Biochemical and histological changes in the liver and gills of Nile tilapia Oreochromis niloticus exposed to Red 195 dye
Abstract
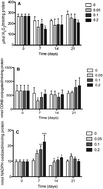
The present study investigates the biochemical and morphological responses induced in the liver and gills of Nile tilapia Oreochromis niloticus by exposure to various Red 195 dye concentrations (0.05, 0.1 and 0.2 mg L−1) for various durations (7, 14 and 21 days). The histology and antioxidant activities of catalase (CAT), glutathione reductase (GR) and glutathione S-transferase (GST) were monitored and evaluated during exposure and after recovery in clean water. The results revealed that CAT activity decreased after 7 and 21 days of exposure to 0.2 mg L−1 concentrations and increased only after 7 days of exposure to high concentrations compared to the liver and gill control samples, respectively. Both organs were, however, noted to undergo a decrease in GST activity after 7 days of exposure to low Red 195 dye concentrations. Compared to the control, the gills of the tilapia exposed to 0.05 and 0.1 mg L−1 concentrations for longer periods underwent an increase in GST activity. Similarly, GR activity was higher in the liver of tilapia exposed to this dye at the sampling days, except for the highest concentration (0.2 mg L−1) after 21 days of exposure. The GR activity in the gills decreased significantly after 7 and 21 days of exposure to 0.05 and 0.1 mg L−1 concentrations, respectively. The results of the recovery group revealed that the liver and gills displayed insignificant differences in antioxidant enzyme activities. The liver and gill tissues of the fish exposed to Red 195 showed several histopathological changes. Liver damages included an increase in cytoplasmic vacuolization, disruption of endothelial lining, metabolic zonation, necrotic cells, rupture of hepatocyte membrane, and decline of higher eosinophilia. The gills also exhibited some necrotic cells, edema, lifting of filaments and lamellar epithelium, and vascular disorders, such as extreme vasodilatation and proliferation of filament epithelium. A correlation between the biochemical and histological changes of the liver and gill tissues was established, attributing the tissue and cell damages to the accumulation of hydrogen peroxide or production of other radicals via a Fenton reaction.

 Please wait while we load your content...
Please wait while we load your content...