Barberry fruit extract assisted in situ green synthesis of Cu nanoparticles supported on a reduced graphene oxide–Fe3O4 nanocomposite as a magnetically separable and reusable catalyst for the O-arylation of phenols with aryl halides under ligand-free conditions
Abstract
In situ synthesis of copper nanoparticles (NPs) supported on a reduced graphene oxide (RGO)–Fe3O4 nanocomposite was carried out with barberry fruit extract as a reducing and stabilizing agent. The morphology and structure of the Cu/RGO–Fe3O4 nanocomposite was fully characterized by means of X-ray diffraction (XRD), Fourier transformed infrared (FT-IR) spectroscopy, field emission scanning electron microscopy (FE-SEM), Raman, energy dispersive X-ray spectroscopy (EDS) and transmission electron microscopy (TEM). Cu/RGO–Fe3O4 was a promising catalyst for the O-arylation of phenols with aryl halides under ligand-free conditions. A diverse range of diaryl ethers were obtained in a good to high yield. Furthermore, due to the magnetic separability and high stability of the composite the catalyst could be separated conveniently from the reaction mixtures by an external permanent magnet and recycled multiple times without loss of catalytic activity.
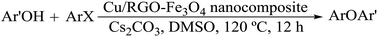

 Please wait while we load your content...
Please wait while we load your content...