A stable Bi2S3 quantum dot–glass nanosystem: size tuneable photocatalytic hydrogen production under solar light†
Abstract
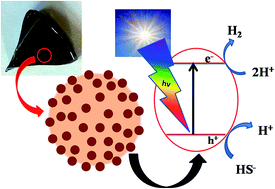
The present work comprises a novel approach to design a bismuth sulfide (Bi2S3) quantum dot (QD) glass nanocomposite system by confining nano-Bi2S3 in a designated glass composition for solar light driven hydrogen (H2) production. Numerous methods have been reported for the synthesis of Bi2S3, however, we have demonstrated the synthesis of Bi2S3 QDs (0.5–0.7%) in silicate glass using the melt and quench method. X-ray diffraction and electron diffraction patterns of the glass nanosystem exhibit an orthorhombic crystallite system of the Bi2S3 QDs. Transmission electron microscopy demonstrates that 3–5 and 7–10 nm size Bi2S3 QDs are distributed homogeneously in a monodispersed form in the glass domain and on the surface with a “partially embedded exposure” configuration. The role of glass on the control of the size and shape of Bi2S3 QDs and their effect on the photocatalytic hydrogen generation has been discussed. The maximum H2 production i.e. 6418.8 μmol h−1 g−1 was achieved for the Bi2S3–glass nanosystem under solar light irradiation. This glass nanosystem shows an excellent photostability against photocorrosion and also has a facile catalytic function. Therefore, even a very small amount of Bi2S3 QDs is able to photodecompose H2S and produce hydrogen under visible light. The salient features of this QD glass nanosystem are reusability after simple washing, enhanced stability and remarkable catalytic activity.

 Please wait while we load your content...
Please wait while we load your content...