Progress in polymeric separators for lithium ion batteries
Abstract
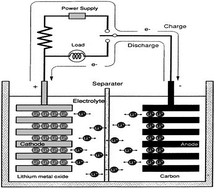
This study reviews the recent developments and the characteristics of polymeric separators used for lithium ion batteries. According to the structure and composition of the separators, they are broadly divided into four types: (1) polyolefin microporous separators, (2) heterochain polymer microporous separators, (3) polymer electrolytes and (4) non-woven separators. In particular, polymer electrolytes were defined as one category of separators for convenient description in this review; these feature intermediates between the two electrodes and possess transport properties comparable with the separator in liquid LIBs. For each category, the structure, characteristics, modification, and performance of separators are described. Finally, guidelines for further improvements in this research are outlined.
- This article is part of the themed collection: Polymers for Electrochemical Energy Storage

 Please wait while we load your content...
Please wait while we load your content...