Hierarchical CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell arrays for high-performance supercapacitors†
Kang Zhanga,
Wei Zengb,
Guanhua Zhangc,
Sucheng Houc,
Fei Wange,
Taihong Wangcd and
Huigao Duan*cd
aSchool of Materials Science and Engineering, Hunan University, Hunan, 410082 P. R. China
bSchool of Chemistry and Chemical Engineering, Hunan University, Hunan, 410082 P. R. China
cSchool of Physics and Electronics, Hunan University, Hunan, 410082 P. R. China. E-mail: duanhg@hnu.edu.cn
dState Key Laboratory for Chem/Biosensing and Chemometrics, Hunan University, Hunan, 410082 P. R. China
eCollege of Science, National University of Defense Technology, Hunan, 410073 P. R. China
First published on 11th August 2015
Abstract
Ternary metal oxides have attracted extensive attention for supercapacitor applications due to their enhanced performance compared to their binary counterparts. In this work, 3D CuCo2O4@NiCo2O4 core/shell nanostructures have been synthesized via a two-step method on nickel foam followed by a post annealing process. Morphological characterizations show that the porous CuCo2O4 nanowires are well covered by NiCo2O4 nanosheets. The hybrid electrode is used as a binder-free electrode, showing a high specific capacitance of 2029 F g−1 (while the areal capacitance is 2.59 F cm−2) at the current density of 10 mA cm−2, higher than the CuCo2O4 electrode. After 4500 cycles, the specific capacitance remains 1548 F g−1, ∼80% of the original capacitance. In particular, the hybrid electrode exhibits great rate capability, the hybrid electrode retains 1551 F g−1 as the current density increases to 30 mA cm−2, which can be ascribed to the excellent electron transport properties of core/shell CuCo2O4@NiCo2O4 structures.
1. Introduction
With the rapid development of electric vehicles and portable devices, there is an urgent need for electrochemical energy storage. Compared to lithium-ion batteries, supercapacitors (SCs) have attracted extensive interest due to their higher power densities, fast charge rate and long cycle life. In general, SCs can be divided into two types defined by the storage mechanism, i.e., electrical double layer capacitors (EDLCs) and pseudocapacitors (PCs). Specifically, EDLCs store the charged ions from an electrolyte onto electrodes forming a double layer structure via a physically adsorbing–desorbing process. Current commercial capacitors mainly belong to EDLCs, which based on carbonaceous materials including carbon aerogel,1,2 carbon fiber,3 carbon nanotube and graphene.4–8 However, EDLCs have suffered from the low energy density which restricts its extensive applications. On the contrary, PCs can provide much higher energy density due to their fast and reversible faradaic redox reactions. Therefore, much more attentions have been focused on the study of PCs recent years.Seeking for better electrode materials is essential to develop PCs. Transition metal oxides,9–11 hydroxides12–14 and sulfides15–17 have been widely reported for PCs owing to their high theoretical capacity, low cost and environmental compatibility. Among the transition metal oxides, Co3O4 has become a promising electrode material because of its high theoretical capacity.11,18–22 However, the relatively low conductivity and high cost restrict its commercial application. In order to overcome these disadvantages and realize better electrochemical performance, some attempts have been made to synthesize ternary oxide via replacing Co in Co3O4 partially with alternative metals (e.g., Zn, Cu, Ni, Mn, and Fe).23–29 Bao et al. synthesized mesoporous ZnCo2O4 nanosheet arrays on Ni foam via a simple hydrothermal method with a ultrahigh specific pseudo-capacitance of 2468 F g−1 at 5 A g−1 and excellent cycling stability.24 Chen and co-workers fabricated porous NiCo2O4 flowerlike nanostructures by a simple hydrothermal and subsequent annealing process with an enhanced specific capacitance of 658 F g−1 at 1 A g−1 compared to Co3O4.25 Obviously, ternary metal oxides which possess multiple oxidation states exhibit a higher supercapacitive performance in comparison with their binary counterparts.
Besides, in order to further enhance the electrochemical ability, lots of electrode materials with hierarchical core/shell nanostructure have been investigated.23,30–36 In a typical core/shell system, the core can provide better conductivity for charge transfer, while the shell can serve as active electrode materials with high surface areas. As a result, researchers have demonstrated that such core/shell nanostructure can realize a higher electrochemical performance due to the synergistic effect. For instance, Huang et al. have shown that hybrid composites Ni(OH)2@NiCo2O4 exhibited a notable increased capacitance and good cycle performance compared with a pristine Ni(OH)2 array.31 In addition, the materials grown on substrate can be directly used as electrode for SCs without using a polymer binder, which can improve the ion diffusion and electron conductivity. Then the better electrochemical performance can be realized.37,38 Qian and co-workers synthesized the ultralight, high-surface-area, multifunctional graphene-based aerogels as binderless monolithic electrodes for supercapacitors with a good electrochemical performance. Yu group reported the free-standing Co–Co(OH)2 composite nanoflakes on 3D nickel foam for supercapacitors, which exhibited a high specific capacitance.
On the basis of the above considerations, we designed and synthesized a hierarchical core/shell structure of CuCo2O4 nanowires@NiCo2O4 nanosheets on Ni foam in this work. We used thin CuCo2O4 nanowires as the scaffold cores to deposit the “shell” composed by NiCo2O4 nanosheets. Taking the advantage of the synergistic effect and 3D core/shell nanostructure, the hybrid electrode exhibited a notable increased electrochemical performance with a high specific capacitance of 2029 F g−1 at the current density of 10 mA cm−2 and an excellent rate capability (61.6% capacity retention at 30 mA cm−2). Moreover, the hybrid electrode retains 76% of the original capacity after 4500 cycles at 10 mA cm−2, which indicates an excellent cycling stability.
2. Experimental
Materials synthesis
All the reagents used in the experiment were analytical grade (purchased from Sinopharm) without further purification. The Ni foam was cleaned by sonication in acetone, ethanol, and deionized (DI) water in sequence for 30 min, respectively.The mass of the samples was weighed by an electronic balance with an accuracy of 0.001 mg. The mass of the pure Ni foam, CuCo2O4 NWAs on Ni foam after hydrothermal process, and CuCo2O4@NiCo2O4 on Ni foam after electrodeposition were noted as M1, M2, and M3, respectively. The mass of CuCo2O4 NWAs (noted as M4) was calculated via the following formulas:
| M4 = M2 − M1, | (1) |
| M5 = M3 − M1. | (2) |
Characterizations
The morphology of as-prepared samples was characterized by scanning electron microscopy (SEM, Hitachi S-4800). Transmission electron microscope (TEM) images, high-resolution transmission electron microscopy (HRTEM) images were obtained with JEM-2100F equipped with Oxford X-Max80 EDS detector. The crystallographic phases of the CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell arrays were observed by X-ray diffraction spectroscopy (XRD Rigaku 2550).Electrochemical measurements
The electrochemical tests were carried out in a three electrode electrochemical cell containing 3 M KOH aqueous solution as the electrolyte. The as-prepared sample was directly used as the working electrode. A saturated calomel electrode (SCE) was used as the reference electrode and a platinum electrode as the counter electrode. The area of the working electrode was made about 1 × 1 cm2. According to the eqn (1) and (2), the mass loading of CuCo2O4 on Ni foam was 0.98 mg cm−2 and the mass loading of CuCo2O4@NiCo2O4 hybrid was 1.28 mg cm−2. The cyclic voltammetry (CV) measurements and galvanostatic charge–discharge measurements were carried out with an electrochemical workstation (CHI 660e). Electrochemical impedance spectroscopy (EIS) measurements were performed by applying an AC voltage with 1 mV amplitude in a frequency ranging from 0.01 Hz to 100 kHz at open circuit potential. The areal capacitance (Ca) and specific capacitance (Cs) were calculated by following equations:
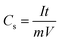 | (3) |
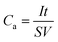 | (4) |
3. Results and discussion
As schematically shown in Fig. 1, the unique hierarchical core/shell structure of CuCo2O4 NWs@NiCo2O4 NSs on Ni foam was synthesized via two-step method with a post-annealing process. | ||
| Fig. 1 Schematic illustration of the fabrication process of 3D hybrid hierarchical CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell arrays on Ni foam. | ||
In this paper, 3D porous Ni foam was used as the substrate. The low-magnification SEM image of Ni foam was shown in Fig. 2a. First, the CuCo2O4 nanowire arrays were grown on Ni foam by a facile hydrothermal and post-annealing process. Fig. 2b shows the close-up image of the interface between the Ni foam and CuCo2O4 nanowires. Fig. 2c and d shows the scanning electron microscopy (SEM) images of the CuCo2O4 nanowires on Ni foam. It is obvious that the Ni foam is covered by the CuCo2O4 nanowires uniformly with an average diameter of ∼100 nm and length of ∼4 μm. Then, the CuCo2O4 nanowire arrays grown on Ni foam were used as the scaffold for the electrodeposition of NiCo2O4 nanosheets, while the NiCo2O4 nanosheets served as the “shell”. The CuCo2O4 nanowires can provide a vast quantity of sites for the growth of NiCo2O4 nanosheets. Fig. 2e and f shows the SEM images of the CuCo2O4@NiCo2O4 core/shell arrays. Obviously, the surface of CuCo2O4 nanowires becomes rough and covered by ultrathin NiCo2O4 nanosheets after the electrodeposition process. As shown in Fig. 2f, the obtained CuCo2O4@NiCo2O4 core/shell arrays are well aligned on Ni foam. The CuCo2O4 nanowire arrays are partially covered by the interconnected NiCo2O4 nanosheet shells, which means that both the “core” and the “shell” can take part in the redox reaction during the charge/discharge process. Typically, core/shell arrayed structures can enhance the surface areas, providing more electrochemical active sites to boost the area capacitance during the reversible redox reaction.
XRD was used to investigate the structural properties of the fabricated structures. To avoid the strong XRD signal of Ni foam substrate, the CuCo2O4 and CuCo2O4@NiCo2O4 were synthesized on carbon cloth for XRD. The successful preparation of CuCo2O4 on carbon cloth was confirmed by XRD. As shown in Fig. S2(a),† all the diffraction peaks can be well indexed to the CuCo2O4 phase (JCPDS no. 1-1155) except the XRD signal resulted from carbon cloth. Similarly, the X-ray diffraction was used to confirm the composition of CuCo2O4@NiCo2O4. As shown in Fig. S2(b),† all the diffraction peaks can be well indexed to the CuCo2O4 phase (JCPDS no. 1-1155) and the spinel NiCo2O4 phase (JCPDS 73-1702).
In order to further verify the structural properties of CuCo2O4 and NiCo2O4, the detailed microstructure and morphology of CuCo2O4 and CuCo2O4@NiCo2O4 hybrid electrode were analyzed by high-resolution transmission electron microscopy (HRTEM). Fig. 3a shows the low-magnification TEM image of as-prepared CuCo2O4 nanowires, demonstrating that the CuCo2O4 nanowires are highly porous structures. Besides, according to the Fig. 3b, the image of selected area electron diffraction (SAED) pattern indicates a good crystallinity of CuCo2O4. Furthermore, the fringe spacing is measured to be 0.28 nm, which is well corresponding to the (220) plane of the spinel CuCo2O4. The SAED result is well consistent with the previous XRD results. Fig. 3c shows the typical TEM image of CuCo2O4@NiCo2O4 hybrid electrode. Obviously, the highly porous CuCo2O4 nanowires are covered by a NiCo2O4 nanosheet layer, forming a unique core/shell nanostructure. It is worth noting that some of the nanosheets might be damaged during the sonication process for preparing the TEM sample. As shown in Fig. 3d, the selected area electron diffraction (SAED) pattern indicates the polycrystalline characteristic of the NiCo2O4 nanosheets. In addition, the fringe spacing is measured to be 0.24 nm, which is well corresponding to the (311) plane of the spinel NiCo2O4 and in accordance with the XRD data.
The “oriented attachment” and “self-assembly” processes, involving a spontaneous self-organization between neighboring particles to share a common crystallographic orientation, dominated the growth of CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell array.39,40 Specifically, the total free energy in the system was reduced by eliminating the surface energy associated with unsatisfied bonds of the nanocrystallines through the bonding between the particles. In our experiments, on the one hand, CuCo2O4 nanowire acts as the backbone to guide the self-assembly of NiCo2(OH)6 (the precursor of NiCo2O4) in aqueous solution during the co-electrodeposition processes. On the other hand, the “oriented attachment” can guide the oriented growth of the nanoparticles. Nanoparticles can attach to the surface of CuCo2O4 to decrease surface energy due to their high surface energy and thermodynamics instability. As shown in Fig. 2e, the nanoparticles attached on the surface of CuCo2O4 gradually grew into the nanosheets with the increase of the co-electrodeposition time. As shown in Fig. S3,† the nanowires would be covered completely by NiCo2O4 finally.
The hierarchical CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell arrays on Ni foam were used as electrode to investigate the pseudocapacitive performance. All electrochemical tests were carried out in a three electrode (a saturated calomel electrode was used as the reference electrode and a platinum electrode as the counter electrode) electrochemical cell containing 3 M KOH aqueous solution as the electrolyte. At first, in order to exclude the contribution of the substrate, the blank measurement of Ni foam was carried out and the CVs curve was shown in Fig. S4.† For comparison, CuCo2O4 nanowire arrays on Ni foam were also investigated by the same method. Fig. 4a shows the cyclic voltammetry (CV) performance of CuCo2O4 and CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell arrays grown on Ni foam with a scan rate of 5 mV s−1. The non-rectangular shape of the CV curve means that the capacitance is from the faradaic reactions of electrode materials.41 Apparently, a pair of redox peaks at 0.21 and 0.34 V are observed for the bare CuCo2O4 electrode, which are due to the Co3+/Co4+ and Cu2+/Cu+ transitions associated with anions OH−. A similar CV curve was observed for the CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell arrays. However, the CV curves of the hybrid electrodes significantly expanded compared to the bare CuCo2O4 electrode, indicating that the CuCo2O4@NiCo2O4 electrode has a larger capacitance.42 The phenomena can be attributed to the deposition of NiCo2O4 nanosheets which enhanced the mass loading and enlarged the surface area of the active materials.
Fig. 4b shows the charge/discharge curves of the CuCo2O4 electrode and CuCo2O4@NiCo2O4 hybrid electrode within the potential range from 0 to 0.42 V at the current density of 10 mA cm−2. The specific capacitance of CuCo2O4@NiCo2O4 hybrid electrode is calculated to be 2029 F g−1, which is higher than the pristine CuCo2O4 electrode (1512 F g−1). Fig. 4c shows the typical CV curves of the CuCo2O4@NiCo2O4 hybrid electrode within the potential range of 0–0.5 V at various scan rates. As the scan rate increases, the anodic peak potential shifts to a higher potential while the cathodic peak potential shifts to a lower potential. It may be resulted from the polarization effect of the electrode.43 Fig. 4d shows the charge/discharge curves of the CuCo2O4@NiCo2O4 hybrid electrode at different current densities ranging from 2 to 30 mA cm−2. According to the eqn (3), the specific capacitances of the hybrid electrode are calculated to be 2517, 2373, 2029, 1767 and 1551 F g−1 at the current densities of 2, 5, 10, 20 and 30 mA cm−2, respectively. Obviously, the specific capacitance decreases with the current density increasing. For comparison, the specific capacitances of pristine CuCo2O4 electrode under the same pretreatment were also tested. As shown in Fig. 5a, the specific capacitances of the pristine CuCo2O4 electrode are calculated to be 2150, 1794, 1512, 1158 and 923 F g−1at the current densities of 2, 5, 10, 20 and 30 mA cm−2, respectively. Evidently, the hybrid electrode shows a much higher specific capacitance compared to the pristine one. Furthermore, when the current density increases from 2 to 30 mA cm−2, the specific capacitance of the hybrid electrode still retains 1551 F g−1 (61.6% of the highest value at 2 mA cm−2), which is much higher than the pristine one (42.9% of the highest value), indicating much better rate performance of the hybrid structures.
The cycling stability is another important factor for the application of supercapacitors.44 The cycling performance of CuCo2O4 electrode and the CuCo2O4@NiCo2O4 hybrid electrode were tested after activation at the current density of 10 mA cm−2. The galvanostatic charge–discharge measurements were conducted in the potential range of 0–0.42 V with 3 M KOH as the electrolyte. As shown in Fig. 5b, the specific capacitance of the hybrid electrode retains 1548 F g−1 which is 76% of the original value after 4500 cycles, while the specific capacitance of the pristine CuCo2O4 electrode retains 1153 F g−1 (75.8% of the original value after the same cycles) at the same current density. That indicates the process of electrochemical deposition increases the specific capacitance while does not affect the cycling stability, which can be further proved by the SEM image, as shown in Fig. 5d, of CuCo2O4@NiCo2O4 hybrid electrode after 4500 cycles. The 3D core/shell nanostructure only changes slightly after 4500 cycles compared with the SEM images before 4500 cycles, almost same as the original one. The corresponding areal capacitances were calculated and shown in Fig. S5.†
To further explain the excellent electrochemical properties of CuCo2O4@NiCo2O4 hybrid electrode, EIS was carried out in a three electrode system. The corresponding impedance Nyquist plots of the pristine CuCo2O4 electrode and CuCo2O4@NiCo2O4 hybrid electrode are shown in Fig. 5c, and the inset shows the blowup image at high frequency. In the low frequency region, where the slope of the curve represents the Warburg impedance, the hybrid electrode shows a more ideal straight line, suggesting more efficient electrolyte and proton diffusion. This result can be attributed to the typical 3D core/shell nanostructure which enlarges the specific surface area and shortens the diffusion distance between the electrolyte and electrode. In the high frequency region, the intersection of the curve at real part Z′ indicates the bulk resistance of the electrochemical system, and the semicircle of the Nyquist diagram represents the faradaic reaction during the charge/discharge processes. From Fig. 5c, the CuCo2O4@NiCo2O4 hybrid electrode shows a low bulk resistance and charge-transfer resistance compared to the pristine CuCo2O4 electrode. The EIS data indicates that the electrodeposition of NiCo2O4 nanosheets improves the electrical conductivity of the hybrid electrode, resulting in enhanced electrochemical properties compared to the bare CuCo2O4.
Table 1 summaries some state-of-the-art results about the different materials for supercapacitors. As a matter of fact, our results are superior to other electrode system. Several contributing factors result in the high specific capacitance, good rate capability and excellent cycling stability of the CuCo2O4@NiCo2O4 core/shell arrays. First, the CuCo2O4 nanowires were directly grown on Ni foam with a robust adhesion. The obtained samples were used as the electrode without using polymer binders, which enhanced the utilization of electrode materials and improved the ion diffusion and electron transport. Second, the NiCo2O4 nanosheets synthesized via the electrodeposition process were well wrapped on the porous CuCo2O4 nanowires, which maintained the cycling stability of the core/shell structure during the charge/discharge process. Third, the porous CuCo2O4 nanowires served as the “core” owing to its outstanding electrical conductivity, while the ultrathin NiCo2O4 nanosheets served as the “shell” with a high theoretical capacity enabled by the multiple oxidation states for the redox reaction. Therefore, the CuCo2O4@NiCo2O4 core/shell structures were able to realize the outstanding supercapacitive performance.
| Materials | Structure | Capacitance | Reference |
|---|---|---|---|
| NiCo2O4@NiCo2O4 | Core/shell nanaoflake arrays | 2.20 F cm−2 (5 mA cm−2) | 36 |
| NiCoSx | Nanosheets | 1418 F g−1 (5 A g−1) | 20 |
| Co3O4 | Nanowires | 754 F g−1 (2 A g−1) | 25 |
| NiCo2O4@MnO2 | Core/shell nanowire arrays | 2.24 F cm−2 (2 mA cm−2) | 31 |
| CuCo2O4@NiCo2O4 | Core/shell | 2.59 F cm−2 (10 mA cm−2), 2029 F g−1 (7.8 A g−1) | This work |
4. Conclusions
In conclusion, we have developed an easy and effective strategy to prepare the unique hierarchical core/shell structure of CuCo2O4 NWs@NiCo2O4 NSs on Ni foam, which can be directly used as the electrode for supercapacitors. The as-prepared electrode shows good electrochemical performance. The capacitance is calculated to be 2029 F g−1 (2.59 F cm−2) when the current density is 10 mA cm−2. Furthermore, when the current density increases from 2 to 30 mA cm−2, the specific capacitance of the hybrid electrode retains 1551 F g−1 (61.6% of the highest value). Besides, the hybrid electrode still delivers a high specific capacitance of 1548 F g−1 (76% of the original value) after 4500 cycles with a current density of 10 mA cm−2. Its outstanding electrochemical performance takes advantages of the material system which have high theoretical capacitance and 3D core/shell nanostructures which are able to shorten the diffusion distance between the electrolyte and electrode, enlarge the specific surface area and offer more active materials. Therefore, the CuCo2O4 nanowire@NiCo2O4 nanosheet core/shell arrays may serve to be a promising candidate as electrode material for supercapacitors.Acknowledgements
We gratefully acknowledge financial support from the National Natural Science Foundation of China (Grant no. 11274107 and 61204109), the Program for New Century Excellent Talents in University (NCET-13-0185), and the Foundation for the authors of National Excellent Doctorial Dissertation of China (201318).References
- J. Li, X. Wang, Q. Huang, S. Gamboa and P. J. Sebastian, J. Power Sources, 2006, 158, 784–788 CrossRef CAS PubMed.
- D. Kalpana, N. G. Renganathan and S. Pitchumani, J. Power Sources, 2006, 157, 621–623 CrossRef CAS PubMed.
- C. Kim, Y. O. Choi, W. J. Lee and K. S. Yang, Electrochim. Acta, 2004, 50, 883–887 CrossRef CAS PubMed.
- K. H. An, W. S. Kim, Y. S. Park, H. J. Jeong, Y. C. Choi, J.-M. Moon, D. J. Bae, S. C. Lim and Y. H. Lee, American Institute of Physics, 2001, 590, 241–244 CAS.
- C. Liu, Z. Yu, D. Neff, A. Zhamu and B. Z. Jang, Nano Lett., 2010, 10, 4863–4868 CrossRef CAS PubMed.
- J. R. Miller, R. A. Outlaw and B. C. Holloway, Science, 2010, 329, 1637–1639 CrossRef CAS PubMed.
- Y. Wang, Z. Shi, Y. Huang, Y. Ma, C. Wang, M. Chen and Y. Chen, J. Phys. Chem. C, 2009, 113, 13103–13107 CAS.
- D. Yu and L. Dai, J. Phys. Chem. Lett., 2010, 1, 467–470 CrossRef CAS.
- T. Cottineau, M. Toupin, T. Delahaye, T. Brousse and D. Bélanger, Appl. Phys. A: Mater. Sci. Process., 2005, 82, 599–606 CrossRef.
- A. L. M. Reddy and S. Ramaprabhu, J. Phys. Chem. C, 2007, 111, 7727–7734 CAS.
- S. Vijayakumar, A. K. Ponnalagi, S. Nagamuthu and G. Muralidharan, Electrochim. Acta, 2013, 106, 500–505 CrossRef CAS PubMed.
- L. Cao, F. Xu, Y. Y. Liang and H. L. Li, Adv. Mater., 2004, 16, 1853–1857 CrossRef CAS PubMed.
- C. C. Hu, J. C. Chen and K. H. Chang, J. Power Sources, 2013, 221, 128–133 CrossRef CAS PubMed.
- U. M. Patil, K. V. Gurav, V. J. Fulari, C. D. Lokhande and O. S. Joo, J. Power Sources, 2009, 188, 338–342 CrossRef CAS PubMed.
- L. Zhang, H. B. Wu and X. W. Lou, Chem. Commun., 2012, 48, 6912–6914 RSC.
- W. Chen, C. Xia and H. N. Alshareef, ACS Nano, 2014, 8, 9531–9541 CrossRef CAS PubMed.
- B. Hu, X. Qin, A. M. Asiri, K. A. Alamry, A. O. Al-Youbi and X. Sun, Electrochim. Acta, 2013, 100, 24–28 CrossRef CAS PubMed.
- S. K. Meher and G. R. Rao, J. Phys. Chem. C, 2011, 115, 15646–15654 CAS.
- Y. Wang, Z. Zhong, Y. Chen, C. T. Ng and J. Lin, Nano Res., 2011, 4, 695–704 CrossRef CAS.
- Z.-S. Wu, W. Ren, L. Wen, L. Gao, J. Zhao, Z. Chen, G. Zhou, F. Li and H.-M. Cheng, ACS Nano, 2010, 4, 3187–3194 CrossRef CAS PubMed.
- X.-h. Xia, J.-p. Tu, Y.-q. Zhang, Y.-j. Mai, X.-l. Wang, C.-d. Gu and X.-b. Zhao, RSC Adv., 2012, 2, 1835–1841 RSC.
- G. Zhang, T. Wang, X. Yu, H. Zhang, H. Duan and B. Lu, Nano Energy, 2013, 2, 586–594 CrossRef CAS PubMed.
- F. Bao, Z. Zhang, X. Liu and X. Zhao, RSC Adv., 2014, 4, 38073 RSC.
- F. Bao, X. Wang, X. Zhao, Y. Wang, Y. Ji, H. Zhang and X. Liu, RSC Adv., 2014, 4, 2393–2397 RSC.
- H. Chen, J. Jiang, L. Zhang, T. Qi, D. Xia and H. Wan, J. Power Sources, 2014, 248, 28–36 CrossRef CAS PubMed.
- J. Du, G. Zhou, H. Zhang, C. Cheng, J. Ma, W. Wei, L. Chen and T. Wang, ACS Appl. Mater. Interfaces, 2013, 5, 7405–7409 CAS.
- Q. Wang, J. Xu, X. Wang, B. Liu, X. Hou, G. Yu, P. Wang, D. Chen and G. Shen, ChemElectroChem, 2014, 1, 559–564 CrossRef PubMed.
- S. G. Mohamed, C. J. Chen, C. K. Chen, S. F. Hu and R. S. Liu, ACS Appl. Mater. Interfaces, 2014, 6, 22701–22708 CAS.
- L.-B. Kong, C. Lu, M.-C. Liu, Y.-C. Luo, L. Kang, X. Li and F. C. Walsh, Electrochim. Acta, 2014, 115, 22–27 CrossRef CAS PubMed.
- L. Bao, J. Zang and X. Li, Nano Lett., 2011, 11, 1215–1220 CrossRef CAS PubMed.
- L. Huang, D. Chen, Y. Ding, Z. L. Wang, Z. Zeng and M. Liu, ACS Appl. Mater. Interfaces, 2013, 5, 11159–11162 CAS.
- G.-R. Li, Z.-L. Wang, F.-L. Zheng, Y.-N. Ou and Y.-X. Tong, J. Mater. Chem., 2011, 21, 4217 RSC.
- Q. Qu, Y. Zhu, X. Gao and Y. Wu, Adv. Energy Mater., 2012, 2, 950–955 CrossRef CAS PubMed.
- A. Vu, Y. Qian and A. Stein, Adv. Energy Mater., 2012, 2, 1056–1085 CrossRef CAS PubMed.
- Y. Qian, A. Vu, W. Smyrl and A. Stein, J. Electrochem. Soc., 2012, 159, A1135–A1140 CrossRef CAS PubMed.
- X. Liu, S. Shi, Q. Xiong, L. Li, Y. Zhang, H. Tang, C. Gu, X. Wang and J. Tu, ACS Appl. Mater. Interfaces, 2013, 5, 8790–8795 CAS.
- Y. Qian, I. M. Ismail and A. Stein, Carbon, 2014, 68, 221–231 CrossRef CAS PubMed.
- Z. Yu, Z. Cheng, S. R. Majid, Z. Tai, X. Wang and S. Dou, Chem. Commun., 2015, 51, 1689–1692 RSC.
- L. Q. Mai, F. Yang, Y. L. Zhao, X. Xu, L. Xu and Y. Z. Luo, Nat. Commun., 2011, 2, 381 CrossRef PubMed.
- M. Niederberger and H. Colfen, Phys. Chem. Chem. Phys., 2006, 8, 3271–3287 RSC.
- H. Wang, Q. Gao and L. Jiang, Small, 2011, 7, 2454–2459 CAS.
- J. W. Lee, A. S. Hall, J.-D. Kim and T. E. Mallouk, Chem. Mater., 2012, 24, 1158–1164 CrossRef CAS.
- J. W. Lee, T. Ahn, J. H. Kim, J. M. Ko and J.-D. Kim, Electrochim. Acta, 2011, 56, 4849–4857 CrossRef CAS PubMed.
- H. Wang, Y. Wang, Z. Hu and X. Wang, ACS Appl. Mater. Interfaces, 2012, 4, 6827–6834 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra11007f |
| This journal is © The Royal Society of Chemistry 2015 |




