Third order NLO properties of corannulene and its Li-doped dimers: effect of concave–convex and convex–convex structures†
Li Wang,
Wen-Yong Wang,
Xin-Yan Fang,
Chang-Li Zhu and
Yong-Qing Qiu*
Institute of Functional Material Chemistry, Faculty of Chemistry, Northeast Normal University, Changchun, Jilin 130024, People's Republic of China. E-mail: qiuyq466@nenu.edu.cn; Fax: +86 431 85098768
First published on 7th September 2015
Abstract
Buckybowls involving π–π interactions offer exciting future opportunities in terms of designing novel smart nonlinear optical (NLO) materials. Dimeric species of corannulene (C20H10), the smallest buckybowl, was considered as a model for the host–guest assemblies, which exhibited the convex–concave stacking of curved conjugated carbon surfaces. Different stacking motifs (concave–convex and convex–convex dimers) of C20H10 dimers that affect the NLO properties are the focus of our study. We performed density functional theory calculations on the structure, binding interactions, electronic absorption spectra and second hyperpolarizabilities of π-stacking dimers of C20H10 and its Li-doped derivative. It was found that the concave–convex dimers exhibit stronger binding interactions because of larger electrostatic interactions and thus are more stable with respect to the convex–convex dimers. The doping of Li ion significantly enhances the orbital interaction between the monomers but slightly affects the spectra and the second hyperpolarizabilities of the dimers. The convex–convex dimers exhibit larger polarizabilities and second hyperpolarizabilities (γzzzz) as a result of enhanced interlayer charge transfer properties. There is an increasing linear relationship between the electronic coupling and γzzzz values. The results presented in this article provide important evidence for the convex–convex stacking motif that enhances the NLO properties of the π-stacking dimers. Thus, controlling molecular stacking is an important way in terms of designing novel smart NLO materials.
1. Introduction
Buckybowls, including fullerenes and carbon nanotubes, have been the subject1,2 of intense experimental and theoretical studies in recent years because of their relation to the formation of intermolecular complexes.3–8 In addition to having a good shape match, buckybowls have large dipole moments due to their curvature that provides enhanced π–π intermolecular interactions,9–15 which are critical in determining crystal packing.16 The crystal packing and resulting charge transfer (CT) properties are found to be strongly connected. A remarkably large range of obvious CT properties can be achieved via crystal engineering of existing molecular backbones.17 The obvious CT properties can cause a large difference between the ground and excited state dipole moments and low-energy CT transitions,18,19 which thus causes the organic molecules to exhibit considerably large nonlinear optical (NLO) activities.20–24 Therefore, the π–π intermolecular interactions, significantly connecting with obvious CT properties, are found to strongly affect the NLO properties. In view of this, buckybowls involving π–π interactions offer exciting future opportunities for designing novel smart NLO materials.The smallest buckybowl corannulene (C20H10, Scheme 1) was first prepared by Lawton and Barth in 1966.25 Motivated by the novel bowl-shape of C20H10, studies of its surfaces have revealed a surface stereochemical principle that facilitates optimal packing motifs of molecules in the ball- and bowl-in-bowl complexation.26 The interesting stacking motifs are the concave–convex and convex–convex assemblies. The potential of the concave–convex stacking motif was recognized early and proposed as a major factor for the gas-phase formation of dimeric species of C20H10.27 The C20H10 dimer was considered as a model system for the host–guest assemblies, exhibiting the convex–concave stacking of curved conjugated carbon surfaces.28 Therefore, differently stacking the motifs of dimeric species of C20H10 is expected to be a method of effectively adjusting the NLO properties.
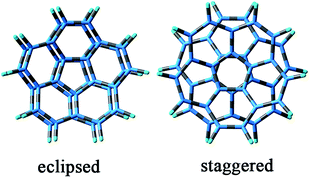
A lot of related studies have focused on the concave–convex eclipsed C20H10 dimer.29–34 Eclipsed and staggered dimers of C20H10 (Scheme 1) have been studied.29 Results have shown that eclipsed conformations are the most stable C20H10 dimers. High level ab initio calculations on the eclipsed concave–convex corannulene dimer yielded a minimum of 15.43 kcal mol−1 at 3.64 Å at the counterpoise corrected QCISD(T)/aug-cc-pVTZ level. For corannulene, the eclipsed concave–convex homodimers is the global minimum.30 In 2008,31 theoretical calculations studied by Andrzej Sygula and Svein Saebø showed that the binding energy of the native concave–convex corannulene dimer is quite substantial (17.2 kcal mol−1 at the best SCS-MP2/cc-pvtz level of theory) with an equilibrium distance of about 3.64 Å. In the same year, it was revealed that the eclipsed concave–convex corannulene dimer yielded a binding energy of 15.5 kcal mol−1 with a monomer–monomer distance of 3.69 Å at the (extrapolated) counterpoise corrected QCISD(T)/aug-cc-pVTZ level.32 In 2012, the benchmark curve shows a minimum of 15.43 kcal mol−1 at 3.64 Å. B3LYP-D3(BJ) exhibited smaller errors, suggesting that B3LYP-D3(BJ) is optimal.33
Based on these results, the eclipsed and staggered configurations of the concave–convex and convex–convex corannulene dimers have been investigated. However, no final stable staggered configuration of the convex–convex corannulene dimer was obtained. The stable eclipsed configuration of the concave–convex corannulene dimer was verified by optimizing the initial staggered configuration in our study. Therefore, the eclipsed stacking of C20H10 and its Li-doped dimers (Scheme 1) were investigated: the concave–convex (1a and 1b) and convex–convex stacking dimers (2a and 2b). Continuing our interests in the research of organic molecular materials with substantial NLO activities,35–38 different stacking motifs (concave–convex and convex–convex dimers) affecting interlayer CT properties and thus adjusting the NLO properties are the focus of the present study. We performed density functional theory (DFT) calculations on the structures, binding interactions, electronic absorption spectra and second hyperpolarizabilities of the four dimers. In particular, in this study, we mainly addressed the following issues: (1) predicting the stability of the concave–convex and convex–convex stacking dimers theoretically, (2) investigating the Li-doped effect on the second hyperpolarizabilities of the dimers, (3) exhibiting new structure-hyperpolarizability relationships and the dependence of the second hyperpolarizabilities on the interlayer CT property, and (4) understanding the nature of the dependence of second hyperpolarizabilities on the interlayer CT properties. We hope this study may evoke attention to design novel and highly efficient third-order NLO molecular materials with excellent building blocks (the convex–convex stacking motifs).
2. Computation details
The choice of a suitable method is crucial to generate reliable and accurate results. The hybrid meta-generalized gradient-approximations M06-2X functional with the double amount of nonlocal exchange (2X) is recommended by Truhlar and Zhao.39,40 This functional is excellent for the calculation of aromatic–aromatic stacking interactions. Therefore, M06-2X was selected to optimize the geometries of the studied dimers. We have adopted the 6-31+G(d,p) basis set for C and H with the exception of 6-311+G(3df) for the Li ion. The large basis set 6-311+G(3df) could more accurately describe the charge transfer and dispersion effects of the metal ion. On the other hand, as for the binding energy, using a large basis set 6-311+G(3df) for the Li cation gave a more accurate binding energy (34.94 kcal mol−1) than 6-31+G(d,p) for the Li cation (34.55 kcal mol−1). Because M06-2X underestimates the binding energies, the use of a large basis set for the Li cation could reduce the error. Each of the geometries is locally stable with all real frequencies.To correct the basis set superposition error (BSSE), the counterpoise (CP) procedure was used to calculate the interaction energy.41 The interaction energy (Eint) can be expressed as the difference between the energy of the dimer and the sum of the energies of the monomers according to the following equation:
| Eint(AB) = E(AB)AB − [E(A)AB + E(B)AB] | (1) |
A proper computational description of the relatively weak dispersion interactions is not trivial. Many people have used different methods to study the binding energy of concave–convex stacking dimers. The results show that the benchmark calculation of 15.5 kcal mol−1 was obtained at the QCISD(T)/aug-cc-pVTZ level.29 M06-2X underestimates when compared to the high level ab initio calculations at the counterpoise corrected QCISD(T)/aug-cc-pVTZ level.34 However, Grimme's B97-D appears to be a promising approach for the routine studies of π–π stacking interactions of large curved carbon networks.29 Therefore, to verify the above mentioned results, interaction energies were computed with the B97-D and M06-2X as well as with the 6-31+G** basis set.
Energy decomposition analysis (EDA) was performed using the energy decomposition scheme of the Amsterdam density functional (ADF) 2012.01 program42,43 at the M06-2X/TZ2P level of theory. We used ADF code for its ease of use and relatively exact treatment of decomposition energy, which offers the total binding energy with respect to the defined fragments. In EDA, the total interaction energy Eint between the interacting fragments is divided into three parts:44–46
| Eint = Eelstat + Epauli + Eorb | (2) |
It is well known that the NLO properties are induced by nonlinear charge displacements that are generated under a strong electric field of light. In the presence of weak and static electric field, the energy of a molecule is a function of the field strength. The energy of the perturbed system is described by expansion as follows:47,48
 | (3) |
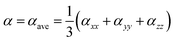 | (4) |
The orientationally averaged second hyperpolarizability (γ) has been calculated using the following expression:
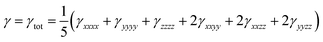 | (5) |
When it comes to the calculation of α and γ, three functionals (M06-2X, CAM-B3LYP and ωB97XD) have been chosen. The M06-2X functional was suited to calculate the γ values of such π-stacking dimers.50 The coulomb-attenuated hybrid exchange-correlation (CAM-B3LYP) functional is a hybrid functional with improved long-range properties.51 The ωB97XD functional is the long-range corrected hybrid density functional with damped atom–atom dispersion corrections, which appears most promising for calculations on π-dimer systems.52 Time-dependent DFT (TD-DFT) has nowadays become a general tool for understanding and predicting the behavior of the electron transition property.53,54 To obtain more insight on the description of the trend of third-order NLO responses, the vertical electronic transition energies between the ground and excited states were calculated using TD-M06-2X.
The transition density matrix (TDM), orbital overlap integral (Sij) and electronic coupling (Vij) were obtained by Multiwfn software Version 3.3.7.55 All the DFT and TD-DFT calculations in this study were carried out using the Gaussian 09W package.56 The reduced density gradient isosurface was plotted using VMD 1.9.1.74.57
3. Results and discussion
3.1 Geometrical structure
The equilibrium structures of the dimers were obtained by the M06-2X functional (Fig. 1). The distance between the center of the five-member ring in the C20H10 monomers of 1a is 3.63 Å (Table 1), which is very close to 3.64 Å, obtained at the QCISD(T)/aug-cc-pVTZ29 and SCS-MP2/cc-pvtz31 levels of theory by Andrzej Sygula and Svein Saebø; this suggests that the M06-2X functional is an efficient and reliable functional to study the dimers. The layer distance of 2a (3.32 Å) is smaller than that of 1a. Similar results are also found between 2b (3.84 Å) and 1b (3.88 Å). This indicates that the layer distance of convex–convex dimers are smaller as compared to that of the concave–convex dimers. However, the concave–convex dimers show much lower energies with respect to the convex–convex dimers, indicating that the concave–convex dimers are more stable.| Dimers | L | Erel | Eaint (B97-D) | Eaint (M06-2X) |
|---|---|---|---|---|
| a Obtained by the Gaussian program. | ||||
| 1a | 3.63 | 0.00 | −15.55 | −13.75 |
| 2a | 3.32 | 11.66 | −8.59 | −2.64 |
| 1b | 3.88 | 0.00 | −45.27 | −34.94 |
| 2b | 3.84 | 10.13 | −36.71 | −25.30 |
Furthermore, we calculated the binding energies (Table 1, Eaint) to understand the stability. It is revealed that M06-2X underestimates when compared to the high level ab initio calculations at the counterpoise corrected QCISD/aug-cc-pVTZ level (15.5 kcal mol−1). Unsurprisingly, the binding energies in corannulene dimer are best reproduced at the B97-D/6-31+G** level (15.5 kcal mol−1), coinciding exactly with that of the benchmark calculations of 15.5 kcal mol−1 at QCISD/aug-cc-pVTZ. The computed results showed that the Eaint (B97-D) values follow the trend: 1a (15.55 kcal mol−1) > 2a (8.59 kcal mol−1) and 1b (45.27 kcal mol−1) > 2b (36.71 kcal mol−1), indicating that the strong binding interactions are beneficial for the stability of the dimerization. Moreover, the interaction energies estimated by the B97-D and M06-2X calculations for the eclipsed concave–convex model of corannulene dimer with various basis sets are summarized in Table S1.† The results show that the better results of B97-D correspond to the smaller basis sets.
3.2 Nature of the interactions in the dimers
To provide further insight into the nature of the intermolecular interactions in these dimeric species, the EDA was performed to arrive at the relative contributions of various energy terms toward the total intermolecular energy. The absolute interaction energies Ebint (Table 2) follow the trend: 1a (13.95 kcal mol−1) > 2a (3.45 kcal mol−1) and 1b (41.62 kcal mol−1) > 2b (23.51 kcal mol−1). It is noted that the Ebint values are quite close to the Eaint (M06-2X) values from Table 1, and their predicted stability sequences are also fully identical (Fig. 2). The slightly different values between Eaint and Ebint may be due to the different basis sets. We also note that all the dimers have negative Eelstst and Eorb values, indicating that the electrostatic and orbital interactions play important role roles in the stability of these cases.The significant contribution of the dispersion interactions term, Eelstst, reveals that the concave–convex dimers (1a and 1b) possess larger Eelstst values when compared to the convex–convex dimers (2a and 2b), which is in line with the Ebint. The difference of Eelstst between concave–convex and convex–convex dimers is large enough (8.2 kcal mol−1 and 11.11 kcal mol−1, respectively) to distinguish them. However, the differences of Eorb and Epauli between concave–convex and convex–convex dimers are considerably small. Therefore, among all the interaction energies, electrostatic interaction, which arises in the molecular crystals of aromatic molecules, plays a pivotal role in determining the energetically accessible stacking motifs. In addition, a linear relationship was founded between Eorb and Eint (Fig. 2), suggesting that the Eorb value makes a major contribution to Ebint. In addition, the Li-doped dimers (1b and 2b) possess relatively larger Ebint values because of the enhancement of Eorb compared with 1a and 2a, suggesting that the doping of Li ion significantly enhances the orbital interaction between the monomers.
To distinguish three different types of noncovalent interactions (i.e., hydrogen-binding, van der Waals interaction and steric hindrance), low-gradient isosurfaces (s = 0.5 a.u.) for the dimers (Fig. 3) were plotted. It is an approach to detect noncovalent interaction in real space, based on the electron density and reduced gradient proposed by Yang et al.58 The surfaces are colored on a red-green-blue scale according to values of sin(λ)ρ, ranging from −0.04 to 0.02 a.u. Blue indicates strong attractive interactions (like hydrogen binding), the transition region (green) indicates typical van der Waals interaction, and red indicates strong steric hindrance.
The interaction region of 1a, marked by large green circle, can be identified as a van der Waals interaction region because the filled-color is green or light brown, meaning that the electron density in this region is low. In the case of 2a, a small green circle is observed between the central five-member rings of the C20H10 monomers. In fact, the weak interaction region in 1a is with more green and light-brown area than those of 2a. Similar comparative results can be observed in 1b and 2b. It can be easily concluded that the π–π interactions between the monomers of the concave–convex dimers (1a and 1b) are stronger with reference to that of the convex–convex dimers (2a and 2b), which is well in agreement with the larger Eint in the concave–convex dimers.
3.3 Electronic absorption spectrum
To obtain a more intuitive description of the band assignments of the electronic absorption spectra and the trends in the NLO behaviors of the studied dimers, TD-DFT calculations were carried out at the M06-2X functional. The excited-state transition energies (eV), oscillator strengths (f), and relevant molecular orbitals of all the complexes in singlet excited-state transitions are summarized in Table S2.† To rationalize the observed spectral properties, the differences in the absorption spectra between the dimers are presented (Fig. 4).The absorption spectrum of 1a is characterized by two broad absorptions (low-energy and high-energy) in the ultraviolet region. Compared with the absorption spectrum of 1a, an additional small peak of the low-energy absorption band appears in the spectrum of 2a at 299.4 nm. Moreover, the relatively lower energy absorption peak of 2a at 253.4 nm with higher oscillator strength was 1 nm red-shifted in relation to the corresponding spectrum of 1a. This indicates that dimer 2a with smaller transition energies is more vulnerable to undergo the electron transition; thus, it exhibits a relatively larger second hyperpolarizability. For Li-doped dimers, the simulated absorption spectrum of 2b is qualitatively quite similar to that of 1b, i.e., it exhibits one high-energy electronic transition absorbing at 221.6 nm (215.2 nm for 1b) along with an intense low-energy electronic transition of higher intensity absorbing at 254.1 nm (266.6 nm for 1b). The low-energy band of 2b is red-shifted with respect to the corresponding electronic transition in the spectrum of 1b, suggesting that 2b may exhibit a relatively large second hyperpolarizability. Note that 1b and 2b show the similar low- and high-energy absorptions as compared to those of 1a and 2a (with exception of different intensity), respectively, indicating that the small amount of Li-doping had an effect on the spectra of the dimers.
3.4 The static polarizability and second hyperpolarizability
For the studied dimers, the first hyperpolarizability (β) vanishes because of their centrosymmetry; thus, the polarizability (α) and the second hyperpolarizability (γ) will be discussed. Tensor components of the γ value were also investigated to understand the fundamental component. Because no experimental or theoretical results of the γ values are currently available for the studied dimers, a comparative discussion of the results (Table 3) obtained by different DFT methods is useful. It can be noted that the α and γ values of M06-2X, ωB97XD and CAM-B3LYP functionals vary within a close margin. To discuss these results in more detail, we use the data of the M06-2X functional to evaluate the trend of the α and γ values.| Dimers | Functionals | 〈R2〉 | α | γ | γxxxx | γyyyy | γzzzz |
|---|---|---|---|---|---|---|---|
| 1a | M06-2X | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 231 231 |
408.4 | 101.4 | 117.5 | 117.6 | 88.1 |
| ωB97XD | — | 407.4 | 118.8 | 121.9 | 122.0 | 149.5 | |
| CAM-B3LYP | — | 408.3 | 110.0 | 127.8 | 127.8 | 89.5 | |
| 2a | M06-2X | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 021 021 |
429.3 | 120.4 | 117.5 | 117.4 | 183.8 |
| ωB97XD | — | 428.1 | 125.4 | 122.5 | 122.5 | 180.5 | |
| CAM-B3LYP | — | 429.2 | 129.1 | 126.9 | 126.9 | 184.8 | |
| 1b | M06-2X | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 170 170 |
404.8 | 77.7 | 89.2 | 89.2 | 69.1 |
| ωB97XD | — | 403.5 | 73.5 | 90.4 | 90.4 | 44.9 | |
| CAM-B3LYP | — | 404.2 | 79.6 | 93.9 | 93.8 | 65.8 | |
| 2b | M06-2X | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 183 183 |
430.4 | 109.8 | 96.3 | 95.8 | 207.3 |
| ωB97XD | — | 429.1 | 97.3 | 98.5 | 98.1 | 134.0 | |
| CAM-B3LYP | — | 431.0 | 106.1 | 101.7 | 101.3 | 164.2 |
The α values (Table 3) of 2a and 2b were estimated to be 429.3 a.u. and 430.4 a.u., which are considerably higher compared to that of 1a and 1b (408.4 a.u. and 404.8 a.u.). This suggests that the convex–convex dimers possess larger α values compared with the concave–convex ones. To provide an original understanding of the α values, we focused on the relative electronic spatial extent 〈R2〉. The 〈R2〉 values in series decrease as follows: 2b (1.4 × 104 a.u.) > 2a (1.2 × 104 a.u.) > 1a (1.0 × 104 a.u.) ≈ 1b (1.0 × 104 a.u.), which is in good accordance with the decreasing order of the α values (Fig. 5). To the best of our knowledge, the 〈R2〉 is a physical property that characterizes the electron density volume around the molecule.59 The more diffuse electron cloud may lead to a larger 〈R2〉 value, resulting in a larger α value.
Moving to the γ values of the dimers, as expected, the convex–convex dimers (2a and 2b) exhibit larger γ values with respect to the concave–convex dimers (1a and 1b) because of smaller transition energies, and their γ values follow the trend: 2a (1.2 × 105 a.u.) > 1a (1.0 × 105 a.u.) and 2b (1.1 × 105 a.u.) > 1b (7.8 × 104 a.u.). The results also reveal that doping with Li ion does not significantly influence the γ values. This is consistent with the information obtained when calculating absorption spectra, namely, the effect of small Li doping on the spectra of the dimers. Considering the tensor components for 1a and 1b, the γxxxx and γyyyy values are the dominating components; whereas, the highest static γ values of 2a and 2b were found for the γzzzz component. This shows that the γzzzz values for the convex–convex dimers (2a and 2b) are larger (of 95.7 a.u. and 138.2 a.u., respectively) with respect to that of concave–convex dimers (1a and 1b). Also note that γzzzz values follow a similar trend to γ values. It is easy to reach the conclusion that the remarkably large γ values of the convex–convex dimers are due to the strong enhancement of the γzzzz longitudinal components. The results indicate that the CT transition along the z-axis between two C20H10 monomers partly takes place, which can be reasonably explained by the TDM as described below.
The molecular second-hyperpolarizability (γ), which evaluates the third-order NLO efficiency, can be predicted from the two-level model60 that links the γ value and the low-lying CT character γ2L as follows:
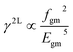 | (6) |
| Dimers | State | λgm | Egm | fgm | γ2L | MO transitions |
|---|---|---|---|---|---|---|
| 1a | 23 | 252.4 | 4.91 | 0.3951 | 0.5470 | H-7 → L (29%), H-6 → L + 1 (29%) |
| 2a | 16 | 253.4 | 4.89 | 0.5323 | 1.0134 | H-5 → L + 1 (38%), H-4 → L (38%) |
| 1b | 19 | 254.1 | 4.88 | 0.6421 | 1.4897 | H-1 → L + 2 (24%), H → L + 3 (22%), H-5 → L + 1 (14%), H-4 → L (12%) |
| 2b | 15 | 266.6 | 4.65 | 0.6503 | 1.9452 | H-5 → L (46%), H-4 → L + 1 (46%) |
To further understand the origin of the transition property, we focused on electron–hole coherence investigated by the transition density matrix (TDM) and the molecular orbitals of the crucial transition states of the four dimers (Fig. S1–S4†). TDM is a helpful tool for analyzing electronic excitation processes and providing additional information on electron–hole coherence of specific excitation.61 The hydrogen atoms have been omitted because they usually have little contribution to the transitions in the TDM.
Inspection of 1a (Fig. 6) reveals that the electron–hole pairs of the 23rd excited state are mainly localized along the diagonal element and are slightly localized along the off-diagonal element. These electronic transitions could be assigned as CT character within the two respective monomers along with less interlayer CT between the two monomers. However, for the 16th excited state of 2a, an increased degree of electron–hole coherence is delocalized along the off-diagonal element, indicating the enhanced interlayer CT between the two monomers. The enhanced interlayer CT in 2a brings about much stronger polarization of C20H10 monomers with respect to 1a. The polarization leads to a strong enhancement of the γzzzz value of 2a compared to 1a. The same result can be observed in the plot of TDM of dimers 1b and 2b. Apparently, the enhanced interlayer CT characters are observed in the convex–convex dimers (2a and 2b), resulting in larger γzzzz values.
 | ||
| Fig. 6 The TDM corresponding to crucial electronic transitions of dimers 1a and 2a (upper layer of the dimer is named as Frag 1, while the lower layer is Frag 2). | ||
In the abovementioned dimers, the low-energy peaks can be assigned to the mixture of intralayer CT within the respective monomers and interlayer CT between the two monomers. In fact, the obvious interlayer CT of the convex–convex dimer is partly due to the large overlap between the orbitals of the two monomers. The orbital overlap integral (Sij) between two monomers is given as follows:62,63
| SIntmoli,j = ∫φMonomer1i(r)φMonomer2j(r)dr, | (7) |
| Vij = CSij(C = −0.716 ± 0.002) | (8) |
This integral is useful in discussions of intermolecular CT.
The absolute Vij order of the HOMOs (Table S3†) of the dimers decreases in the order: 2b (0.975 a.u.) > 2a (0.080 a.u.) > 1a (0.009 a.u.) > 1b (0.003 a.u.). Interestingly, there was an increasing linear relationship between electronic coupling (Vij) and γzzzz values (Fig. 7). This is due to the fact that the interlayer orbital overlap provides the pathway for the redistribution of electronic charges, thus enhancing the interlayer CT characteristics. Thus, large γzzzz values rely on the enhancement of electronic coupling (Vij) between HOMOs of the two monomers of the convex–convex dimers (2a and 2b).
 | ||
| Fig. 7 Relationship between the γzzzz values and the corresponding |Vij| values for the four dimers. | ||
3.5 The frequency-dependent second hyperpolarizability
The frequency-dependent second polarizabilities relevant to the photoelectric Kerr's effect, γEOKE (−ω; ω, 0, −0), and the second harmonic generation, γSHG (−2ω; ω, ω, 0), were performed at the M06-2X/6-31+G(d, p) (6-311+G(3df) basis set for the Li ion) level of theory. Generally, γSHG is measured with a fundamental incident wavelength that has a second harmonic considerably far from the absorption bands to avoid the over measurement of γ values due to resonance effects. Thus, according to the maximum absorption of 2a at about 299.4 nm, we investigated the frequency dispersion at a wavelength of 1.340 mm (ω = 0.0340 a.u.) and 1.907 mm (ω = 0.0239 a.u.). There was a tiny increase in the frequency dependent γ values (Table 5). For example, frequency dependent γEOKE and γSHG values of 1a increased by a factor of ∼1.03 and ∼1.07, respectively, when the frequency of the incident field increased from 0.0239 a.u. to 0.0340 a.u. Similar results can be observed for γEOKE and γSHG values of all dimers, implying a slight influence of frequency on the γEOKE and γSHG values.| Dimers | γEOKE (−ω; ω, 0, −0) | γSHG (−2ω; ω, ω, 0) | ||
|---|---|---|---|---|
| 1907 nm | 1340 nm | 1907 nm | 1340 nm | |
| 1a | 99.5 | 102.0 | 104.1 | 111.9 |
| 2a | 121.6 | 125.6 | 128.9 | 143.4 |
| 1b | 75.9 | 77.8 | 79.4 | 84.8 |
| 2b | 105.5 | 109.3 | 112.3 | 124.5 |
4. Conclusion
In this study, the binding interactions of the dimers have been systematically investigated using the energy decomposition scheme. It was revealed that the concave–convex dimers exhibit stronger binding interactions and thus are more stable as compared to convex–convex dimers. Among all interaction energies, electrostatic interaction plays a pivotal role in determining the energetically accessible stacking motifs. In addition, doping with Li ion significantly enhances the orbital interaction between the monomers, but has slight effects on the spectra and the second hyperpolarizabilities of the dimers. Importantly, the convex–convex dimers exhibit more obvious interlayer charge transfer, leading to larger second hyperpolarizabilities (γzzzz) as compared to the concave–convex dimers. This provides important evidence for the convex–convex stacking way for enhancing the NLO properties of the π-dimers. Thus, the convex–convex stacking dimers may be designed as novel smart NLO materials and a new structure–property correlation can be obtained for optimizing the second hyperpolarizability. Interestingly, due to the obvious interlayer charge transfer properties that result from a large overlap and electronic coupling between HOMOs of the monomers, there is an increasing linear relationship between electronic coupling and γzzzz values. The result, concerning the estimate of the contribution of the electronic coupling and orbital overlap, will be helpful in the qualitative application of such convex–convex stacking models as part of excellent NLO materials.Acknowledgements
The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China (No. 21173035).References
- M. Yamada, K. Ohkubo, M. Shionoya and S. Fukuzumi, J. Am. Chem. Soc., 2014, 136, 13240–13248 CrossRef CAS PubMed.
- G. Casella and G. Saielli, New J. Chem., 2011, 35, 1453–1459 RSC.
- J. Kang, D. Miyajima, Y. Itoh, T. Mori, H. Tanaka, M. Yamauchi, Y. Inoue, S. Harada and T. Aida, J. Am. Chem. Soc., 2014, 136, 10640–10644 CrossRef CAS PubMed.
- L. Zoppi, L. Martin-Samos and K. K. Baldridge, J. Am. Chem. Soc., 2011, 133, 14002–14009 CrossRef CAS PubMed.
- Q. Stöckl, D. Bandera, C. S. Kaplan, K. H. Ernst and J. S. Siegel, J. Am. Chem. Soc., 2014, 136, 606–609 CrossRef PubMed.
- T. Bauert, L. Zoppi, G. Koller, A. Garcia, K. K. Baldridge and K. H. Ernst, J. Phys. Chem. Lett., 2011, 2, 2805–2809 CrossRef CAS.
- T. Bauert, L. Zoppi, G. Koller, J. S. Siegel, K. K. Baldridge and K. H. Ernst, J. Am. Chem. Soc., 2013, 135, 12857–12860 CrossRef CAS PubMed.
- Q. Zhang, K. Kawasumi, Y. Segawa, K. Itami and L. T. Scott, J. Am. Chem. Soc., 2012, 134, 15664–15667 CrossRef CAS PubMed.
- L. Zoppi, L. M. S. Layla and K. K. Baldridge, Acc. Chem. Res., 2014, 47, 3310–3320 CrossRef CAS PubMed.
- W. Xiao, D. Passerone, P. Ruffieux, K. Aït. Mansour, O. Gröning, E. Tosatti, J. S. Siegel and R. Fasel, J. Am. Chem. Soc., 2008, 130, 4767–4771 CrossRef CAS PubMed.
- R. L. Parc, P. Hermet, S. Rols, D. Maurin, L. Alvarez, A. Ivanov, J. M. Quimby, C. G. Hanley, L. T. Scott and J. L. Bantignies, J. Phys. Chem. C, 2012, 116, 25089–25096 Search PubMed.
- A. Karton, B. Chan, K. Raghavachari and L. Radom, J. Phys. Chem. A, 2013, 117, 1834–1842 CrossRef CAS PubMed.
- T. F. Headen, C. A. Howard, N. T. Skipper, M. A. Wilkinson, D. T. Bowron and A. K. Soper, J. Am. Chem. Soc., 2010, 132, 5735–5742 CrossRef CAS PubMed.
- S. M. Mathew, J. T. Engle, C. J. Ziegler and C. S. Hartley, J. Am. Chem. Soc., 2013, 135, 6714–6722 CrossRef CAS PubMed.
- E. R. Frey, A. Sygula and N. I. Hammer, J. Chem. Educ., 2014, 91, 2186–2190 CrossRef CAS.
- M. C. R. Delgado, E. G. Kim, D. A. d. S. Filho and J. L. Bredas, J. Am. Chem. Soc., 2010, 132, 3375–3387 CrossRef PubMed.
- S. Sanyal, A. K. Manna and S. K. Pati, ChemPhysChem, 2014, 15, 885–893 CrossRef CAS PubMed.
- W. Y. Wang, N. N. Ma, C. H. Wang, M. Y. Zhang, S. L. Sun and Y. Q. Qiu, J. Mol. Graphics Modell., 2014, 48, 28–35 CrossRef CAS PubMed.
- H. L. Xu, Z. R. Li, D. Wu, B. Q. Wang, Y. Li, F. L. Gu and Y. Aoki, J. Am. Chem. Soc., 2007, 129, 2967–2970 CrossRef CAS PubMed.
- W. Y. Wang, N. N. Ma, S. L. Sun and Y. Q. Qiu, Organometallics, 2014, 33, 3341–3352 CrossRef CAS.
- C. H. Wang, N. N. Ma, X. X. Sun, S. L. Sun, Y. Q. Qiu and P. J. Liu, J. Phys. Chem. A, 2012, 116, 10496–10506 CrossRef CAS PubMed.
- M. Y. Zhang, C. H. Wang, W. Y. Wang, N. N. Ma, S. L. Sun and Y. Q. Qiu, J. Phys. Chem. A, 2013, 117, 12497–12510 CrossRef CAS PubMed.
- W. Y. Wang, N. N. Ma, S. L. Sun and Y. Q. Qiu, Phys. Chem. Chem. Phys., 2014, 16, 4900–4910 RSC.
- N. Ma, L. Yan, W. Guan, Y. Qiu and Z. Su, Phys. Chem. Chem. Phys., 2012, 14, 5605–5612 RSC.
- W. E. Barth and R. G. Lawton, J. Am. Chem. Soc., 1966, 88, 380–381 CrossRef CAS.
- T. Bauert, K. K. Baldridge, J. S. Siegel and K. H. Ernst, Chem. Commun., 2011, 47, 7995–7997 RSC.
- H. Becker, G. Javahery, S. Petrie, P. C. Cheng, H. Schwarz, L. T. Scott and D. K. Bohme, J. Am. Chem. Soc., 1993, 115, 11636–11637 CrossRef CAS.
- M. L. Christian, G. Stefan, K. Lesya and S. Andrzej, Phys. Chem. Chem. Phys., 2010, 12, 7091–7097 RSC.
- T. Janowski, P. Pulay, A. A. Sasith Karunarathna, A. Sygula and S. Saebø, Chem. Phys. Lett., 2011, 512, 155–160 CrossRef CAS PubMed.
- P. A. Denis, Chem. Phys. Lett., 2011, 516, 82–87 CrossRef CAS PubMed.
- A. Sygula and S. Saebø, Int. J. Quantum Chem., 2009, 109, 65–72 CrossRef CAS PubMed.
- D. Josa, J. Rodriguez Otero and E. M. Cabaleiro Lago, Phys. Chem. Chem. Phys., 2011, 13, 21139–21145 RSC.
- M. R. Kennedy, L. A. Burns and C. D. Sherrill, J. Phys. Chem. A, 2012, 116, 11920–11926 CrossRef CAS PubMed.
- D. Josa, J. Rodriguez Oteroand, E. M. Cabaleiro Lago and M. Rellan Pineiro, Chem. Phys. Lett., 2013, 557, 170–175 CrossRef CAS PubMed.
- C. G. Liu, Y. Q. Qiu, Z. M. Su, G. C. Yang and S. L. Sun, J. Phys. Chem. C, 2008, 112, 7021–7028 CAS.
- N. N. Ma, S. L. Sun, C. G. Liu, X. X. Sun and Y. Q. Qiu, J. Phys. Chem. A, 2011, 115, 13564–13572 CrossRef CAS PubMed.
- N. N. Ma, S. J. Li, L. K. Yan, Y. Q. Qiu and Z. M. Su, Dalton Trans., 2014, 43, 5069–5075 RSC.
- L. Wang, W. Y. Wang, N. N. Ma, D. M. Tian, J. Wang and Y. Q. Qiu, J. Mol. Graphics Modell., 2015, 55, 33–40 CrossRef CAS PubMed.
- Y. Zhao and D. Truhlar, Theor. Chem. Acc., 2008, 120, 215–241 CrossRef CAS.
- Y. Zhao and D. G. Truhlar, Acc. Chem. Res., 2008, 41, 157–167 CrossRef CAS PubMed.
- L. M. da Costa, S. R. Stoyanov, S. Gusarov, X. Tan, M. R. Gray, J. M. Stryker, R. Tykwinski, J. W. de M. Carneiro, P. R. Seidl and A. Kovalenko, Energy Fuels, 2012, 26, 2727–2735 CrossRef CAS.
- ADF 2012.01 SCM, Theoretical Chemistry, Vrije Universiteit, Amsterdam, http://www.scm.com Search PubMed.
- C. Fonseca Guerra, J. G. Snijders, G. te Velde and E. J. Baerends, Theor. Chem. Acc., 1998, 99, 391–403 Search PubMed.
- M. Contreras, E. Osorio, F. Ferraro, G. Puga, K. J. Donald, J. G. Harrison, G. Merino and W. Tiznado, Chem.–Eur. J., 2013, 19, 2305–2310 CrossRef CAS PubMed.
- G. Gayatri, Y. Soujanya, I. Fernández, G. Frenking and G. N. Sastry, J. Phys. Chem. A, 2008, 112, 12919–12924 CrossRef CAS PubMed.
- A. Diefenbach, F. M. Bickelhaupt and G. Frenking, J. Am. Chem. Soc., 2000, 122, 6449–6458 CrossRef CAS.
- Y. Q. Qiu, H. L. Fan, S. L. Sun, C. G. Liu and Z. M. Su, J. Phys. Chem. A, 2008, 112, 83–88 CrossRef CAS PubMed.
- W. Y. Wang, X. F. Du, N. N. Ma, S. L. Sun and Y. Q. Qiu, J. Mol. Model., 2013, 19, 1779–1787 CrossRef CAS PubMed.
- J. Tong, Y. Li, D. Wu, Z. R. Li and X. R. Huang, J. Phys. Chem. A, 2010, 114, 5888–5893 CrossRef CAS PubMed.
- A. Alparone, Chem. Phys. Lett., 2011, 514, 21–25 CrossRef CAS PubMed.
- A. Pedone, J. Chem. Theory Comput., 2013, 9, 4087–4096 CrossRef CAS.
- U. Salzner and A. Aydin, J. Chem. Theory Comput., 2011, 7, 2568–2583 CrossRef CAS.
- D. Casanova, F. P. Rotzinger and M. Grätzel, J. Chem. Theory Comput., 2010, 6, 1219–1227 CrossRef CAS.
- S. Meng, E. Kaxiras, M. K. Nazeeruddin and M. Grätzel, J. Phys. Chem. C, 2011, 115, 9276–9282 CAS.
- T. Lu and F. W. Chen, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, et al., Gaussian 09, Revision D.01, Gaussian, Inc., Wallingford, CT, 2009 Search PubMed.
- W. Humphrey, A. Dalke and K. Schulten, VMD: visual molecular dynamics, J. Mol. Graphics, 1996, 14, 33–38 CrossRef CAS.
- E. R. Johnson, S. Keinan, M. S. Paula, C. G. Julia, A. J. Cohen and W. Yang, J. Am. Chem. Soc., 2010, 132, 6498–6506 CrossRef CAS PubMed.
- D. Scuderi, A. Paladini, M. Satta, D. Catone, S. Piccirillo, M. Speranza and A. G. Guidoni, Phys. Chem. Chem. Phys., 2002, 4, 4999–5003 RSC.
- K. Hatua and P. K. Nandi, J. Phys. Chem. A, 2013, 117, 12581–12589 CrossRef CAS PubMed.
- W. Y. Wang, Y. H. Kan, L. Wang, S. L. Sun and Y. Q. Qiu, J. Phys. Chem. C, 2014, 118, 28746–28756 CAS.
- E. W. Castner, D. Kennedy and R. J. Cave, J. Phys. Chem. A, 2000, 104, 2869–2885 CrossRef CAS.
- E. W. Schlag, D. Y. Yang, S. Y. Sheu, H. L. Selzle, S. H. Lin and P. M. Rentzepis, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 9849–9854 CrossRef CAS PubMed.
- A. Troisi and G. Orlandi, J. Phys. Chem. B, 2002, 106, 2093–2101 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra09864e |
| This journal is © The Royal Society of Chemistry 2015 |