New approach for post-functionalization of meso-formylporphyrins†
Kirill P. Birin*a,
Yulia G. Gorbunova ab and
Aslan Yu. Tsivadzeab
ab and
Aslan Yu. Tsivadzeab
aA.N. Frumkin Institute of Physical Chemistry and Electrochemistry RAS, Leninsky Prosp. 31, bldg. 4, Moscow, 119071, Russia. E-mail: kirill.birin@gmail.com
bN.S. Kurnakov Institute of General and Inorganic Chemistry RAS, Leninsky Prosp. 31, Moscow, 119991, Russia. E-mail: yulia@igic.ras.ru
First published on 28th July 2015
Abstract
A strategy for the post-functionalization of readily accessible meso-formylporphyrins affording meso-(2-imidazolyl)-porphyrins, containing substituents at the 4,5-positions of the imidazole fragment is developed. The reaction of 5-formyl-10,20-diarylporphyrins with phenanthrene- or phenanthroline-5,6-dione and ammonium acetate provides 5-(areneimidazol-2-yl)-10,20-diarylporphyrins with high yields. This methodology is used to prepare a set of representatives of this new type of substituted porphyrins. The distribution of the frontier orbitals of this class of compounds is evaluated by means of DFT calculations.
Post-modification of meso-arylporphyrins,1–4 which are readily available on the gram scale, provides access to a variety of functionalized compounds with multiple applications. Introduction of different substituents to porphyrin meso- and β-positions allows modification of the chemical, physicochemical and coordination properties of the resulting compounds, which determines their wide applications as molecular tectons for supramolecular chemistry,5–9 in solar cell development,10 catalysis,11,12 molecular electronics,13 photovoltaics,14 molecular recognition,15–17 etc. Among the various approaches for modification of porphyrin molecules, the application of the Vilsmeier–Haack reaction occupies an outstanding position.18 Formyl derivatives can be prepared efficiently from various metal porphyrins,19 but examples of further employment of this functional group is limited because of its low reactivity. Interaction of formylporphyrins with phosphorus ylides,20–22 the reduction of the carbonyl group,23 formation of acetals,24 interaction with Grignard reagents22 and condensations with CH-acidic compounds25,26 are described. In contrast, other reactions, typical for aromatic aldehydes, proceed with formylporphyrins notably different. For example, while oximes can be prepared from formylporphyrins by interaction with hydroxylamine, the related interaction with amines produces Schiff-bases with vanishing yields and the starting material is recovered.27 The examples of chemical transformations of the formylporphyrins, in which the carbonyl group acts as a part of the constructed functional center of the molecule are virtually unknown. The application of formylporphyrins as carbonyl components in the heterocyclic condensations is limited to examples of interaction of β-formylporphyrin with acetylarenes under Kroehnke reaction conditions, providing substituted β-pyridylporphyrins in mixture with corresponding chalcones and substituted benzoporphyrins.28
The interaction of formylporphyrins with α-diketones providing 2,4,5-substituted imidazoles has not been described by now either for β- or for meso-formylporphyrins. Nevertheless this type of junction of the porphyrin and peripheral fragment is attractive for design of semi-rigid polytopic systems. The rigidity of the molecule along its axis with preservation of rotational lability of the fragments allows design of self-adapting molecular tectons for supramolecular chemistry. Moreover, the absence of the conjugation of the fragments, bridged by imidazole moiety, diminishes their mutual influence, allowing straightforward design of polytopic systems with desired properties. The balance between rigidity and conformational lability of the bridging fragment allows precise tuning of the electronic structure and self-association of the polytopic tectons under investigation. The introduction of heterocyclic fragment to the porphyrin allows modification of the coordination and physical–chemical properties of the obtained conjugate.
Meso-(2-imidazolyl)-porphyrins up to date were prepared from 2-formylimidazole or meso-(2-imidazolyl)-dipyrromethane29–31 and consequently did not contain substituents at the 4,5-positions of the heterocycle. The only exception is the preparation of porphyrins, containing benzimidazol-2-yl substituents,32 from corresponding 2-formyl-benzimidazole. Various meso-(2-imidazolyl)-porphyrins were found to be promising compounds for catalytic oxidation reactions,30,33,34 self-assembly of supramolecular architectures,35–39 two-photon absorption in organic media and in water,35,37 non-linear optics,38 intramolecular energy transfer39 and modeling of photosynthetic “special pair”.31,40
With this diversity of possible applications for functionalized meso-(2-imidazolyl)-porphyrins we focused on development of the general synthetic strategy towards meso-(2-imidazolyl)-porphyrins, containing substituents at the 4,5-positions of the imidazole fragment. In present work we report efficient approach for condensation of meso-formylporphyrins and aromatic α-diketones with formation of imidazole heterocyclic bridge between the fragments.
Two diarylporphyrins, containing phenyl or mesityl groups at 5,15-positions and two aromatic α-diketones – phenanthrene-5,6-dione and 1,10-phenanthroline-5,6-dione were selected for investigation. The application of two different diketones reveals their reactivity in the interaction under discussion, while two different types of meso-substituents of the porphyrin macrocycle allow to evaluate the influence of their bulkiness onto the reaction pathway. The studied sequence of chemical transformations is shown at Scheme 1. The formylation of copper(II) porphyrins 1Cu and 2Cu was performed similar to published procedure20 with minor modification. The preparation of the Vilsmeier reagent in dichloroethane solution and decrease of Vilsmeier reagent/porphyrin ratio allowed to increase the yield of corresponding formylporphyrins 3Cu–4Cu up to 98%. The demetalation of 3Cu under acidic conditions20 provided 3H2 nearly quantitatively. The obtained meso-formylporphyrins 3H2, 3Cu, 4Cu were further involved into interaction with α-diketones A and B.
While equimolar interaction of diketones A or B with typical aromatic aldehydes proceeds smoothly and provides high to quantitative yields of the corresponding substituted imidazoles,41–44 in present case we encountered several peculiarities. Thus, the interaction of formylporphyrin 3Cu with equimolar amount of diketone A and 10-fold excess of NH4OAc upon reflux in CHCl3/AcOH mixture did not provide complete conversion of the starting material, as revealed by TLC and MALDI-TOF MS. The stepwise addition of A (3 equiv.) and NH4OAc (30 equiv.) is unavoidable to achieve complete conversion of 3Cu to condensation product upon reflux for 3 days. It is noteworthy, that simultaneous application of 3 equivalents of α-diketone A and 30-fold excess of NH4OAc does not allow complete conversion of 3Cu. We attribute this peculiarity to self-condensation of α-diketones under reaction conditions, which occurs faster than the condensation with meso-formylporphyrin. The treatment of A with 10-fold excess of NH4OAc upon reflux in CHCl3/AcOH mixture results in formation of inattributable mixture of aromatic compounds, while the starting α-diketone could hardly be detected in the mixture after 24 hours of reflux (Fig. S1–S3†). Eventually, formation of 5Cu and 7Cu requires 3-fold excess of A, while 6-fold excess of B is required for preparation of 6Cu and 8Cu. The interaction of 3H2 with A (3 equiv.) under found conditions resulted in formation of corresponding free-base porphyrin 5H2 with 95% yield, but the reaction rate was lower compared to copper complexes and reflux for 5 days was required to achieve complete conversion.
Meso-imidazolylporphyrins, containing phenyl groups in 10,20-positions (5Cu, 6Cu), are found to be virtually insoluble in organic media, even in CHCl3, CH2Cl2 and their mixtures with MeOH, that makes their purification impossible. 5H2 demonstrates slightly increased solubility, allowing its characterization by UV-vis and NMR, but it is also inapplicable for chromatography. In contrast, mesityl-substituted analogues are sufficiently soluble in common organic solvents and can be easily purified by column chromatography. In order to solubilize 5H2, 5Cu, 6Cu we have determined the conditions for their N-alkylation. These compounds were treated with 1-bromobutane in DMF, acetone and acetonitrile in the presence of K2CO3. It was found, that alkylation proceeds smoothly only in DMF at 100 °C and its yield is estimated to be nearly quantitative since the overall yields of 9Cu and 10Cu for two stages are quite similar to the ones of 7Cu and 8Cu.
The prepared meso-(2-imidazolyl)-porphyrins 5Cu, 7Cu–10Cu are found to be sufficiently stable to survive the treatment with mixture of trifluoroacetic and sulfuric acids (7/1) providing corresponding free-base porphyrins with high yields. A mild demetalation procedure with sulfuric acid in CH2Cl2 is described,45 but its application did not allow conversion of metal complexes to free-base porphyrins in our case. Moreover, it should be mentioned, that porphyrins, containing N-alkylimidazole fragment are more stable upon acidic demetalation, than non-alkylated ones, that is revealed by higher yields of 9H2 (97%) and 10H2 (95%), compared to 7H2 (88%) and 8H2 (87%).
All the synthesized compounds were sufficiently characterized by UV-vis spectroscopy and MALDI-TOF MS, while the structures of the free-base porphyrins were additionally proved by NMR (Fig. S4–S30†). The UV-vis spectroscopy reveals the sets of absorption bands, typical for 10,20-diarylporphyrins. Additional low-intensity absorptions are present in the 250–350 nm region and it can be attributed to areneimidazole transitions (Fig. S22–S30†). The expected peaks are observed in the mass-spectra, demonstrating the well-defined isotopic patterns (Fig. S13–S21†).
The analysis of NMR spectra in CDCl3 revealed that behavior of areneimidazole NH-protons depends on the origin of the appended aromatic fragment (Fig. 1). The interpretation of the spectra was based on the intensity and multiplicity of the signals as well as 1H–1H correlation patterns (Fig. S6, S8 and S12†). In the case of imidazophenanthrene-appended porphyrins 7H2 resolved sets of multiplets are observed, corresponding to phenanthrene protons and imidazole NH-proton. The observed set reveals the localization of NH-proton and dissymmetry of the phenanthrene fragment. In contrast, broadening of imidazophenanthroline fragment was revealed in the spectrum of 8H2. In this case the signal of NH-proton is not observed and we attribute this behaviour to inter- or intramolecular proton exchange between imidazole and phenanthroline nitrogen atoms. Moreover, a sharp signal of porphyrin inner NH-protons is observed in all cases, revealing that these protons do not participate the exchange. Moreover, in the case of 10H2 in which imidazole nitrogen is alkylated, a resolved set of resonances of phenanthroline fragment is observed.
 | ||
| Fig. 1 Aromatic regions of 1H NMR spectra (300 MHz, CDCl3) of 7H2, 8H2 and 10H2 (● = Hmeso, ◆ = phenanthrene or phenanthroline protons). | ||
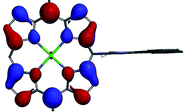
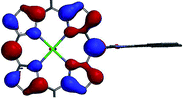
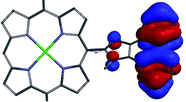
A DFT analysis of the molecular orbitals of the synthesized compounds has been performed in order to reveal the mutual influence of the electronic structures of the aromatic fragments of the molecule. The calculations were carried out at B3LYP/6-31G(d) level of theory with model zinc 5,15-dimesitylporphyrin, bearing 10-imidazophenanthrene fragment, substituted with methyl group at nitrogen atom (Table 1). In this case the orthogonal orientation of aromatic planes was found to be the most favorable. It was found that localization of the frontier orbitals of the molecule depends on the respective orientation of porphyrin ring and areneimidazole unit. The first two unoccupied orbitals (LUMO and LUMO+1) are localized predominantly at porphyrin ring in tilted conformation and exclusively in orthogonal one. HOMO is distributed homogeneously between porphyrin macrocycle and aromatic moiety in tilted conformation, while in orthogonal one it is localized at porphyrin cycle exclusively. Moreover, an orbital HOMO−3 can be found, that is localized exclusively at polyaromatic fragment regardless the skew angle between aromatic planes.
The observed behavior of the molecular orbitals reveals the possibility of the electron and charge transfer processes upon photoexcitation of the molecule with orthogonal orientation of aromatic planes. The energy gap for transitions HOMO−3 → LUMO and HOMO−3 → LUMO+1 correspond to 391 nm and 381 nm wavelengths, respectively. Since B3LYP functional tends to overestimate the energy of transitions up to 0.5 eV,46 it can be presumed that the corresponding absorption bands are overlapped with Soret bands in the UV-vis spectra of the synthesized compounds.
Conclusions
We have developed a new strategy for the post-functionalization of readily accessible meso-formylporphyrins. This approach allows introduction of 2-imidazolyl fragment, fused with functional polyaromatic moiety at 4,5-positions. This transformation represents the first example of utilization of meso-formylporphyrins as carbonyl components in the heterocyclic condensation, despite the low reactivity of meso-formyl group. A set of compounds is synthesized with this approach and their further transformations are demonstrated. The preliminary DFT calculations have revealed the promising electronic properties of the meso-areneimidazolyl-porphyrins for further investigation of charge and electron transfer properties.Acknowledgements
The authors are grateful to the Council of the Russian President for support of young scientists (grant MK-272.2014.3).Notes and references
- J. A. S. Cavaleiro, A. C. Tome and M. G. P. M. S. Neves, Meso-tetraarylporphyrin derivatives: new synthetic methodologies, in Handbook of porphyrin science, ed. K. M. Kadish, K. M. Smith and R. Guilard, 2010, vol. 2, pp. 193–294 Search PubMed.
- Synthesis and modifications of porphyrinoids: in Topics in Heterocyclic Chemistry, v. 33, ed. R. Paolesse, Springer-Verlap, Berlin, Heidelberg, 2014 Search PubMed.
- I. P. Beletskaya, V. S. Tyurin, A. Uglov, C. Stern and R. Guilard, Survey of Synthetic Routes for Synthesis and Substitution in Porphyrins, in Handbook of porphyrin science, ed. K. M. Kadish, K. M. Smith and R. Guilard, 2012, vol. 23, pp. 81–279 Search PubMed.
- N. N. Sergeeva, M. O. Senge and A. Ryan, Organometallic C–C Coupling Reactions for Porphyrins, in Handbook of porphyrin science, ed. K. M. Kadish, R. Guilard and K. M. Smith, 2010, vol. 3, pp. 325–365 Search PubMed.
- V. Bulach and M. W. Hosseini, Porphyrin-Based Tectons in Molecular Tectonics, in Handbook of porphyrin science, ed. K. M. Kadish, K. M. Smith and R. Guilard, 2012, vol. 13, pp. 299–390 Search PubMed.
- I. Beletskaya, V. S. Tyurin, A. Y. Tsivadze, R. Guilard and C. Stern, Chem. Rev., 2009, 109, 1659–1713 CrossRef CAS PubMed.
- Y. Y. Enakieva, A. G. Bessmertnykh, Y. G. Gorbunova, C. Stern, Y. Rousselin, A. Y. Tsivadze and R. Guilard, Org. Lett., 2009, 11, 3842–3845 CrossRef CAS PubMed.
- E. V. Vinogradova, Y. Y. Enakieva, S. E. Nefedov, K. P. Birin, A. Y. Tsivadze, Y. G. Gorbunova, A. G. Bessmertnykh Lemeune, C. Stern and R. Guilard, Chem.–Eur. J., 2012, 18, 15092–15104 CrossRef CAS PubMed.
- A. A. Sinelshchikova, S. E. Nefedov, Y. Y. Enakieva, Y. G. Gorbunova, A. Y. Tsivadze, K. M. Kadish, P. Chen, A. Bessmertnykh-Lemeune, C. Stern and R. Guilard, Inorg. Chem., 2013, 52, 999–1008 CrossRef CAS PubMed.
- L.-L. Li and E. W.-G. Diau, Chem. Soc. Rev., 2012, 42, 291–304 RSC.
- R. De Paula, I. C. M. S. Santos, M. M. Q. Simões, M. G. P. M. S. Neves and J. A. S. Cavaleiro, J. Mol. Catal. A: Chem., 2015, 404–405, 156–166 CrossRef CAS PubMed.
- M. Goswami, V. Lyaskovskyy, S. R. Domingos, W. J. Buma, S. Woutersen, O. Troeppner, I. Ivanović-Burmazović, H. Lu, X. Cui, X. P. Zhang, E. J. Reijerse, S. DeBeer, M. M. van Schooneveld, F. F. Pfaff, K. Ray and B. de Bruin, J. Am. Chem. Soc., 2015, 137, 5468–5479 CrossRef CAS PubMed.
- M. Jurow, A. E. Schuckman, J. D. Batteas and C. M. Drain, Coord. Chem. Rev., 2010, 254, 2297–2310 CrossRef CAS PubMed.
- J. Chen, S. Ko, L. Liu, Y. Sheng, H. Han and X. Li, New J. Chem., 2015, 39, 3736–3746 RSC.
- S. Norrehed, P. Polavarapu, W. Yang, A. Gogoll and H. Grennberg, Tetrahedron, 2013, 69, 7131–7138 CrossRef CAS PubMed.
- K.-I. Hong, H. Yoon and W.-D. Jang, Chem. Commun., 2015, 51, 7486–7488 RSC.
- A. Brown and P. D. Beer, Dalton Trans., 2012, 41, 118–129 RSC.
- G. V. Ponomarev, Chem. Heterocycl. Compd., 1994, 30, 1444–1465 CrossRef.
- J. W. Buchler, C. Dreher and G. Herget, Eur. J. Org. Chem., 1988, 1988, 43–54 Search PubMed.
- T. Morotti, M. Pizzotti, R. Ugo, S. Quici, M. Bruschi, P. Mussini and S. Righetto, Eur. J. Inorg. Chem., 2006, 2006, 1743–1757 CrossRef PubMed.
- O. B. Locos and D. P. Arnold, Org. Biomol. Chem., 2006, 4, 902–916 CAS.
- K. Dahms, M. O. Senge and M. B. Bakar, Eur. J. Org. Chem., 2007, 2007, 3833–3848 CrossRef PubMed.
- N. V. Tkachenko, H. Lemmetyinen, J. Sonoda, K. Ohkubo, T. Sato, H. Imahori and S. Fukuzumi, J. Phys. Chem. A, 2003, 107, 8834–8844 CrossRef CAS.
- C. M. Carcel, J. K. Laha, R. S. Loewe, P. Thamyongkit, K.-H. Schweikart, V. Misra, D. F. Bocian and J. S. Lindsey, J. Org. Chem., 2004, 69, 6739–6750 CrossRef CAS PubMed.
- C. Muthiah, M. Taniguchi, H.-J. Kim, I. Schmidt, H. L. Kee, D. Holten, D. F. Bocian and J. S. Lindsey, Photochem. Photobiol., 2007, 83, 1513–1528 CrossRef CAS PubMed.
- J. Warnan, L. Favereau, F. Meslin, M. Severac, E. Blart, Y. Pellegrin, D. Jacquemin and F. Odobel, ChemSusChem, 2012, 5, 1568–1577 CrossRef CAS PubMed.
- G. V. Ponomarev, Chem. Heterocycl. Compd., 1996, 32, 1263–1280 CrossRef.
- N. M. M. Moura, M. A. F. Faustino, M. G. P. M. S. Neves, A. M. S. Silva, A. C. Tome and J. A. S. Cavaleiro, Chem. Commun., 2012, 48, 6142 RSC.
- J. Bhaumik, Z. Yao, K. E. Borbas, M. Taniguchi and J. S. Lindsey, J. Org. Chem., 2006, 71, 8807–8817 CrossRef CAS PubMed.
- R. D. Paula, M. M. Q. Simões, M. G. P. M. S. Neves and J. A. S. Cavaleiro, Catal. Commun., 2008, 10, 57–60 CrossRef PubMed.
- H. Ozeki, A. Nomoto, K. Ogawa, Y. Kobuke, M. Murakami, K. Hosoda, M. Ohtani, S. Nakashima, H. Miyasaka and T. Okada, Chem.–Eur. J., 2004, 10, 6393–6401 CrossRef CAS PubMed.
- L. R. Milgrom, P. J. Dempsey and G. Yahioglu, Tetrahedron, 1996, 52, 9877–9890 CrossRef CAS.
- R. De Paula, M. M. Q. Simões, M. G. P. M. S. Neves and J. A. S. Cavaleiro, J. Mol. Catal. A: Chem., 2011, 345, 1–11 CrossRef CAS PubMed.
- Y. Miyazaki, A. Satake and Y. Kobuke, J. Mol. Catal. A: Chem., 2008, 283, 129–139 CrossRef CAS PubMed.
- K. Ogawa, H. Hasegawa, Y. Inaba, Y. Kobuke, H. Inouye, Y. Kanemitsu, E. Kohno, T. Hirano, S.-I. Ogura and I. Okura, J. Med. Chem., 2006, 49, 2276–2283 CrossRef CAS PubMed.
- F. Hajjaj, Z. S. Yoon, M.-C. Yoon, J. Park, A. Satake, D. Kim and Y. Kobuke, J. Am. Chem. Soc., 2006, 128, 4612–4623 CrossRef CAS PubMed.
- K. Ogawa, A. Ohashi, Y. Kobuke, K. Kamada and K. Ohta, J. Phys. Chem. B, 2005, 109, 22003–22012 CrossRef CAS PubMed.
- K. Ogawa, T. Zhang, K. Yoshihara and Y. Kobuke, J. Am. Chem. Soc., 2002, 124, 22–23 CrossRef CAS PubMed.
- N. Nagata, Y. Kuramochi and Y. Kobuke, J. Am. Chem. Soc., 2009, 131, 10–11 CrossRef CAS PubMed.
- M. Morisue, N. Haruta, D. Kalita and Y. Kobuke, Chem.–Eur. J., 2006, 12, 8123–8135 CrossRef CAS PubMed.
- M. Mao, J.-B. Wang, Z.-F. Xiao, S.-Y. Dai and Q.-H. Song, Dyes Pigm., 2012, 94, 224–232 CrossRef CAS PubMed.
- N. N. Sergeeva, M. Donnier-Marechal, G. M. Vaz, A. M. Davies and M. O. Senge, Bioorg. Med. Chem. Lett., 2011, 21, 4385–4388 CrossRef CAS PubMed.
- Z. Wang, P. Lu, S. Chen, Z. Gao, F. Shen, W. Zhang, Y. Xu, H. S. Kwok and Y. Ma, J. Mater. Chem., 2011, 21, 5451–5456 RSC.
- Y. Ooyama, H. Kumaoka, K. Uwada and K. Yoshida, Tetrahedron, 2009, 65, 8336–8343 CrossRef CAS PubMed.
- C. M. B. Carvalho, M. G. P. M. S. Neves, A. C. Tome, F. A. A. Paz, A. M. S. Silva and J. A. S. Cavaleiro, Org. Lett., 2011, 13, 130–133 CrossRef CAS PubMed.
- E. Baerends, G. Ricciardi, A. Rosa and S. Van Gisbergen, Coord. Chem. Rev., 2002, 230, 5–27 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Synthetic procedures and all spectral data. See DOI: 10.1039/c5ra13532j |
| This journal is © The Royal Society of Chemistry 2015 |