Photoexcited state chirality transfer. Hidden tunability of circularly polarized luminescent binaphthyl–anthracene tandem molecular systems†
Sayaka Kitamuraa,
Kazuki Nakabayashia,
Takashi Wakabayashia,
Nobuo Tajimab,
Michiya Fujiki*c and
Yoshitane Imai*a
aDepartment of Applied Chemistry, Faculty of Science and Engineering, Kinki University, 3-4-1 Kowakae, Higashi-Osaka, Osaka 577-8502, Japan. E-mail: y-imai@apch.kindai.ac.jp
bComputational Materials Science Center, National Institute for Materials Science 1-2-1 Sengen, Tsukuba, Ibaraki 305-0047, Japan
cGraduate School of Materials Science, Nara Institute of Science and Technology, Takayama, Ikoma, Nara 630-0192, Japan. E-mail: fujikim@ms.naist.jp
First published on 24th July 2015
Abstract
Circularly polarized luminescent properties of C2-symmetrical binaphthyl molecular systems bearing two achiral anthracenes as luminophores were tuned by solely exploiting photoexcited-state chirality transfer from the binaphthyl to anthracenes when open- or closed-type binaphthyl structure as non-luminophore were chosen.
Introduction
Although many organic luminophores are utilized in organic electroluminescence, lasing and optoelectronic devices, chiral luminophores exhibiting circularly polarized luminescence (CPL) with a high quantum efficiency are rarely reported.1,2C2-symmetrical binaphthyl is one of the more prominent building blocks, facilitating the introduction of chiral motifs into achiral molecular units in the ground state.2 Additionally, the binaphthyl moiety itself, though a good luminogen, reveals CPL and photoluminescence (PL) signals in the UV region reflecting from photoexcited chirality.2 Previously, we reported that, in certain binaphthyl chromophores with rotational freedom of C–C single bond between the naphthyls, chiroptical signs in circular dichroism (CD) signals are determined solely by the dihedral angle between two binaphthyl units whilst the chromophores possess the same axial chirality.3 In this case, C2-chiral binaphthyl itself acts as a CPL luminophore. Chiroptical sign inversion between CD and CPL spectra was observed, depending on the degree of the rotational freedom.
This knowledge and understanding prompted us to investigate in more detail the ground- and photoexcited-state chiralities, revealed by chiroptical signs in the CPL and CD signals of binaphthyl chromophore directly connected to two achiral anthracene luminophores. A comparative study between the CD and CPL signals of tandem chiral luminophores, e.g., binaphthyl with multiple achiral luminophores, is rare to date. Actually, analysis of tandem chiral luminophores is especially difficult due to the presence of conformational freedom of C–C bonds and plural dihedral angles.2–5
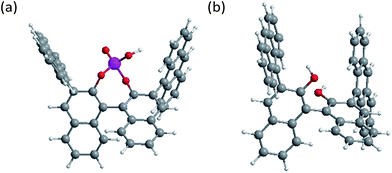
Herein, we demonstrate the hidden tunable CPL properties of binaphthyl-driven luminophores connected to the two achiral anthracenes, in which marked difference in photoexcited- and ground-state chiralities are reflected by CPL and CD signals and their signs. Two types of chiral luminophore systems were designed: closed-style and open-style. The former is 3,3′-bis(9-anthracenyl)-1,1-binaphthyl-2,2′-diyl-hydrogen phosphate (1, Fig. 1) and the latter is 3,3′-bis(9-anthracenyl)-1,1′-bis-2-naphthol (2, Fig. 1). Generally, we cannot fully predict their chiroptical properties because the chiral and achiral units are remotely positioned and these units have many rotatable C–C single bonds (Fig. 1).
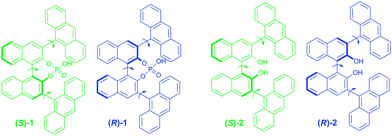 | ||
| Fig. 1 C2-symmetrical binaphthyl-anthracene luminophores, closed-type 1 and open-type 2 with three rotational C–C bonds. | ||
Results and discussion
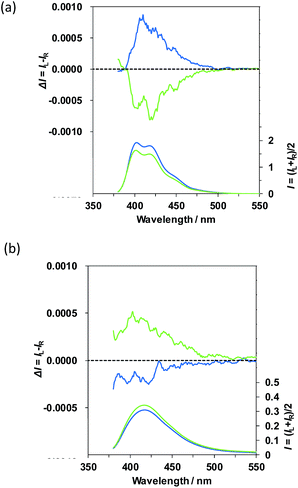
Firstly, the solution-state PL and CPL properties of (R)-1 and (R)-2 were compared to know their photoexcited chiralities in a homogeneous solution. The most probable PL phenomena of luminescent molecules containing two luminophores are the occurrence of intra-luminophore PL quenching, intra-energy transfer and excimer-like redshift emission.Evidently, (R)-1 and (R)-2 exhibited PL from the anthracenes at a concentration of 1.0 × 10−4 M in N,N-dimethylformamide (DMF), as indicated by the blue lines in the lower panels of Fig. 2a for 1 and Fig. 2b for 2. Although the PL maxima (λem) for (R)-1 and (R)-2 were almost similar, i.e., 420 nm of (R)-1 and 417 nm of (R)-2, the absolute PL quantum yields (ΦF), 0.59 for (R)-1 and only 0.01 for (R)-2, were markedly different from each other. This extremely low ΦF value of (R)-2 originates from significant ro-vibrational deactivation modes due to rotational C–C bond freedom characteristic of open-style binaphthyl in (R)-2. This is evident from a larger Stokes shift of ≈30 nm, as can be seen in Fig. 2 and 3.
As expected, (R)-1 and (R)-2 revealed CPL signals on the order of |gem| ≈ (0.4–1.2) × 10−3, where the dimensionless Kuhn's anisotropy in the photoexcited state (gem) is defined as: gem = 2(IL − IR)/(IL + IR), where IL and IR denote the quantum yields of the left-handed and right-handed CPL under excitation of unpolarized light, respectively. The CPL spectra of (R)-1 and (R)-2 in DMF are given as the blue lines of the upper panels in Fig. 2. Surprisingly, although (R)-1 and (R)-2 commonly possess the same axial chirality, their CPL signs are opposite; (R)-1 is positive whereas (R)-2 is negative. To verify whether these signs arise from unresolved artefacts, the CPL spectra of (S)-1 and (S)-2 in DMF were acquired in a similar manner (Fig. 2, green lines).
The CPL spectra of (R)-1 and (S)-1 adopt nearly mirror images of each other. The CPL spectra in absolute magnitude of (R)-2 and (S)-2 are, however, slightly different, due to unresolved reasons. The |gem| value was ≈4.3 × 10−4 at 415 nm for (R)-1 and (S)-1. The |gem| values of (R)-2 and (S)-2 are ≈0.7 × 10−3 (at 415 nm) and ≈1.2 × 10−3 (at 415 nm), respectively. These results led to an idea that the CPL sign, deriving from certain chirally reorganized geometry between two remote anthracene luminophores (chirality sergeants) under the 350 nm excitation, is controllable by the choice of open- or closed-style binaphthyl unit (chirality commander). The axial chirality of binaphthyl in the ground state is not the deterministic factor in controlling photoexcited chirality. The sergeants may not always obey a left-right hand from the commander. Obtaining a luminophore with the opposite chirality is often difficult. Thus, the choice of the open- or closed-style chiral-gen should offer a unique opportunity to control photoexcited chirality, proven by the sign inversion of CPL signals, without any use of the counterpart chirality. This idea may contribute to the design of more elaborate chiral molecular emitters.
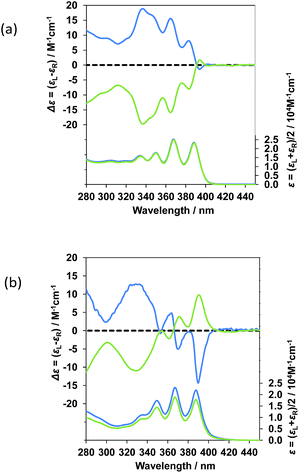
For comparison, the ground-state chirality of 1 and 2 was verified by means of CD and unpolarized UV spectra in dilute solution. CD and UV spectra of (R)-1 and (R)-2 in DMF were acquired as shown in Fig. 3 (indicated by blue lines).
(R)-1 and (R)-2 in DMF have several well-resolved CD bands in the range of 340 nm to 410 nm, arising from π–π* vibronic transitions (0–0, 0–1, 0–2, 0–3 and so on) of the two achiral anthracenes (Fig. 3). This suggests that the binaphthyl C2-chilarity efficiently transfers to the two achiral, but remote-positioned anthracene chromophores in the ground state. The CD band profiles between (S)-1 and (R)-1 displayed an ideal mirror image relationship. Similarly, the CD band profiles between (S)-2 and (R)-2 had nearly a mirror image relationship.
To quantitatively evaluate the absolute CD magnitude, we used the dimensionless Kuhn's anisotropy factor in the ground state, defined as gabs = Δε/ε. The |gabs| values of 1 and 2 were ≈1.1 × 10−4 at 394 nm (the first Cotton band) and ≈7.2 × 10−4 at 391 nm (the first Cotton band), respectively. The |gabs| value of 2 is enhanced by ≈7 times, relative to that of 1. These |gabs| values and their CD signs at the first Cotton band in the ground state, however, were not identical to the corresponding |gem| values and their CPL signs in the photoexcited state. The |gem| value of 1 is enhanced by ≈4 times compared to the corresponding |gabs| value at the first Cotton band. The |gem| value of (R)-2 is almost identical to the corresponding |gabs| value of (R)-2 at the first Cotton band, while the |gem| value of (S)-2 is twice relative to the |gabs| value of (S)-2 at the first Cotton band.
Actually, CPL signs of (R)-1 and (R)-2 in the photoexcited state are negative and positive, respectively, although their CD signs in the ground state are commonly negative. Evidently, these results demonstrated that the ground state chirality and photoexcited state chirality are not solely determined by the binaphthyl chirality. Whether the open-type with C–C rotational freedom or the close-type with greatly restricted C–C rotational freedom might be critical structural factors to control ground and photoexcited chiralities. These results will bring a conceptually novel tandem molecular chromophore–luminophore system, offering hidden ground-state chirality. In other word, C2-symmetrical binaphthyl moiety acts as non-luminescent chirality commander and two achiral luminescent anthracenes obey chirality sergeants, but do not always follow his/her hand in some case.
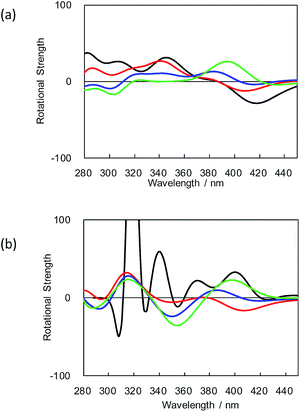
To theoretically clarify those differences in the CPL and CD characteristics of closed-style 1 and open-style 2, the CD spectra of (R)-2 were calculated as functions of two dihedral angles. Fig. 4 displays the calculated CD spectra of (R)-2. These spectra were simulated by varying the dihedral angle θ1 ((O)C–C–C–C(O)) of the binaphthyl unit and θ2 ((O)C–C–C–C) between anthracene and naphthalene of binaphthyl.
Evidently, the CD sign of (R)-2 completely inverted when θ1 altered.2c,3 From a comparison between Fig. 4a (θ1 = −60°) and Fig. 4b (θ1 = −120°), this significant alteration of (R)-2 might occur, depending on the ground- and photoexcited states. Moreover, these simulations indicate that the first Cotton CD sign of (R)-2 is changeable by angle θ2 between binaphthyl and anthracene (θ2 = 60° for black curve, θ2 = 80° for red curve, θ2 = 100° for blue curve, and θ2 = 120° for green curve), as given in Fig. 4a and b.
The theoretical equilibrium structures of 1 (ref. 2c) and 2 were shown in Fig. 5. (R)-1 had θ1 = −56° and θ2 = +98° and +102° (Fig. 5a), while (R)-2 had θ1 = −59° and θ2 = +77° and +94° (Fig. 5b). These equilibrium structures suggest that the first Cotton CD bands around ≈400 nm of (R)-1 and (R)-2 have commonly a negative sign.
Although chirally oriented conformations between anthracene and binaphthyl units of 1 and 2 in the ground state are very similar, their photoexcited-state chiralities might be very different, as proven by inversion in CPL signs whilst the same sign in the first Cotton CD bands is evident both experimentally and theoretically. These simulated CD spectra infer that sign inversion of CPL spectra between 1 and 2 originates from the considerable changes in θ1 and θ2 angles in the ground and photoexcited states.
At the moment, our theoretical explanation is qualitative and very speculative because no further consideration of electron–phonon couplings in the ground and photoexcited states. In a previous paper, we showed dihedral angle dependent sign inversion in CD spectrum and redshift/blueshift in UV spectra of hypothetical binapthyl moiety as a model of (R)-1.2c,3 For comparison, the current simulation tells us of how two dihedral angles θ1 and θ2 significantly alter their CD spectra and wavelength dependent signs of (R)-2.
Previously, chiral π-conjugated molecular assemblies showed chiroptical (CD and CPL) inversion, that is significantly dependent on the nature of good non-polar (chloroform) or poorer non-polar (dichloroethane) solvents in the dark condition.1c In this case, CD sign at the first Cotton band (a longer wavelength edge) is identical to CPL sign at the first Cotton band (a shorter wavelength edge). The chiroptical properties may have passive function.
Aiming at designing more elaborate active function, e.g., photon-induced chirality in the photoexcited state, followed by spontaneous relaxation process to restore the ground-state chirality,4–7 the choice of the opposite and/or same signs in the ground-state and photoexcited-state molecular chiralities can be exploited as advanced security systems for future global communication.4 As prototype molecules,2–5 the binaphthyl chirality transfer to achiral luminophores may offer unique enciphering and deciphering molecular coding and decoding system:4 one is the positive-sign CD and negative-sign CPL signals from luminophores with the same chirality, whilst the other is the negative-sign CD and negative-sign CPL signals of luminophores with the same chirality. The present knowledge and understanding of our chiral tandem molecules should stimulate to design various sophisticated organic molecular systems, enabling physically and chemically controlled active and passive CPL functions in future.2,7
Experimental
General methods
Compounds (R)-1, (S)-1, (R)-2 and (S)-2 were purchased from Sigma-Aldrich Japan (Tokyo, Japan). Chloroform (CHCl3) and N,N-dimethylformamide (DMF) used for crystallisation and optical measurements was purchased from Wako Pure Chemical Industries (Osaka, Japan).Measurement of the CPL and photoluminescence spectra
Photoluminescence (PL) spectra and absolute PL quantum yields in DMF were obtained with an absolute PL quantum yield measurement system (Hamamatsu Photonics C9920-02, Hamamatsu, Japan) under air atmosphere at room temperature. The DMF solutions of (R)-1 and (R)-2 (1.0 × 10−4 M, pathlength 10 mm) were excited at 350 nm.The CPL and PL spectra in DMF solution were measured using a JASCO CPL-200 spectrofluoropolarimeter (Tokyo, Japan) at room temperature. The instrument used a scattering angle of 0° under excitation of unpolarised, monochromated incident light. DMF solutions of 1.0 × 10−4 M were excited at 340 nm for 1 and 330 nm for 2. Bandwidths for excitation and emission for the high ΦF (R)-1 and (S)-1 were 10 nm/10 nm, respectively. However, to obtain good S/N ratio CPL spectra, bandwidths for the (R)-2 and (S)-2 should be 3000 μm/3000 μm, respectively, due to the extremely low ΦF value. Pathlength was commonly 2.0 mm to keep 63–70% transmittance under the 340 nm excitation for 1 and 330 nm excitation for 2. Scanning speed for CPL/PL measurement was 50 nm min−1 with time constant 8 s of PMT during acquisitions of 4–8 times.
Measurement of CD and UV-vis absorption spectra
The circular dichroism (CD) and UV-vis absorption spectra of (R)-1, (S)-1, (R)-2 and (S)-2 in DMF solution (1.0 × 10−4 M) were measured using a JASCO J-820 spectropolarimeter at room temperature. The pathlength is 2 mm.Theoretical calculations
The geometry of (R)-2 was optimised using the hybrid density functional theory (B3LYP functional).8 The excitation energies and rotational strengths of the geometry-optimised molecules were calculated using the time-dependent density functional theory (TD-DFT)9 with the B3LYP functional. In all the calculations, the cc-pVDZ basis set was used.10 The program Gaussian03 (ref. 11) was used for quantum chemical calculations.Conclusions
We compared CD and CPL spectra of open- and close-types of chiral binaphthyl moieties linked to achiral anthracene luminophores in DMF. The CPL sign due to anthracene luminophores was efficiently controlled by the choice of open- or closed-style units with the scenario of binaphthyl chirality transfer to the achiral luminophores.Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research (No. 15K05489) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan, a MEXT-Supported Program for the Strategic Research Foundation at Private Universities, 2014–2018, and the KDDI Foundation.Notes and references
- (a) J. Shinar, Organic Light-Emitting Devices, Springer, New York, Berlin, Heidelberg, 2004 Search PubMed; (b) K. Müllen and U. Scherf, Organic Light-Emitting Devices, Wiley-VCH, Weinheim, 2006 Search PubMed; (c) A. Satrijo, S. C. J. Meskers and T. M. Swager, J. Am. Chem. Soc., 2006, 128, 9030–9031 CrossRef CAS PubMed; (d) C. Jeanne and R. Regis, Dalton Trans., 2008, 6865–6876 Search PubMed; (e) S. Kappaun, C. Slugovc and J. M. Emil, Int. J. Mol. Sci., 2008, 9, 1527–1547 CrossRef CAS PubMed; (f) G. Lakhwani, R. A. J. Janssen and S. C. J. Meskers, J. Phys. Chem. A, 2009, 113, 10891–10894 CrossRef CAS PubMed; (g) K. Milan and O. Franc, Acc. Chem. Res., 2009, 42, 235–248 CrossRef PubMed; (h) W.-Y. Wong and C.-L. Ho, J. Mater. Chem., 2009, 19, 4457–4482 RSC; (i) M. Wolffs, N. Delsuc, D. Veldman, N. V. Anh, R. M. Williams, S. C. J. Meskers, R. A. J. Janssen, I. Huc and A. P. H. J. Schenning, J. Am. Chem. Soc., 2009, 131, 4819–4829 CrossRef CAS PubMed; (j) K.-L. Chan, P. Sonar and A. Sellinge, J. Mater. Chem., 2009, 19, 9103–9120 RSC; (k) F. Laquai, Y.-S. Park, J.-J. Kim and T. Basche, Macromol. Rapid Commun., 2009, 30, 1203–1231 CrossRef CAS PubMed; (l) C. Wang, J. Zhang, K. Ryu, A. Badmaev, L. G. D. Arco and C. Zhou, Nano Lett., 2009, 9, 4285–4291 CrossRef CAS PubMed; (m) M.-K. Wei, C.-W. Lin, C.-C. Yang, Y.-W. Kiang, J.-H. Lee and H.-Y. Lin, Int. J. Mol. Sci., 2010, 11, 1527–1545 CrossRef CAS PubMed; (n) G. Duran-Sampedro, A. R. Agarrabeitia, I. Garcia-Moreno, L. Gartzia-Rivero, S. de la Moya, J. Bañuelos, I. López-Arbeloa and M. J. Ortiz, Chem. Commun., 2015, 51, 11382–11385 RSC , and references cited therein.
- (a) T. Kawai, K. Kawamura, H. Tsumatori, M. Ishikawa, M. Naito, M. Fujiki and T. Nakashima, ChemPhysChem, 2007, 8, 1465–1468 CrossRef CAS PubMed; (b) H. Tsumatori, T. Nakashima and T. Kawai, Org. Lett., 2010, 12(10), 2362–2365 CrossRef CAS PubMed; (c) T. Amako, T. Kimoto, N. Tajima, M. Fujiki and Y. Imai, Tetrahedron, 2013, 69, 2753–2757 CrossRef CAS PubMed; (d) T. Amako, T. Kimoto, N. Tajima, M. Fujiki and Y. Imai, RSC Adv., 2013, 3, 6939–6944 RSC; (e) B. A. S. Jose, J. Yan and K. Akagi, Angew. Chem., Int. Ed., 2014, 53, 10641–10644 CrossRef PubMed; (f) E. M. Sánchez-Carnerero, F. Moreno, B. L. Maroto, A. R. Agarrabeitia, M. J. Ortiz, B. G. Vo, G. Muller and S. de la Moya, J. Am. Chem. Soc., 2014, 136, 3346–3349 CrossRef PubMed; (g) J. Kumar, T. Nakashima, H. Tsumatori and T. Kawai, J. Phys. Chem. Lett., 2014, 5, 316–321 CrossRef CAS; (h) Y. Kitayama, T. Amako, N. Suzuki, M. Fujiki and Y. Imai, Org. Biomol. Chem., 2014, 12, 4342–4346 RSC; (i) Y. Kitayama, K. Nakabayashi, T. Wakabayashi, N. Tajima, M. Fujiki and Y. Imai, RSC Adv., 2015, 5, 410–415 RSC; (j) T. Amako, K. Nakabayashi, A. Sudo, M. Fujiki and Y. Imai, Org. Biomol. Chem., 2015, 13, 2913–2917 RSC; (k) Y. Kono, K. Nakabayashi, S. Kitamura, R. Kuroda, M. Fujiki and Y. Imai, Tetrahedron, 2015, 71, 3985–3989 CrossRef CAS PubMed.
- (a) T. Kinuta, N. Tajima, M. Fujiki, M. Miyazawa and Y. Imai, Tetrahedron, 2012, 68, 4791–4796 CrossRef CAS PubMed; (b) T. Kimoto, N. Tajima, M. Fujiki and Y. Imai, Chem.–Asian J., 2012, 7, 2836–2841 CrossRef CAS PubMed.
- T. Amako, K. Nakabayashi, N. Suzuki, S. Guo, N. A. A. Rahim, T. Harada, M. Fujiki and Y. Imai, Chem. Commun., 2015, 51, 8237–8240 RSC.
- N. Taniguchi, K. Nakabayashi, T. Harada, N. Tajima, M. Shizuma, M. Fujiki and Y. Imai, Chem. Lett., 2015, 44, 598–600 CrossRef CAS.
- M. Fujiki, Y. Donguri, Y. Zhao, A. Nakao, N. Suzuki, K. Yoshida and W. Zhang, Polym. Chem., 2015, 6, 1627–1638 RSC.
- For a recent review, see., E. M. Sánchez-Carnerero, A. R. Agarrabeitia, F. Moreno, B. L. Maroto, G. Muller, M. J. Ortiz and S. de la Moya, Chem.–Eur. J., 2015 DOI:10.1002/chem. 201501178.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS PubMed.
- M. E. Casida, Recent Advances in Density Functional Methods, ed. D. P. Chong, World Scientific, Singapore, 1995, Vol 1 Search PubMed.
- (a) T. H. Dunning Jr., J. Chem. Phys., 1989, 90, 1007–1023 CrossRef PubMed; (b) D. E. Woon and T. H. Dunning Jr., J. Chem. Phys., 1993, 98, 1358–1371 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery Jr., T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez and J. A. Pople, Gaussian03, Rev.C.02, Gaussian, Inc., Wallingford, CT, 2004 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details of PL, CD, and CPL spectra and theoretical calculations. See DOI: 10.1039/c5ra10716d |
| This journal is © The Royal Society of Chemistry 2015 |