODcr etc. means were calculated from three independent runs of the reactor, with values given as mean ± standard deviation throughout the text.
3. Results and discussion
3.1. Optimization of process parameters
Treatment was carried out under different conditions of ozone dosage, UV fluence rate and pH to achieve the maximum CODcr removal rate (shown in Fig. 2).In order to determine the effect of the ozone dosage on CODcr removal, ozone concentrations were tested at 6, 12, 24, 36, 48, 60 and 72 mg L−1 with UV fluence rate, initial pH and reaction time being fixed at 50 mW cm−2, 8.0 and 120 min respectively. Fig. 2a shows that the CODcr removal increased from 9.5% to 56.7% as the ozone dosage was increased from 6 to 48 mg L−1. Much less improvement in CODcr removal (3.4%) was observed when the ozone dosage was further increased to 72 mg L−1.
The influence of UV fluence rate on the CODcr removal was investigated at 50, 70, 90, 114 and 139 mW cm−2 with ozone dosage of 48 mg L−1, initial pH of 8.0 and a reaction time of 120 min. Fig. 2b shows that the CODcr removal increased from 58.7% to 85.4% as the UV fluence rate was increased from 50 to 90 mW cm−2. The CODcr removal rate increased slightly when the UV fluence rate was increased continuously. Because of the limited benefit at the higher UV fluence rate, the UV fluence rate of 90 mW cm−2 was selected for further optimization.
According to the literature,11,12 pH is a crucial parameter that influences contaminant removal during O3/UV treatment. Since the type of oxidant generated greatly depends on reaction pH,21 the effect of the initial pH on CODcr removal in the microbubble-ozonation/UV system was tested in the range of 3 to 11, while ozone dosage, UV fluence rate and reaction time were kept at 48 mg L−1, 90 mW cm−2 and 120 min, respectively. Fig. 2c shows that the CODcr removal rate reached its highest (87.3%) at the pH of 8.0, and then decreased as pH was further increased. There are two well-established oxidation pathways for ozone: ozone direct oxidation and indirect oxidation by hydroxyl radicals generated from ozone decomposition.22,23 Direct oxidation, the predominate form of ozone oxidation in acidic media, has a greater selectivity for organics than hydroxyl radical oxidation.24 In contrast, the indirect oxidation by non-selective hydroxyl radicals is the main oxidation pathway of ozonation under alkaline conditions.25 At neutral pH, both oxidation pathways contribute to the degradation of bio-refractory matter.26 Therefore, the above described relatively lower CODcr removal at acidic to neutral pH than that at pH of 8.0 can be attributed to the relatively weaker oxidation ability of ozone molecules than hydroxyl radicals which was generated from ozone-OH− reaction. Besides, at the acidic to neutral pH, free radicals are not formed, which results in few free hydroxyl radicals forming. In general, the generation rate of hydroxyl radicals in an alkaline medium (pH > 8.0) can be greater than that at pH of 8.0,27 thus, higher contaminants degradation should be achieved. However, since copolymers (such as acrylate and acrylamide) formed under an alkaline medium during hydroxyl radical oxidation, the wastewater will be much more difficult to remove than the parent compound,28 which could be the reason for the decline of CODcr removal when the pH was further increased. The optimal pH value was determined to be 8.0.
The effect of the reaction time on the CODcr removal rate was investigated at ozone dosage of 48 mg L−1, UV fluence rate of 90 mW cm−2 and an initial pH of 8.0. Fig. 2d shows that the CODcr removal rate gradually increased to 87.5% at 120 min. Further, prolonging the reaction for a further 1 h caused only a 4.2% increase in CODcr removal. In consideration of the economic cost of an increased reaction time, we consider the optimal reaction time of this process to be 120 min.
3.2. Biodegradability improvement under optimal condition
The removal of contaminants in terms of CODcr, BOD5, TOC, UV254, and BOD5/CODcr ratio were investigated under the above optimized reaction conditions (i.e., ozone dosage of 48 mg L−1, UV fluence rate of 90 mW cm−2, initial pH value of 8.0 and reaction time of 120 min) (Fig. 3).Fig. 3 shows that the CODcr decreased by 87.6% (from 322 ± 25 to 39.8 ± 3.6 mg L−1), and UV254 by 87.7% (from 0.42 ± 0.05 to 0.05 ± 0.004 Abs per cm), over the course of the reaction. BOD5 decreased from 59 ± 16 to 23.5 ± 7.1 mg L−1 in 30 min, and then slightly increased and maintained a stable level for the remaining reaction. TOC decreased from 113 ± 19 to 31.8 ± 7.4 mg L−1, indicating that approximately 63.0% of TOC was eliminated in the combined treatment process. Further, the calculated BOD5/CODcr ratio decreased from 0.18 ± 0.02 to 0.10 ± 0.02 in the first 30 min, and then increased to 0.47 ± 0.03 after 120 min, demonstrating an increased biodegradability. The variation in BOD5/CODcr is similar to that found by Martins et al.15 through the Fenton exudation process applied to phenolic wastewater. This above described result can be supported by the variation of toxicity in wastewater, where the toxicity of treated wastewater was only 13.6% of that of raw wastewater (EC50 of raw sample was 17.5%). In addition, the improved biodegradability of the wastewater could also be explained by the removal of bio-refractory organic compounds in the microbubble-ozonation/UV combined process. Therefore, the changes of contaminants in raw and treated wastewater were verified by GC-MS analysis. Organic compounds identified in the raw wastewater through GC-MS (Fig. 4a) are summarized in Table 2.
 | ||
| Fig. 4 GC-MS chromatographs of (a) raw wastewater and (b) treated by combined microbubble-ozonation/UV process. | ||
| No. | Retention time (min) | Chemicals | Similarity (%) | Area (mean ± S.D.) | Removal efficiency (%) (mean ± S.D.) |
|---|---|---|---|---|---|
| 1 | 7.492 | Toluene | 92 | 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 455 ± 1901 455 ± 1901 |
99.3 ± 0.5 |
| 2 | 9.783 | Ethyl-benzene | 90 | 52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 645 ± 1102 645 ± 1102 |
94.7 ± 0.7 |
| 3 | 10.000 | 1,2-Dimethyl-benzene | 88 | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 014 ± 1637 014 ± 1637 |
97.9 ± 1.1 |
| 4 | 10.508 | Ethenyl-benzene | 90 | 703![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 290 ± 16 290 ± 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 134 134 |
100 ± 0.0 |
| 5 | 11.142 | N,N-Dimethylacetamide | 81 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 103 ± 304 103 ± 304 |
100 ± 0.0 |
| 6 | 12.083 | Benzenol | 93 | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 964 ± 989 964 ± 989 |
99.0 ± 0.8 |
| 7 | 12.642 | Decane | 95 | 43![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 010 ± 710 010 ± 710 |
78.5 ± 1.3 |
| 8 | 13.742 | Sulfurous acid, hexyl octyl ester | 86 | 43![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 853 ± 1104 853 ± 1104 |
85.9 ± 1.9 |
| 9 | 13.950 | 2-Methyleneglutaronitrile | 92 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 243 ± 380 243 ± 380 |
90.8 ± 1.3 |
| 10 | 15.733 | 2-Phenyl-tridecane | 84 | 112![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 885 ± 2493 885 ± 2493 |
94.5 ± 1.6 |
| 11 | 16.367 | Pentadecane | 96 | 205![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 338 ± 3390 338 ± 3390 |
79.3 ± 1.7 |
| 12 | 16.742 | Oxalic acid, 4-chlorophenyl octyl ester | 82 | 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 842 ± 855 842 ± 855 |
99.3 ± 1.5 |
| 13 | 16.983 | Adipic acid, ethyl methyl ester | 85 | 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 622 ± 883 622 ± 883 |
88.4 ± 1.9 |
| 14 | 18.717 | Heptadecane | 91 | 189![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 164 ± 4264 164 ± 4264 |
84.8 ± 1.3 |
| 15 | 19.567 | Hexadecane | 91 | 165![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 455 ± 3067 455 ± 3067 |
82.3 ± 2.3 |
| 16 | 20.900 | Eicosane | 92 | 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 726 ± 1653 726 ± 1653 |
88.0 ± 0.9 |
| 17 | 21.242 | 2,4-Di-tert-butylphenol | 88 | 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 303 ± 1254 303 ± 1254 |
97.8 ± 1.1 |
| 18 | 22.383 | Heneicosane | 92 | 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 895 ± 1717 895 ± 1717 |
88.1 ± 2.4 |
| 19 | 23.333 | Pentacosane | 92 | 669![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 806 ± 15 806 ± 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 393 393 |
90.0 ± 2.1 |
| 20 | 23.800 | Dotriacontane | 93 | 588![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 049 ± 9993 049 ± 9993 |
92.1 ± 1.5 |
| 21 | 25.300 | Tetracosane | 92 | 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 405 ± 2026 405 ± 2026 |
89.0 ± 1.2 |
| 22 | 26.925 | Hexatriacontane | 93 | 161![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 151 ± 4117 151 ± 4117 |
93.8 ± 1.4 |
| 23 | 27.117 | Tetracontane | 91 | 578![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 067 ± 12 067 ± 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 530 530 |
95.6 ± 1.1 |
These compounds include 11 alkanes (accounting for 68.6% total peak area), 5 aromatic compounds (accounting for 25.3% total peak area), and 3 esters (accounting for 2.8% total peak area). A small number of phenols (2, 2.5%), an organic nitrile (1, 0.4%), and an amide (1, 0.3%) were also identified. Among these long-chain alkane compounds, the carbon numbers of alkanes (except decane) varied from 15 to 40, which are bio-refractory for natural water bodies.29 Both aromatic compounds and organic nitriles are toxic and are difficult to eliminate through natural biodegradation.30 Therefore, the poor biodegradability of the raw wastewater probably is due to the presence of these bio-refractory organic compounds.
By comparison to Fig. 4a, the chromatograph of Fig. 4b shows that most organic pollutants were significantly removed after the microbubble-ozonation/UV treatment. The compounds remaining in the effluent were long-chain alkanes, nevertheless, more than 87.0% of alkane peak area was eliminated. Based on overall performance, it can be concluded that organic compounds with high molecular weight were converted into smaller molecules with easier biodegradable characteristics, which is supported by the improved biodegradability and decreased toxicity of the effluent. This result is similar to that derived by Gong et al.31 who reported the organic contaminants in treated samples was easy to degrade with the analysis of BOD5 and toxicity. Therefore, the microbubble-ozonation/UV combined process is an effective technique for the treatment of secondary wet-spun acrylic fiber manufacturing wastewater.
3.3. Synergistic effect
 | ||
| Fig. 5 The removal efficiency of CODcr and UV254 in microbubble-ozonation/UV combined process and separated processes. Error bars represent standard deviation of three runs. | ||
Fig. 5 shows that UV irradiation alone only reduced 5.0% CODcr of the wastewater in 120 min; the CODcr slightly increased in the first 60 min. The UV254 removal rate of UV irradiation alone was 14.1% in 120 min. The microbubble-ozonation achieved better removal rates for CODcr and UV254 (i.e., 23.2% and 50.5%, respectively), which could be ascribed to greater amounts of hydroxyl radicals being generated during the collapse of ozone microbubbles as well as the self-decomposition of ozone in water at the pH of 8.1,2,32,33 The microbubble-ozonation/UV combined process removed CODcr by 92.5% and UV254 by 83.4%, which is much higher than the sum of individual removal rates of the two separated processes.
The synergistic efficiency of microbubble-ozonation UV process is defined as:
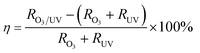 | (A) |
| Items | CODcr removal (%) | UV254 removal (%) | Improvement of BOD5/CODcr |
|---|---|---|---|
| a Note: the improvement of BOD5/CODcr was the D-value between the BOD5/CODcr at 120 min and the initial BOD5/CODcr. | |||
| Microbubble-ozonation alone | 23.2 | 50.5 | 0.09 |
| UV irradiation alone | 5.0 | 14.1 | 0.03 |
| Microbubble-ozonation/UV combined process | 92.5 | 83.4 | 0.29 |
| Synergistic efficiency (%) | 228 | 29 | 142 |
Table 3 indicates that the microbubble-ozonation/UV combined process has a good performance with the synergistic efficiencies in CODcr and UV254 removal and BOD5/CODcr improvement in 120 min reaction being 228%, 29%, and 142%, respectively. Based on a great number of hydroxyl radicals in microbubble-ozonation, the introduced UV irradiation technology can also effectively enhance the production of hydroxyl radicals from ozone.34 Therefore, the significant improvement in contaminant degradation in the combined process is achieved. In addition, the activation energy of the reaction could also be reduced by UV irradiation, contributing to organic contaminant degradation.
 | ||
| Fig. 6 The influence of t-BuOH concentration on CODcr removal for the combined microbubble-ozonation/UV process; error bars represent standard deviation of three runs. | ||
Fig. 6 shows that the CODcr removal was significantly inhibited by the increase of t-BuOH concentration (the contribution of t-BuOH to CODcr was totally eliminated with the result of a control test). The previous CODcr removal rate of 92.5% rapidly decreased to 40.1% at t-BuOH concentrations of 0 and 50 mg L−1, respectively. When the concentration of t-BuOH was increased continuously, the CODcr removal inhibited slightly, which could be considered as all of the hydroxyl radicals produced in microbubble-ozonation/UV process was scavenged by t-BuOH of 50 mg L−1. The hydroxyl radical would have contributed approximately 56.6% of the CODcr removal.
The relative amount of hydroxyl radicals produced in the ozone water, UV water and the combined ozone/UV water under different conditions was compared with the fluorescence intensity of HTA (produced from hydroxyl radical-NaTA reaction) recorded with 3D-EEM (Fig. 7). The quantities of hydroxyl radicals of the samples from microbubble-ozonation process alone was much higher than that from the macrobubble-ozonation process and UV irradiation alone, which lead to a better achievement (described above) for the microbubble-ozonation process alone. A similar result was derived by Chu et al.2 who reported that the fluorescence intensity of the samples from the microbubble system was much higher than that from the bubble contactor. In addition, there was almost no fluorescence detected in the presence of the hydroxyl radical scavenger t-BuOH of 50 mg L−1 in the ozone water sample, which also supported the results of the inhibitation concentration of t-BuOH for hydroxyl radical during microbubble-ozonation/UV process. Interestingly, the quantities of hydroxyl radicals in the microbubble-ozonation/UV combined process during the treatment of the wastewater were significantly higher than the summation of these in microbubble-ozonation alone and UV alone: the peak area of the former was 1.8 times higher than the summation of the latter two processes. This result clearly shows that UV irradiation and microbubble-ozonation have a synergetic effect in the degradation of the contaminants in the wastewater.
The ozone decomposition rates of the microbubble-ozonation/UV combined process, microbubble-ozonation alone, and macrobubble-ozonation alone can be well fitted with first-order reaction kinetics (Fig. 8) with rate constant (kd) being 0.5233 min−1, 0.1264 min−1 and 0.0651 min−1, respectively. The kd of microbubble-ozonation alone was 1.9 times higher than that in the macrobubble-ozonation alone, indicating that the use of microbubbles can effectively improve the ozone decomposition rate. This conclusion is consistent with Liu et al.3 who found that the dissolved oxygen concentration in ozone-microbubble system was much higher than that in air-microbubble system, which was ascribed to the higher transformation from ozone to oxygen of the former at higher ozone decomposition rate. The kd of microbubble-ozonation/UV combined process was 4.1 times higher than that of the microbubble-ozonation alone. Chang et al.40 also found that UV-C radiation in the ozonation process can promote the decomposition of O3 to form ˙OH radical. Therefore, the much higher ozone decomposition rate in the combined process can explain why more hydroxyl radicals were generated in this process, which can further decompose the organic contaminates in wastewater.
4. Conclusion
For the selected secondary wet-spun acrylic fiber manufacturing wastewater, 87.6% of CODcr, 68.0% of BOD5, 71.9% of TOC, and 87.7% of UV254 can be removed by the microbubble-ozonation/UV combined process under optimal conditions (ozone dosage of 48 mg L−1, UV fluence rate of 90 mW cm−2, initial pH value of 8.0, and reaction time of 120 min). Biodegradability (BOD5/CODcr) was also improved from 0.18 to 0.47, which was ascribed to the degradation of refractory contaminants such as alkanes and aromatic compounds. A significant synergetic effect existed between UV irradiation and microbubble ozonation for the removal of CODcr and UV254, and the improvement of BOD5/CODcr, which can be attributed to an acceleration in ozone decomposition and an enhancement of hydroxyl radical generation.Acknowledgements
The authors gratefully acknowledge the financial support of the Major Science and Technology Program for Water Pollution Control and Treatment (2012ZX07201002-6). Thanks also give to Kikuchi ECO-Earth Co. Ltd (Tokyo, Japan) for providing the microbubble generator and China Oil HBP Science & Technology Co., Ltd for providing the wastewater.References
- T. Zheng, Q. Wang, T. Zhang, Z. Shi, Y. Tian, S. Shi, N. Smale and J. Wang, J. Hazard. Mater., 2015, 287, 412–420 CrossRef CAS PubMed.
- L. B. Chu, X. H. Xing, A. F. Yu, Y. N. Zhou, X. L. Sun and B. Jurcik, Chemosphere, 2007, 68, 1854–1860 CrossRef CAS PubMed.
- S. Liu, Q. H. Wang, T. C. Sun, C. Wu and Y. Shi, J. Chem. Technol. Biotechnol., 2012, 87, 206–215 CrossRef CAS PubMed.
- L. B. Chu, S. T. Yan, X. H. Xing, A. F. Yu, X. L. Sun and B. Jurcik, Chemosphere, 2008, 72, 205–212 CrossRef CAS PubMed.
- T. E. Agustina, H. M. Ang and V. K. Vareek, J. Photochem. Photobiol., C, 2005, 6, 264–273 CrossRef CAS PubMed.
- A. Agarwal, W. J. Ng and Y. Liu, Chemosphere, 2011, 84, 1175–1180 CrossRef CAS PubMed.
- P. Li, M. Takahashi and K. Chiba, Chemosphere, 2009, 75, 1371–1375 CrossRef CAS PubMed.
- M. Takahashi, K. Chiba and P. Li, J. Phys. Chem. B, 2007, 111, 1343–1347 CrossRef CAS PubMed.
- C. Gottschalk, J. A. Libra and A. Saupe, in Application of Ozone in Combined Processes, Wiley-VCH Verlag GmbH & Co. KGaA, 2009, pp. 267–343 Search PubMed.
- S. Osbeck, R. H. Bradley, C. Liu, H. Idriss and S. Ward, Carbon, 2011, 49, 4322–4330 CrossRef CAS PubMed.
- M. S. Lucas, J. A. Peres and G. Li Puma, Sep. Purif. Technol., 2010, 72, 235–241 CrossRef CAS PubMed.
- K. Wang, J. Guo, M. Yang, H. Junji and R. Deng, J. Hazard. Mater., 2009, 162, 1243–1248 CrossRef CAS PubMed.
- J. R. Bolton, Water Res., 2000, 34, 3315–3324 CrossRef CAS.
- F. S. Wei, W. Q. Qi, Z. G. Sun, Y. R. Huang and Y. W. Shen, in Water and wastewater monitoring and analysis method, China Environmental Science Press, Beijing, 2002 Search PubMed.
- R. C. Martins, A. F. Rossi and R. M. Quinta-Ferreira, J. Hazard. Mater., 2010, 180, 716–721 CrossRef CAS PubMed.
- R. Gehr, M. Wagner, P. Veerasubramanian and P. Payment, Water Res., 2003, 37, 4573–4586 CrossRef CAS.
- W. A. Appa, in Standard methods for the examination of water and wastewater, American Public Health Association, American Water Work Association, Water Environment federation, Washington, DC, 21st edn, 2005 Search PubMed.
- G. J. Price and E. J. Lenz, Ultrasonics, 1993, 31, 451–456 CrossRef CAS.
- M. Sahni and B. R. Locke, Ind. Eng. Chem. Res., 2006, 45, 5819–5825 CrossRef CAS.
- R. W. Matthews, Radiat. Res., 1980, 83, 27–41 CrossRef CAS.
- S. Jagadevan, N. J. Graham and I. P. Thompson, J. Hazard. Mater., 2013, 244–245, 394–402 CrossRef CAS PubMed.
- J. Suave, H. J. José and R. F. P. M. Moreira, Ozone: Sci. Eng., 2014, 36, 560–569 CrossRef CAS PubMed.
- J. B. Parsa and S. H. Negahdar, Sep. Purif. Technol., 2012, 98, 315–320 CrossRef PubMed.
- J. Lv, Y. Li and Y. Song, Water Res., 2013, 47, 4993–5002 CrossRef CAS PubMed.
- H. Kusic, N. Koprivanac and A. L. Bozic, Chem. Eng. J., 2006, 123, 127–137 CrossRef CAS PubMed.
- E. J. Rosenfeldt, K. G. Linden, S. Canonica and U. von Gunten, Water Res., 2006, 40, 3695–3704 CrossRef CAS PubMed.
- J. Hoigne and H. Bader, Water Res., 1976, 10, 377–386 CrossRef CAS.
- L. B. Krentsel, Y. V. Kudryavtsev, A. I. Rebrov, A. D. Litmanovich and N. A. Platé, Macromolecules, 2001, 34, 5607–5610 CrossRef CAS.
- K. Tong, Y. Zhang, G. Liu, Z. Ye and P. K. Chu, Int. Biodeterior. Biodegrad., 2013, 84, 65–71 CrossRef CAS PubMed.
- F. Widdel and R. Rabus, Curr. Opin. Biotechnol., 2001, 12, 259–276 CrossRef CAS.
- C. Gong, Z. Zhang, H. Li, D. Li, B. Wu, Y. Sun and Y. Cheng, J. Hazard. Mater., 2014, 274, 465–472 CrossRef CAS PubMed.
- P. Li, M. Takahashi and K. Chiba, Chemosphere, 2009, 77, 1157–1160 CrossRef CAS PubMed.
- M. Takahashi, J. Phys. Chem. B, 2005, 109, 21858–21864 CrossRef CAS PubMed.
- T. Garoma and M. D. Gurol, Environ. Sci. Technol., 2004, 38, 5246–5252 CrossRef CAS.
- U. von Gunten and J. Hoigne, Environ. Sci. Technol., 1994, 28, 1234–1242 CrossRef CAS PubMed.
- E. C. Wert, F. L. Rosario-Ortiz and S. A. Snyder, Environ. Sci. Technol., 2009, 43, 4858–4863 CrossRef CAS.
- M. S. Elovitz and U. von Gunten, Ozone: Sci. Eng., 1999, 21, 239–260 CrossRef CAS PubMed.
- C. Gottschalk, J. A. Libra and A. Saupe, in Ozone Applications, Wiley-VCH Verlag GmbH & Co. KGaA, 2009, pp. 27–65 Search PubMed.
- J. Hoigné, in Chemistry of aqueous ozone and transformation of pollutants by ozonation and advanced oxidation processes, Springer, 1998, pp. 83–141 Search PubMed.
- C. Chang, C. Trinh, C. Chiu, C. Chang, S. Chiang, D. Ji, J. Tseng, C. Chang and Y. Chen, J. Taiwan Inst. Chem. Eng., 2014, 49, 100–104 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2015 |





