Benzothiazepines: chemistry of a privileged scaffold
Debasmita Saha
,
Garima Jain
and
Anuj Sharma
*
Department of Chemistry, Indian Institute of Technology Roorkee, Roorkee-247667, India. E-mail: anujs77@gmail.com; Tel: +91-1332-284751
First published on 6th August 2015
Abstract
Benzothiazepines are one such class of heterocyclic scaffolds with celebrated biological activities. The flooding of the pharmaceutical market with cardiovascular drugs like diltiazem, clentiazem and siratiazem bearing the benzothiazepine core confirms the biological importance of the core. This review presents a comprehensive vision of the various synthetic tactics adopted till now to afford benzothiazepine cores.
1. Introduction
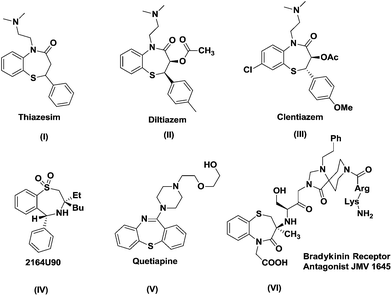
Medicinal chemistry is related to the discovery, development, interpretation and identification of the mechanism of action of biologically active compounds at the molecular level.1 The organic synthesis of new compounds plays a vital role in this tedious process of drug discovery. Particularly, the development of privileged heterocyclic scaffolds is an ever burgeoning area of research in medicinal chemistry. The term ‘privileged structure’ being coined by Evans et al. in 1988 in relation to the heterocycle 1,4-benzodiazepine-2-one was defined as “a single molecular framework able to provide ligands for diverse receptors”. The molecular framework of a ‘privileged structure’ has versatile binding extensions that enable it to be projected as a potent and selective ligand for a range of different biological targets.2Benzothiazepines are one such class of heterocyclic scaffolds with celebrated biological activities in the central nervous system and other therapeutic actions.3 After years of thorough research in the area of this heterocyclic core, benzothiazepines have firmly stood as a “drug prejudice core” due to their presence in a wide range of bioactive compounds like antimicrobials, antifungals, Ca2+ antagonists, CNS depressants, antiplatelet aggregation inhibitors, anti-HIV drugs, calmodulin antagonists and bradykinin receptor antagonists.4
The therapeutic journey of benzothiazepines can be traced back to the entry of the anti-depressant “thiazesim” into the pharmaceutical market followed by diltiazem, clentiazem, and siratiazem, the cardiovascular drugs of this family.5 Further optimization of the substituents around the benzothiazepine nucleus resulted in many drugs like quetiapine fumarate and thiazesim for treating CNS disorders, 2164U90 as a bile acid active transport system inhibitor and JMV1645 as a bradykinin receptor antagonist (Fig. 1).6 This seven membered ring system is also widely acclaimed as an eminent cardiotherapeutic and psychotherapeutic scaffold.7
The pharmaceutical prominence of thiazepines has encouraged some reviews to be documented in the literature.8 These reviews are primarily focused on the compilation of reports on all of the activities associated with the benzothiazepine nucleus and few have been organized from a synthetic perspective. Limited compiled literature reports on the synthetic advances towards various possible thiazepine nuclei motivated us to script this document.
Consequently, this review presents a systematic assemblage of more than 60 research articles and reviews to provide a comprehensive vision of the various synthetic tactics adopted till now to afford benzothiazepine cores.
2. Classification of benzothiazepines
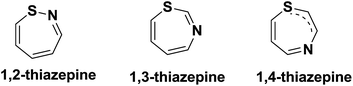
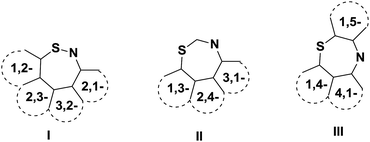
Benzothiazepines are benzo-fused analogues of a seven membered thiazepine ring. The three structural isomers of thiazepines are shown in Fig. 2.Interestingly, several structures of this important seven membered core involve three instead of two annulated rings in which the seven membered thiazepine is commonly sandwiched between a benzene ring and another aromatic or heteroaromatic ring. The role of an additional ring to the bicyclic benzothiazepine core is to restrict the inversion of the seven membered ring which improves the thermodynamic profile of the scaffold. Both bicyclic and tricyclic forms of thiazepines can exist in various isomeric forms. The ten possible structural isomers of benzothiazepine including six dibenzothiazepines are depicted in Fig. 3.
To date, most of the reported compounds have been from the type III scaffold for the reasons of sheer abundance of pharmaco-profiling of this class of compounds. The present review article summarizes the synthetic progress of all three types of scaffold and their various subtypes.
3. Biological activities of benzothiazepines
As mentioned before, both benzothiazepines and tricyclic thiazepines have been reported in the domain of pharmaco-active compounds with profound biological activities. In several cases, these compounds have been known to cross the blood–brain barrier with ease and subsequently used as psycho-active drug candidates. All pharmacologically active thiazepine scaffolds after the aforementioned work are shown in Table 1.| S. no | Compound name | Structure | Use | Reference |
|---|---|---|---|---|
| 1 | 2,4-Diaryl-2,3-dihydro-1,5-benzothiazepine |  |
Antifungal | 9 and 10 |
| 2 | 1,1-Dioxide-2,4-diaryl-2,3-dihydro-1,5-benzothiazepine | 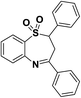 |
Antifungal | 9 and 10 |
| 3 | (±)-cis-2-(4-Methoxyphenyl)-3-hydroxy-7-chloro-8-methoxy-1,5-benzothiazepine-4-(5H)-one | 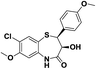 |
Antimicrobial | 11 |
| 4 | 6,9-Dichloro-2,3-dihydro-1,5-benzothiazepine-4-(5H)-ones | 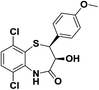 |
Antimicrobial | 11 |
| 5 | (2S,3S)-5-(2-(Dimethylamino)ethyl)-2-(4-methoxyphenyl)-4-oxo-2,3,4,5-tetrahydrobenzo[b][1,4]thiazepin-3-yl benzoate | 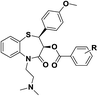 |
Calmodulin receptor | 12 |
| 6 | MB2 | 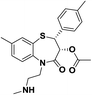 |
Antiplatelet aggression inhibitor | 13 |
| 7 | MB3 | 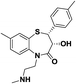 |
Antiplatelet aggression inhibitor | 13 |
| 8 | 5-(4-Bromophenyl)-1,3-diphenyl-4,5-dihydro-3H-benzo[b][1,2,4]oxadiazolo[4,5-d][1,4]thiazepine | 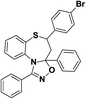 |
CNS depressant | 14 |
| 9 | 4-Chloro-7-[(diethylcarbomyl)oxy]-6-phenylpyrrolo[2,1-d]-[1,5]-benzothiazepine | 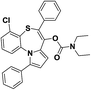 |
CNS depressant | 15 |
| 10 | CRD-401 (+)-cis-3-(Acetyloxy)-5-[[2,2-dimethylamino]ethyl]-2,3-dihydro-2-(4-methoxyphenyl)-1,5-benzothiazepine-4(5H)-one | 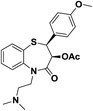 |
Ca2+ antagonist | 16 |
| 11 | TA-3090 (+)-(2S,3S)-3-Acetoxy-8-chloro-5-(2-(dimethylamino)ethyl)-2,3-dihydro-2-(4-methoxyphenyl)-1,5-benzothiazepine-4-(5H)-one | 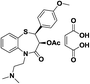 |
Ca2+ antagonist | 16 |
| 12 | CV-5975 (R)-3-[(S)-1-Carboxy-5-(4-piperidyl)pentyl]amino-4-oxo-2,3,4,5-tetrahydro-1,5-benzothiazepine-5-acetic acid | 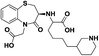 |
ACE inhibitor | 17 |
| 13 | JMV1116 |  |
Bradykinin receptor | 18 |
4. 1,2-Benzothiazepines
4.1 Introduction
1,2-Thiazepines are a class of privileged skeletons labelled as drug-like compounds. Among all, the core structure of benzo[f][1,2]thiazepine dioxide exhibits remarkable biological activities and is present in major antidepressants. For example, tianeptine (trade names: Stablon, Coaxil, Tatinol) has been a popular antidepressant drug. Benzo[f][1,2]thiazepine dioxide derivatives are marked as human immunodeficiency virus protease inhibitors, calcium sensing receptor agonists and farnesyltransferase inhibitors.19 For instance, 2,3,4,5-tetrahydro-4-methyl-1,2-benzothiazepine 1,1-dioxide showed HIV-1 protease inhibitory activity. Dibenzo[c,f][1,2]thiazepine was revealed as the first LC-MS based metabolite for antidepressants. Several synthetic protocols have been reported in the literature to access this biodynamic core, most of which have been taken into account in this document.4.2 Synthesis of 1,2-benzothiazepines
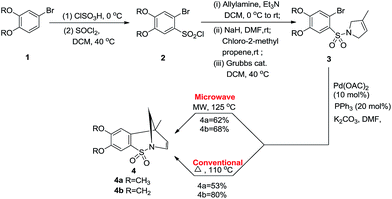
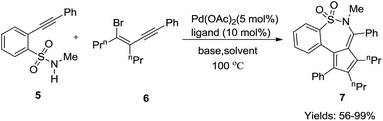
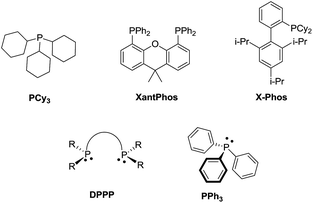
For example, Geoghegan et al. generated a series of regioisomeric cyclic sulphonamides by reacting 4-bromo-1,2-dialkoxybenzene 1 with chlorosulphonic acid resulting in the corresponding sulfonic acid which transformed into 2-bromo-4,5-dialkoxybenzene-1-sulfonyl chloride 2 by reacting with thionyl chloride; amination and further incorporation of a Grubb’s catalyst promoted ring closing metathesis to generate intermediate 3, which is the key product to access tri- or bi-cyclic sulfonamide 4 with an intramolecular Mizoroki–Heck coupling reaction (Scheme 1). Both conventional and microwave conditions were explored and it was found that 4a was obtained in good yields under MW conditions whereas 4b provided better yields under conventional conditions. This method worked well for substrates having an electron donating group attached to the benzene ring.20H. Wang et al., performed a one pot tandem reaction to synthesize 6H-benzo[f]cyclopenta[d]1,2-thiazepine-5,5-dioxides 7 using Pd(0) via double carbopalladation of 2-(2-alkynyl)benzenesulfonamide 5 and 2-alkynylvinyl bromide 6 (Scheme 2). This reaction takes place specifically in the presence of a class of phosphine ligands shown in Fig. 4. This method yielded fifteen thiazepine dioxide derivatives in good to excellent yields (56–99%).21
Mechanistically as depicted in Scheme 3, first the oxidative addition of Pd(0) to 2-alkynylvinyl bromide generated the Pd(II) species A, which underwent coordination to the triple bond of 2-(2-alkynyl)benzenesulfonamide to afford intermediate B. The further intramolecular insertion of a triple bond produced intermediate C which then provided the expected benzo[f][1,2]thiazepine dioxide 7 via a C–N coupling.
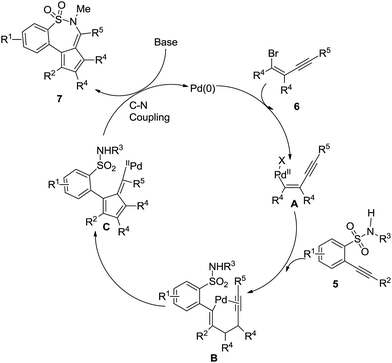 | ||
| Scheme 3 Mechanistic pathway of the formation of 6H-benzo[f]cyclopenta[d]1,2-thiazepine-5,5-dioxides. | ||
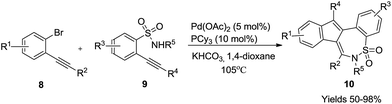
Along similar lines, Xiao and coworkers (Scheme 4) also provided the synthesis of benzo[f][1,2]thiazepine dioxides 10 using 2-alkynylbromobenzene 8 as the starting substrate. Here PCy3 was used as the phosphine ligand along with KHCO3 as the base in 1,4-dioxane. This methodology furnished twenty-three derivatives in good to excellent yields (50–98%).22 The reaction goes via an analogous mechanistic pathway similar to Scheme 3.
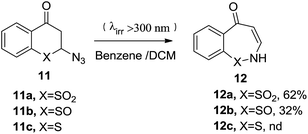
Earlier in 1980, Still et al. (Scheme 5) incorporated photolytic conditions (λ > 300 nm) in benzene or DCM to synthesize 2H-benzo[f]-1,2-thiazepin-5-one-1,1-dioxide 12a (62%) from 2-azido-4-thiochromanone-1,1-dioxide 11a in good yield. This methodology turned out to be tricky for isolating 2-azidothiochroman-4-one-1-oxide 12b from the starting sulphoxide analogue 11b and was a failure for sulphide analogue 11c.23
Abramovitch et al., (Scheme 6) performed a thermal decomposition of 3-(2,6-dichlorophenyl)propanesulfonylazide 13 to furnish 6,9-dichloro-1,3,4,5-tetrahydrobenzo[c][1,2]thiazepine 2,2-dioxide 15. Also, flash vacuum pyrolysis (FVP) of 3-(2-mesityl)propanesulfonylazide 16 followed by a 1,2-methyl shift afforded 6,8,9-trimethyl-1,3,4,5-tetrahydrobenzo[c][1,2]thiazepine-2,2-dioxide 17 in lower yields (25–34%). The mechanistic details point towards the formation of an intermediate 3-(2,6-dichlorophenyl)propanesulfonyl nitrene 14 which further undergoes intermolecular cyclisation followed by a 1,2-Cl shift in Freon 113 under an inert atmosphere to afford 6,9-dichloro-1,3,4,5-tetrahydrobenzo[c][1,2]thiazepine-2,2-dioxide 15.24
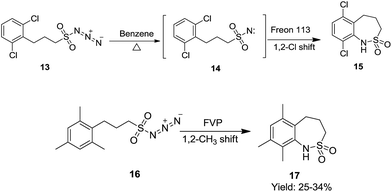 | ||
| Scheme 6 Synthesis of 6,9-dichloro-1,3,4,5-tetrahydrobenzo[c][1,2]thiazepine-2,2-dioxide and 6,8,9-trimethyl-1,3,4,5-tetrahydrobenzo[c][1,2]thiazepine-2,2-dioxide. | ||
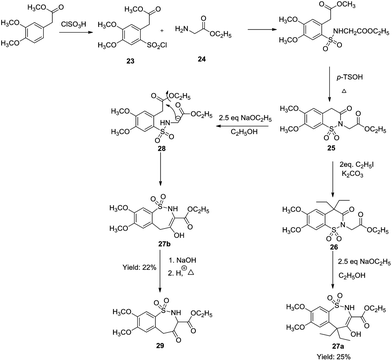
Similarly, Khalaj & Adibpour via ring expansion converted 1,2-benzothiazine 25 to hydroxy-1,2-benzothiazepines 27a and 27b (Schemes 7 & 8). These hydroxy-1,2-benzothiazepines were synthesized by the reaction of ethyl methyl 2-(2-chlorosulfonyl-4,5-dimethoxyphenyl) acetate 20 with N-phenyl acetamide 21 which itself was prepared from the reaction of ethyl 2-aminoacetate 18 with aniline 19.
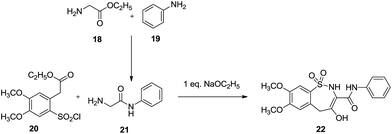 | ||
| Scheme 7 Synthesis of 7,8-dimethoxy-4-hydroxy-2,5-dihydrobenzo[f][1,2]thiazepine-1,1-dioxide-3-carboxanilide. | ||
A series of benzothiazines starting with 25 were synthesized, which were treated with sodium ethoxide in ethanol for the subsequent substitution of the alkoxy group through nucleophilic substitution via an anion generated by the abstraction of the benzylic hydrogen of the acetate group. This eventually led to the formation of 7,8-dimethoxy-4-hydroxy-2,5-dihydrobenzo[f][1,2]thiazepine-1,1-dioxide-3-carboxanilide 22. This approach produced only two derivatives 27a and 27b in lower yields (25% and 22% respectively).25
5. 1,3-Benzothiazepines
5.1 Introduction
1,3-Thiazepines can be described as seven membered cyclic thiourea derivatives. These derivatives are an important class of compounds with triggered biological activities26–29 such as nitric oxide synthase inhibitors. The 1,3-thiazepine core is present in Omapatrilat which is currently in phase IV of clinical trials. Omapatrilat lowers blood pressure by inhibiting the activity of the angiotensin converting enzyme (ACE), which causes blood vessels to constrict. The added advantage of this drug is the inhibition of the neutral endopeptidase enzyme (NEP), which causes the relaxation of blood vessels.30,315.2 Synthesis of 1,3-benzothiazepines
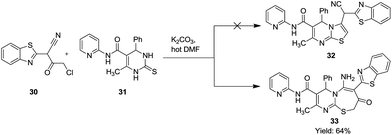
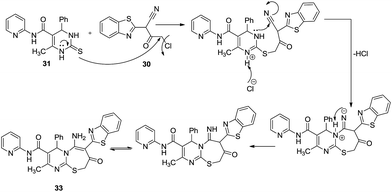
In an interesting example, Mohamed et al. attempted to synthesize pyridine and pyrimidine derivative 32 containing the benzothiazole moiety but serendipitously formed [2,1-b][1,3]thiazepine-3-carboxamide 33 (Scheme 9). They treated 1-(2-benzothiazolyl)-1-cyano-3-chloroacetone 30 with 6-methyl-4-phenyl-N-(pyridin-2-yl)-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-carboxamide 31 in the presence of anhydrous K2CO3/hot DMF to obtain 15 title compounds in good yield (64%).32Mechanistic details as depicted in Scheme 10 explain the formation of a 7-membered ring proceeded by the nucleophilic substitution of the α-halo group in 30 resulting in a C–S bond, where nitrogen further behaved as a nucleophile to attack the cyanide carbon resulting in the target molecule 33.
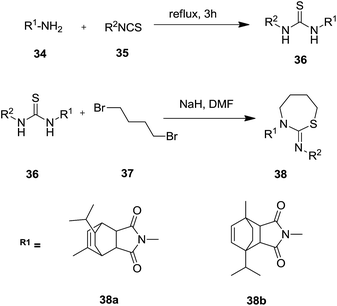
Afterwards, Struga et al. (Scheme 11) synthesized 14 thiourea derivatives of 36 which were used as starting substrates to react with 1,4-dibromobutane 37 to obtain the 1,3-thiazepine derivatives of 38, 10-isopropyl-8-methyl-4-aza-tricyclo[5.2.2.02,6]undec-8-ene-3,5-dione 38a in moderate to good yields (38% to 66%) and 1-isopropyl-7-methyl-4-aza-tricyclo[5.2.2.02,6]undec-8-ene-3,5-dione 38b in good yields (62% to 85%).33,34
The mechanism illustrated in Scheme 12 explains that the nucleophilic substitution of the dihalo compound is the cause of the formation of the target product.
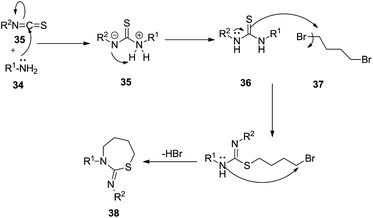 | ||
| Scheme 12 Mechanistic aspects of the formation of 10-isopropyl-8-methyl-4-aza tricyclo[5.2.2.02,6]undec-8-ene-3,5-dione-1,3 benzothiazepine derivatives. | ||
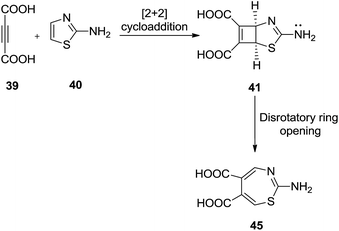
Alajarìn et al. carried out a thermal transformation of (1S, 5R)-3-amino-2-thia-4-azabicyclo[3.2.0]hepta-3,6-diene-6,7-dicarboxylic acid 41 into 2-amino-1,3-thiazepine-5,6-dicarboxylic acid 42. Prior to this transformation, compound 41 was prepared by [2 + 2] cycloaddition of but-2-ynedioic acid 39 and thiazol-2-amine 40 (ref. 35) (Scheme 13).
A six electron five centre thermally allowed electrocyclic disrotatory ring opening mechanism was proposed to produce cis-1,3-thiazepine 42 in good yield, starting with fused cyclobutene with five membered ring 41 which contribute electronically and geometrically in the polar disrotatory ring opening. Polar groups present in the ring reduce the energy barrier for the disrotation. The reaction did not perform well in the absence of trifluoroacetic acid and formed pyridine in the place of 1,3-thiazepine (Scheme 14).
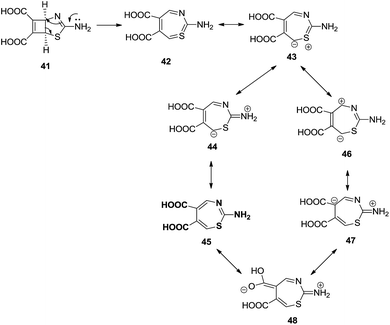 | ||
| Scheme 14 Ring opening mechanism of the bicyclic compound forming 2-amino-1,3-thiazepine-5,6-dicarboxylic acid. | ||
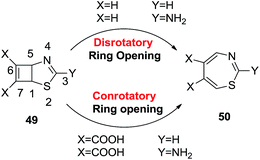
In continuation, Silva López et al. extended the above work in order to explain the mechanism more clearly (Scheme 15). Their aim was to observe a more favourable product between the Woodward–Hoffman (W–H) and anti-W–H pathways. They found that the mechanistic pathway strives between the disrotatory transition state and double-bond isomerization depending on the substituents around the bicyclic structure. They suggested that the bicyclo[3.2.0]hepta-3,6-diene 49 moiety adorned with heteroatoms at the 2 and 4 positions, an electron withdrawing group at the cyclobutene core and an electron donating amine forming an isothiourea group on the five membered ring enhanced the energy profile of the W–H conrotatory pathway ensuring the formation of target moiety 50.36
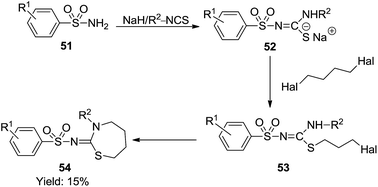
Rudorf and Cleve described a thiocarbamoylation protocol of sulfonamides 51 with isothiocyanates allowing access to 3-phenyl-2-(p-toluenesulfonylimino)-perhydro-1,3-thiazepine 54. As depicted in Scheme 16, sulfonamides treated with isothiocyanates in the presence of NaH produced the corresponding sodium salts 52 which was further alkylated and cyclized by nucleophilic substitution with dihalobutane in very low yield (15%).37
Mente and Heine employed a reaction of 2-vinylaziridine 56 with phenyl isothiocyanate 55 in ether at 0 °C to furnish 2-anilino-4,7-dihydro-1,3-thiazepine 57 in moderate yield (59%). Subsequently, applying the same method with 2-vinyl aziridine 56 and sodium p-chlorothiobenzoylthioglycolate 58 in a stirred ether–water mixture gave low-melting 2-(p-chlorophenyl)-4,7-dihydro-1,3-thiazepine 59 which decomposed after some time38 (Scheme 17).
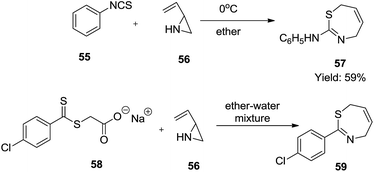 | ||
| Scheme 17 Synthesis of 2-anilino-4,7-dihydro-1,3-thiazepine and 2-(p-chlorophenyl)-4,7-dihydro-1,3-thiazepine. | ||
The mechanistic pathway in Scheme 18 postulated the nucleophilic attack of the amine group on the carbon atom which shifted the electron density of the double bond towards the nitrogen followed by cyclization to the 7-membered ring.
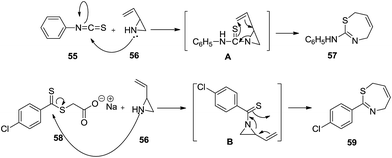 | ||
| Scheme 18 Mechanistic routes to 2-anilino-4,7-dihydro-1,3-thiazepine and 2-anilino-4,7-dihydro-1,3-thiazepine. | ||
6. 1,4-Benzothiazepines
6.1 Introduction
There are several reports available for the synthesis of 1,4-benzothiazepine. We have split this section into two parts based on the cyclic nature of the products:(a) Bicyclic thiazepines.
(b) Tricyclic thiazepines.
6.2 Synthesis of bicyclic 1,4-benzothiazepines
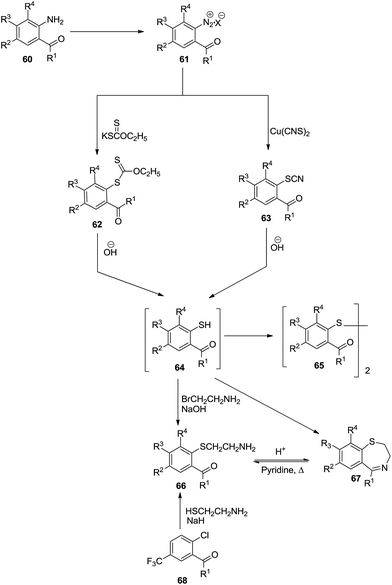
Sternbach et al. constructed 2,3-dihydro-1,4-benzothiazepines 67 through various transformations (Scheme 19). The multi-step synthesis started with the diazotization of o-amino substituted aromatic compounds 60 followed by a Sandmeyer-type reaction using potassium xanthate or copper thiocyanate. Furthermore, base hydrolysis and nucleophilic substitution with 2-bromoethanamine resulted in substituted o-mercapto ketones 66 which underwent ring closure in the presence of pyridine to give 2,3-dihydro-l,4-benzothiazepines 67 in moderate yield (41%).39Dölling et al. devised a methodology to synthesize only two thiazepine derivatives 72, namely 4-methyl-2-methylthio-5(4H)-oxopyrido[3,2-f][1,4]thiazepine-3-carbonitrile and 2-ethylthio-4-methyl-5(4H)-oxopyrido[3,2-f][1,4]thiazepine-3-carbonitrile in 42% and 12% yields respectively via a multistep reaction sequence40 (Scheme 20).
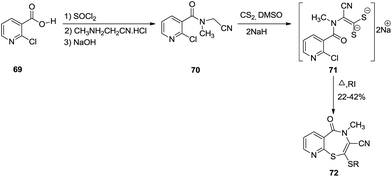
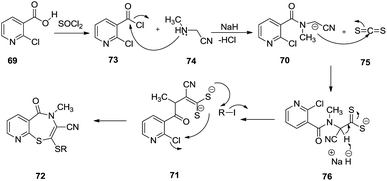
Mechanistically, 2-chloronicotinic acid 69 subsequently reacted with thionyl chloride and 2-(methylamino)acetonitrile resulting in 2-chloro-N-(cyanomethyl)-N-methylnicotinamide 70. Two equivalents of sodium hydride were used to abstract the more acidic proton to generate a thiolate anion on reaction with carbon disulphide with succeeding cyclisation via intermolecular aromatic nucleophilic substitution into the desired 1,4-benzothiazepine 72. This reaction failed for nitro substituted chloronicotinic acid as in this case intermediate 71 failed to undergo cyclisation into benzothiazepine, instead forming the benzothiazole derivative (Scheme 21).
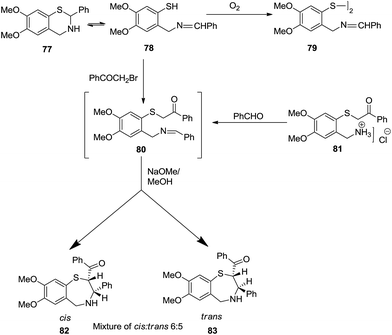
Similarly, Fodor et al. reported two strategies for the formation of a mixture of two diastereomers of 2-benzoyl-3-phenyl-7,8-dimethoxy-2,3,4,5-tetrahydro-l,4-benzothiazepine, 82 and 83. One of them included the conversion of a methanolic solution of dihydro-1,3-thiazine 77 into disulphide 79 on exposure to air. The open ring tautomeric intermediate 78 on reaction with 2-bromoacetophenone formed a mixture of 1,4-benzothiazepine diastereomers via cyclisation of the intermediate 80 to yield slightly more cis isomer 82 than trans 83 (75%). On the other hand, reaction of 2-benzoylmethylthio-4,5-dimethoxybenzylamine hydrochloride 81 with benzaldehyde in the presence of NaOMe in MeOH, yielded the diastereomers in a cis![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) trans ratio of 6
trans ratio of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (ref. 41) (Scheme 22).
5 (ref. 41) (Scheme 22).
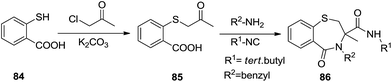
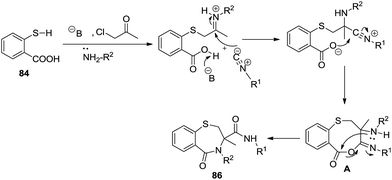
Nowadays, the significance of multicomponent reactions is flourishing in current research scenarios. Complete utilization of three or more components present in the reaction mixture, atom economy, efficiency, environmental friendliness and selectivity are some of the advantages of MCRs. Mironov et al. indulged in the benefits of MCRs in a one-pot four component Ugi reaction in the liquid phase synthesis of twelve 1,4-benzothiazepin-5-ones 86 in good yields (36–90%) by condensing thiosalicylic acid 84, 1-chloropropan-2-one, tert-butyl isocyanide and benzylamine. In this reaction, bromoacetophenone as an acetyl halide input did not work well despite chloroacetophenone being a success. Also the use of methanol as a polar solvent is the prime requirement of the reaction beside electron donating group substituted isocyanides providing incremental yields42 (Scheme 23).
A detailed mechanism of the Ugi reaction to afford 1,4-thiazepinone derivatives is illustrated in Scheme 24. It first explains the base promoted proton abstraction followed by nucleophilic substitution with 1-chloropropan-2-one. Further condensation of the amine with the keto functionality generates the iminium ion on which the attack of isocyanide forms cyclic intermediate A which undergoes acyl transfer to afford the final product.
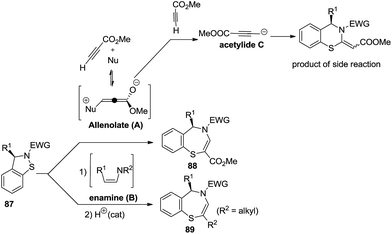
C. Spitz et al. anticipated two pathways for the synthesis of 1,4-benzothiazepines. One pathway explains the formation of the target compounds being facilitated by simultaneous bond breaking and bond formation via a ring expansion mechanism, in which cyclic sulfenamides 87 reacted with allenolate A derived from the Michael addition of neutral organic nucleophiles to methyl propionate. The second pathway involved trapping cyclic sulfenamides 87 with enamines B followed by an acid catalyzed condensation reaction to give 1,4-benzothiazepines 89 (Scheme 25). Electron withdrawing groups attached to the nitrogen atom of 87 favoured the product formation. Trapping intermediates A and B proved extremely troublesome, failure of which resulted in a side reaction generating acetylide C from the allenolate intermediate A which on reaction with cyclic sulfenamides 87 led to 1,3-benzothiazines. This methodology provided 100% conversion with high efficiency and atom economy.43
A plausible mechanism for the above reaction is described in Scheme 26. This explains that the initially formed zwitterionic allenolate A attacks the electrophilic sulfur atom of sulfenamide 87 to form the corresponding zwitterion D which in turn cyclizes after an addition–elimination reaction into the final 1,4-benzothiazepine product.
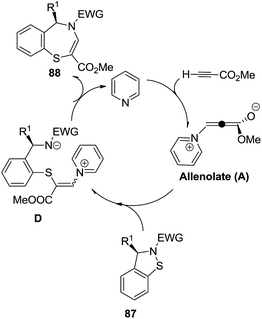 | ||
| Scheme 26 Mechanistic details for the synthesis of 1,4-benzothiazepine from cyclic sulfenamides and an allene. | ||
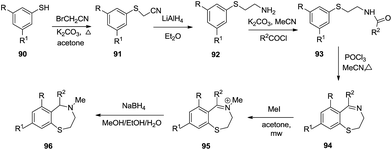
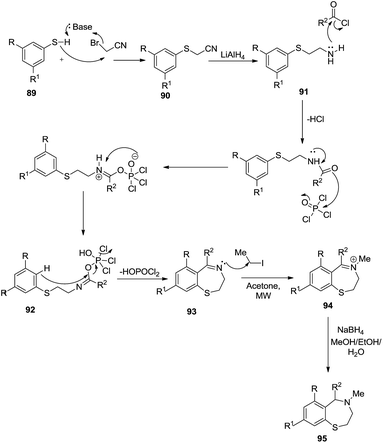
Voskressensky et al. utilized tandem nucleophilic substitution and a Bischler–Napieralski reaction to synthesise 2,3-dihydro-1,4-benzothiazepines 96. In this multistep reaction, alkylation of substituted thiophenols 90 with bromoacetonitrile generated cyanomethylthioethers 91 followed by reduction, acylation and finally cyclization to afford 8-methoxy-5-phenyl-2,3-dihydro-1,4-benzothiazepines 94 in low yield (11%) and further reduction led to the formation of 8-methoxy-4-methyl-2,3,4,5-tetrahydro-1,4-benzothiazepines 96 in good yield (92%).44 This reaction was facilitated by electron donating groups adorned to the backbone structure of starting material (Scheme 27).
The detailed mechanism for the above reaction as depicted in Scheme 28 first describes the alkylation of the starting substituted thiophenol with bromoacetonitrile to cyanomethylthioethers. Reduction of the nitrile group using lithium aluminum hydride gave amines which were converted to the N-acyl derivatives using acyl chlorides. Cyclization of 92 under Bischler–Napieralski conditions gave dihydrobenzothiazepines. The dihydro derivatives were quaternized with methyl iodide under microwave irradiation to give the quaternary salts, reduction of which by sodium borohydride in methanol gave 2,3,4,5-tetrahydro-1,4-benzothiazepines.
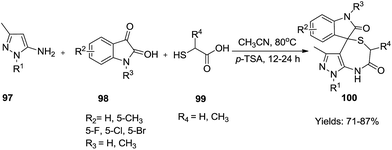
Shi et al. reported a one-pot 3-CR of 5-amino-3-methylpyrazoles 97, isatins 98 and thioacids 99 through a 3-(5-aminopyrazol-3-yl)-3-hydroxy-2-oxindoline intermediate (Baylis–Hillman type adduct) for the synthesis of spiro[indoline-3,4-pyrazolo[3,4-e][1,4]thiazepine-dione derivatives 100 in high yields (71–87%) (Scheme 29).45
6.3 Synthesis of tricyclic 1,4-benzothiazepines
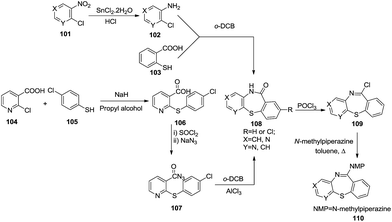
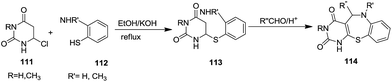
In 1994, Liegios et al. synthesized nine N-methylpiperazinopyrido-[1,4]benzothiazepine derivatives 110 in good yields via a pyrido-[1,4]benzothiazepinone intermediate 108 which undergoes chlorination in the presence of POCl3 to form compound 109 which finally affords the target product 110. The intermediary lactam 108 has been synthesized by two different methods. Reaction of 4-chloro-3-aminochloropyridine 102 with thiosalicylic acid 103 gave lactam 108. On the other hand, 2-chloronicotinic acid 104 was treated with 4-chlorothiophenol 105 to provide (4-chlorophenyl)thiolnicotinic acid 106 which got transformed into acyl azide 107. Thermal decomposition of this azide yielded the corresponding isocyanate which in the presence of aluminum chloride yielded lactam 108 (ref. 46) (Scheme 30).In 1995, Johnson and Maruendat performed the reaction of 6-chlorouracils 111 with substituted 2-aminothiophenols 112 under refluxing conditions with KOH in ethanol to afford 6-(arylthio)uracils 113 which were further treated with appropriate aldehydes under acidic conditions to yield the desired pyrimido[5,4-f]benzo[1,4]thiazepines 114 via an intramolecular Mannich-type cyclization. This synthetic route generated ten thiazepine derivatives in good to excellent yields (56–95%) and tolerated a wide variety of substituted substrates such as methyl, phenyl and p-nitro phenyl benzaldehydes47 (Scheme 31).
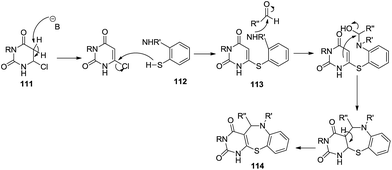
The mechanism of the above reaction is depicted in Scheme 32. It describes the initial base promoted proton abstraction followed by nucleophilic substitution with substituted 2-aminothiophenol to obtain the intermediate 113 which causes a nucleophilic attack on benzaldehyde followed by cyclisation to fabricate the final pyrimido benzothiazepine derivative.
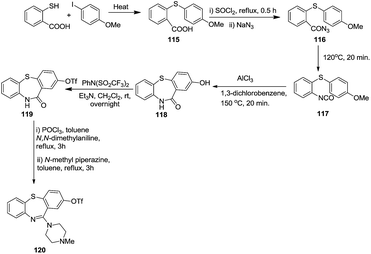
In 1999, Laio et al.synthesized 2-TfO-11-(4-methylpiperazinyl)-dibenzo[b,f][1,4]thiazepine 120 from 4-iodoanisole and 2-carboxythiophenol which was transformed into acid intermediate 115 and then azide 116. This was then converted into the final product 120 via 2-hydroxylactum intermediate 118 and isocyanate intermediate 117. Triflate derivatives induce less oxidative metabolism in comparison to hydroxy or methoxy groups due to their electron-withdrawing effect and lipophilicity. So to enhance the pharmacological profile of this core, this group specially focussed on the synthesis of triflate derivatives in moderate yields (45%)48 (Scheme 33).
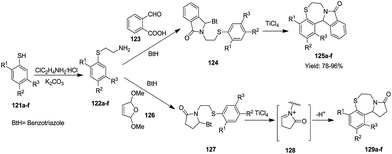
In 2001, Katrizky et al. synthesized six 1,4-benzothiazepines via a multistep reaction sequence in good yields (78–96%). Treatment of substituted thiophenols 121a–f with 2-chloroethylaminehydrochloride and excess potassium carbonate in DCM provided 2-(arylsulfonyl)ethylamines 122a–f. This series of amines 122a–f, when treated with 2-carboxybenzaldehyde 123 and 2,5-dimethoxy-2,5-dihydrofuran 126 in the presence of the nucleophile benzotriazole, gave intermediate compounds 124 and 127 respectively. These intermediates in the presence of a Lewis acid underwent cyclisation with the elimination of benzotriazole to afford two series of 1,4-benzothiazepine derivatives 125a–f and 129a–f (ref. 49) (Scheme 34).
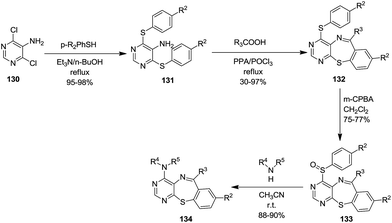
Fu and co-workers described the synthesis of tricyclic pyrimido[4,5-b][1,4]benzothiazepines from 5-amino-4,6-bis-(arylthio)pyrimidines and carboxylic acids via Bischler–Napieralski-type reactions (Scheme 35). Substitution of pyrimidines 130 with a thiophenol yielded 5-amino-4,6-bisphenylthiopyrimidines 131 which under refluxing in PPA/POCl3 (Bishler–Napieralski conditions) gave the final benzothiazepine skeletons 132. The aryl sulfide group of the resulting 4-arylthiopyrimido[4,5-b][1,4]benzothiazepines was subjected to selective oxidation and subsequent nucleophilic substitution to produce derivatives of 134 in good yields (88–90%).50 Here, the bisphenylthio products 131 could be obtained in substantial amounts due to the high nucleophilicity of the thio group. The reaction yields were more sensitive to carboxylic acid (R3) compared to substitution on the thiophenol ring. Aromatic carboxylic acids provided higher yields compared to aliphatic ones. An improvement in yields was observed in the presence of electron-donating groups on the aromatic acid ring whereas electron-withdrawing groups proved detrimental to the yields.
The mechanistic attributes of this reaction shown in Scheme 36 portray the acylation of bis-(phenylthio) compounds 131 to give products 131a which tautomerise to form 131b. These were converted into imidoyl chlorides 131c when treated with POCl3, which in turn transformed into corresponding nitrilium salts 131d. These nitrilium salt underwent an intramolecular electrophilic substitution on the phenyl ring and a subsequent elimination of hydrogen chloride to yield the final thiazepine skeletons 132.
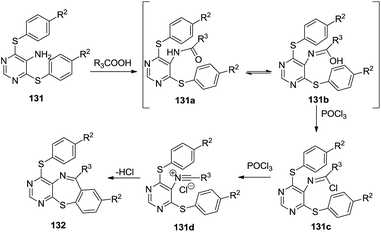 | ||
| Scheme 36 Mechanistic details of the Bischler–Napieralski cyclisation forming 4-arylthiopyrimido[4,5-b][1,4]benzothiazepines. | ||
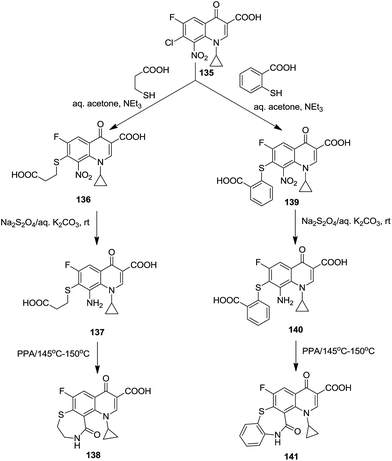
In 2007 Zahra et al. synthesized substituted [1,4]thiazepino[2,3-h]quinolone carboxylic acids (138 and 141) with a PPA-catalyzed thermal lactamization of the respective 8-amino-7-[(2-carboxyethyl)thio]-1,4-dihydroquinoline-3-carboxylic acids (137 and 140).51
Mechanistically, here 3-mercaptopropionic acid acts as a ‘sulfur’ nucleophile which undergoes nucleophilic aromatic substitution, SNAr, facilitated by the presence of the electron withdrawing fluoro, keto and nitro groups. Further, compound 137 or 140 underwent lactamization upon heating with polyphosphoric acid (PPA) to afford a tricyclic system (Scheme 37).
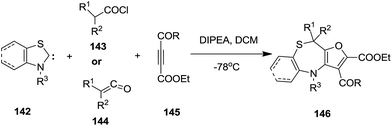
In 2007 Chen Ma et al. developed a highly efficient synthesis strategy to furo[2,3-c][1,4]thiazepine cores 146 via a three component reaction between thiazole or benzothiazole carbenes 142, disubstituted ketenes 144 and activated alkynes 145 (ref. 52) (Scheme 38).
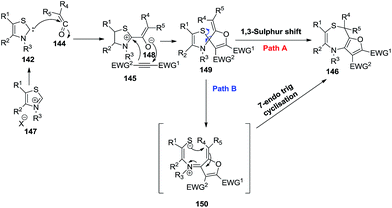
A plausible mechanistic pathway is depicted in Scheme 39. At first, thiazole carbenes 142, generated in situ from thiazolium salts 147, attacked the disubstituted ketenes 144 to give zwitterions 148, which underwent oxa-Michael addition with activated alkynes 145 subsequently getting converted into spirocycle intermediates 149 through intramolecular annulation. Ring-transformation of spirocycles 149 might take place via concerted pathway A or stepwise pathway B. Path A describes the ring expansion of 148 proceeding to form fluorothiazepines 146 via a [1,3]-sigmatropic sulfur shift. On the other hand, path B illustrates the ring opening of 149 to reactive thiolates 150, which subsequently underwent a 7-endo-trig cyclization to produce thiazepine rings 146.
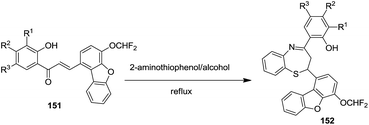
In 2009, Randhavane et al. synthesized chalcones of 4-difluoromethoxy-dibenzofuran-1-carboxaldehydes 151 which were converted into corresponding dihydro-benzo[1,4]thiazepines 152 by refluxing with 2-aminothiophenol53 (Scheme 40).
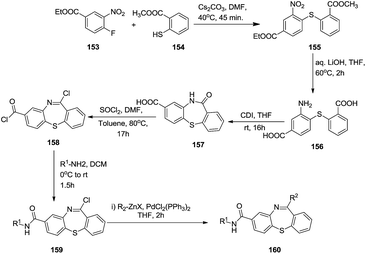
Later on, Olsson et al. synthesized lactam 157 from commercially available starting materials over three sequential steps. Subsequent diversification of the lactam yielded amides 159 and a Negishi cross-coupling reaction afforded the final benzo[1,4]thiazepine-8-carboximides 160.54 Detailed description of the mechanism revealed an initial nucleophilic substitution of methyl 2-mercaptobenzoate with ethyl 4-fluoro-3-nitrobenzoate to generate bisphenylmonothio product 155. Further reduction of the nitro group followed by cyclisation in the presence of CDI in THF gave the lactam 157. Further chlorination in the presence of SOCl2, yielded imidoyl chloride 158 and subsequent nucleophilic substitution of an amine provided compounds 159. Palladium-catalysed Negishi cross-coupling of imidoyl chlorides 159 with Grignard’s reagent produced the final compounds 160 (Scheme 41).
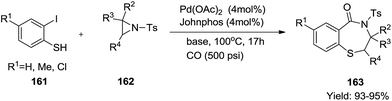
Similarly, Zeng et al. devised an efficient domino procedure for the synthesis of 1,4-benzothiazepin-5-ones 163 from simple and readily accessible N-tosyl aziridines 162 and o-iodothiophenols 161 (ref. 55) (Scheme 42).
The reaction of the N-tosyl aziridine derivative with substituted 2-iodothiophenol furnished the corresponding 1,4-thiazepinones in excellent yields (93–95%). The versatility of the used starting materials demonstrated that this transformation could tolerate both electron donating (p-Me) and electron-withdrawing (p-Cl) groups on the phenyl ring of the thiophenol.
Newton et al. in 2011 carried out a SNAr nucleophilic substitution of thiol 164 with arylflouride 165, which upon further reduction via a three step process was converted to methyl 10-(3-chlorobenzyl)-11-oxo-10,11-dihydrodibenzo[b,f][1,4] thiazepine-8-carboxylates 168, and following N-alkylation gave the final products 169 (ref. 56) (Scheme 43).
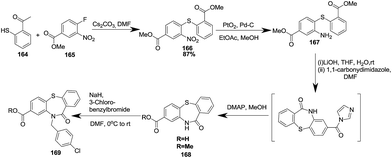 | ||
| Scheme 43 Synthesis of methyl 10-(3-chlorobenzyl)-11-oxo-10,11-dihydrodibenzo[b,f][1,4]thiazepine-8-carboxylate. | ||
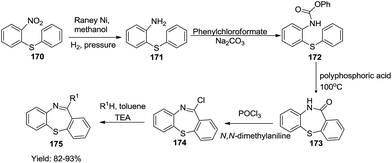
In continuation, Mahale et al. designed a reaction sequence involving the hydrogenation of 2-nitrodiphenylsulphide 170 which formed carbamate 172 on reaction with phenylchloroformate in the presence of a base. Carbamate 172 underwent cyclisation into 173 which upon treatment with POCl3 formed imino chloride 174. Here the reaction proceeded via the formation of an iminium ion generated in situ by the removal of a chloride ion. This chloride ion is scavenged by acid scavengers to drive the reaction forward and obtain the final product dibenzo[b,f][1,4]thiazepines 175.57 This reaction accounted for sufficient versatility to generate 1,4-dibenzothiazepines in good yields (82–93%) (Scheme 44).
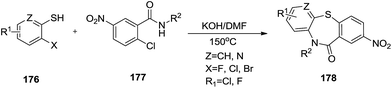
Remarkably, Zhao et al. established an efficient metal free method for the synthesis of a library of dibenzo[b,f][1,4]thiazepin-11(10H)-ones 178 via Smiles rearrangement in excellent isolated yields (Schemes 45 and 46).58 Here, substituted 2-halobenzenethiols 176 and N-alkyl-2-chloro-5-nitrobenzamides 177 were reacted using KOH in DMF to effortlessly obtain the title compounds.
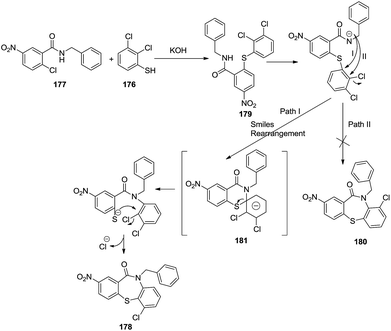
The probable mechanism for the above reaction could be described as in Scheme 46. The reaction of 2,3-dichlorobenzenethiol with N-benzyl-2-chloro-5-nitro benzamide yielded compound 180 which may proceed towards completion via two paths. Path II afforded the direct intramolecular nucleophilic substitution product 179. By contrast, path I could lead to intermediate 181 via Smiles rearrangement. The imido nitrogen underwent intramolecular nucleophilic attack on the carbonium ion followed by further migration of the spiro-sulfur, proceeding through the “Meisenheimer Complex” 181, with an intramolecular nucleophilic displacement of chlorine by the sulfur anion yielding the desired cyclic product 178.
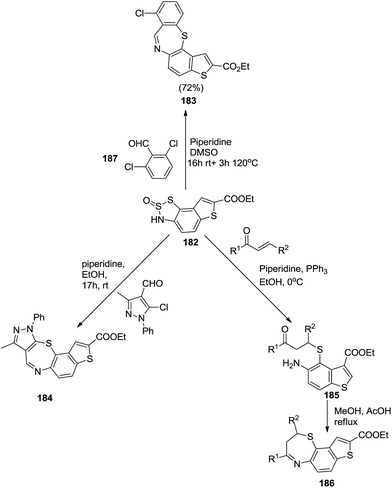
Interestingly, W. Van Snick et al. used 3H-thienobenzodithiazole-2-oxide 182 as a 2-aminothiphenol precursor in the synthesis of benzothiazepines (Scheme 47). Similarly, the reaction of 3H-thienobenzodithiazole-2-oxide 182 with 2,6-dichlorobenzaldehyde 187 resulted in the formation of thienobenzothiazepine derivative 183 (ref. 59) by intramolecular nucleophilic aromatic substitution reaction of the sulfur atom to the 2-position of the intermediate imine. Also the reaction of chalcones with 3H-thienobenzodithiazole-2-oxide 182 produced dihydrothienobenzothiazepines 186. Similarly, the reaction of chloroformylpyrazoles with precursor 182 generated new benzothiazepines 184.
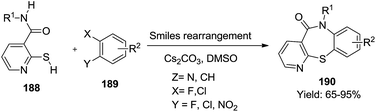
Recently, Yang et al. synthesized a series of twenty-five 1,4-thiazepin-5(4H)-one 190 derivatives via a transition metal-free one-pot Smiles rearrangement process at room temperature.60 Here, thiazepine scaffolds were obtained through the reaction of N-substituted 2-mercaptonicotinamides 188 and substituted benzenes 189 at room temperature in good to excellent yields (65–95%) (Scheme 48).
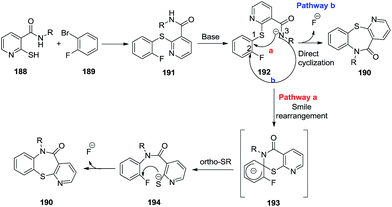
On the basis of the previous literature on the Smiles rearrangement, a plausible reaction mechanism is depicted in Scheme 49. Reaction of substrates 188 and 189 produced the intermediates 191 through a nucleophilic substitution reaction. The formation of carboxamide anions 192 then resulted in the target compounds by two ways. Pathway a formed intermediates 193 via Smiles rearrangement followed by an intramolecular nucleophilic substitution with a loss of a fluorine atom, leading to the corresponding products 190. On the other hand, pathway b led to direct intramolecular cyclization to form the products 190.
7. 1,5-Benzothiazepines
7.1 Introduction
Exclusively, 1,5-benzothiazepines have been the object of immense importance and investigation in the field of medicinal chemistry. Currently, 1,5-benzothiazepines such as diltiazem, thiazesim and clentiazem are amongst the most widely used drugs in the treatment of cardiovascular disorders.7.2 Synthesis of 1,5-benzothiazepines
A very typical method to construct 1,5-benzothiazepine skeletons is via the reaction of 2-aminothiophenol with α,β-unsaturated carbonyl compounds. The generality and scope of this approach has been represented in Fig. 5. Some of the schemes have been explained in detail as follows: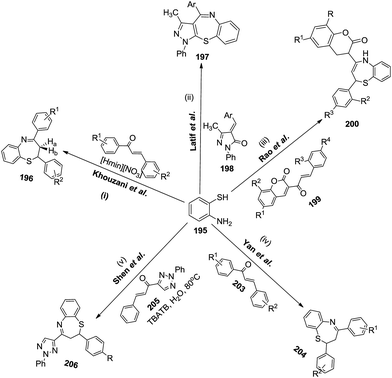 | ||
| Fig. 5 Reaction of 2-aminothiophenol with α,β-unsaturated carbonyl compounds to afford 1,5-benzothiazepine derivatives. | ||
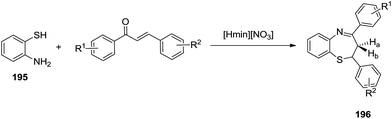
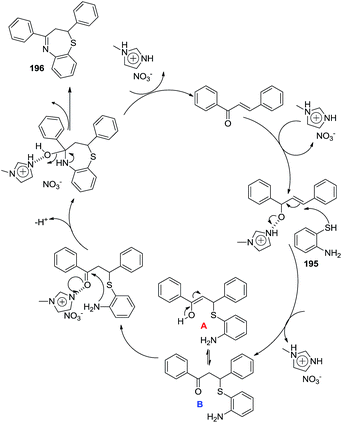
(i) Initially, Khouzani et al. carried out an eco-friendly synthesis of thirty-six 1,5-benzothiazepine derivatives 196 in good yields via a one-pot cyclocondensation reaction of o-aminothiophenol 195 with α,β-unsaturated carbonyl compounds through a [4 + 3] annulation reaction in the presence of N-methylimidazolium nitrate, [Hmim][NO3], as a Brønsted acidic ionic liquid. Comparative studies proved that this catalytic system is superior to the ones previously reported in terms of the amount of catalyst, cost and availability of the precursors used for the preparation of the catalyst61 (Scheme 50).
Mechanistically, the reaction proceeded initially by intermolecular hydrogen bonding promoted by the [Hmim][NO3] ionic liquid that activated the chalcone towards nucleophilic attack by the sulfur atom of 2-aminothiophenol to afford intermediate A. A 1,3-H shift on intermediate A occurs to give isomeric keto form B that cyclized to give the seven-membered ring product (Scheme 51).
(ii) In 2004, Latif et al. reported the synthesis of 4-aryl-3-methyl-1-phenylpyrazolo[3,4-b][1,5]benzothiazepines 197 from 4-arylidene-3-methyl-1-phenylpyrazole-5-one 198 and 2-aminobenzenethiol 195. The products obtained were in moderate yields (50–55%)62 (Scheme 52).
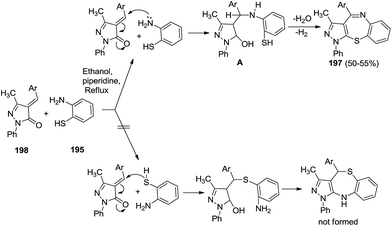 | ||
| Scheme 52 Mechanistic pathway of the formation of 4-aryl-3-methyl-1-phenylpyrazolo[3,4-b][1,5]benzothiazepines. | ||
The mechanistic pathway proceeded via Michael addition of anilinic nitrogen to the α,β-unsaturated ketone resulting in the formation of intermediate A which subsequently loses a water molecule to form the target compounds 197. Alternatively, if the thiol functionality underwent Michael addition first, it subsequently resulted in a different product whose formation has not been confirmed through characterization techniques.
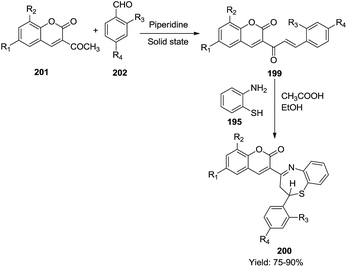
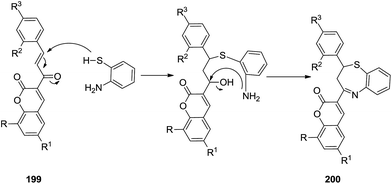
(iii) Along similar lines, Rao and his co-workers successfully generated chalcones 199 by the condensation of 3-acetyl coumarins 201 with various aromatic aldehydes 202 (Schemes 53 and 54). These chalcones 199 were utilised in situ to react with 2-aminothiphenol to generate 2-aryl-4-[2H-2-oxo[1]benzopyran-3-yl]-2,3-dihydro and 2,5-dihydro-1,5-benzothiazepines 200 in a one pot manner in excellent yields (75–90%).63
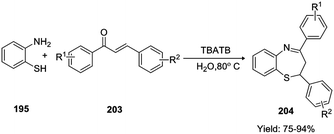
(iv) Yan et al. performed the reaction of 2-aminothiophenol 195 with chalcone 203, using water as solvent and TBATB as the phase transfer catalyst (Scheme 55) to afford the final 1,5-benzothiazepines 204 in good to excellent yields (75–94%).64
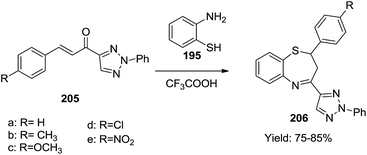
(v) In 2010, Shen and co-workers synthesized ten azeto[2,1-d][1,5]benzothiazepinone derivatives (Scheme 56) using 4-acetyl-2-phenyl-1,2,3-triazole as the starting substrate. Condensation with various aromatic aldehydes produced 2-phenyl-1,2,3-triazole-4-yl-α,β-unsaturated ketones 205 which subsequently underwent cyclisation with 2-aminothiphenol 195 to afford the corresponding 2,4-disubstituted-1,5-benzothiazepines 206 in good yields (75–85%).65
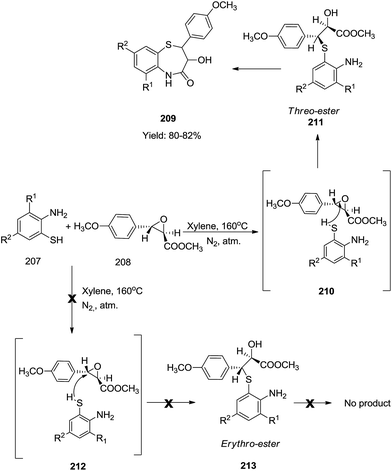
Similarly, Singh and co-workers described the synthesis of (±)-cis-2(4-methoxyphenyl)-3-hydroxy-1,5-benzothiazepinones 209 by the condensation of chloro substituted 2-amino-benzenethiol 207 with methyl-(±)-trans-3(4-methoxyphenyl)glycidate 208 in xylene under an inert atmosphere in good yields (80–82%)66 (Scheme 57).
A mechanistic interpretation as shown in Scheme 57 was derived by performing control experiments. This method exclusively preferred the cis product over the trans product. The formation of the cis product took place via the threo-ester which upon cyclization yielded the desired compounds 209. Whereas the trans-isomer did not form via the erythro-ester under similar conditions.
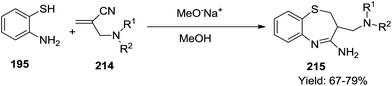
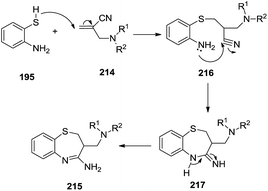
In 2002, Yaccoubi et al. performed the reaction of 2-aminothiophenol 195 with 2-dialkylaminomethylpropenenitriles 214 to afford corresponding benzothiazepines 215 (Scheme 58) in good yields (67–79%). The mechanistic pathway for this reaction has been determined by isolating some intermediates as depicted in Scheme 59. Firstly, the Michael addition of 2-aminothiol to dialkylpropenenitrile 214 formed the intermediate 216 which underwent intramolecular cyclisation to form the final product 215.67
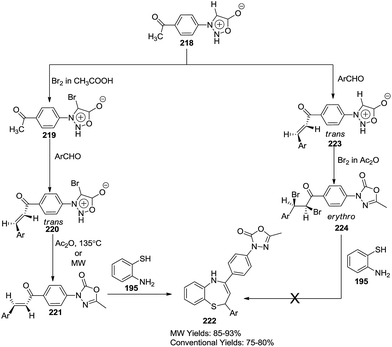
Kamble et al., devised a synthesis of 1,5-benzothiazepines integrated with 5-methyl-2-oxo-3-phenyl-4-1,3,4-oxadiazoles using 4-acetylphenylsydnone 218 and 2-aminothiophenol 195 as the starting substrates. Microwave and conventional synthesis conditions for this method have also been explored, and infer that microwave conditions (85–93% yield) fostered better results in comparison to conventional conditions (75–80% yield)68 (Scheme 60).
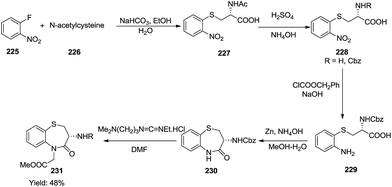
Slade and co-workers devised a synthetic strategy to synthesize chiral 1,5-benzothiazepinones 231 using o-fluoronitrobenzene 225 and N-acetylcysteine 226 as the starting precursors (Scheme 61).69 Initial nucleophilic substitution of o-fluoronitrobenzene with N-acetylcysteine affords S-(o-nitropheny1)-N-acetylcysteine which is further deacetylated and converted to the Cbz derivative. It then undergoes ring closure to a lactum using 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimidehydrohloride in DMF to afford the title compound 231 in moderate yield (48%).
Conclusively, Scheme 62 summarizes the reaction of 2-aminothiophenol with various unsaturated ketones to afford diverse substituted benzothiazepines. The reactions are described as follows:
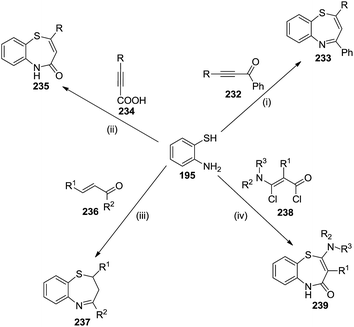 | ||
| Scheme 62 Synthesis of 1,5-benzothiazepine derivatives via Michael addition of 2-aminothiphenol with α,β-unsaturated ketones. | ||
(i) 2-Aminothiphenol reacts with alkynones 232 in a mixture of hot methanol and acetic acid to obtain 2,4-disubstituted 1,5-benzothiazepines 233.70,71
(ii) Also, 1,5-benzothiazepin-4(5H)-ones were synthesized by heating 2-aminothiophenol with either propiolic acid or its β-substituted derivatives 234 to yield the appropriate 1,5-benzothiazepines 235.72–74
(iii) Reaction of 2-aminothiophenol and α,β-unsaturated ketones 236 gives 2,3-disubstituted 2,3-dihydro-1,5-benzothiazepines 237.75
(iv) Reaction of 2-aminothiophenol with α-chloro-β-chlorocarbonyl enamines 238 in the presence of pyridine produced 2,3-disubstituted 1,5-benzothiazepin-4(5H)-ones 239.76
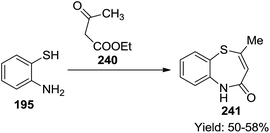
Also, 2-methyl-1,5-benzothiazepin-4(5H)-one 241 was synthesized in moderate yields (50–58%) by the reaction of 2-aminothiophenol with ethylacetoacetate 240 in xylene at refluxing temperature (Scheme 63).75,77
A series of 4-aryl-2,3-dihydro-1,5-benzothiazepines 244 has been synthesized by the reaction of 2-aminothiophenol with either N,N-disubstituted (2-aminoethyl)aryl ketones 243 (ref. 78) or β-haloketones 242 (ref. 79) (Scheme 64).
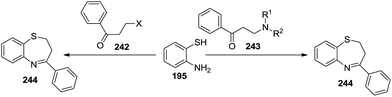 | ||
| Scheme 64 Synthesis of 4-aryl-2,3-dihydro-1,5-benzothiazepines from N,N-disubstituted (2-aminoethyl)aryl ketones. | ||
Likewise, benzo[b][1,5]thiazepines (247, 248) were prepared via the reaction of o-aminothiophenol 195 with 3-(bis(methylthio)methylene)-pentane-2,4-dione 245 or 2-(bis(methylthio)methylene)-3-carbonitrile 246 respectively80 (Scheme 65).
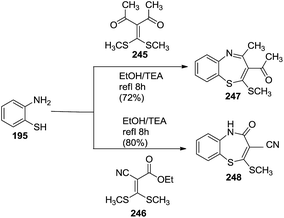 | ||
| Scheme 65 Synthesis of benzo[b][1,5]thiazepines from 3-(bis(methylthio)methylene)-pentane-2,4-dione and 2-(bis(methylthio)methylene)-3-carbonitrile. | ||
8. Conclusion
Over the past years the privileged structure concept has emerged as a rewarding approach in the arena of drug discovery and development. In this review, benzothiazepines have been showcased as a growing and increasingly important class of heterocyclic ring system with celebrated drug-like properties and versatile binding properties. Early developments in the arena of benzothiazepines were subjected to the synthesis of various derivatives and their biological evaluation. This stemmed into the discovery of some potent drugs such as thiazesim, dilteazem and quetipine fumerate as the mainstays of anti-psychotic therapeutics. Remarkably, the last decade has witnessed substantial developments in the chemistry of benzothiazepines resulting in a variety of innovative and interesting reactions to construct this core. The present survey evidently unites the scattered synthesis links to provide a comprehensive outlook of all the synthesis tactics implemented to date. Though many efficient synthesis procedures providing access to the benzothiazepine core are already known, it is expected that research to be performed in the coming years will uncover interesting aspects in the fields discussed in this review.Acknowledgements
This work was financially supported by the Department of Science and Technology (DST), Govt. of India (Grant No. SR/FT/CS-55/2011). Both the authors D. S. and G. J. gratefully acknowledge MHRD for the award of a research fellowship.References
- K. D. Mjos and C. Orvig, Chem. Rev., 2014, 114, 4540 CrossRef CAS PubMed.
- B. E. Evans, K. E. Rittle, M. G. Bock, R. M. DiPardo, R. M. Freidinger, W. L. Whitter, G. F. Lundell, D. F. Veber and P. S. Anderson, J. Med. Chem., 1988, 31, 2235 CrossRef CAS.
- G. C. Morton, J. M. Salvino, R. F. Labaudiniere and T. F. Herpin, Tetrahedron Lett., 2000, 41, 3029 CrossRef CAS.
- (a) S. Kawakita, M. Kinoshita, H. Ishikawa, T. Kagoshima, R. Katori, K. Ishikawa and Y. Hirota, Clin. Cardiol., 1991, 14, 53 CrossRef CAS PubMed; (b) H. M. Geyer, N. Watzman and J. P. Buckley, J. Pharm.Sci., 1970, 59, 964 CrossRef CAS PubMed.
- S. K. Bagal, A. D. Brown, P. J. Cox, K. Omoto, R. M. Owen, D. C. Pryde, B. Sidders, S. E. Skerratt, E. B. Stevens, R. I. Storer and N. A. Swain, J. Med. Chem., 2013, 56, 593 CrossRef CAS PubMed.
- N. Ye, J. L. Neumeyer, R. J. Baldessarini, X. Zhen and A. Zhang, Chem. Rev., 2013, 113, 123 CrossRef PubMed.
- J. B. Bariwal, K. D. Upadhyay, A. T. Manvar, J. C. Trivedi, J. S. Singh, K. S. Jain and A. K. Shah, Eur. J. Med. Chem., 2008, 43, 2279 CrossRef CAS PubMed.
- (a) B. C. Sekhar, Acta Chim. Slov., 2014, 61, 651 CAS; (b) A. P. G Nikalje, M. Ghodkeand and S. Tiwari, Asian J. Res. Chem., 2013, 6, 182 Search PubMed; (c) R. K. Gill, N. Aggarwal, J. Kumari, M. Kumari, P. Kaur, M. Kaur, A. Rani, A. Bansal, A. Shah and J. Bariwal, Chem.-Biol. Interact., 2013, 3, 146 CAS; (d) A. Levai, J. Heterocycl. Chem., 2000, 37, 199 CrossRef CAS PubMed.
- K. P. Jadhav and B. D. Ingle, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 1983, 22, 180 Search PubMed.
- V. M. Barot, M. R. Patel and H. B. Naik, Asian J. Chem., 2001, 13, 347 CAS.
- A. K. Swati and L. S. Prakash, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 1996, 35, 1221 Search PubMed.
- T. Suzuki, M. Ohashi, O. Takaiti and S. Harigaya, Arzneim. Forsch., 1994, 44, 3 CAS.
- A. Odawara, K. Kikkawa, M. Katoh, H. Toryu, T. Shimazaki and Y. Sasaki, Circ. Res., 1996, 78, 643 CrossRef CAS PubMed.
- G. D. Sarro, A. Chimirri, A. D. Sarro, R. Gitto, S. Grasso and M. Zappala, Eur. J. Med. Chem., 1995, 30, 925 CrossRef.
- G. Campiani, V. Nacci, I. Fiorini, M. P. D. Filippis, A. Garofalo, S. M. Ciani, G. Greco, E. Novellino, C. Manzoni and T. Mennini, Eur. J. Med. Chem., 1997, 32, 241 CrossRef CAS.
- H. Ishikawa, M. Matsushima, H. Matsui, A. Honjo, M. Hayashi, T. Shindo, T. Morifuji and M. Okaybayashi, Arzneim. Forsch., 1978, 28, 402 CAS.
- Y. Inada, K. Itoh, K. Kamiya, H. Sugihara and K. Nishikawa, Jpn. J. Pharmacol., 1988, 47, 135 CrossRef CAS.
- M. Amblard, I. Daffix, P. Bedos, G. Berge, D. Pruneau, J. L. Paquet, J. M. Luccarini, P. Belichard, P. Dodey and J. Martinez, J. Med. Chem., 1999, 42, 4185 CrossRef CAS PubMed.
- (a) S. Kasper and B. S. McEwen, CNS Drugs, 2008, 22, 15 CrossRef CAS; (b) A. K. Ganguly, S. S. Alluri, D. Caroccia, D. Biswas, C. H. Wang, E. Kang, Y. Zhang, A. T. Mc Phail, S. S. Carroll, C. Burlein, V. Munshi, P. Orth and C. Strickland, J. Med. Chem., 2011, 54, 7176 CrossRef CAS PubMed; (c) L. Kiefer, T. Gorojankina, P. Dauban, H. Faure, M. Ruat and R. H. Dodd, Bioorg. Med. Chem. Lett., 2010, 20, 7483 CrossRef CAS PubMed; (d) P. Gilleron, N. Wlodarczyk, R. Houssin, A. Farce, G. Laconde, J. F. Goossens, A. Lemoine, N. Pommery, J. P. Henichart and R. Millet, Bioorg. Med. Chem. Lett., 2007, 17, 5465 CrossRef CAS PubMed.
- K. Geoghegan, S. Smullen and P. Evans, J. Org. Chem., 2013, 78, 10443 CrossRef CAS PubMed.
- H. Wang, Y. Luo, X. Hou and J. Wu, Dalton Trans., 2013, 4410 RSC.
- Q. Xiao, Y. Luo, Y. Yan, Q. Ding and J. Wu, RSC Adv., 2013, 3, 5779 RSC.
- I. W. J. Still and T. S. Leong, Can. J. Chem., 1980, 58, 369 CrossRef CAS.
- R. A. Abramovitch, A. O. Kress, S. P. McManus and M. R. Smith, J. Org. Chem., 1984, 49, 3114 CrossRef CAS.
- A. Khalaj and N. Adibpour, Synth. Commun., 2008, 38, 3662 CrossRef CAS PubMed.
- H. Strobel, H. Bohn, O. Klingler, U. Schindler, K. Schoenafinger, and G. Zoller, Eur. Pat. EP 718 294, 1996.
- Y. Shinji, O. Hidekazu and W. Karekiyo, Eur. Pat. EP 717 040, 1996.
- S. K. Shah, S. K. Grant, M. Maccoss, K. Shankaran, H. Qi, and R. N. Guthikonda, PCT Int. WO 96 14842, 1996.
- J. Rongione, R. Brown and R. Dwight, US Pat., 6300503 2001.
- J. A. Robl, C. Q. Sun, J. Stevenson, D. E. Ryono, L. M. Simpkins, M. P. Cimarusti, T. Dejneka, W. A. Slusarchyk, S. Chao, L. Stratton, R. N. Misra, M. S. Bednarz, M. M. Asaad, H. S. Cheung, B. E. Abboa-Offei, P. L. Smith, P. D. Mathers, M. Fox, T. R. Schaeffer, A. A. Seymour and N. C. Trippodo, J. Med. Chem., 1997, 40, 1570 CrossRef CAS PubMed.
- N. G. Delaney, J. C. Barrish, R. Neubeck, S. Natarajan, M. Cohen, G. C. Rovnyak, G. Huber, N. Murugesan, R. Girotra and E. Sieber-McMaster, Bioorg. Med. Chem. Lett., 1994, 4, 1783 CrossRef CAS.
- K. S. Mohamed, N. M. Abdulaziz and A. A. Fadda, J. Heterocycl. Chem., 2013, 50, 645 CrossRef CAS PubMed.
- M. Struga, J. Kossakowski, A. E. Koziol, E. Kedzierska, S. Fidecka, P. L. Colla, C. Ibba, G. Collu, G. Sanna, B. Secci and R. Loddo, Eur. J. Med. Chem., 2009, 44, 4960 CrossRef CAS PubMed.
- M. Struga, J. Kossakowski, B. Miroslaw, A. E. Koziol and A. Zimniak, J. Heterocycl. Chem., 2009, 46, 298 CrossRef CAS PubMed.
- M. Alajarin, J. Cabrera, A. Pastor, P. S. Andrada and D. Bautista, J. Org. Chem., 2006, 71, 5328 CrossRef CAS PubMed.
- C. S. Lopez, O. N. Faza and A. R. de Lera, J. Org. Chem., 2009, 74, 2396 CrossRef CAS PubMed.
- W. Rudorf and D. Cleve, Sulfur Lett., 2002, 25, 105 CrossRef CAS PubMed.
- P. G. Mente and H. W. Hein, J. Org. Chem., 1971, 36, 3076 CrossRef.
- L. H. Sternbach, H. Lehr, E. Reeder, T. Hayes and N. Steiger, J. Org. Chem., 1965, 30, 2812 CrossRef CAS.
- W. Dolling, M. Biedermann and H. Hartung, Eur. J. Org. Chem., 1998, 1237 CrossRef.
- L. Fodor, J. Szabo, G. Bernath and P. Sohar, Tetrahedron Lett., 1995, 36, 753 CrossRef CAS.
- M. A. Mironov, M. N. Ivantsova, M. I. Tokareva and V. S. Mokrushin, Russ. Chem. Bull., 2004, 53, 1232 CrossRef CAS.
- C. Spitz, V. Reboul and P. Metzner, Tetrahedron Lett., 2011, 52, 6321 CrossRef CAS PubMed.
- L. G. Voskressensky, S. V. Akbulatov, T. N. Borisova, L. N. Kulikova, A. V. Listratova, E. A. Sorokina and A. V. Varlamov, Chem. Heterocycl. Compd., 2013, 49, 331 CrossRef CAS.
- H. Chen and D. Shi, J. Comb. Chem., 2010, 12, 571 CrossRef CAS PubMed.
- J. F. Liegeois, F. A. Rogister, J. Bruhwyler, J. Damas, T. P. Nguyen, M. Inarejos, E. M. G. Chleide, M. G. A. Mercier and J. E. Delarge, J. Med. Chem., 1994, 37, 519 CrossRef CAS.
- H. Maruendat and F. Johnson, J. Med. Chem., 1995, 38, 2145 CrossRef.
- Y. Liao, B. J. Venhuis, N. Rodenhuis, W. Timmerman and H. Wikstrom, J. Med. Chem., 1999, 42, 2235 CrossRef CAS PubMed.
- A. R. Katritzky, Y. Xu, H. He and S. Mehta, J. Org. Chem., 2001, 66, 5590 CrossRef CAS PubMed.
- R. Fu, X. Xu, Q. Dang and X. Bai, J. Org. Chem., 2005, 70, 10810 CrossRef CAS PubMed.
- M. H. Al-Huniti, M. M. El-Abadelah, J. A. Zahra, S. S. Sabri and A. Ingendoh, Molecules, 2007, 12, 1558 CrossRef CAS.
- H. Ding, Y. Zhang, M. Bian, W. Yao and C. Ma, J. Org. Chem., 2008, 73, 578 CrossRef CAS PubMed.
- P. Randhavane and B. Karale, J. Heterocycl. Chem., 2009, 46, 732 CrossRef CAS PubMed.
- H. Pettersson, A. Bulow, F. Ek, J. Jensen, L. K. Ottesen, A. Fejzic, J. Ma, A. L. D. Tredici, E. A. Currier, L. R. Gardell, A. Tabatabaei, D. Craig, K. McFarland, T. R. Ott, F. Piu, E. S. Burstein and R. Olsson, J. Med. Chem., 2009, 52, 1975 CrossRef CAS PubMed.
- F. Zeng and H. Alper, Org. Lett., 2010, 12, 5567 CrossRef CAS PubMed.
- G. L. Newton, N. Buchmeier, J. J. L. Clair and R. C. Fahey, Bioorg. Med. Chem., 2011, 19, 3956 CrossRef CAS PubMed.
- G. D. Mahale, Y. M. Kolekar, A. Kumar, D. Singh, K. M. Kodam and S. B. Waghmode, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 2011, 50, 1196 Search PubMed.
- Y. Zhao, Q. Dai, Z. Chen, Q. Zhang, Y. Bai and C. Ma, ACS Comb. Sci., 2013, 15, 130 CrossRef CAS PubMed.
- W. V. Snick, Y. K. Aibuldinov and W. Dehaen, Tetrahedron, 2013, 69, 4176 CrossRef PubMed.
- B. Yang, X. Tan, R. Guo, S. Chen, Z. Zhang, X. Chu, C. Xie, D. Zhang and C. Ma, J. Org. Chem., 2014, 79, 8040 CrossRef CAS PubMed.
- H. L. Khouzani, P. Tamjidi, I. Mohammadpoor-Baltork, M. Yaeghoobi, N. A. Rahman, A. R. Khosropour, M. Moghadam, S. Tangestaninejad, V. Mirkhani, M. H. Habibi, A. Kashima and T. Suzuki, J. Heterocycl. Chem., 2014, 51, 138 CrossRef PubMed.
- F. M. A. E. Latif and E. A. E. Rady, Phosphorus, Sulfur Silicon Relat. Elem., 2004, 179, 215 CrossRef PubMed.
- V. R. Rao and M. M. M. Reddy, Phosphorus, Sulfur Silicon Relat. Elem., 2006, 181, 461 CrossRef CAS PubMed.
- Y. Yan, X. Yang and L. Wu, Phosphorus, Sulfur Silicon Relat. Elem., 2012, 187, 573 CrossRef CAS PubMed.
- S. Shen, J. Ye and F. Liu, Phosphorus, Sulfur Silicon Relat. Elem., 2010, 185, 2366 CrossRef CAS PubMed.
- G. Singh, N. Kumar, A. K. Yadav and A. K. Mishra, Phosphorus, Sulfur Silicon Relat. Elem., 2002, 177, 621 CrossRef CAS PubMed.
- F. Yaccoubi, M. L. E. Efrit and H. Zantour, Phosphorus, Sulfur Silicon Relat. Elem., 2002, 177, 2321 CrossRef CAS PubMed.
- R. R. Kamble and B. S. Sudha, Phosphorus, Sulfur Silicon Relat. Elem., 2008, 183, 1691 CrossRef CAS PubMed.
- J. Slade, J. L. Stanton, D. B. David and G. C. Mazzenga, J. Med. Chem., 1985, 28, 1517 CrossRef CAS.
- W. Ried and E. Konig, Liebigs Ann. Chem., 1972, 755, 24 CrossRef CAS PubMed.
- A. S. Nahmanovits, T. E. Glomova, G. G. Skrovtsova, T. N. Komarova, B. I. Skrobogatova and U. A. Mansurov, Izv. Akad. Nauk SSSR, Ser. Khim., 1982, 1371 Search PubMed.
- V. I. Avramenko, V. S. Fedorenko, Z. F.Sholomko and N. Ya Bozhanova, Khim. Geterosikll. Soedin., 1978, 1049 CAS.
- J. Rokach and P. Hamel, J. Chem. Soc., Chem. Commun., 1979, 789 Search PubMed.
- J. Krapcho and C. F. Turk, J. Med. Chem., 1966, 39, 191 CrossRef.
- W. Ried and W. Marx, Chem. Ber., 1957, 90, 2683 CrossRef CAS PubMed.
- R. Buyle and H. G. Vieche, Tetrahedron, 1969, 25, 3453 CrossRef CAS.
- A. K. Gupta and U. C. Pant, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 1981, 20, 157 Search PubMed.
- (a) K. Hiedeg and O. Hanskovszky, Acta Chim. Acad. Sci. Hung., 1966, 50, 403 Search PubMed; (b) K. Hiedeg and O. Hanskovszky, Acta Chim. Acad. Sci. Hung., 1968, 56, 405 Search PubMed; (c) K. Hiedeg and O. Hanskovszky, Acta Chim. Acad. Sci. Hung., 1973, 75, 137 Search PubMed.
- (a) L. K. Muskalo, Zh. Obshch. Khim., 1958, 28, 742 Search PubMed; (b) J. C. Lancelot, B. Letosis, C. Saturnio and M. Robba, Synth. Commun., 1991, 21, 1901 CrossRef CAS PubMed.
- M. A. A. Mohamed, Synth. Commun., 2011, 41, 331 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |