Rapidly accomplished femtomole soluble CD40 ligand detection in human serum: a “green” homobifunctional agent coupled with reduced graphene oxide-tetraethylene pentamine as platform
Abstract
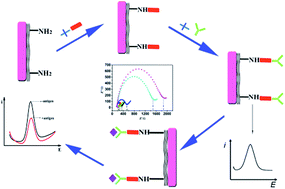
The analysis of soluble CD40 ligand (sCD40L), which is present at significant levels in the blood of patients with cardiovascular disease, can reveal the severity of the disease at its early stage. However, the current biomarker detection techniques exhibit poor detection limits. To accomplish this main challenge, herein, we demonstrate an assay based on a novel modified electrochemical immunosensor for the ultrasensitive assay of sCD40L in human serum, which uses β-cyclodextrin (CD) and reduced graphene oxide-tetraethylene (rGO-TEPA) as a platform. rGO-TEPA contains a great number of amino groups and has excellent conductivity, which makes it a promising material for application in electrochemical biosensor development. To further improve the solubility and stability of rGO-TEPA, CD was selected. The CD decorated rGO-TEPA film not only improved the electron transfer but also provided more amino-groups for the immobilization of antibodies. For speeding up the immobilization of antibodies, the amine-modified electrodes were functionalized by a “green” conjugation route using a lower toxicity homobifunctional 1,4-phenylene diisothiocyanate (PDITC) linker. This is the first study that challenges electrochemical immunosensors with CD-rGO-TEPA-PDITC as a platform for the detection of biomarkers. Under optimal conditions, sCD40L could be assayed in the range of 0.25 to 50 pg mL−1 with detection limits of 83.3 fg mL−1 (S/N = 3). We demonstrate excellent specificity and show that the proposed assay accurately detects the protein of interest. The results were in agreement with an enzyme linked immunosorbent assay, suggesting that the electrochemical immunosensor may possess potential towards use in clinical applications of the proposed immunosensor.

 Please wait while we load your content...
Please wait while we load your content...