Adsorption behavior and removal mechanism of arsenic on graphene modified by iron–manganese binary oxide (FeMnOx/RGO) from aqueous solutions†
Jin Zhua,
Zimo Loua,
Yu Liua,
Ruiqi Fua,
Shams Ali Baigab and
Xinhua Xu*a
aDepartment of Environmental Engineering, College of Environmental and Resource Sciences, Zhejiang University, Hangzhou 310058, People's Republic of China. E-mail: xuxinhua@zju.edu.cn; Fax: +86-571-88982031; Tel: +86-571-88982031
bDepartment of Environmental Sciences, COMSATS Institute of Information Technology (CIIT), Abbottabad 22060, Pakistan
First published on 4th August 2015
Abstract
Iron–manganese binary oxide (FeMnOx) is considered highly effective for arsenic adsorption, however, the agglomeration effect hindered its practical application. In this study, graphene has been used as a supporting matrix to disperse FeMnOx due to its huge specific surface area, and the synthesized novel composite adsorbent (FeMnOx/RGO) was employed for arsenic removal. Results demonstrated that FeMnOx/RGO (mass ratio of FeMnOx to FeMnOx/RGO nanocomposites is 45%) has larger specific surface area (411 m2 g−1) in comparison with bare FeMnOx, and showed 10.16 mg As g−1 FeMnOx and 11.49 mg As g−1 FeMnOx adsorption capacities for As(III) and As(V), respectively, with 1 mg L−1 initial concentration. Increased in the initial concentration to 7 mg L−1, the adsorption capacities of As(III) and As(V) reached to 47.05 mg As g−1 FeMnOx and 49.01 mg As g−1 FeMnOx, respectively. The removal process perfectly obeys pseudo second-order kinetic model for both As(III) and As(V). And PO43− was found to strongly inhibit arsenic adsorption. Furthermore, adsorption tests and characterization analyses confirmed that MnO2 played a key role on the oxidation of As(III), while iron(III) oxide was found crucial to As(V) removal. Electrostatic interaction and surface complexation mechanisms involved in the adsorption. These findings suggested that the adsorbent could be used in real arsenic-contaminated water treatment.
1. Introduction
Arsenic is one of the most toxic and carcinogenic chemical elements and its contamination in natural waters has become a worldwide problem.1 Long-term exposure to drinking water containing arsenic can give rise to cancers of skin, lungs, liver, kidney and bladder, as well as hypertension and cardiovascular disease.2 To minimize associated health risks, the World Health Organization (WHO) and US Environmental Protection Agency (USEPA) have set a more stringent standard value for arsenic in drinking water from 0.05 to 0.01 mg L−1 in 1990s.3,4 Inorganic forms of arsenic mainly exist in natural water environments. The inorganic forms, arsenite (As(III)) and arsenate (As(V)), are thought to be more toxic than the organic arsenic species. In general, As(III) exists in groundwater, while As(V) is dominate in surface water bodies.5–7 In addition, As(III) is more soluble and toxic than As(V) and it is less efficient to be removed because of its mobility in water and low affinity towards adsorbents.8In recent years, due to growing concerns of arsenic contamination in water bodies, numerous methods have been developed and adsorption is considered to be one of the most promising technologies due to its simple operation and low cost.9,10 Nowadays, a variety of materials have been used as adsorbents for arsenic removal, such as granular ferric hydroxide,11 Fe–Mn binary oxide,12 activated carbon,13 alumina,14 and so on. Among these materials, Fe–Mn binary oxide has been widely studied for arsenic remediation.15,16 The combination can take full advantages of the excellent oxidation potential of manganese dioxide for As(III) and the superior adsorption ability of iron(III) oxide for arsenic removal from aqueous solutions. However, bare Fe–Mn binary oxide exhibits high tendency to agglomerate, so that the adsorption properties will be weakened, limiting its full-scale application. In order to overcome this problem, providing a carrier such as multiwalled carbon nanotubes,17 activated carbon,18 graphene,19 and wheat straw20 to disperse particles of Fe–Mn binary oxide seems to be a better alternative for enhanced arsenic removal.
Graphene, a two-dimensional structure of sp2-bonded carbon with only one-atom thick, possesses huge theoretical specific surface area (2630 m2 g−1), good electrical conductivity, high thermal conductivity and intrinsic mobility.21 Compared to other carbon-based materials, such as activated carbon and multiwalled carbon nanotubes, graphene sheets have higher specific surface area, which provides more active adsorption sites for Fe–Mn binary oxide loading. Furthermore, their superior electrical conductivity can promote electron transport between Fe–Mn binary oxide and targeted contaminants in the adsorption process.22 Therefore, utilizing graphene as a supporter for adsorbents' synthesis attracts its wide application in pollutant remediation measures.
In our previous study, honeycomb briquette cinders (HBC) were used as a carrier to coat with Fe3O4 and MnO2 for arsenic removal, certifying that Fe3O4 and MnO2 had good affinity to arsenic.23 Nanoscale zero-valent iron (nZVI) assembled on magnetic Fe3O4/graphene for Cr(VI) removal was studied to confirm that graphene was an excellent supporter to disperse and stabilize nZVI particles.22 Therefore, in this study, graphene modified by iron–manganese binary oxide (FeMnOx/RGO) was synthesized and to reduce the associated costs, graphene was derived from exfoliation of graphite oxide followed by reduction. Characterization techniques including TEM, XRD, FTIR, BET and XPS were applied to characterize the adsorbent composites. Effect of loading ratios of Fe–Mn binary oxide was studied to explain the role of graphene and examine the removal efficiency of As(III) and As(V). Adsorption isotherms and adsorption kinetics were performed to explore the adsorbent properties. Effects of competing ions on As(III)/As(V) were also investigated. And effects of initial pH, various molar ratios of iron to manganese on graphene were carried out to identify the possible removal mechanism.
2. Materials and methods
2.1. Materials
Chemicals used in this study were of analytical grade except hydrochloric acid (GR). Graphite powder and hydrazine hydrate were purchased from Aladdin Reagent Database Inc., China. Potassium permanganate (KMnO4), sulfuric acid (H2SO4) 98%, sodium nitrate, hydrogen peroxide (H2O2) 30%, ammonia, ferrous sulfate heptahydrate (FeSO4·7H2O), manganese(II) sulfate monohydrate (MnSO4·H2O), sodium hydroxide and sodium hypochlorite were purchased from Sinopharm Chemical Reagent Co., Ltd, China.As(III) and As(V) stock solutions (1000 mg L−1) were prepared by dissolving appropriate amount of sodium arsenite (NaAsO2) and sodium arsenate dodecahydrate (Na3AsO4·12H2O) in purified water, respectively. Thiourea, L-ascorbic acid and potassium borohydride were utilized for arsenic detection.
2.2. Preparation of RGO
Graphite oxide was synthesized through a modified Hummer's method.24 In brief, 120 mL concentrated sulfuric acid was added into a 500 mL three-neck flask and then the flask was put in water bath containing ice bags with continuous stirring at 200 rpm. At 10 °C, 5.0 g graphite powder, 2.5 g NaNO3 and 15 g KMnO4 were added separately into the flask. After removing ice bags, the suspension was stirred overnight until its color turned into light gray and the mixture became pasty. Then 150 mL water was slowly added into the mixture to dilute the paste and the reaction temperature was heated to 98 °C for 2 h. Later on, 50 mL H2O2 was added and the mixture continued stirring for another 30 min. The product was washed by 5% HCl, ethanol and deionized water, and then dried in vacuum oven at 60 °C.Approximately 1.6 g synthesized graphitic oxide was re-dispersed in 800 mL deionized water and exfoliated by ultrasonication for 1.5 h to obtain graphene oxide. Subsequently, 19.2 mL hydrazine solution (50%) and 22.72 mL ammonia solution (25%) were added to reduce graphene oxide into graphene. The reaction was held for 3 h at 98 °C and then the mixture was centrifuged, rinsed by deionized water and filtered through a 0.45 μm filter membrane. Finally, the product was dried in vacuum at 60 °C and stored for subsequent experiments.
2.3. Preparation of FeMnOx/RGO
The fabrication of FeMnOx/RGO was followed by a co-precipitation process16 with some modifications. Firstly, 0.2 g graphene was dispersed in deionized water under ultrasonic wave for 1.5 h to obtain uniform black suspension, followed by the addition of desired amounts of KMnO4 and FeSO4·7H2O under vigorous stirring. At the same time, 5 M NaOH was added to adjust pH value to 7–8. Then the mixture was continuously stirred for 2 h at 60 °C and aged at room temperature for 12 h. Finally, the material was separated, rinsed and dried for further use.When the molar ratio of Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Mn was 3
Mn was 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, different mass ratios of generated Fe–Mn binary oxide on graphene to the whole nanocomposites were prepared, mass ratios of FeMnOx to FeMnOx/RGO nanocomposites are 14%, 45% and 76.5%, respectively. For instance, to obtain FeMnOx/RGO with mass ratio of FeMnOx 45%, 0.079 g KMnO4 and 0.4170 g FeSO4·7H2O were added for reaction. Fe–Mn binary oxide (FeMnOx) was also synthesized for comparison using the method employed by Zhang et al.16
1, different mass ratios of generated Fe–Mn binary oxide on graphene to the whole nanocomposites were prepared, mass ratios of FeMnOx to FeMnOx/RGO nanocomposites are 14%, 45% and 76.5%, respectively. For instance, to obtain FeMnOx/RGO with mass ratio of FeMnOx 45%, 0.079 g KMnO4 and 0.4170 g FeSO4·7H2O were added for reaction. Fe–Mn binary oxide (FeMnOx) was also synthesized for comparison using the method employed by Zhang et al.16
Using FeMnOx/RGO (nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3
nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, mass ratio of FeMnOx to FeMnOx/RGO nanocomposites is 45%) as a standard, different molar ratios of Fe
1, mass ratio of FeMnOx to FeMnOx/RGO nanocomposites is 45%) as a standard, different molar ratios of Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Mn were prepared. As the amount of FeSO4·7H2O was 0.4170 g, change the dosage of KMnO4 to make nFe
Mn were prepared. As the amount of FeSO4·7H2O was 0.4170 g, change the dosage of KMnO4 to make nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3
nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, 3
0.5, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 3
2 and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, respectively. In contrast, while 0.079 g KMnO4 was added, the dosage of FeSO4·7H2O was changed to make nFe
4, respectively. In contrast, while 0.079 g KMnO4 was added, the dosage of FeSO4·7H2O was changed to make nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 0.5
nMn = 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 4
1 and 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively.
1, respectively.
In order to better understand the removal mechanism, FeOx/RGO (0.2 g RGO, 0.4170 g FeSO4·7H2O) and MnOx/RGO (0.2 g RGO, 0.0507 g MnSO4·H2O, 0.0316 g KMnO4) were synthesized to compare with FeMnOx/RGO. Theoretically, the amount of obtained MnO2 in MnOx/RGO was the same as that in FeMnOx/RGO.
2.4. Characterization and analytical methods
TEM (JEM-1230, JEOL, Japan) analyses were conducted to know the morphological structures of bare graphene, FeMnOx and FeMnOx/RGO. XRD analysis (X'pert PRO analytical B.V., Netherlands) of the samples was performed in 2θ scale. The specific surface area and porosity data were analyzed by a BET analyzer (ASIC-2, USA). Functional groups and valence states of elements were investigated by FTIR (IRaffinity-1, SHIMADZU, Japan) and XPS (Kratos Axis Ultra DLD, SHIMADZU, Japan), respectively.PZC determination of RGO, FeMnOx and FeMnOx/RGO was obtained using a Zeta Meter 3.0 (Zetasizer 3000 HSa, UK). 0.01 g adsorbent was added into five 100 mL conical flasks with 50 mL deionized water, respectively. And the mixture was under ultrasonic wave for 20 min to obtain uniform suspension. Then adjust pH values to 2, 4, 6, 8 and 10, respectively. So zeta potentials under different pH values could be measured.
Total arsenic concentration was measured by an AFS-230E atomic fluorescence spectrophotometer (Beijing Kechuang Haiguang Instrument Company, China). Solution of 5% L-ascorbic acid and 5% thiourea was employed to act as a reducing reagent.
2.5. Batch adsorption experiments
Adsorption isotherms were conducted at temperature 25 °C and pH 7.0 ± 0.1. Adsorbent dose of 0.01 g was added into 50 mL 0.2, 0.6, 1.0, 3.0, 5.0, 7.0 mg L−1 As(III) or As(V) solution, respectively. And then the suspension was mixed by a rotary shaker (SUKUN, SKY-110WX, Shanghai, China) at 180 rpm. Samples were taken out using syringe and filtered through a 0.45 μm filter membrane after 24 h for arsenic detection.Kinetic studies were examined by batch experiments at different time intervals from 5 min to 24 h with 1 mg L−1 initial arsenic concentration and 0.2 g L−1 adsorbent dose. Effects of competing ions (SO42−, HCO3−, PO43−) on arsenic removal were investigated by varying the ionic concentration ranges (0–10 mM). Effect of initial pH on arsenic removal was tested by changing pH values from 3–11. Other conditions were kept the same as for isotherm experiments. All batch adsorption experiments were performed in triplicate, and the average values were reported.
2.6. Adsorbent desorption and regeneration
In order to study the regeneration potential of the sorbent, five consecutive adsorption–regeneration cycles were performed. 0.01 g adsorbent was added into 50 mL 100 μg L−1 As(III) or As(V) solution for adsorption and then 0.1 M NaOH + 0.1 M NaClO was used as the regenerant.3. Results and discussion
3.1. Characterization of adsorbents
TEM images reveal the morphological structures of RGO, FeMnOx as well as FeMnOx/RGO with different loading ratios of Fe–Mn binary oxide. The folding nature of graphene sheets was clearly visible in Fig. 1a. It can be observed that graphene possesses crumpled and silk wave-like structure, confirming that the reduced graphite oxide was successfully formed. Fig. 1b shows that particles of iron–manganese binary oxide agglomerated severely with each other, which could result in a dramatic decline in the functions of Fe–Mn binary oxide for arsenic removal. Irregular shapes of Fe–Mn binary oxide particles were found to attach tightly on graphene sheets (Fig. 1c–e). They were well dispersed on the surface of RGO, however, the average diameter increased with the increase of FeMnOx loading ratios. It can be clearly seen that the particle size of Fe–Mn binary oxide assembled on graphene was less than 10 nm when the mass ratio of FeMnOx to FeMnOx/RGO nanocomposites was 14%. However, the Fe–Mn binary oxide diameter was found to be 20–50 nm when the mass ratio was 45%. As the loading ratio achieved 76.5%, the average diameter (50–80 nm) was further increased, indicating the occurrence of a slight aggregation effect. Therefore, FeMnOx/RGO with appropriate loading ratios of Fe–Mn binary oxide could be more conductive to arsenic adsorption. | ||
| Fig. 1 TEM images of (a) RGO, (b) FeMnOx, (c) FeMnOx/RGO 14%*, (d) FeMnOx/RGO 45%* and (e) FeMnOx/RGO 76.5%*. *: mass ratios of Fe–Mn binary oxide to FeMnOx/RGO nanocomposites. | ||
According to Table S1,† the specific surface areas of RGO, Fe–Mn binary oxide were measured to be 548 m2 g−1 and 360 m2 g−1, respectively. In the presence of graphene possessing higher surface area, the specific surface area of FeMnOx/RGO became larger compared to the unsupported Fe–Mn binary oxide.25 And when the mass ratios of FeMnOx to FeMnOx/RGO increased, the surface area decreased because more active sites were occupied by FeMnOx particles. The BET surface area of FeMnOx/RGO was 411 m2 g−1, which was obtained from the nitrogen adsorption–desorption isotherms in Fig. S1.† A typical type-IV curve was observed, which was characteristic for mesoporous materials.26 Further, the isotherms exhibited a H2 hysteresis loop at a relative pressure from 0.4, implying that the adsorbent was a porous structure.27,28 According to Barrett–Joyner–Halenda (BJH) equation, the mean pore size in FeMnOx/RGO was 5.62 nm and the total pore volume was measured to be 0.59 cm3 g−1 (Table S1†), which was similar to the result of graphene, indicating that no major effect on porous structure occurred during the loading process, and bare FeMnOx also had a porous structure and high surface area, which was effective to arsenic adsorption.
Fig. S2† presents XRD patterns of RGO, FeMnOx and FeMnOx/RGO. As shown in Fig. S2a,† two typical peaks at 2θ = 25.0°, 44.0° corresponded to (002) and (100) reflections of graphene sheets.22 In Fig. S2b,† no obvious crystalline peak emerged, indicating that iron oxide and manganese oxide mainly existed in an amorphous form. The results were consistent with studies reported by Zhang et al.,12 and the formation of crystalline iron(III) oxides and manganese oxide could be blocked by their coexistences during the preparation process. After combining graphene with Fe–Mn binary oxide, reductions in the diffraction peaks of graphene were noticed (Fig. S2c†), which might be attributed to more disordered stacking and less agglomeration of graphene in the synthesized composite.29
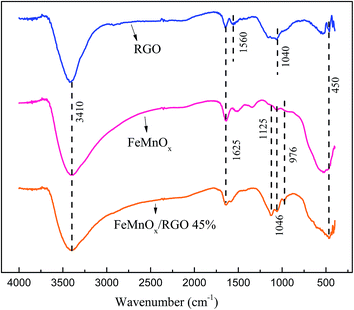
FTIR spectrums of RGO, FeMnOx and FeMnOx/RGO are shown in Fig. 2. And the magnification of relevant peaks from 400 to 1900 cm−1 wavenumber is given in Fig. S3.† Peak at λ value of 3410 cm−1 was assigned to O–H stretching vibrations in water molecules.30 The band appeared at 1625 cm−1 could be attributed to the hydroxyl deformation in water molecules indicating the presence of physisorbed water on the adsorbents.31 In the spectra of RGO, a broad band at 1040 cm−1 was the typical characteristic of C–O stretching vibration, and the presence of a weak band at 1560 cm−1 was assigned to the skeletal vibration of the unoxidized graphite,30 demonstrating that most part of graphene oxide has been reduced to RGO. The absorption band at 450 cm−1 corresponding to Mn–O vibration19 revealed the existence of residual KMnO4. Three small peaks appeared at 1125 cm−1, 1046 cm−1 and 976 cm−1 in FeMnOx and FeMnOx/RGO were assigned to the bending vibrations of Fe–OH,32 indicating the successful formation of iron oxide. The band at 450 cm−1 corresponding to Mn–O vibration strengthened after coating with Fe–Mn binary oxide on graphene,19 which revealed that particles of manganese oxide were integrated onto the basal plane of graphene.
3.2. Effect of loading ratios of Fe–Mn binary oxide
Fig. 3 compares the effect of different mass ratios of Fe–Mn binary oxide to FeMnOx/RGO nanocomposites for (a) As(III) and (b) As(V) adsorption. As presented in Fig. 3a, the adsorption capacity of FeMnOx/RGO for As(III) removal improved as the mass ratio of Fe–Mn binary oxide to FeMnOx/RGO increased. However, in Fig. 3b, there was little difference in the removal efficiency of As(V) when the mass ratios were 45% and 76.5%, indicating that higher proportion of Fe–Mn binary oxide might hinder As(V) adsorption on FeMnOx/RGO nanocomposites due to particle aggregation effect. This phenomenon could also be inferred from TEM images in Fig. 1. Moreover, Fig. 3 shows that FeMnOx/RGO had lower adsorption ability for both As(III) and As(V) removal as the mass ratio of FeMnOx was 14%, because the composition of Fe–Mn binary oxide containing in this nanocomposite was also lower.Langmuir and Freundlich isotherm models were employed to describe the data of arsenic adsorption. The Langmuir model33 and the Freundlich model34 are expressed as follows:
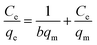 | (1) |
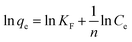 | (2) |
The fitting data of different kinds of sorbents are listed in Table 1. It can be seen that the Freundlich isotherm matched well with As(III) adsorption on FeMnOx/RGO with various ratios of Fe–Mn binary oxide and the values of n were all between 1 and 1019, indicating favorable adsorption on the sorbents. In contrast, As(V) adsorption was better described by the Langmuir model.
| Mass ratios of FeMnOx | Arsenic | Langmuir model | Freundlich model | ||||
|---|---|---|---|---|---|---|---|
| b (L mg−1) | qm (mg g−1) | R2 | n | KF (mg g−1) (mg−1)1/n | R2 | ||
| 14% | As(III) | 2.50 | 6.01 | 0.9824 | 3.18 | 3.48 | 0.9848 |
| As(V) | 2.81 | 6.44 | 0.9900 | 3.16 | 3.81 | 0.9861 | |
| 45% | As(III) | 3.46 | 22.42 | 0.9827 | 2.27 | 14.42 | 0.9963 |
| As(V) | 17.31 | 22.22 | 0.9928 | 5.34 | 20.50 | 0.8955 | |
| 76.5% | As(III) | 77.50 | 32.26 | 0.9846 | 3.62 | 44.05 | 0.9947 |
| As(V) | 15.38 | 22.42 | 0.9670 | 5.91 | 19.59 | 0.9119 | |
In order to determine the optimal loading ratios of Fe–Mn binary oxide more persuasively, Table 2 summarized the arsenic adsorption capacity of FeMnOx/RGO with different loading ratios of Fe–Mn binary oxide when the initial arsenic concentration was 7 mg L−1. Meanwhile, RGO and bare Fe–Mn binary oxide were used for comparison. From Table 2, it was obvious that RGO had subtile influence on arsenic removal, which meant that graphene only acted as a favorable carrier to disperse iron and manganese oxides and provided high specific surface area for Fe–Mn binary oxide loading. As can be clearly seen in Table 2, the adsorption capacities of Fe–Mn binary oxide assembled onto RGO with different loading ratios for both As(III) and As(V) removal were much higher than bare Fe–Mn binary oxide. Therefore, using RGO as a template can effectively enhance the adsorption ability of Fe–Mn binary oxide. The possible reason might be that RGO could prevent the particles of Fe–Mn binary oxide from aggregating due to its high specific surface area, so that active adsorption sites increased with the help of graphene, resulting in the enhancement of adsorption capacity.
| Adsorbent | Mass ratios of FeMnOx | Arsenic | qe |
|---|---|---|---|
| RGO | 0% | As(III) | 0.003 (mg As g−1) |
| As(V) | 0.001 (mg As g−1) | ||
| Bare FeMnOx | 100% | As(III) | 33.59 (mg As g−1 FeMnOx) |
| As(V) | 23.97 (mg As g−1 FeMnOx) | ||
| FeMnOx/RGO | 14% | As(III) | 40.19 (mg As g−1 FeMnOx) |
| As(V) | 44.16 (mg As g−1 FeMnOx) | ||
| FeMnOx/RGO | 45% | As(III) | 47.05 (mg As g−1 FeMnOx) |
| As(V) | 49.01 (mg As g−1 FeMnOx) | ||
| FeMnOx/RGO | 76.5% | As(III) | 42.12 (mg As g−1 FeMnOx) |
| As(V) | 30.31 (mg As g−1 FeMnOx) |
As shown in Table 2, FeMnOx/RGO owned the maximum equilibrium adsorption capacities for As(III) and As(V), which were found to be 47.05 mg As g−1 FeMnOx and 45.95 mg As g−1 FeMnOx. Furthermore, Table 3 compares arsenic adsorption capacity with some adsorbent materials in references, it showed that the synthesized FeMnOx/RGO in our study possessed excellent adsorption capacity for arsenic removal. Thus, FeMnOx/RGO (nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3
nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, mass ratio of FeMnOx to FeMnOx/RGO nanocomposites is 45%) was used for further experiments.
1, mass ratio of FeMnOx to FeMnOx/RGO nanocomposites is 45%) was used for further experiments.
| Adsorbent | Adsorbent dose (g L−1) | Initial concentration (mg L−1) | Adsorption capacity (mg g−1) | Ref. | |
|---|---|---|---|---|---|
| As(III) | As(V) | ||||
| FeMnOx/RGO | 0.2 | 7 | 22.17 | 22.05 | This paper |
Fe3O4–RGO–MnO2 (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8) 8) |
0.5 | 10 | 14.04 | 12.22 | 19 |
| Magnetic wheat straw | 0.5 | 28 | 3.898 | 8.062 | 20 |
HBC–Fe3O4–MnO2 (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
0.2 | 0.8 | 2.42 | 1.45 | 23 |
| Fe coated mesoporous carbon | 3.0 | 24 | 5.96 | 5.15 | 36 |
| Bead cellulose loaded with iron oxyhydroxide | 1.9 | 7.5 | 4.09 | 4.55 | 37 |
| Magnetite2–RGO | 0.2 | 7 | 13.10 | 5.83 | 38 |
| Magnetic biochar | 0.2 | 0.8 | 2.0 | 3.1 | 39 |
3.3. Sorption kinetics
Effect of contact time on As(III) or As(V) uptake onto the FeMnOx/RGO nanocomposites is shown in Fig. 4. The adsorption process was initially rapid in first 2 h for both As(III) and As(V), after which the adsorption capacity remained slowdown and reached to an equilibrium state within 12 h. As the initial arsenic concentration was 1 mg L−1, the equilibrium adsorption capacities of the adsorbent for As(III) and As(V) were 4.57 mg g−1 and 5.17 mg g−1, respectively. Therefore, it was observed that FeMnOx/RGO exhibited faster and greater adsorption ability for As(V) than As(III).The pseudo-first-order kinetic model40 and pseudo-second-order kinetic model41 were used to describe the experimental data. The linear forms are presented as follows:
ln(qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t
| (3) |
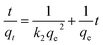 | (4) |
The results in Table 4 indicated that the pseudo-second-order model (R2 ≥ 0.9989) fitted perfectly with the experimental data for both As(III) and As(V). What's more, the calculated qe values derived from the pseudo-second-order model were close to the experimental qe (Table 4). That is to say, the rate-limiting step of arsenic adsorption on FeMnOx/RGO was chemical adsorption between the adsorbent and the adsorbate rather than a mass transfer in solutions.42
| Arsenic | Pseudo-first-order kinetic model | Pseudo-second-order kinetic model | Experimental data | ||||
|---|---|---|---|---|---|---|---|
| k1 (min) | qe (mg g−1) | R2 | k2 (g mg−1 min−1) | qe (mg g−1) | R2 | qe (mg g−1) | |
| As(III) | 0.0035 | 2.04 | 0.9112 | 7.53 × 10−3 | 4.59 | 0.9989 | 4.57 |
| As(V) | 0.0095 | 2.17 | 0.9742 | 1.52 × 10−2 | 5.24 | 0.9999 | 5.18 |
3.4. Effect of competing ions
Fig. 5 presents the influence of various competing ions (SO42−, HCO3−, PO43−) on As(III) and As(V) removal by FeMnOx/RGO. It can be seen that SO42− at different concentrations affected little on arsenic removal efficiency. Whereas, HCO3− and PO43− at the studied concentration range showed significant influence on arsenic removal.HCO3− inhibited As(V) adsorption more remarkably than As(III), because As(V) usually exists as anionic species in aqueous solutions.14 Thus, HCO3− and As(V) would compete for the active adsorption sites on the adsorbent to form more stable surface complexes with iron (oxy)hydroxides.43
As presented in Fig. 5, when the concentration of PO43− increased to 10 mM, the removal efficiency of As(III) and As(V) remained only 31.26% and 1.42%, respectively. The reason could be that PO43− had stronger affinity towards iron(oxy)hydroxides than arsenic, and it can form inner-sphere surface complexes with hydroxyl groups more easily, resulting in low removal efficiency of arsenic.44,45
3.5. Removal mechanism
The removal mechanisms of As(III) and As(V) exist differences due to their various species in water. Herein, the mechanism could be explained under the following aspects:As presented in Fig. 6, the removal efficiency of As(III) maintained over 80% in the pH between 3 and 9 and it dropped when pH > 9, indicating that part of neutral As(III) was converted into anionic As(V), which brought out electrostatic repulsion between negatively charged sorbent's surface and anionic As(V), together with the competition for active adsorption sites by OH−.
Variations of pH values before and after arsenic removal were investigated and presented in Table S2.† It can be seen that in acidic environment, pH slightly increased after arsenic adsorption, whereas the pH decreased under alkaline conditions demonstrating the possible release of H+ from the surface of sorbent.39 There were minor variations between pH values before and after arsenic removal as the initial pH value was around 7, showing the material was stable in real water treatment and it was considered to be a promising adsorbent for arsenic removal. Our results were in agreement with the study reported by Feng et al.47
 | ||
Fig. 7 XPS spectra of (a) Fe2p and (b) Mn2p: (1) nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 0.5 nMn = 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, (2) nFe 1, (2) nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 1 nMn = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, (3) nFe 1, (3) nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3 nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, (4) nFe 0.5, (4) nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3 nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, (5) nFe 1, (5) nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3 nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, (6) nFe 2, (6) nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3 nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, (7) nFe 4, (7) nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 4 nMn = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 1. | ||
Fig. 8 presents the influence of various molar ratios of iron to manganese in As(III) and As(V) adsorption. In Fig. 8a, when the proportion of MnO2 increased, the adsorption ability of the sorbent for As(III) removal also enhanced, indicating the oxidation ability of MnO2. However, too much amount of MnO2 (nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3
nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) resulted in a dramatic decline in the adsorption capacity for As(III), which might be the reason that MnO2 occupied too many adsorption sites, inhibiting As(III) adsorption. There was a little difference in the removal efficiency of As(V) as nFe
4) resulted in a dramatic decline in the adsorption capacity for As(III), which might be the reason that MnO2 occupied too many adsorption sites, inhibiting As(III) adsorption. There was a little difference in the removal efficiency of As(V) as nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3
nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, 3
0.5, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 3
2 and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4. While FeMnOx/RGO (nFe
4. While FeMnOx/RGO (nFe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nMn = 3
nMn = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) held the highest adsorption capacity among the other adsorbents (Fig. 8b). The results demonstrated that MnO2 had subtle effect on As(V) adsorption and 3
1) held the highest adsorption capacity among the other adsorbents (Fig. 8b). The results demonstrated that MnO2 had subtle effect on As(V) adsorption and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was an appropriate proportion of iron to manganese for arsenic removal. In contrast, when changing the amount of iron oxide, the difference of adsorption capacity for As(V) was more obvious (Fig. 8d) as compared to As(III) (Fig. 8c), which revealed that iron(III) oxide owned the dominant availability for As(V) removal.
1 was an appropriate proportion of iron to manganese for arsenic removal. In contrast, when changing the amount of iron oxide, the difference of adsorption capacity for As(V) was more obvious (Fig. 8d) as compared to As(III) (Fig. 8c), which revealed that iron(III) oxide owned the dominant availability for As(V) removal.
To understand the role of iron and manganese oxides more clearly, Fig. 9 compares the adsorption ability of FeOx/RGO and MnOx/RGO with FeMnOx/RGO for As(III) and As(V) adsorption. From Fig. 9, the adsorption capacity of adsorbents for both As(III) and As(V) was in the following order: FeMnOx/RGO > FeOx/RGO > MnOx/RGO. It was obvious that MnO2 had quite lower adsorption ability for As(III) and As(V); however, MnO2 was found highly effective for As(III) oxidation. It can be seen in Fig. 10 that after reaction with As(III) solution, MnOx/RGO showed a peak at 48.5 eV, corresponding to As(V), while the peak in XPS spectra of FeOx/RGO was also the characteristic peak of As(III) (44.3 eV).31,49 Thus, MnO2 could transform As(III) into As(V) easily during the adsorption process and FeOx/RGO had higher adsorption capacity for As(V) rather than As(III) (Fig. 9), confirming iron(III) oxide played a key role on As(V) removal.
Data fitted by Langmuir model showed that the sum of adsorption capacities of FeOx/RGO and MnOx/RGO for As(III) removal was lower than the maximum adsorption amount of As(III) by FeMnOx/RGO (5.96 mg g−1 + 2.95 mg g−1 = 8.91 mg g−1 < 22.42 mg g−1). Moreover, the results of As(V) (12.14 mg g−1 + 3.41 mg g−1 = 15.55 mg g−1 < 22.22 mg g−1) were in agreement with that of As(III) (Table S3†), confirming that Fe–Mn binary oxide possessed synergistic effects for As(III) and As(V) removal. Thus, combination of both iron oxide and manganese oxide on graphene was very effective and beneficial for arsenic removal.
To summarize the possible mechanism of As(III) adsorption on FeMnOx/RGO, the schematic diagram of removal mechanism is shown in Fig. 11. As the adsorbent was added into As(III) solution, the part of As(III) was converted into As(V) by MnO2. The reaction pathway involved two steps containing the reduction of Mn(IV) to Mn(III) and then Mn(III) to Mn(II):31
| MnO2 + H3AsO3 + 2H+ = Mn2+ + H3AsO4 + H2O | (5) |
| 2MnO2 + H3AsO3 = 2MnOOH* + H3AsO4 | (6) |
| 2MnOOH* + H3AsO3 + 4H+ = 2Mn2+ + H3AsO4 + 3H2O | (7) |
Furthermore, As(V) would diffuse into the aqueous solution because of higher concentration on the surface of MnO2.23 Then As(V) together with the minority of As(III) was more attractive to iron(III) oxide attached tightly on graphene. As a consequence, hydroxyl groups on the surface of iron oxide could be replaced by As(III)/As(V) via ligand exchange, forming the inner-sphere surface complexes.50,51 Thus, arsenic was eventually adsorbed on FeMnOx/RGO.
3.6. Regeneration and reusability
Relevant experiments about the reusability of this adsorbent were performed. Fig. 12 reveals arsenic removal efficiency on regenerated FeMnOx/RGO at initial concentration 100 μg L−1. In the five treatment cycles, the majority of As(III) or As(V) was removed and As(III) removal efficiency was always higher than 90%, indicating that the residual concentration of As(III) was still under the standard concentration 10 ppb after the fifth adsorption. These results also indirectly demonstrated that the adsorbed arsenic could be successfully desorbed from FeMnOx/RGO using 0.1 M NaOH + 0.1 M NaClO as the regenerant. Therefore, this adsorbent possesses good regeneration potential and it will be effectively and consecutively applied in real treatment of arsenic-contaminated water in future. | ||
| Fig. 12 Five consecutive adsorption–regeneration cycles on FeMnOx/RGO for As(III) and As(V) removal. | ||
4. Conclusions
In summary, FeMnOx/RGO has been successfully prepared and employed for As(III) and As(V) removal from aqueous solutions. Due to huge specific surface area of graphene, Fe–Mn binary oxide was finely dispersed onto the surface of RGO, providing a large number of adsorption active sites for arsenic removal. Results demonstrated that MnO2 possessed excellent oxidative ability for As(III), while iron(III) oxide was quite efficient to As(V) removal. High removal efficiency by FeMnOx/RGO confirmed that this material could be promising in large scale applications.Acknowledgements
The authors acknowledge for the financial support of the National Natural Science Foundation of China (No. 21477108 and 21277119).References
- D. Mohan and C. U. Pittman, J. Hazard. Mater., 2007, 142, 1–53 CrossRef CAS PubMed.
- V. M. Boddu, K. Abburi, J. L. Talbott, E. D. Smith and R. Haasch, Water Res., 2008, 42, 633–642 CrossRef CAS PubMed.
- WHO, Guidelines for Drinking Water Quality, Switzerland, Geneva, 4th edn, 2011 Search PubMed.
- USEPA, Arsenic Treatment Technology Evaluation Handbook for Small System, EPA 816-R-03-014, Washington, D.C., 2000 Search PubMed.
- C. K. Jain and I. Ali, Water Res., 2000, 34, 4304–4312 CrossRef CAS.
- P. L. Smedley and D. G. Kinniburgh, Appl. Geochem., 2002, 17, 517–568 CrossRef CAS.
- K. Zhang, V. Dwivedi, C. Y. Chi and J. S. Wu, J. Hazard. Mater., 2010, 182, 162–168 CrossRef CAS PubMed.
- T. A. Saleh, S. Agarwal and V. K. Gupta, Appl. Catal., B, 2011, 106, 46–53 CAS.
- D. Ranjan, M. Talat and S. H. Hasan, J. Hazard. Mater., 2009, 166, 1050–1059 CrossRef CAS PubMed.
- M. Jang, S. H. Min, T. H. Kim and J. K. Park, Environ. Sci. Technol., 2006, 40, 1636–1643 CrossRef CAS.
- K. Banerjee, G. L. Amy, M. Prevost, S. Nour, M. Jekel, P. M. Gallagher and C. D. Blumenschein, Water Res., 2008, 42, 3371–3378 CrossRef CAS PubMed.
- G. S. Zhang, J. H. Qu, H. J. Liu, R. P. Liu and R. C. Wu, Water Res., 2007, 41, 1921–1928 CrossRef CAS PubMed.
- P. Mondal, C. Balomajumder and B. Mohanty, J. Hazard. Mater., 2007, 144, 420–426 CrossRef CAS PubMed.
- T. F. Lin and J. K. Wu, Water Res., 2001, 35, 2049–2057 CrossRef CAS.
- R. J. Gohari, W. J. Lau, T. Matsuura and A. F. Ismail, Sep. Purif. Technol., 2013, 118, 64–72 CrossRef PubMed.
- G. S. Zhang, H. J. Liu, R. P. Liu and J. H. Qu, J. Hazard. Mater., 2009, 168, 820–825 CrossRef CAS PubMed.
- P. Tamilarasan and S. Ramaprabhu, AIP Conf. Proc., 2012, 1447, 321–322 CrossRef CAS PubMed.
- Z. G. Liu, F. S. Zhang and R. Sasai, Chem. Eng. J., 2010, 160, 57–62 CrossRef CAS PubMed.
- X. B. Luo, C. C. Wang, S. L. Luo, R. Z. Dong, X. M. Tu and G. S. Zeng, Chem. Eng. J., 2012, 187, 45–52 CrossRef CAS PubMed.
- Y. Tian, M. Wu, X. B. Lin, P. Huang and Y. Huang, J. Hazard. Mater., 2011, 193, 10–16 CrossRef CAS PubMed.
- Y. W. Zhu, S. Murali, W. W. Cai, X. S. Li, J. W. Suk, J. R. Potts and R. S. Ruoff, Adv. Mater., 2010, 22, 3906–3924 CrossRef CAS PubMed.
- X. S. Lv, X. Q. Xue, G. M. Jiang, D. L. Wu, T. T. Sheng, H. Y. Zhou and X. H. Xu, J. Colloid Interface Sci., 2014, 417, 51–59 CrossRef CAS PubMed.
- J. Zhu, S. A. Baig, T. T. Sheng, Z. M. Lou, Z. X. Wang and X. H. Xu, J. Hazard. Mater., 2015, 286, 220–228 CrossRef CAS PubMed.
- W. S. Hummers and R. E. Offeman, J. Am. Chem. Soc., 1958, 80, 1339 CrossRef CAS.
- X. L. Wu, L. Wang, C. L. Chen, A. W. Xu and X. K. Wang, J. Mater. Chem., 2011, 21, 17353 RSC.
- A. Yürüm, Z. Ö. Kocabaş-Ataklı, M. Sezen, R. Semiat and Y. Yürüm, Chem. Eng. J., 2014, 242, 321–332 CrossRef PubMed.
- S. W. Liu, S. H. Kang, G. Z. Wang, H. J. Zhao and W. P. Cai, J. Colloid Interface Sci., 2015, 458, 94–102 CrossRef PubMed.
- G. Srinivas, Y. W. Zhu, R. Piner, N. Skipper, M. Ellerby and R. Ruoff, Carbon, 2010, 48, 630–635 CrossRef CAS PubMed.
- Z. S. Wu, W. C. Ren, L. Wen, L. B. Gao, J. P. Zhao, Z. P. Chen, G. M. Zhou, F. Li and H. M. Cheng, ACS Nano, 2010, 4, 3187–3194 CrossRef CAS PubMed.
- C. B. Wang, J. W. Zhou, J. Ni, Y. L. Cheng and H. Li, Chem. Eng. J., 2014, 253, 130–137 CrossRef CAS PubMed.
- G. S. Zhang, J. H. Qu, H. J. Liu, R. P. Liu and G. T. Li, Environ. Sci. Technol., 2007, 41, 4613–4619 CrossRef CAS.
- Y. Zhang, M. Yang, X. M. Dou, H. He and D. S. Wang, Environ. Sci. Technol., 2005, 39, 7246–7253 CrossRef CAS.
- I. Langmuir, J. Am. Chem. Soc., 1918, 40, 1361–1403 CrossRef CAS.
- H. Freundlich, Z. Phys. Chem., 1906, 57, 385–470 CAS.
- K. Fytianos, E. Voudrias and E. Kokkalis, Chemosphere, 2000, 40, 3–6 CrossRef CAS.
- Z. M. Gu and B. L. Deng, Environ. Eng. Sci., 2007, 24, 113–121 CrossRef CAS.
- X. J. Guo and F. H. Chen, Environ. Sci. Technol., 2005, 39, 6808–6818 CrossRef CAS.
- V. Chandra, J. Park, Y. Chun, J. W. Lee, I. Hwang and K. S. Kim, ACS Nano, 2010, 4, 3979–3986 CrossRef CAS PubMed.
- S. A. Baig, J. Zhu, N. Muhammad, T. T. Sheng and X. H. Xu, Biomass Bioenergy, 2014, 71, 299–310 CrossRef CAS PubMed.
- Y. S. Ho, Scientometrics, 2014, 59, 171–177 Search PubMed.
- Y. S. Ho and G. McKay, Adsorpt. Sci. Technol., 1998, 16, 243–255 CAS.
- L. S. Tan, J. Xu, X. Q. Xue, Z. M. Lou, J. Zhu, S. A. Baig and X. H. Xu, RSC Adv., 2014, 4, 45920–45929 RSC.
- L. J. Dong, P. V. Zinin, J. P. Cowen and L. C. Ming, J. Hazard. Mater., 2009, 168, 626–632 CrossRef CAS PubMed.
- J. C. Hsu, C. J. Lin, C. H. Liao and S. T. Chen, Chemosphere, 2008, 72, 1049–1055 CrossRef CAS PubMed.
- X. G. Meng, G. P. Korfiatis, S. Bang and K. W. Bang, Toxicol. Lett., 2002, 133, 103–111 CrossRef CAS.
- E. Lombi, W. W. Wenzel and R. S. Sletten, J. Plant Nutr. Soil Sci., 1999, 162, 451–456 CrossRef CAS.
- L. Y. Feng, M. H. Cao, X. Y. Ma, Y. S. Zhu and C. W. Hu, J. Hazard. Mater., 2012, 217–218, 439–446 CrossRef CAS PubMed.
- J. Lu, X. L. Jiao, D. R. Chen and W. Li, J. Phys. Chem. C, 2009, 113, 4012–4017 CAS.
- C. Wang, H. J. Luo, Z. L. Zhang, Y. Wu, J. Zhang and S. W. Chen, J. Hazard. Mater., 2014, 268, 124–131 CrossRef CAS PubMed.
- R. L. Vaughan and B. E. Reed, Water Res., 2005, 39, 1005–1014 CrossRef CAS PubMed.
- X. J. Guo, Y. H. Du, F. H. Chen, H. S. Park and Y. N. Xie, J. Colloid Interface Sci., 2007, 314, 427–433 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra11601e |
| This journal is © The Royal Society of Chemistry 2015 |