Synthesis and controlled curcumin supramolecular complex release from pH-sensitive modified gum-arabic-based hydrogels†
Adriana P. Gerola*ab,
Danielle C. Silvaab,
Sandra Jesuscd,
Rui A. Carvalhoe,
Adley F. Rubirab,
Edvani C. Munizb,
Olga Borgescd and
Artur J. M. Valente*a
aCQC, Department of Chemistry, University of Coimbra, 3004-535 Coimbra, Portugal. E-mail: avalente@ci.uc.pt; adrianapassarella@yahoo.com.br; Fax: +351 239 827703; Tel: +351 239854459
bGrupo de Materiais Poliméricos e Compósitos, GMPC, Chemistry Department, Maringá State University, 87020-900 Maringá, Paraná, Brazil
cFaculty of Pharmacy, University of Coimbra, Coimbra, Portugal
dCNC, Center for Neuroscience and Cell Biology, University of Coimbra, 3004-517 Coimbra, Portugal
eDepartment of Life Sciences, University of Coimbra, 3004-535 Coimbra, Portugal
First published on 16th October 2015
Abstract
Curcumin (CUR) is a polyphenolic compound including a beta-diketone moiety, which is associated with numerous pharmacological activities, but applications are limited due to its low water solubility. Thus, in this work some inclusion complexes of CUR with alpha-cyclodextrin (α-CD) and beta-cyclodextrin (β-CD) were prepared using different host![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) guest proportions to improve drug solubilization in biological fluids. The formation of these complexes was confirmed by 1H NMR and thermogravimetric analysis. The stoichiometries of the CUR/α-CD and CUR/β-CD complexes were 1
guest proportions to improve drug solubilization in biological fluids. The formation of these complexes was confirmed by 1H NMR and thermogravimetric analysis. The stoichiometries of the CUR/α-CD and CUR/β-CD complexes were 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and the association constants were 344 mol−1 L and 7.2 × 107 mol−2 L2 for α-CD and β-CD, respectively. The major stability of the CUR/β-CD complex is justified by an inclusion of the aromatic ring inside the CD cavity, whilst in the case of α-CD-complexes the interactions occur via H-bridges, showing the latter complexes’ slow exchange on the NMR time-scale. Even so, the solubility of curcumin complexes is clearly controlled by the solubility of CDs, showing the highest solubility for CUR/α-CD complexes. Hydrogels of modified gum arabic containing CUR/α-CD (1
2 and the association constants were 344 mol−1 L and 7.2 × 107 mol−2 L2 for α-CD and β-CD, respectively. The major stability of the CUR/β-CD complex is justified by an inclusion of the aromatic ring inside the CD cavity, whilst in the case of α-CD-complexes the interactions occur via H-bridges, showing the latter complexes’ slow exchange on the NMR time-scale. Even so, the solubility of curcumin complexes is clearly controlled by the solubility of CDs, showing the highest solubility for CUR/α-CD complexes. Hydrogels of modified gum arabic containing CUR/α-CD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) were obtained and used for controlled release of CUR in simulated intestinal fluid (SIF) and simulated gastric fluid (SGF). The kinetics of release was pH-responsive and the percentage of CUR released was ca. 97% in SIF and 6.7% in SGF. For the toxicity studies on undifferentiated Caco-2 cells, IC50s of 63.4 ± 14.4 μg mL−1 and 85.2 ± 14.9 μg mL−1 for CUR and CUR/α-CD (1
4) were obtained and used for controlled release of CUR in simulated intestinal fluid (SIF) and simulated gastric fluid (SGF). The kinetics of release was pH-responsive and the percentage of CUR released was ca. 97% in SIF and 6.7% in SGF. For the toxicity studies on undifferentiated Caco-2 cells, IC50s of 63.4 ± 14.4 μg mL−1 and 85.2 ± 14.9 μg mL−1 for CUR and CUR/α-CD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4), respectively, were obtained. The toxicity of these samples on differentiated Caco-2 cells was lower than on undifferentiated cells. Additionally, the CUR incorporated into hydrogels showed no toxic effects on differentiated and undifferentiated Caco-2 cells, indicating the pharmaceutical potential of three-dimensional matrices of GAm for controlled release of CUR complexed with cyclodextrin.
4), respectively, were obtained. The toxicity of these samples on differentiated Caco-2 cells was lower than on undifferentiated cells. Additionally, the CUR incorporated into hydrogels showed no toxic effects on differentiated and undifferentiated Caco-2 cells, indicating the pharmaceutical potential of three-dimensional matrices of GAm for controlled release of CUR complexed with cyclodextrin.
1. Introduction
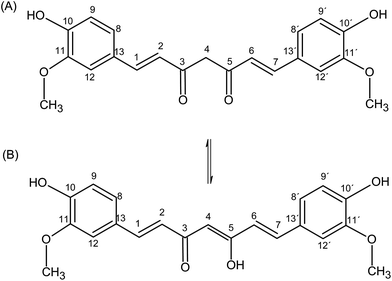
Curcumin {(1E,6E)-1,7-bis(4-hydroxy-3-methoxy phenyl)-1,6-heptadiene-3,5-dione} (CUR) is a yellow polyphenolic compound, with a very low solubility in aqueous media, occurring in the dried rhizomes of turmeric (Curcuma longa).1 Curcumin has two aromatic rings with phenolic –OH groups connected by an α,β-unsaturated-β-diketone (Fig. 1). This structure is able to undergo keto–enol tautomerism. Curcumin has been used as a natural food coloring agent2 and it is associated with numerous pharmacological applications,3 including healing, antimicrobial, antiviral, anti-inflammatory, antioxidant, antiproliferative,4 hypocholesterolemic and anticarcinogenic properties.5–7 In addition, curcumin has shown significant activity for the treatment of chronic diseases such as type II diabetes,8 rheumatoid arthritis,9 multiple sclerosis and Alzheimer’s disease.10–12 However, the main drawback for its administration is its low solubility in aqueous solution (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = 3.29).13 In order to overcome this limitation, different formulations have been developed allowing CUR solubilization in aqueous media. The use of liposomes,14 polymeric micelles,15 microemulsions,16 hydrogels,17 and cyclodextrins18–21 are just some examples of approaches used to enhance the solubility of CUR in aqueous solutions and thus improving its biological applications.20
P = 3.29).13 In order to overcome this limitation, different formulations have been developed allowing CUR solubilization in aqueous media. The use of liposomes,14 polymeric micelles,15 microemulsions,16 hydrogels,17 and cyclodextrins18–21 are just some examples of approaches used to enhance the solubility of CUR in aqueous solutions and thus improving its biological applications.20
Cyclodextrins (CD) are cyclic oligosaccharides in the shape of truncated cones made up of several units of α-glucopyranose linked by α-1,4 glucoside bonds. The number of glucose units (e.g., 6, 7, and 8 for α-, β-, and γ-CD, respectively) defines the width of the cavity and the flexibility of the compound.22 The CD structure provides an external hydrophilic region, where primary and secondary –OH groups are located, as well as a relatively hydrophobic cavity.23 The main feature of cyclodextrins is the cavity that enables them to form inclusion complexes with guest molecules of appropriate size, shape, and polarity.
Host–guest like complexes may improve the properties of the guest molecule, e.g. the drug, in aqueous media, such as solubility enhancement,24 bioavailability and stability improvement.25–27 Although CD can act as efficient drug carrier, providing a controlled and sustained release, avoiding undesirable toxic effects28 for some applications, such as oral administration, the use of those complexes is rather limited.
Hydrogels are 3D crosslinked hydrophilic polymers swollen in water or in the aqueous environment, used, e.g., for biomedical applications such as drug delivery systems, in situ gels and tissue scaffolds.29 Besides the general properties of hydrogels, such as elastomeric consistency and high permeability, for such applications they must be biocompatible, biodegradable and non-toxic. Polysaccharide-based hydrogels are materials constituted by polymers of natural origin that fulfil many of the requirements needed to act as matrices for drug delivery.30 However, the high solubility of polysaccharides in aqueous media is often responsible for the premature release of solutes. Modified gum arabic (M-GA)-based hydrogels are pH-responsive, showing structural integrity at acidic conditions, and aging at moderately basic pH; these features make these gels highly recommended for colon-specific controlled release systems.31
In this manuscript, we describe the synthesis and characterization of CUR/α-CD and CUR/β-CD supramolecular complexes at different CUR/CD molar ratios. Although the β-CD complexes show a higher binding constant, the highest solubility values (in water) were obtained using α-CD. Thus, the obtained complex was incorporated in a pre-gel solution of M-GA. Some chemical and morphological characteristics of the resulting composites were studied. The release kinetics of CUR/α-CD from the hydrogel matrices to simulated body fluids (gastric – SGF – and intestinal – SIF) has also been evaluated. The cytotoxicity of the samples on differentiated and undifferentiated Caco-2 cells was analyzed.
2. Experimental
2.1. Materials
Curcumin (purity > 98%) was purchased from Fisher Chemical; dimethyl sulfoxide (DMSO) and sodium persulfate (Na2S2O8, 99%) were purchased from Fluka. Gum arabic (GA) from Acacia tree, alpha-(α-CD) and beta-cyclodextrin (β-CD), with purities higher than 98% and 97%, respectively, were obtained from Sigma-Aldrich. Ethyl alcohol (95%) and tetrahydrofuran (THF) were purchased by Applichem Panreac and hydrochloric acid (HCl, 37%) was obtained from Carlo Erba. Glycidyl methacrylate (GMA, 97%) was supplied by Acros Organics. These reagents were used without further purification. All solutions were prepared using Millipore-Q water.2.2. Characterization of curcumin in homogeneous systems
Electronic absorption spectra of curcumin in different solvents (a mixture of water/THF (50%, v/v), Milli-Q© water, simulated gastric fluid (SGF, 2.0 g NaCl and 7.0 mL of concentrated hydrochloric acid in 1000 mL of distilled water, pH 1.2) and simulated intestinal fluid (SIF, 6.80 g of KH2PO4, and 77 mL of an aqueous solution of NaOH 0.20 mol L−1 in 1000 mL of distilled water, pH 6.8)) were recorded using a Shimadzu, UV-2450 spectrophotometer in a mixture of water/THF (50%, v/v), Milli-Q© water, SGF, and SIF. In order to obtain the molar absorption coefficient for CUR aqueous solutions, the following methodology was adopted: different concentrations of CUR were obtained by adding aliquots of a stock solution ([CUR] = 0.054 mol L−1 in DMSO) to water, by keeping the percentage of DMSO at levels below 1% (DMSO/water, v/v). The molar absorption coefficient of CUR has been computed from the Lambert–Beer law. All spectra were obtained at 25.0 °C.The CUR solubility in SIF, SGF and Milli-Q© water, at 25.0 °C, was also evaluated by preparing supersaturated solutions; the obtained mixtures were sonicated for 10 min. Then the samples were centrifuged for 5 min at 3800 rpm (Centrifuge 1-1SPK Sigma) and an aliquot of the supernatant was removed and mixed with an equal volume of THF to obtain a water/THF (50%, v/v) mixture. When necessary, dilutions were done with water/THF mixtures. UV-visible spectra (350 nm to 700 nm) were recorded and the CUR concentration calculated by using a molar absorption coefficient equal to 37.9 × 103 L mol−1 cm−1 (for absorbance at λ = 425 nm).
2.3. CUR/CD supramolecular complexes
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 384 data-points, a number of scans of 98 and a relaxation delay of 1 s. Chemical shifts (δ) were obtained taking as reference (δ = 0) the internal reference. Further NMR characterization of the CUR/α-CD and CUR/β-CD complexes was made using a Varian 600 MHz spectrometer and a 3 mm indirect detection probe. Highly resolved 1H NMR spectra were obtained for the two complexes, both in D2O and in deuterated DMSO (DMSO-D6), using quantitative acquisition parameters, namely an interpulse delay of 10 s and a radiofrequency observation pulse of 30°. This ensures full relaxation of all 1H nuclei in the sample and a quantitative analysis of all resonances. Other parameters included a spectral width of 6000 Hz and a number of scans of 128, to allow observation of the resonances due to the aromatic rings of curcumin. Further characterization of the interaction between CUR and CD was made using ROESY spectra. In D2O the amount of dissolved CUR relative to CD was very low, rendering this 2D analysis impossible for the CUR/β-CD system. For the CUR/α-CD the acquisition and interpretation of the ROESY spectra was possible. To gain further insight to the interaction a ROESY analysis was also attempted for the complexes dissolved in DMSO-D6. Acquisition parameters for ROESY spectra included a magnetization mixing period of 200 ms to allow magnetization transfer, a 6000 Hz spectral width, 2048 data points in F2 and 512 data points in F1. Before Fourier transformation, Gaussian functions were applied in both dimensions.
384 data-points, a number of scans of 98 and a relaxation delay of 1 s. Chemical shifts (δ) were obtained taking as reference (δ = 0) the internal reference. Further NMR characterization of the CUR/α-CD and CUR/β-CD complexes was made using a Varian 600 MHz spectrometer and a 3 mm indirect detection probe. Highly resolved 1H NMR spectra were obtained for the two complexes, both in D2O and in deuterated DMSO (DMSO-D6), using quantitative acquisition parameters, namely an interpulse delay of 10 s and a radiofrequency observation pulse of 30°. This ensures full relaxation of all 1H nuclei in the sample and a quantitative analysis of all resonances. Other parameters included a spectral width of 6000 Hz and a number of scans of 128, to allow observation of the resonances due to the aromatic rings of curcumin. Further characterization of the interaction between CUR and CD was made using ROESY spectra. In D2O the amount of dissolved CUR relative to CD was very low, rendering this 2D analysis impossible for the CUR/β-CD system. For the CUR/α-CD the acquisition and interpretation of the ROESY spectra was possible. To gain further insight to the interaction a ROESY analysis was also attempted for the complexes dissolved in DMSO-D6. Acquisition parameters for ROESY spectra included a magnetization mixing period of 200 ms to allow magnetization transfer, a 6000 Hz spectral width, 2048 data points in F2 and 512 data points in F1. Before Fourier transformation, Gaussian functions were applied in both dimensions.Thermogravimetric analysis (TGA) of pure samples (CUR, α-CD and β-CD) and inclusion complexes (CUR/α-CD and CUR/β-CD), prepared at different molar ratios, was carried out on a thermo-microbalance thermogravimetric analyzer TG 209 F3 Tarsus®, from Netzsch Instruments. Samples (ca. 10 mg) were heated at 10 °C min−1 from 30 °C to 475 °C, under a nitrogen gas at a flow rate of 20 mL min−1.
2.4. Solubility of inclusion complexes
Solubility studies were carried out at 25.0 °C by adding an excess of the CUR/CD complex to a given volume (5 mL) of Milli-Q© water. The mixtures were then immersed in an ultrasonic bath for 10 min and subsequently centrifuged. To determine the concentration of CUR/CD solubilized, an aliquot of the supernatant was mixed with an equal volume of THF. The electronic absorption spectra were recorded and the CUR concentration was determined by the molar absorption coefficient at 425 nm (37.9 × 103 L mol−1 cm−1).2.5. Modification of gum arabic (GA) with glycidyl methacrylate (GMA)
The chemical modification of GA with GMA was performed following the methodology described by Maior and coworkers.33 Initially, the GA was dissolved (7.5 g) in Milli-Q© water (50 mL) at 65 °C. After the complete solubilization of GA, 1.0 mL of GMA was added to the solution (pH 3.5). The mixture was kept under stirring at 65 °C for 12 h. Then, the modified gum arabic (M-GA) was precipitated with an excess of ethanol and filtered under reduced pressure. The precipitated M-GA was lyophilized (Labconco Freeze) for 12 h and characterized by 1H NMR spectroscopy in D2O using TPS as the internal reference. 1H NMR spectra were obtained using a 400 MHz spectrometer, as described in Section 2.3.2, and chemical shifts (δ) were obtained in relation to the internal reference.2.6. M-GA/CUR/α-CD hydrogel composites: synthesis and characterization
The synthesis of α-cyclodextrin-containing M-GA hydrogels was adapted from a methodology previously described by Reis et al.31 A pre-gel solution was prepared by initially dissolving the complex CUR/α-CD (10% w/w) and the initiator Na2S2O8 (10 mmol L−1) in an aqueous solution of M-GA (15% w/v).The mixture was stirred at room temperature for 30 min and 900 μL of the solution was transferred to a cell culture plate of 24 wells. The hydrogel matrices were prepared by free radical polymerization of the pre-gel solution by placing the culture plate inside the oven (Heraeus) at 75 °C for 90 min. The hydrogels obtained were carefully removed from the molds and washed with water, showing a macroscopic homogeneous structure with a yellowish colour.
The surface morphology of M-GA and M-GA/CUR/α-CD hydrogels have been analyzed by scanning electron microscopy (SEM) with a JEOL model 5310 scanning microscope operating under low vacuum at 10 kV. The gels, after swelling in SGF and SIF solutions, were submitted to a fast cryogenic treatment by plunging the gel samples into liquid nitrogen for 10 s to 20 s, and then they were left in a freeze dryer for 48 h before being coated with a gold film.
In addition, solid state diffuse reflectance measurements were recorded on a Cary 5000 UV-Vis-NIR spectrometer equipped with an integrating sphere (200 nm to 1800 nm) accessory.
2.7. CUR release kinetics from hydrogels
In vitro release studies of CUR from CUR/α-CD-containing M-GA hydrogels were performed. All experiments were carried out at 37 °C by using a thermostatic bath Multistirrer 6 from Velp Scientifica and under constant stirring (200 rpm). The procedure consists of the immersion of the hydrogel matrix sample, containing 0.135 g of M-GA and 0.0135 g of CUR/α-CD complex, in 10 mL of either simulated gastric fluid (SGF) or simulated intestinal fluid (SIF) previously equilibrated at 37 °C. Fluids were prepared according to United States Pharmacopeia 37 (USP37) with one exception: enzymes were not added to the fluids. At defined intervals, aliquots of the supernatant (0.5 mL) were collected and replaced by an equal volume of SGF or SIF. Then, an equal volume of THF is added to the collected aliquots to obtain a water/THF (50% v/v) solvent mixture. The amount of CUR release is quantified by UV-vis spectrophotometry, by measuring the absorbance at 425 nm, and using the molar absorption coefficient equal to 37.9 × 103 L mol−1 cm−1.| Ct/C∞ = ktn | (1) |
| Q1/3 = Q01/3 − kCt | (2) |
The release kinetics of the CUR, in the complexed form, from the M-GA-based matrices was evaluated by using the Lagergren first-order and pseudo-second order rate equations,37 which can be written, respectively, in their linear form as
| ln(C∞ − Ct) = ln(C∞) − k1t | (3) |
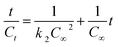 | (4) |
2.8. Cytotoxicity studies
Caco-2 cells were seeded in a 96-well plate at a density of 2 × 105 cells per well in 100 μL of Dulbecco’s modified Eagle medium (DMEM) and incubated for 24 h. On the next day, the medium was replaced by equal volume of fresh medium and formulations were added to a final concentration of CUR from 0.4 μg mL−1 to 363.3 μg mL−1. Hydrogels without CUR were tested with concentrations, from 35 μg mL−1 to 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μg mL−1. After incubation for 24 h at 37 °C, 20 μL of 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) 5 mg mL−1 in phosphate buffered saline (PBS, pH = 7.4) was added to each well and incubated for more 2 h at 37 °C. Finally, the medium was removed and the formazan crystals produced by metabolic active cells were solubilized with DMSO (200 μL per well) and the optical density measured at 540 nm and at 630 nm as the reference wavelength.
000 μg mL−1. After incubation for 24 h at 37 °C, 20 μL of 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) 5 mg mL−1 in phosphate buffered saline (PBS, pH = 7.4) was added to each well and incubated for more 2 h at 37 °C. Finally, the medium was removed and the formazan crystals produced by metabolic active cells were solubilized with DMSO (200 μL per well) and the optical density measured at 540 nm and at 630 nm as the reference wavelength.
Cytotoxicity was also evaluated in the differentiated Caco-2 cell line. Cells were seeded in a 48-well plate at a density of 8 × 104 cells per well in 250 μL of DMEM and incubated for 14 to 21 days allowing cells to differentiate. The medium was replaced by fresh medium every two days and formulations were added to a final concentration from 5.7 μg mL−1 to 363.3 μg mL−1 and control experiments of the hydrogel in the absence of CUR were performed. After incubation for 24 h, 40 μL of MTT was added and plates were incubated for 3 h at 37 °C. The culture medium was discarded and formazan crystals were dissolved as described for undifferentiated cells.
The viability of non-treated cells (control) was defined as 100% and the relative cell viability (%) calculated using the eqn (5):
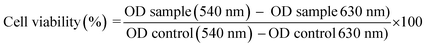 | (5) |
3. Results and discussion
3.1. Curcumin solubility
Electronic absorption spectra of curcumin, at different concentrations, were obtained in different media: SIF, SGF, Milli-Q© water and water/THF (50%, v/v) – see representative spectra in Fig. 2. In aqueous media (SIF, SGF, Milli-Q© water) the CUR showed a maximum absorption band at 425 nm and a shoulder at 353 nm. The presence of this shoulder in acidic and neutral buffer solutions can be assigned to the stabilization of the CUR in the twisted β-diketo form.38 On the other hand, the spectrum of CUR in the water/THF (50%, v/v) shows only a maximum absorbance at ca. 425 nm (no shoulder at 350 nm is observed). This can be explained by the occurrence of the CUR keto–enol form.39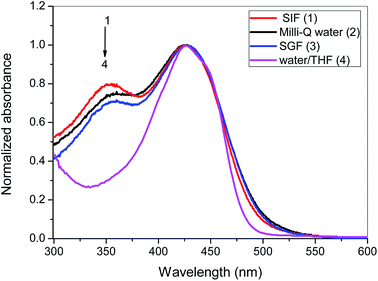 | ||
| Fig. 2 Normalized absorption spectra of CUR (9.0 × 10−5 mol L−1) in Milli-Q© water, SIF, SGF and water/THF (50%, v/v) mixture, at 25.0 °C. | ||
Molar absorption coefficient (ε) values for CUR at different media were determined by the Beer–Lambert law and are summarized in the Table 1. For aqueous systems (SGF, SIF and Milli-Q© water), the ε values are quite similar and range from 1.68 × 103 to 2.63 × 103 L mol−1 cm−1. However, the molar absorption coefficient for CUR in the water/THF (50%, v/v) mixed solvent is one order of magnitude higher: 37.9 × 103 L mol−1 cm−1.31 For those reasons this solvent mixture will be used for quantification of curcumin where needed.
| λ/nm | ε × 103/L mol−1 cm−1 | S/μmol L−1 | R2 | |
|---|---|---|---|---|
| Milli-Q© water | 424 | 2.63 (±0.03) | 0.58 | 0.999 |
| SGF | 427 | 2.39 (±0.08) | 1.20 | 0.993 |
| SIF | 423 | 1.68 (±0.06) | N.d. | 0.989 |
| Water/THF (50%, v/v) | 425 | 37.9 (±0.85) | Soluble | 0.995 |
Solubilities of curcumin, at 25 °C, in different solvents were also evaluated and are described in Table 1.
Curcumin is insoluble in SIF, while the solubilities in Milli-Q© water and SGF are 0.58 μmol L−1 and 1.20 μmol L−1, respectively. These results are in agreement with the data reported elsewhere, showing that CUR is practically insoluble in neutral and acid aqueous solutions due to their neutral and hydrophobic form (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = 3.29).13
P = 3.29).13
The low CUR solubility in SGF and SIF are thus limiting factors for the bioavailability of curcumin either for in vitro or in vivo release tests. Thus, it becomes necessary to use alternative strategies to overcome this problem, such as, for example, the formation of inclusion complexes with cyclodextrins40,41 as will be discussed in the following sections.
3.2. Solubility of curcumin in the presence of cyclodextrins
The formation of these complexes will provide relevant modifications to the properties of the complexed (guest) molecules. The considerable increase of solubility of a guest molecule in aqueous media is an area where CD performance stands out.42Two different curcumin-containing supramolecular complexes using α- and β-CD at different molar ratios have been prepared. Since the β-CD solubility in water (1.85 g/100 mL, 25 °C) is ca. eight times lower than that for α-CD (14.5 g/100 mL, 25 °C),43 the samples of CUR/β-CD complexes in water were prepared in smaller concentrations ([CUR] = 1.06 × 10−4 mol L−1) than for CUR/α-CD complexes ([CUR] = 1.34 × 10−3 mol L−1) to ensure their complete solubilization in an aqueous medium.
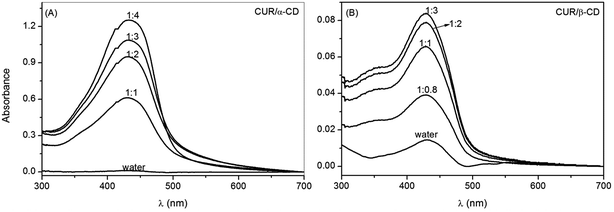
Electronic absorption spectra of aqueous solutions of curcumin with α-CD and β-CD, at different molar ratios ([CUR]/[CD] = r), are shown in Fig. 3. It can be seen that by increasing the amount of cyclodextrin in the solution (by decreasing the r value) an increase of the curcumin maximum absorption intensity occurs, indicating an increase of CUR solubilization by host–guest interaction with CD (evidence for this will be discussed later). It should be noted that CD does not show absorbance in the UV region.
Table 2 shows the absolute solubility of curcumin complexed with α-CD and β-CD, and the corresponding solubility efficiencies (SE). The analysis of the solubilization efficiency of the complexes shows that the highest SE is attained for the CUR/α-CD complex with r = 0.25, corresponding to an increase in the CUR solubility of 172 times. In the case of CUR/β-CD the maximum SE is reached for the molar ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and drops off for higher molar ratios. The larger value of SE for the CUR/α-CD complex compared to CUR/β-CD is due to the greater solubility of alpha-CD in the aqueous environment.
3, and drops off for higher molar ratios. The larger value of SE for the CUR/α-CD complex compared to CUR/β-CD is due to the greater solubility of alpha-CD in the aqueous environment.
| CUR/CD | CUR/α-CD | CUR/β-CD | ||
|---|---|---|---|---|
| [CUR]/μmol L−1 | SEa | [CUR]/μmol L−1 | SEa | |
| a SE (solubilization efficiency) is calculated as the ratio of CUR/CD solubility to the CUR solubility in pure water. | ||||
| Pure water | 0.58 | — | 0.58 | — |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 0.5 |
13.1 | 22.6 | 5.30 | 9.1 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
23.0 | 39.7 | 13.5 | 23.3 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
54.0 | 93.1 | 24.0 | 41.4 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
75.0 | 129.3 | 32.0 | 55.2 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
100 | 172.4 | 21.0 | 36.2 |
Since the solubilization of CUR in an aqueous medium is a precondition for release studies in biological fluids, the CUR/α-CD complex, at r = 0.25, was chosen for the preparation of hydrogels in further assays.
3.3. Interactions of curcumin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) cyclodextrin as seen by NMR
cyclodextrin as seen by NMR
To have a deeper assessment on the interaction mechanism between curcumin and cyclodextrins, NMR studies were carried out. As the assignment of 1H NMR for α- and β-CD has been reported in the literature,44–46 their spectra will not be discussed here.
The 1H NMR spectrum of a CUR/α-CD solution, obtained with the complex prepared with a molar ratio r = 1.00 ([α-CD] = 3.73 μmol L−1), shows slightly upfield shifts (Δδ = δ(CUR/CD) − δ(CD)) for H3 (wide side of cavity) and H5 (narrow side of cavity), of −0.02 and −0.02 ppm, respectively. These Δδ values demonstrate an involvement of these protons in host–guest interactions through predominantly hydrophobic interactions.47 Following the same procedure for the CUR/β-CD complex (using a r = 1, [β-CD] = 3.33 μmol L−1) a downfield shift of +0.01 ppm is observed for both H3 and H5 protons. The smaller Δδ obtained for inner CD protons in the complex, compared with those found for α-CD complex, can be justified for the smaller inner diameter of the α-CD (4.9 Å) compared with that for β-CD (6.2 Å); thus, it should be expected that internal protons of α-CD are more sensitive to environmental changes and, consequently, higher Δδ values are found. Besides, Δδ(H3) and Δδ(H5) values can also be used to assess if the incorporation of a guest inside the CD cavity is partial (Δδ(H3) > Δδ(H5)) or total (Δδ(H3) ≤ Δδ(H5)).48 Although the discussion based on chemical shifts has demonstrated that curcumin is efficiently complexed by CD, further 2D-ROESY experiments were carried out.
1H-NMR plus 2D-ROESY spectra, in deuterated water (D2O) and in deuterated dimethyl sulfoxide (DMSO-D6), were acquired for each supramolecular adduct. As stated before, complexes were formed by mixing curcumin with CDs in a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4. In the 1H-NMR spectra run in D2O, the resonances due to each CD were overwhelming compared with those attributed to CUR. This suggests a preferable dissolution of free CD and a very small contribution due to the complex formation between each CD and curcumin. This, combined with the much lower solubility of β-CD itself, renders the CUR/β-CD system essentially unsuitable for ROESY analysis. The amount of CUR present in solution is too low to be adequately detected/analyzed and the ROESY spectra become fully dominated by the β-CD resonances. In the CUR/α-CD system, the sensitivity and dynamic range issues were not as limiting and a ROESY spectrum was acquired (Fig. 4A) that allowed the detection of correlations between aromatic resonances due to CUR (12,12′, 8,8′, and 9,9′) and the H3 resonance of α-CD, denoting the formation of a true inclusion complex. Some weaker NOEs were also observed between the olefinic protons of CUR (1,7 and 2,6) and protons H6 of the α-CD, suggesting the presence of a second complex. This second interaction does not support the formation of a true inclusion complex but solely the formation of hydrogen bonds between CUR carbonyls and the –OH6 in the narrower rim of the CDs. These two sets of intramolecular NOE interactions could also be consistent, although this is very unlikely, with the occurrence in D2O of a complex with a 2
4. In the 1H-NMR spectra run in D2O, the resonances due to each CD were overwhelming compared with those attributed to CUR. This suggests a preferable dissolution of free CD and a very small contribution due to the complex formation between each CD and curcumin. This, combined with the much lower solubility of β-CD itself, renders the CUR/β-CD system essentially unsuitable for ROESY analysis. The amount of CUR present in solution is too low to be adequately detected/analyzed and the ROESY spectra become fully dominated by the β-CD resonances. In the CUR/α-CD system, the sensitivity and dynamic range issues were not as limiting and a ROESY spectrum was acquired (Fig. 4A) that allowed the detection of correlations between aromatic resonances due to CUR (12,12′, 8,8′, and 9,9′) and the H3 resonance of α-CD, denoting the formation of a true inclusion complex. Some weaker NOEs were also observed between the olefinic protons of CUR (1,7 and 2,6) and protons H6 of the α-CD, suggesting the presence of a second complex. This second interaction does not support the formation of a true inclusion complex but solely the formation of hydrogen bonds between CUR carbonyls and the –OH6 in the narrower rim of the CDs. These two sets of intramolecular NOE interactions could also be consistent, although this is very unlikely, with the occurrence in D2O of a complex with a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry in which one CD molecule would interact with the aromatic moiety of the CUR and the second CD would interact with the keto or eventually enol forms of CUR. The coexistence of keto- and enol forms of CUR is easily appreciated in the proton spectra run in DMSO-D6 (Fig. 4B). The –OH resonance at ∼6.05 ppm, due to the enol form of curcumin, shows up as a double resonance, with different intensities and consistent with the occurrence in this solvent of a slow exchange equilibria between complexed and free CUR. This is very easily appreciated in the CUR/α-CD system since the –OH2 and –OH3 resonances due to complexed CD also appear at distinct chemical shifts from their counterparts due to free CD. In the CUR/α-CD system only the –OH resonance due to the enol form of CUR is distinguishable and no distinct –OH2 and OH3 resonances are detected. This information, together with the absence of any ROESY intramolecular correlations in the DMSO-D6 solvent, suggest that the solvent itself blocks the entrance of CUR in the cavities of both CDs and that the non-inclusion adduct formed with the α-CD is more stable than the one established with β-CD.
1 stoichiometry in which one CD molecule would interact with the aromatic moiety of the CUR and the second CD would interact with the keto or eventually enol forms of CUR. The coexistence of keto- and enol forms of CUR is easily appreciated in the proton spectra run in DMSO-D6 (Fig. 4B). The –OH resonance at ∼6.05 ppm, due to the enol form of curcumin, shows up as a double resonance, with different intensities and consistent with the occurrence in this solvent of a slow exchange equilibria between complexed and free CUR. This is very easily appreciated in the CUR/α-CD system since the –OH2 and –OH3 resonances due to complexed CD also appear at distinct chemical shifts from their counterparts due to free CD. In the CUR/α-CD system only the –OH resonance due to the enol form of CUR is distinguishable and no distinct –OH2 and OH3 resonances are detected. This information, together with the absence of any ROESY intramolecular correlations in the DMSO-D6 solvent, suggest that the solvent itself blocks the entrance of CUR in the cavities of both CDs and that the non-inclusion adduct formed with the α-CD is more stable than the one established with β-CD.
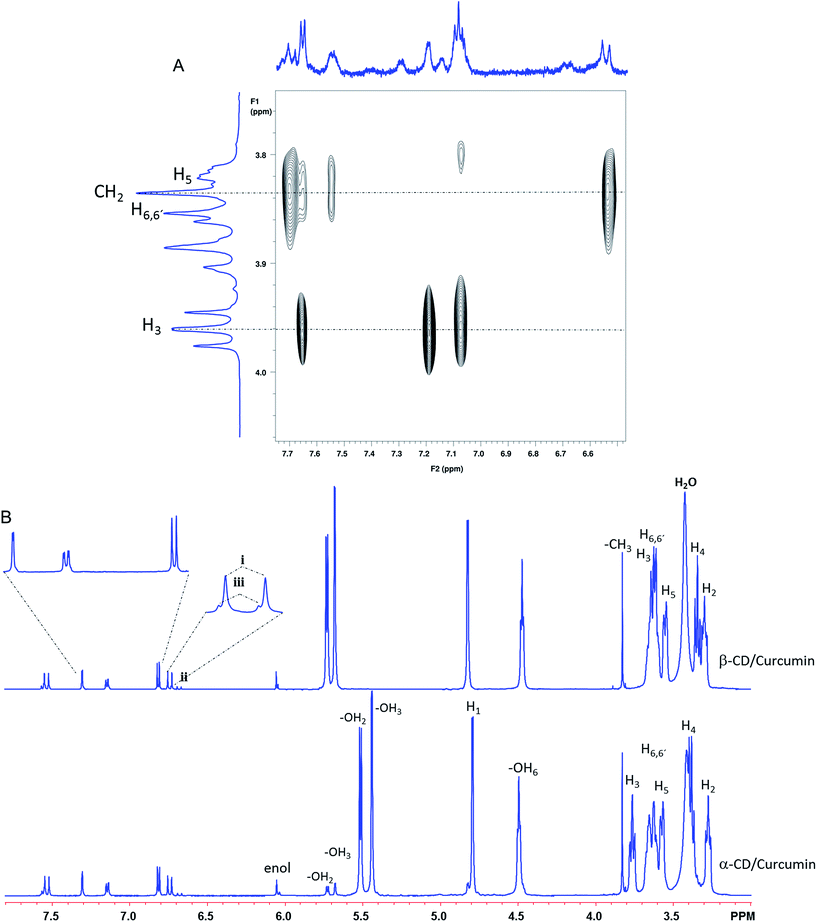 | ||
Fig. 4 (A) Expansion from the 2D ROESY spectrum of a mixture of CUR/α-CD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) in D2O, with respective projections. (B) 1H NMR spectra of CUR/β-CD (top) and CUR/α-CD (bottom) run in DMSO-D6. 4) in D2O, with respective projections. (B) 1H NMR spectra of CUR/β-CD (top) and CUR/α-CD (bottom) run in DMSO-D6. | ||
Recently, based on 2D NMR ROESY analysis, Jahed et al.49 demonstrated that CUR interacts with β-CD forming 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (CUR
2 (CUR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CD) complexes; following this, our studies were focused on CUR/α-CD systems. Our results for this system are compatible with a 1
CD) complexes; following this, our studies were focused on CUR/α-CD systems. Our results for this system are compatible with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex formation in D2O and the occurrence of a true inclusion complex. Nevertheless this interaction is strongly affected by the solvent and in DMSO-D6 no true inclusion complexes were observed. In fact, with this solvent, there is an alteration in the host–guest exchange regimen and a slow exchange is detected in the CUR/α-CD system.
1 complex formation in D2O and the occurrence of a true inclusion complex. Nevertheless this interaction is strongly affected by the solvent and in DMSO-D6 no true inclusion complexes were observed. In fact, with this solvent, there is an alteration in the host–guest exchange regimen and a slow exchange is detected in the CUR/α-CD system.
3.4. Association of curcumin and cyclodextrins
Taking into consideration the discussion carried out in the previous section and data in particular for CUR/β-CD complexation, reported elsewhere,49–52 it is suggested that 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (CUR/CD) complexes can be formed. Despite some limitations of the Benesi–Hildebrand method,27,53 it still remains the easiest method to predict the stoichiometry of association and to compute the corresponding overall binding constants.54 The modified Benesi–Hildebrand method for a 1
2 (CUR/CD) complexes can be formed. Despite some limitations of the Benesi–Hildebrand method,27,53 it still remains the easiest method to predict the stoichiometry of association and to compute the corresponding overall binding constants.54 The modified Benesi–Hildebrand method for a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) n (CUR
n (CUR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CD) stoichiometry is given by the eqn (6):
CD) stoichiometry is given by the eqn (6):
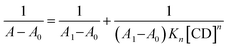 | (6) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexation whilst for the α-CD system the best correlation favours a 1
1 complexation whilst for the α-CD system the best correlation favours a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexation with CUR. These conclusions are in close agreement with the rationale based on NMR data.
1 complexation with CUR. These conclusions are in close agreement with the rationale based on NMR data.
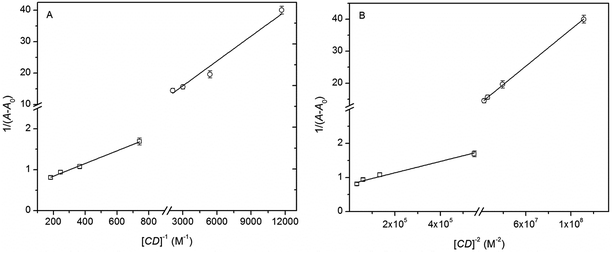 | ||
Fig. 5 Benesi–Hildebrand plots for CUR/α-CD (squares) and CUR/β-CD (circles), assuming a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (A) and a 1 1 (A) and a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (B) stoichiometry. 2 (B) stoichiometry. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 CUR/CD stoichiometries computed by fitting eqn (6) to experimental data showed in the Fig. 5
2 CUR/CD stoichiometries computed by fitting eqn (6) to experimental data showed in the Fig. 5
| n = 1 | n = 2 | |||||
|---|---|---|---|---|---|---|
| A1 − A0 | K1/M−1 | R2 | A1 − A0 | K2/M−2 | R2 | |
| α-CD | 1.87 (±0.03) | 344 (±26) | 0.9913 | 1.24 (±0.06) | 4.8 (±0.7) × 105 | 0.9507 |
| β-CD | 0.12 (±0.02) | 3083 (±625) | 0.96551 | 0.072 (±0.001) | 7.2 (±0.1) × 107 | 0.9994 |
Another issue, relevant for the objectives of the present work, is how these binding constants are related to the higher solubility of CUR/α-CD than CUR/β-CD complexes. From simple mathematical grounds it can be seen that the dominant factor for higher concentrations of CUR in the complex form is the solubility of CDs themselves. This supports the choice of using α-CD-based complexes for encapsulation of CUR into hydrogel matrices in order guarantee a higher CUR concentration in the formulation.
3.5. Thermogravimetric analysis (TGA)
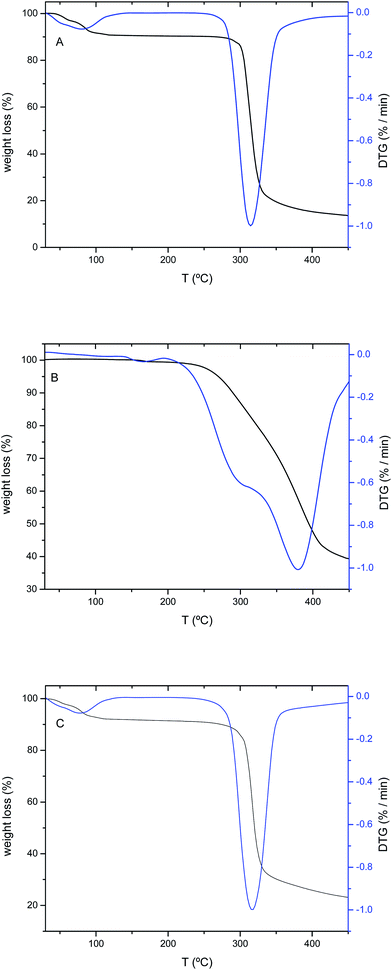
Bearing in mind the potential use of CUR/CD complexes for pharmaceutical applications, the complexes were also characterized by thermogravimetric analysis. The corresponding thermograms were compared with those of pure CDs and CUR. A representative thermogram, and the corresponding DTG, of α-CD, CUR and CUR/α-CD (at r = 0.25) is shown in Fig. 6. The thermograms for β-CD and CUR/β-CD are not shown. Thermograms for pure CDs show two characteristic steps: the first decomposition step (40 °C to 140 °C) is related to the mass loss caused by crystalline water, whilst the second stage, occurring from 250 °C to 350 °C, is due to CD decomposition,55 with temperatures at the maximum degradation rate (Tmax) equal to 314 °C and 318 °C, for α- and β-CD, respectively.On the other side, the CUR thermogram shows a small mass loss at 164 °C, related to the dehydroxylation of –OH groups by elimination of two water molecules, followed by two Tm values at 301 °C and 380 °C.
The thermograms for CUR/α-CD (independent of r values) are similar to that found for α-CD, but the Tm is equal to 317 °C. Such an improvement in the thermal stability is probably due to the formation of complexes with the stabilization of the CD structure.56 Such an increase in the thermal stability is not followed by CUR/β-CD complexes, where Tm values are equal to 316 °C (for r = 1 and 0.5) and 318 °C (for r = 0.33). In this case, the thermal degradation temperatures of the complexes are similar (or even slightly lower) to that observed for β-CD (Tm = 318 °C) suggesting the existence of a host–guest complex.
3.6. Characterization of curcumin complex-containing modified gum arabic hydrogels
The 1H NMR spectra of GA and M-GA are shown in Fig. S1 (ESI†). The resonances at δ 6.20 ppm and 5.79 ppm are assigned to the vinyl hydrogens of the methacryloyl group, while the signal δ 1.96 ppm is related to the methyl hydrogens of the methacryloyl group.31The hydrogels of M-GA containing the CUR/α-CD complex in a molar ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4), were prepared and characterized by SEM and diffuse reflectance.
4), were prepared and characterized by SEM and diffuse reflectance.
Fig. 7 shows the micrographs of M-GA without and with CUR/α-CD (A and B, respectively). The surface morphology of the M-GA hydrogel (Fig. 7A) shows well-defined pores. It should be noted that the pores are well defined and, in general, are not interconnected. In the presence of the CUR-based complex (Fig. 7B) the pores are bigger and form in some places a water lily-like structure, suggesting that a phase-separation is taking place.
 | ||
| Fig. 7 Scanning electron micrographs of (A) M-GA hydrogel and (B) M-GA/CUR/α-CD hydrogel membranes. ×1500. | ||
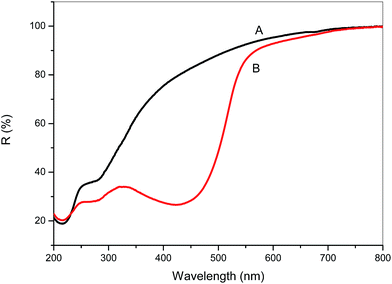
The presence of curcumin inside the hydrogel matrix was also confirmed by diffuse reflectance spectroscopy. Fig. 8 shows the effect of the incorporation of curcumin in the UV-visible spectrum of the M-GA matrix. The presence of CUR/α-CD leads to a significant change in the reflectance band at wavelengths ranging between 350 and 520 nm, with a maximum reflection at 425 nm.
3.7. Release kinetics of CUR from M-GA hydrogel composites
As it was stated before, our intention is to take advantage of GA as pH-responsive gel to evaluate its possible use as a matrix for drugs acting at the colon and allowing oral administration. Following that, the effect of pH on the release profiles of α-CUR/CD, from M-GA (15% w/v)/CUR/α-CD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 10% w/w), to SIF (pH 1.2) and SGF (pH 6.8) solutions has been studied. Fig. 9 shows the release kinetics of the CUR inclusion complex. At first we can infer that, depending on the pH, the release profile of CUR is significantly different. At pH 1.2 the cumulative release of CUR (for the sake of simplicity the complex is just labelled as CUR) is only 6.7%; on the other hand the cumulative release of CUR for simulated intestinal fluid is equal to 96.4% (taking the amount of CUR initially incorporated inside the gel). However, this release occurs via two well defined steps: at the beginning (say, between 0 min and 1080 min) there occurs a release of 21.5%, followed by a second step characterized by a cumulative release equal to 74.9%. This behavior clearly suggests a pH-responsive behavior. This can be related to the deprotonation of the carboxylate groups present in the GA polysaccharide chain above the pKa (≈4.03),31 mainly generating COO−. Thus, in SIF the repulsive forces between the anions of the ionized groups of glucuronic acid segments promote hydrogel swelling and erosion and, consequently, enhance the CUR release.
4, 10% w/w), to SIF (pH 1.2) and SGF (pH 6.8) solutions has been studied. Fig. 9 shows the release kinetics of the CUR inclusion complex. At first we can infer that, depending on the pH, the release profile of CUR is significantly different. At pH 1.2 the cumulative release of CUR (for the sake of simplicity the complex is just labelled as CUR) is only 6.7%; on the other hand the cumulative release of CUR for simulated intestinal fluid is equal to 96.4% (taking the amount of CUR initially incorporated inside the gel). However, this release occurs via two well defined steps: at the beginning (say, between 0 min and 1080 min) there occurs a release of 21.5%, followed by a second step characterized by a cumulative release equal to 74.9%. This behavior clearly suggests a pH-responsive behavior. This can be related to the deprotonation of the carboxylate groups present in the GA polysaccharide chain above the pKa (≈4.03),31 mainly generating COO−. Thus, in SIF the repulsive forces between the anions of the ionized groups of glucuronic acid segments promote hydrogel swelling and erosion and, consequently, enhance the CUR release.
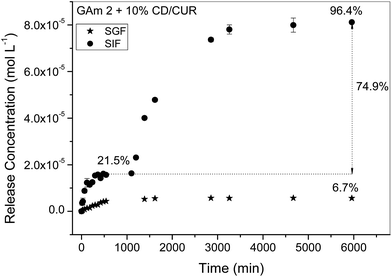 | ||
| Fig. 9 Release kinetics of CUR from M-GA (15% w/v) hydrogels and CUR/α-CD (10% w/w) in SGF and SIF at 37.0 °C. The initial CUR concentration inside the hydrogel is equal to 1.31 × 10−4 mol L−1. | ||
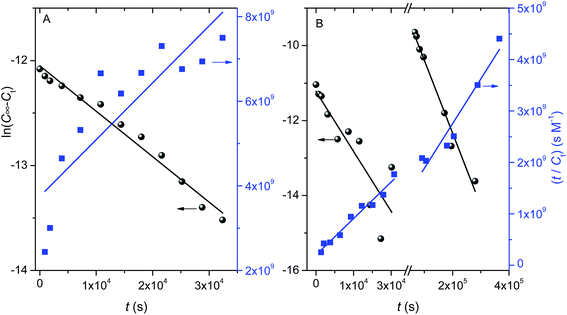
To gain further insight into the release mechanism, the experimental data were fitted to eqn (1). The analysis of the fitting parameter n shows that the release process in SGF is anomalous or non-Fickian, indicting that CUR release occurs due to a combination of polymer macromolecular chain relaxation of the matrix and Fickian transport; the latter occurs when the release is only dependent on the drug concentration gradient. Despite the limited number of experimental data points used to calculate n values for SIF systems, it is possible to conclude that the release at times lower than 18.3 h (1st step) has a mechanism similar to that observed for SIF; in the 2nd step the release changes to a Case II transport mechanism, characterized by a constant release rate, i.e., independent of drug concentration, suggesting that the CUR deliver system is swelling-controlled.57 This second stage behavior can also be due to matrix erosion as it was found for similar systems.58,59 It should also be stressed that such a hypothesis is also confirmed by the goodness of the fit (not shown) of the Hixson–Crowell equation (eqn (2)) to experimental data representative of the second stage release (R2 = 0.9863).
Support for this also comes from the analysis of the surface morphology of M-GA/CUR/α-CD hydrogel membranes after immersion in SGF (pH 1.2) and SIF (pH 6.8) (Fig. 10A and B, respectively). In the case of gels immersed at pH 1.2 solution, the surface shows well-defined pores, although with smaller diameters than those observed in Fig. 7B, with holes at the bottom of cavity; however, the gel membrane immersed in SIF shows quite a rough entropic surface, showing that the degradation of gel structure is taking place.
 | ||
| Fig. 10 Scanning electron micrographs of M-GA/CUR/α-CD hydrogel membranes previously swelled in (A) SGF and (B) SIF (×1500). | ||
The kinetics analysis shows that the CUR release is also dependent on pH. Fig. 11 shows plots of the fitting of eqn (3) and (4) to experimental data of release kinetics. From this analysis, and from the analysis of fitting parameters summarised in Table 4, it can be seen that at pH 1.2 the pseudo-first order model shows a better correlation coefficient (Fig. 11A). This clearly suggests that the free volume (and consequently the diffusion) has, most probably, a predominant role in the release of CUR. However, for SIF systems the release follows a pseudo-second order kinetic model (Fig. 11B and Table 4). This can be concluded from the assessment of correlation coefficients and C∞ values as well. Indeed, C∞ values computed from eqn (4) (1.8 × 10−5 M and 1.2 × 10−4 M, for the 1st and 2nd release stages, respectively) match well with the corresponding experimental values: 2.8 × 10−5 M and 1.26 × 10−4 M, respectively. This indicates that physisorption is not the rate-limiting step for the release of CUR from M-GA-based matrices;60 i.e., it suggests that diffusion is governed by the relaxation of polymer chains in the swollen state, thus facilitating drug release outside the matrix,61,62 in agreement with the anomalous and Case-II mechanisms described by eqn (1). It is also worth noting that the rate constant decreases ca. 20 times from the 1st to the second stage (13 s−1 M−1 to 0.68 s−1 M−1, respectively). This can be justified by the increase in the CUR concentration gradient (once a great majority of CUR remains inside gel after the 1st stage), leading to an increase of CUR–CUR interactions63 and, consequently, the resistance to the “hydrodynamic” flux tends to increase; another possible justification is a higher mobility (and relaxation) of polymeric chain, as a consequence of erosion, contributing for a higher (drag) resistance to the CUR release. Both these effects can contribute to a decrease in k2 values for the 2nd stage when compared to the first one.
| Eqn | SGF | SIF | ||
|---|---|---|---|---|
| 1st stage, t < 1098 min | 2nd stage, t > 1098 min | |||
| a Values inside brackets are standard deviations of the average. | ||||
| (1) | n | 0.65 (±0.03) | 0.62 (±0.20) | 0.96 (±0.18) |
| R2 | 0.9887 | 0.8159 | 0.9345 | |
| (3) | k1 (10−5 s−1) | 4.3 (±0.2) | 12.8 (±2.5) | 1.9 (±0.1) |
| C∞ (M) | 5.8 (±0.2) × 10−6 | 1.3 (±0.5) × 10−5 | 2.1 (±0.4) × 10−4 | |
| R2 | 0.9780 | 0.7283 | 0.9774 | |
| (4) | k2 (s−1 M−1) | 4.8 (±1.5) | 13 (±3) | 0.68 (±0.12) |
| C∞ (M) | 7.4 (±1.5) × 10−6 | 1.8 (±0.1) × 10−5 | 1.2 (±0.2) × 10−4 | |
| R2 | 0.7172 | 0.9689 | 0.9834 | |
3.8. Cytotoxicity assays on the Caco-2 cells
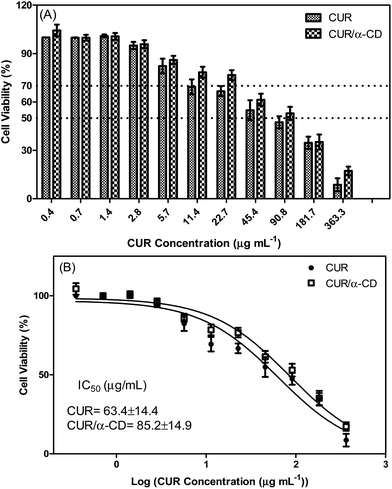
The human intestinal Caco-2 cell line has been used as a model of the intestinal barrier64 and it can also be used to evaluate the cytotoxicity of formulations that were designed to be administered by the oral route. Cells are grown in vitro, under standard culture conditions, in the absence of inducers of differentiation mimicking enterocytes that line mucosal surfaces. In this study, CUR in cyclodextrin formulations and a hydrogel of M-GA were placed in contact with cells for 24 h and cell viability was evaluated. The different samples were plated according to the CUR concentration. The cell viability (%) for CUR and CUR/α-CD as a function of CUR concentration is shown in Fig. 12A. For concentrations of CUR below 2.8 μg mL−1 the cellular viability was about 100%, while for higher concentrations of CUR there was a decrease in cell viability with increasing concentration of curcumin in contact with the cells. A cell viability of 70%, from which samples are considered toxic, was observed for the samples whose CUR concentration is higher than 22.7 μg mL−1. The inhibitory concentration of 50% (IC50) for cells was estimated using the GraphPad Prism program (Fig. 12B). The IC50 values for CUR and CUR/α-CD were 63.4 ± 14.4 μg mL−1 and 85.2 ± 14.9 μg mL−1, respectively. Moreover, the dose–response curves from same assays with Caco-2 cells (Fig. 12B) showed similar profiles and close IC50 values; however, when the Student’s t-test was applied to the curves, using the GraphPad Prism program, the results obtained showed that the curves are significantly different at P < 0.05. Therefore, the CUR complexed with α-CD has a lower toxicity (higher IC50). The association of the CUR with α-CD is thus shown to be very advantageous, as it not only increased CUR solubility but also decreased cytotoxicity.The cytotoxicity assays of hydrogels containing CUR/CD complexes were performed maintaining the same CUR concentrations described in previous test. A control experiment was performed to evaluate the cytotoxicity of the hydrogel in the absence of the complex. Fig. S2 (ESI†) shows cell viability as a function of hydrogel concentration for M-GA/CUR/α-CD and M-GA hydrogels. The results were very similar for both samples and showed no significant differences (P < 0.05). The M-GA hydrogel and the M-GA/CUR/α-CD hydrogel samples had no toxic effect on Caco-2 cells until the hydrogel concentration of 18 mg mL−1 was reached (viability > 70%). A significant decrease in cell viability was observed for samples with high concentrations (36 mg mL−1) and may be explained by the formation of a gel barrier between the cells and the culture medium that prevents passage of gases and nutrients to the cells. However, this concentration is extremely high and for this reason both samples were not considered cytotoxic to the undifferentiated Caco-2 cells.
Thus, unlike the CUR/α-CD samples, the CUR/α-CD complex in the hydrogel showed no cytotoxicity towards Caco-2 cells, enabling the application of this material for pharmaceutical purposes.
3.9. Assays with differentiated Caco-2 cells
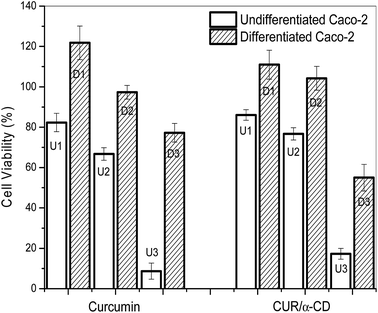
The cell differentiation was made to simulate the conditions in the human intestine and to mimic as close as possible the physical defensive properties of the intestinal mucosa.64,65 The cell viability of differentiated Caco-2 cells was evaluated in the presence of 4 different formulations CUR, CUR/α-CD, and M-GA and M-GA/CUR/α-CD, each one at 3 different concentrations ([CUR] = 5.7 μg mL−1, 22.7 μg mL−1 and 363.3 μg mL−1).The hydrogel sample without CUR was used as control for the three dimensional matrix of the M-GA/CUR/α-CD samples. The CUR cell viability in the three dimensional matrix (M-GA/CUR/α-CD) on differentiated Caco-2 cells presented the same profile observed for undifferentiated Caco-2 cells, this means that there was no cytotoxicity for the two lower concentrations ([CUR] = 5.7 μg mL−1 and 22.7 μg mL−1), while for high CUR concentration (363.3 μg mL−1) the decreased of cell viability is due to the high amount of three dimensional matrix in contact with the cells (not shown). In addition, the cytotoxic effect of the CUR and CUR/α-CD complex was evaluated on differentiated Caco-2 cells (D) and undifferentiated cells (U) (Fig. 13). The curcumin concentrations in the samples were 5.7 μg mL−1, 22.7 μg mL−1 and 363.3 μg mL−1, shown in Fig. 13 as U1, U2 and U3, respectively for the undifferentiated cells and D1, D2 and D3, respectively, for differentiated cells. For both samples CUR and CUR/α-CD, at different CUR concentrations, there was less toxicity of the samples on the differentiated cells compared to undifferentiated Caco-2 cells. A special remarkable difference was observed for the high curcumin concentration, associated or not-associated with α-CD. The differentiation process was conducted in order to mimic the existing conditions in the intestine. Therefore it can be considered that the evaluation of the formulation cytotoxicity with differentiated cells would be closer to the real situation. Additionally, differentiated Caco-2 cells are able to express morphological and functional features, related to the transport and barrier function of small intestinal enterocytes. Theoretically, a higher sensitivity would be expected upon differentiation and this feature would be translated with a higher formulation toxicity profile.66 However there are several reports where this feature was not observed.66 This is the case with the present study, a low cytotoxicity of CUR and CUR/α-CD on differentiated cells was observed. A possible explanation for this result is related to the time (14–21 days) required for cell differentiation that most probably was not sufficient to complete differentiation and consequently to acquire higher cell sensitivity. On the other side, the time (14–21 days) was sufficient to allow the cells to form a barrier that would allow a better protection against the effects of the formulation. Finally, since during cell differentiation the number of cells per well increases, the ratio number of cells![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) formulation mass is consequently different, when comparing the two assays (differentiated and undifferentiated cells).
formulation mass is consequently different, when comparing the two assays (differentiated and undifferentiated cells).
In summary, it is apparent that the cytotoxicity of CUR e CUR/α-CD samples was lower in the differentiated cells. On the other hand, the complex incorporated into the hydrogels showed no toxicity on differentiated and undifferentiated Caco-2 cells, indicating the potential of three-dimensional matrices as carrier system for CUR and cyclodextrins.
4. Conclusions
The complexation of CUR with α-CD and β-CD in different CUR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CD molar ratios is a viable alternative to solve the low solubility of CUR in biological fluids. The obtained supramolecular complexes of CUR/α-CD and CUR/β-CD showed 1
CD molar ratios is a viable alternative to solve the low solubility of CUR in biological fluids. The obtained supramolecular complexes of CUR/α-CD and CUR/β-CD showed 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 stoichiometries; although the latter complex shows the highest binding constant which can be justified by steric factors, since the aromatic ring fits well into the β-CD cavity, the CUR/α-CD complex formed through H-bridge interactions (K = 344 mol−1 L) shows the highest solubility, justified by the highest solubility of α-CD. In order to use this complex further for pharmacological applications, the optimization of the complex solubility was studied and it was concluded that the highest solubility of CUR is reached for CUR/α-CD (1
2 stoichiometries; although the latter complex shows the highest binding constant which can be justified by steric factors, since the aromatic ring fits well into the β-CD cavity, the CUR/α-CD complex formed through H-bridge interactions (K = 344 mol−1 L) shows the highest solubility, justified by the highest solubility of α-CD. In order to use this complex further for pharmacological applications, the optimization of the complex solubility was studied and it was concluded that the highest solubility of CUR is reached for CUR/α-CD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) complex stoichiometry, which is in agreement with the low binding constant of this complex. The formation of supramolecular complexes CUR/CD was confirmed by 1H NMR and thermogravimetric analysis.
4) complex stoichiometry, which is in agreement with the low binding constant of this complex. The formation of supramolecular complexes CUR/CD was confirmed by 1H NMR and thermogravimetric analysis.
Taking into account that the main aim of this work was to find out the best formulation for oral administration of curcumin and having in mind the colon as the target, the chemical modification by methacrylation of gum arabic was carried out and, consequently hydrogels of M-GA containing CUR/α-CD (at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (mol/mol)) were produced. The incorporation of curcumin was followed by spectroscopic measurements and the morphology of the obtained gels was followed by SEM. The release kinetics of curcumin from the hydrogel matrices at different pH values by using SI and SG fluids were analysed. The release kinetics and release mechanism are pH-dependent, showing a high stability of the matrix and low curcumin release at SGF and a high curcumin release efficiency in the SIF due to the degradation of the hydrogel matrix, as can be observed by SEM micrographs. In addition, cytotoxicity assays of samples on undifferentiated and differentiated Caco-2 cells showed that the complexes of CUR/α-CD incorporated into hydrogels are non-toxic. With these results we found, to the best of our knowledge, an efficient methodology to increase the bioavailability of curcumin, and the formulation using the M-GA hydrogel as matrix allows the triggering of curcumin release in SIF conditions.
4 (mol/mol)) were produced. The incorporation of curcumin was followed by spectroscopic measurements and the morphology of the obtained gels was followed by SEM. The release kinetics of curcumin from the hydrogel matrices at different pH values by using SI and SG fluids were analysed. The release kinetics and release mechanism are pH-dependent, showing a high stability of the matrix and low curcumin release at SGF and a high curcumin release efficiency in the SIF due to the degradation of the hydrogel matrix, as can be observed by SEM micrographs. In addition, cytotoxicity assays of samples on undifferentiated and differentiated Caco-2 cells showed that the complexes of CUR/α-CD incorporated into hydrogels are non-toxic. With these results we found, to the best of our knowledge, an efficient methodology to increase the bioavailability of curcumin, and the formulation using the M-GA hydrogel as matrix allows the triggering of curcumin release in SIF conditions.
Acknowledgements
A. P. G. and D. C. S. are grateful for the scholarship provided by CAPES/CSF. Financial support from FCT (Portugal) and CNPq (Brazil), or FCT/CAPES application (Proc. No. 329/13), allowing scientific cooperation between Brazilian and Portuguese research groups, is gratefully acknowledged. This work was also partially funded by FEDER funds through the Operational Programme Competitiveness Factors – COMPETE and national funds by FCT under strategic project UID/NEU/04539/2013.References
- D. Patra and C. Barakat, Spectrochim. Acta, Part A, 2011, 79, 1034–1041 CrossRef CAS PubMed.
- A. Jitoe-Masuda, A. Fujimoto and T. Masuda, Curr. Pharm. Des., 2013, 19, 2084–2092 CAS.
- P. Anand, A. B. Kunnumakkara, R. A. Newman and B. B. Aggarwal, Mol. Pharmaceutics, 2007, 4, 807–818 CrossRef CAS PubMed.
- B. B. Aggarwal, C. Sundaram, N. Malani and H. Ichikawa, Adv. Exp. Med. Biol., 2007, 595, 1–75 CrossRef PubMed.
- C. Araújo and L. Leon, Mem. Inst. Oswaldo Cruz, 2001, 96, 723–728 CrossRef.
- S. N. Jangle, Pravara Med. Rev., 2010, 2, 4–7 Search PubMed.
- V. Pongrakhananon and Y. Rojanasakul, in Advances in Cancer Therapy, ed. H. Gali-Muhtasib, INTECH Open Access Publisher, 2011 Search PubMed.
- D. W. Zhang, M. Fu, S. H. Gao and J. L. Liu, J. Evidence-Based Complementary Altern. Med., 2013, 2013, 636053 Search PubMed.
- C.-H. Hsu and A.-L. Cheng, in The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease, ed. B. B. Aggarwal, Y.-J. Surh and S. Shishodia, Springer Science+Business Media, LLC, New York, 2007 Search PubMed.
- A. Darvesh, R. Carroll, A. Bishayee, N. Novotny, W. Geldenhuys and C. V. D. Schyf, Expert Opin. Invest. Drugs, 2012, 21, 1123–1140 CrossRef CAS PubMed.
- J. J. Bright, in The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease, ed. B. B. Aggarwal, Y.-J. Surh and S. Shishodia, Springer Science+Business Media, LLC, New York, 2007 Search PubMed.
- G. M. Cole, B. Teter and S. A. Frautschy, Adv. Exp. Med. Biol., 2007, 595, 197–212 CrossRef PubMed.
- M. L. Lestari and G. Indrayanto, Profiles of drug substances, excipients, and related methodology, Academic Press, San Diego, 2014, vol. 39, pp. 113–204 Search PubMed.
- M. Hasan, N. Belhaj, H. Benachour, M. Barberi-Heyob, C. J. Kahn, E. Jabbari, M. Linder and E. Arab-Tehrany, Int. J. Pharm., 2014, 461, 519–528 CrossRef CAS PubMed.
- L. Liu, L. Sun, Q. Wu, W. Guo, L. Li, Y. Chen, Y. Li, C. Gong, Z. Qian and Y. Wei, Int. J. Pharm., 2013, 443, 175–182 CrossRef CAS PubMed.
- Y. Xiao, X. Chen, L. Yang, X. Zhu, L. Zou, F. Meng and Q. Ping, J. Agric. Food Chem., 2013, 61, 3654–3660 CrossRef CAS PubMed.
- E. J. Da-Lozzo, R. C. Moledo, C. D. Faraco, C. F. Ortolani-Machado, T. M. Bresolin and J. L. Silveira, Carbohydr. Polym., 2013, 93, 279–284 CrossRef CAS PubMed.
- T. Harada, L. Giorgio, T. J. Harris, D. T. Pham, H. T. Ngo, E. F. Need, B. J. Coventry, S. F. Lincoln, C. J. Easton, G. Buchanan and T. W. Kee, Mol. Pharmaceutics, 2013, 10, 4481–4490 CrossRef CAS PubMed.
- V. R. Yadav, S. Suresh, K. Devi and S. Yadav, AAPS PharmSciTech, 2009, 10, 752–762 CrossRef CAS PubMed.
- O. Naksuriya, S. Okonogi, R. M. Schiffelers and W. E. Hennink, Biomaterials, 2014, 35, 3365–3383 CrossRef CAS PubMed.
- S. Wang, M. Tan, Z. Zhong, M. Chen and Y. Wang, J. Nanomater., 2011, 2011, 1–8 Search PubMed.
- G. Crini, Chem. Rev., 2014, 114, 10940–10975 CrossRef CAS PubMed.
- K. A. Connors, Chem. Rev., 1997, 97, 1325–1357 CrossRef CAS PubMed.
- T. Loftsson and M. E. Brewster, J. Pharm. Sci., 2012, 101, 3019–3032 CrossRef CAS PubMed.
- B. Medronho, A. J. M. Valente, P. Costa and A. Romano, Colloid Polym. Sci., 2014, 292, 885–894 CAS.
- S. V. Kurkov and T. Loftsson, Int. J. Pharm., 2013, 453, 167–180 CrossRef CAS PubMed.
- A. J. Valente and O. Soderman, Adv. Colloid Interface Sci., 2014, 205, 156–176 CrossRef CAS PubMed.
- G. Castronuovo and M. Niccoli, Bioorg. Med. Chem., 2006, 14, 3883–3887 CrossRef CAS PubMed.
- A. Vashist, A. Vashist, Y. K. Guptac and S. Ahmad, J. Mater. Chem. B, 2014, 2, 147–166 RSC.
- T. Coviello, P. Matricardi, C. Marianecci and F. Alhaique, J. Controlled Release, 2007, 119, 5–24 CrossRef CAS PubMed.
- A. V. Reis, M. R. Guilherme, O. A. Cavalcanti, A. F. Rubira and E. C. Muniz, Polymer, 2006, 47, 2023–2029 CrossRef CAS.
- C. S. Mangolim, C. Moriwaki, A. C. Nogueira, F. Sato, M. L. Baesso, A. M. Neto and G. Matioli, Food Chem., 2014, 153, 361–370 CrossRef CAS PubMed.
- J. F. Maior, A. V. Reis, E. C. Muniz and O. A. Cavalcanti, Int. J. Pharm., 2008, 355, 184–194 CrossRef CAS PubMed.
- R. W. Korsmeyer, R. Gumy, E. Doelker, P. Buri and N. A. Peppas, Int. J. Pharm., 1983, 15, 25–35 CrossRef CAS.
- D. Costa, A. J. Valente, M. G. Miguel and J. Queiroz, Langmuir, 2011, 27, 13780–13789 CrossRef CAS PubMed.
- A. W. Hixson and J. H. Crowell, Ind. Eng. Chem., 1931, 23, 1160–1168 CrossRef CAS.
- Y. Asci, U. Acikel and Y. S. Acikel, Environ. Technol., 2012, 33, 1857–1868 CrossRef CAS PubMed.
- R. Benassi, E. Ferrari, S. Lazzari, F. Spagnolo and M. Saladini, J. Mol. Struct., 2008, 892, 168–176 CrossRef CAS.
- A. Dutta, B. Boruah, A. K. Manna, B. Gohain, P. M. Saikia and R. K. Dutta, Spectrochim. Acta, Part A, 2013, 104, 150–157 CrossRef CAS PubMed.
- P. R. K. Mohan, G. Sreelakshmi, C. V. Muraleedharan and R. Joseph, Vib. Spectrosc., 2012, 62, 77–84 CrossRef CAS.
- M. M. Yallapu, M. Jaggi and S. C. Chauhan, Colloids Surf., B, 2010, 79, 113–125 CrossRef CAS PubMed.
- M. E. Brewster and T. Loftsson, Adv. Drug Delivery Rev., 2007, 59, 645–666 CrossRef CAS PubMed.
- R. Singh, N. Bharti, J. madan and S. N. Hiremath, J. Pharm. Sci. Technol., 2010, 2, 171–183 CAS.
- Z.-Z. Li, Q.-X. Guo, T. Ren, X.-Q. Zhu and Y.-C. Liu, J. Inclusion Phenom. Mol. Recognit. Chem., 1993, 15, 37–42 CrossRef CAS.
- N. Funasaki, S. Ishikawa and S. Neya, J. Phys. Chem. B, 2003, 107, 10094–10099 CrossRef CAS.
- H.-J. Schneider and F. H. V. Rudiger, Chem. Rev., 1998, 98, 1755–1785 CrossRef CAS PubMed.
- M. Fathy and M. Sheha, Pharmazie, 2000, 55, 513–517 CAS.
- F. B. T. Pessine, A. Calderini and G. L. Alexandrino, in Magnetic Resonance Spectroscopy, ed. D. Kim, InTech, 2012, pp. 237–264 Search PubMed.
- V. Jahed, A. Zarrabi, A. K. Bordbar and M. S. Hafezi, Food Chem., 2014, 165, 241–246 CrossRef CAS PubMed.
- N. M. Patro, A. Sultana, K. Terao, D. Nakata, A. Jo, A. Urano, Y. Ishida, R. N. Gorantla, V. Pandit, K. Devi, S. Rohit, B. K. Grewal, E. M. Sophia, A. Suresh, V. K. Ekbote and S. Suresh, J. Inclusion Phenom. Macrocyclic Chem., 2014, 78, 471–483 CrossRef CAS.
- K. N. Baglole, P. G. Boland and B. D. Wagner, J. Photochem. Photobiol., A, 2005, 173, 230–237 CrossRef CAS.
- B. Tang, L. Ma, H.-Y. Wang and G.-Y. Zhang, J. Agric. Food Chem., 2002, 50, 1355–1361 CrossRef CAS PubMed.
- G. Chalumot, C. Yao, V. Pino and J. L. Anderson, J. Chromatogr. A, 2009, 1216, 5242–5248 CrossRef CAS PubMed.
- H. A. Benesi and J. H. Hildebran, J. Am. Chem. Soc., 1949, 71, 2703–2707 CrossRef CAS.
- Y. Bai, J. Wang, M. Bashari, X. Hu, T. Feng, X. Xu, Z. Jin and Y. Tian, Thermochim. Acta, 2012, 541, 62–69 CrossRef CAS.
- M. M. Yallapu, M. Jaggi and S. C. Chauhan, Macromol. Biosci., 2010, 10, 1141–1151 CrossRef CAS PubMed.
- R.-H. Juang and D. Storey, J. Controlled Release, 2003, 89, 375–385 CrossRef CAS PubMed.
- D. Costa, A. J. M. Valente, M. G. Miguel and B. Lindman, Colloids Surf., A, 2011, 391, 80–87 CrossRef CAS.
- D. L. Sitta, M. R. Guilherme, E. P. da Silva, A. J. Valente, E. C. Muniz and A. F. Rubira, Colloids Surf., B, 2014, 122, 404–413 CrossRef CAS PubMed.
- Y. S. Ho, J. Hazard. Mater., 2006, 136, 681–689 CrossRef CAS PubMed.
- S. Ekici and D. Saraydin, Drug Delivery, 2004, 11, 381–388 CrossRef CAS PubMed.
- M. Montiel-Herrera, A. Gandini, F. M. Goycoolea, N. E. Jacobsen, J. Lizardi-Mendoza, M. Recillas-Mota and W. M. Arguelles-Monal, Carbohydr. Polym., 2015, 128, 220–227 CrossRef CAS PubMed.
- M. K. Hazra, S. Roy and B. Bagchi, J. Chem. Phys., 2014, 141, 18C501 CrossRef PubMed.
- L.-S. L. Gan and D. R. Thakkerb, Adv. Drug Delivery Rev., 1997, 23, 77–98 CrossRef CAS.
- Y. Sambuy, I. D. Angelis, G. Ranaldi, M. L. Scarino, A. Stammati and F. Zucco, Cell Biol. Toxicol., 2005, 21, 1–26 CrossRef CAS PubMed.
- M. Puerto, S. Pichardo, A. Jos and A. M. Camean, Arch. Toxicol., 2010, 84, 405–410 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra14331d |
| This journal is © The Royal Society of Chemistry 2015 |