[(Y1−xGdx)0.95Eu0.05]2(OH)5NO3·nH2O (0 ≤ x ≤ 0.50) layered rare-earth hydroxides: exfoliation of unilamellar and single-crystalline nanosheets, assembly of highly oriented and transparent oxide films, and greatly enhanced red photoluminescence by Gd3+ doping†
Qi Zhua,
Ji-Guang Li*ab,
Xiaodong Lia,
Yang Qic and
Xudong Suna
aKey Laboratory for Anisotropy and Texture of Materials (Ministry of Education), School of Materials and Metallurgy, Northeastern University, Shenyang, Liaoning 110819, China
bAdvanced Materials Processing Unit, National Institute for Materials Science, Namiki 1-1, Tsukuba, Ibaraki 305-0044, Japan. E-mail: LI.Jiguang@nims.go.jp; Tel: +81-29-860-4394
cInstitute of Materials Physics and Chemistry, School of Sciences, Northeastern University, Shenyang, Liaoning 110819, China
First published on 15th July 2015
Abstract
Well crystallized layered rare-earth hydroxide (LRH) crystals of [(Y1−xGdx)0.95Eu0.05]2(OH)5NO3·nH2O (0 ≤ x ≤ 0.50), with a tens of micron-sized lateral, have been hydrothermally synthesized in the presence of the mineralizer NH4NO3. Smaller LRH particles, expanding in the ab plane, and a shrinked interlayer distance (c/2) were observed for a higher Gd content. The interaction between the adjacent hydroxide main layers of the LRH has been weakened by inserting water insoluble oleate anions into the interlayer via hydrothermal treatment. Unilamellar and single-crystalline nanosheets with lateral sizes close to those of the parent crystals and a thickness of ∼1.76 nm were delaminated by dispersing the oleate-containing LRH in toluene. Through a quasi-topotactic phase transformation, highly [111] oriented [(Y1−xGdx)0.95Eu0.05]2O3 (0 ≤ x ≤ 0.50) films with a transmittance of up to 81% and a thickness of ∼55 nm have been constructed through spin-coating colloidal suspension of the exfoliated nanosheets on quartz, followed by calcination at 800 °C. Gd3+ doping leads to greatly enhanced red emission of Eu3+ at 615 nm and obvious red shifting of the charge transfer excitation band (CTB) owing to the smaller electronegativity of Gd3+. The asymmetry factor of luminescence, I(5D0 → 7F2)/I(5D0 → 7F1), remains nearly constant at ∼4.9 up to x = 0.05, followed by a steady increase to ∼8.2 at x = 0.50, which has been ascribed to the splitting of the C1 symmetry from distorted S6 lattice sites at x > 0.05. Gd3+ doping has little influence on the fluorescence lifetime of the 615 nm emission, which was determined to be ∼1.6 ± 0.1 ms.
1. Introduction
Nanosheets of layered inorganic compounds are ideal building blocks for the construction of inorganic or hybrid organic–inorganic multifunctional films owing to their special two-dimensional (2D) morphologies.1–5 Also owing to this peculiar morphology, an oriented nanosheet film may be assembled to introduce additional or greatly enhanced functionalities.1–4 Orientation dependent luminescence behaviors were previously found by Ida1 for the colloidal solutions of K2Ln2Ti3O10, KLnNb2O7, and RbLnTa2O7 perovskite nanosheets (Ln: lanthanide). Greatly enhanced luminescent properties were also observed by us for the two-dimensional Y2O3:Eu3+ nanofilms of high [111] orientation.6 In addition, layered inorganic compounds may be potentially delaminated into unilamellar nanosheets or nanosheets of a few-layer thickness for the further construction of various nanostructures, particularly transparent functional films.7 Therefore, delaminating layered compounds into nanosheets has attracted much attention.In the family of layered inorganic compounds, the layered rare-earth hydroxides (LRHs) of RE2(OH)5(Am−)1/m·nH2O (RE: rare-earth ions; A: intercalated anions)8,9 are a new group of important anion-type layered materials, which have attracted immediate attention for their potential application in luminescent films using the ultra-thin LRH nanosheets as building blocks, due to the unique properties of the rare-earth elements. To date, exfoliation and exfoliation-free synthesis are the main employed techniques to produce 2D LRH nanosheets.8,10 Recently, LRHs crystals have been exfoliated into nanosheets via anion exchange with dodecylsulfate (C12H25OSO3−, DS−) at room temperature, followed by mechanical agitation in formamide.11 Transparent Gd2O3:Eu films were prepared by allowing the formamide suspension containing submicron-sized LRH nanosheets to dry on a substrate.11 In addition, delamination of directly synthesized Eu(OH)3−xDSx was subsequently performed in formamide, and hybrid films were fabricated by combining the exfoliated nanosheets with Ti1.84O4 nanosheets, which showed strong red emission due to an antenna effect of the titanium oxide sheets.12 Exfoliation-free synthesis was proposed by capping the crystal growth in thickness with tetrabutylammonium ions (TBA+, (C4H9)4N+) in a hydrothermal reaction, and highly luminescent Y2O3:Eu3+ films were also obtained through annealing of the precursor films made with submicron LRH nanosheets.6 Unfortunately, the resultant nanosheets are all submicron sized, and larger ones seem impossible. Very recently, tens of micron-sized unilamellar nanosheets have been efficiently delaminated by us from sub-millimeter sized LRH crystals via hydrothermal-assisted processing.13–15 A significantly faster anion exchange and higher extent of dodecylsulfate (C12H25OSO3−, DS−)14 and oleate (C17H33COO−)15 intercalation were observed for hydrothermal treatment than those of ambient processing. The exfoliated ultra-large nanosheets are ideal building blocks for oriented luminescent films, and meanwhile hydrothermal-assisted processing provides an efficient way for anion exchange and intercalation.
Two-dimensional nanophosphors of high quality play an important role in the miniaturization and intelligentization of opto-electronic components, so the design and preparation of highly luminescent films are of great importance. In this work, (Y,Gd,Eu)2O3 nano-phosphor films of high [111] orientation have been constructed via self-assembly of exfoliated LRH nanosheets followed by proper annealing, and greatly enhanced luminescence was observed by simultaneous Gd3+ doping. Hydrothermal-assisted exfoliation was employed for the delamination of Y/Gd/Eu solid-solution LRH crystals into unilamellar nanosheets for further luminescent film construction. The Y/Gd/Eu system was mainly chosen here due to the fact that Y2O3:Eu3+ is the most important red phosphor, finding wide application in areas such as fluorescent lamps, white light emitting diodes (white LEDs), plasma display panels (PDPs), flat panel displays (FDPs), field emission displays (FEDs), and cathode ray tubes (CRTs).16–18 Furthermore, the smaller electronegativity of Gd3+ (1.20) than that of Y3+ (1.22) allows an easier charge transfer (CT) from the 2p orbital of O2− to the 4f orbital of Eu3+, thus leading to an enhanced intensity of the CT/PL bands as well as a red-shift of the CT band edge.19–22 We believe that the outcomes of this work would have wide implications for phosphors and layered inorganic materials, and the obtained transparent oxide films may have potential application in “see-through” displays, LED chips, and so on.11
2. Experimental details
The starting rare-earth sources for the LRH synthesis are Y2O3, Gd2O3, and Eu2O3, all 99.99% pure products from Huizhou Ruier Rare-Chem. Hi-Tech. Co. Ltd (Huizhou, China). Analytical grade nitric acid (HNO3, 63 wt%), ammonium hydroxide solution (NH3·H2O, 25 wt%), and ammonium nitrate (NH4NO3, 99.0% pure) were purchased from Shenyang Chemical Reagent Factory (Shenyang, China). The nitrate solution of RE3+ was prepared by dissolving the corresponding oxide with a slightly excessive amount of nitric acid, followed by evaporation at ∼90 °C to dryness to remove superfluous acid. Synthesis of large LRH crystals for the Y/Gd/Eu ternary system was conducted via hydrothermal reaction (180 °C for 48 h) in the presence of NH4NO3, as described in our previous paper.13 Hydrothermal-assisted anion exchange, exfoliation, and film construction were conducted as previously described.14,15 An oxide film was obtained by calcining the nanosheet film in flowing oxygen (200 mL min−1) at 800 °C for 4 h.Phase identification was performed by X-ray diffractometry (XRD, Model PW3040/60, Philips, Eindhoven, Netherlands) operating at 40 kV/40 mA using nickel filtered Cu Kα radiation and a scanning speed of 4.0° min−1 (2θ). Lattice constants were calculated from the XRD patterns using the software package X’Pert HighScore Plus version 2.0 (PANanalytical B.V., Almelo, Netherlands). Morphologies of the products were observed via field emission scanning electron microscopy (FE-SEM, Model JSM-7001F, JEOL, Tokyo, Japan) and transmission electron microscopy (TEM, Model JEM-2000FX, JEOL, Tokyo). Fourier transform infrared (FTIR) spectroscopy (Model Spectrum RXI, Perkin-Elmer, Shelton, Connecticut, USA) of the pristine and anion-exchanged LRHs was performed by the standard KBr method. A Nanosurf easyScan 2 atomic force microscope (AFM, Switzerland) was employed to obtain topographical images of the nanosheets. Optical properties of the phosphor films were measured at room temperature with a UV-Vis spectrophotometer (Lambda-750S, Perkin-Elmer) for transmittance and with an LS-55 fluorescence spectrophotometer (Perkin-Elmer) for photoluminescence excitation (PLE) and emission (PL).
3. Results and discussion
Fig. 1 shows XRD patterns of the hydrothermal products with various amounts of Gd3+, where all the peaks can be well indexed to a layered phase8 in each case, as previously reported for Ln2(OH)5NO3·nH2O LRHs.8,9,13 No impurity was detectable, indicating a direct crystallization of the homogeneous solid solution. The x = 0 sample exhibits a series of strong (00l) and sharp (220) diffractions, suggesting that the resultant LRH is ordered for its hydroxide main layers. The samples processed with a Gd content of x = 0.05–0.20 similarly show the (220) diffraction of LRH, but splitting of the (002) and (004) diffractions was clearly observed. The above results imply that the samples contain two phases of different basal spacings. Taking the x = 0.05 sample as an example, the basal spacings calculated from the two (002) diffractions are 0.90 and 0.84 nm, different from the single one of 0.92 nm for the x = 0 sample. Dehydration contributes to the appearance of the LRH phase with a shorter basal spacing. In the LRH structure, water molecules are directly coordinated to the Ln atoms to form coordination polyhedrons, instead of being intercalated between the layers, and the loss of hydration water shortens the basal spacing.9 The high hydration phase is thereafter referred to as HH (larger basal spacing), while the low hydration one is referred to as LH. The intensity ratio of ILH002 to IHH002 steadily increases from 1.12 for x = 0.05 to 2.01 for x = 0.20, indicating an increased content of the LH phase for higher Gd doping. Only the LH phase was observed for the x = 0.50 sample, with a basal spacing of 0.84 nm. The hydration number of Ln2(OH)5NO3·nH2O was reported to decrease towards a larger Ln3+ (for 8-fold coordination, Y3+, Gd3+, and Eu3+ have ionic sizes of 0.1019, 0.1053, and 0.1066 nm, respectively),23 while a smaller number of coordinated water would lead to an appreciable contraction in the basal spacing.9 This is the main reason for the evolution of two phases. As the (220) diffraction is related to the metal-to-metal distance in the host layer, replacing Y3+ with larger Gd3+ would have thus contributed to the plane expansion in compliance with the steady shifting of the (220) diffraction towards a lower angle in the XRD pattern. The interplanar spacing of (220) was found to increase from ∼0.308 nm for the x = 0 sample to ∼0.315 nm for the x = 0.50 sample. | ||
| Fig. 1 XRD patterns of the [(Y1−xGdx)0.95Eu0.05]2(OH)5NO3·nH2O (0 ≤ x ≤ 0.50) LRH solid solutions hydrothermally synthesized at 180 °C for 48 h. | ||
Fig. 2 shows particle morphologies of the [(Y1−xGdx)0.95Eu0.05]2(OH)5NO3·nH2O solid solutions for some typical compositions, from which the effect of Gd3+ doping is clearly discernable. The crystals are hexagons, but the particle size successively decreases at a higher Gd content. The majority of particles in Fig. 2a have a lateral size of ∼30 μm for x = 0, which gradually decreases to ∼1 μm in Fig. 2d for x = 0.50. Such a tendency might be attributed to the higher nucleation rate of Gd3+ than that of Y3+ owing to the lanthanide contraction.19,21,24
 | ||
| Fig. 2 FE-SEM morphology images of [(Y1−xGdx)0.95Eu0.05]2(OH)5NO3·nH2O: (a) x = 0, (b) x = 0.10, (c) x = 0.20, and (d) x = 0.50. | ||
The interlayer NO3− ions are readily exchangeable with other inorganic and small organic anions at room temperature, and can also be completely replaced by larger water-soluble/insoluble organic anions with the help of hydrothermal treatment.14,15,22 Our previous work showed that oleate anions may move into the interlayers of LRH via “wriggle intercalation”14,15 to completely replace NO3− and yield the oleate derivative of Ln2(OH)5(C17H33COO)(C17H33COOH)y·nH2O (Ln = Y, Tb, Eu)15 with multiple interlayer spacings, which were also found in this work (Fig. S1a and b†). The (00l) reflections shift to the lower-angle side while the (220) reflections are at the same position (Fig. S1a†), indicating the expanded interlayer. At the same time, several sets of (00l) reflections were indeed observed for anion exchange products in Fig. S1a,† suggesting the existence of multi interlayer distances. In Fig. S1b,† the strong absorption peak at 1384 cm−1 is characteristic of an uncoordinated nitrate anion and is comparable to other layered hydroxides containing free interlayer nitrate groups.14,15 After anion exchange, the vibration of nitrate is no longer observable. Instead, two intense bands appeared at ∼1566 and 1459 cm−1, which are assignable to the stretching modes of carboxyl (COO−).15 The strong absorptions at ∼2926 and 2856 cm−1 are due to the asymmetric and symmetric CH2 stretching vibrations, respectively, whereas the weak band at ∼3010 cm−1 is assignable to the stretching mode of the terminal CH3 group of the hydrocarbon tail.15 The above results confirmed a complete replacement of the interlayer nitrate by oleate. Owing to the massive insertion of oleate anions into the interlayer under hydrothermal conditions, the interlayer distance has been significantly expanded, leading to cracks of different gaps along the thickness direction (Fig. S1c†). The thickness of the LRH platelets has thus been expanded from ∼1 to 5–10 μm, as shown in Fig. S1c.†
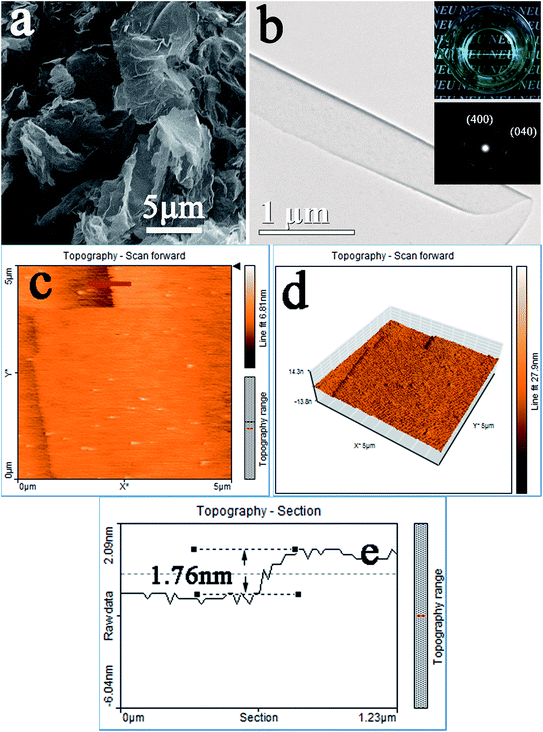
Obviously, the swollen structure remains long-range ordered but the interlayer interaction should have been significantly weakened. Mechanical agitation in a medium may impose a transverse sliding force on the swollen phase, leading to exfoliation of LRHs. Dispersing the LRH-oleate powder in toluene indeed yielded a transparent colloidal suspension via constant and slow magnetic stirring for 16 h, with a clearly observable Tyndall effect under laser beam irradiation (inset in Fig. 3b), indicating the delamination of LRH-oleate. Most of the exfoliated nanosheets have lateral sizes of ≥10 μm (Fig. 3a), close to their starting LRH crystals. The uniform contrast in the TEM image (Fig. 3b) of the individual nanosheet implies that the nanosheet is rather uniform in thickness. The TEM image also indicates that the nanosheets are very smooth, clean, and free of fragmentation. The well-arranged spot-like patterns observed via selected area electron diffraction (SAED) imply that the nanosheet under observation is well crystallized and is of single crystallinity (inset in Fig. 3b). From the SAED pattern, the cell parameters are determined as a ≈ 1.28 and b ≈ 0.72 nm, which are in close agreement with those of the bulk LRH.9 The nanosheet was estimated to be ∼1.76 nm thick from the AFM height profile (Fig. 3e), indicating that it is primarily unilamellar. The thickness is larger than the crystallographic thickness of ∼1 nm for unilamellar nanosheets,13 possibly owing to the surface chemical adsorption of oleate and toluene molecules. At the same time, AFM observation indicated that the nanosheets are very flat and smooth (Fig. 3c and d). The nanosheets showed a significantly larger lateral size and a more unabridged shape than those of the ones produced previously, which are thus ideal building blocks for highly oriented luminescent films.
Owing to their significantly larger sizes, the nanosheets tend to orient themselves along the crystallographic c-direction perpendicular to the substrate surface to introduce special or enhanced functionalities. Depositing 500 μL colloidal nanosheets (solid loading: ∼2.5 vol%) on a quartz substrate followed by spin-coating (3500 rpm for 1 min) has produced highly [001] oriented films via self-assembly of the nanosheets (Fig. 4a). As there are oleate anions and toluene molecules on the surfaces of the positively charged nanosheets, the nanosheets tend to assemble themselves into new layered materials similar to LRH, and thus the (00l) reflection was observed from the LRH film. Calcining the LRH films at 800 °C for 4 h yielded cubic-structured [(Y1−xGdx)0.95Eu0.05]2O3 (0 ≤ x ≤ 0.50) films (Fig. 4a) through a quasi-topotactic phase transformation.6 Thus, the oxide film shows strong (222) and very weak non-(222) reflections, indicating a high [111] orientation. The amorphous diffraction in the range of 15–27° is assigned to the contribution from the quartz substrate, mainly due to the rather thin film (the thickness has been determined hereinafter). Also owing to the same reason, widened (222) diffractions were observed, though the actual crystallinity is good as observed in our previous work.6,14,15 Calculations with the (222) diffraction yielded an expanded lattice constant of a at a higher Gd content (x value). The cell constant steadily increased from ∼1.0649 nm for x = 0 to 1.0725 nm for x = 0.20 and then to ∼1.0810 nm for x = 0.50, in compliance with the steady shifting of the (222) diffraction towards a lower angle with increasing x (Fig. 4a). This is owing to the larger ionic size of Gd3+ than that of Y3+. The oxide films are flat and significantly denser than those made with submicron-sized nanosheets,6 showing the great advantages of a larger sheet size (Fig. 4b). The oxide film was estimated to have a thickness of up to ∼55 nm via AFM analysis (Fig. 4c–e), and exhibits a high transmittance of up to 81% (bare quartz: ∼94%, Fig. 4f) in the visible wavelength region (500–800 nm).
Fig. 5 shows PLE/PL spectra of the highly [111] oriented [(Y1−xGdx)0.95Eu0.05]2O3 (0 ≤ x ≤ 0.50) films. The strong PLE band with a maximum at ∼237–244 nm is ascribed to the transition by charge transfer (CT), that is the electronic transition from the 2p orbital of O2− to the 4f orbital of the Eu3+ activator.25,26 Compared with the reported values of ∼250 nm for Y2O3:Eu3+ powder,6,9,13,19 a blue shift of the CT band centre was clearly observed for the [111] oriented films, owing to the fact that the (222) facets are closely packed low-energy planes.6 Successive red-shifting of the CT band center from ∼237 to 244 nm with increasing Gd content was also observed (Fig. 5). During the charge transfer excitation, the O2− 2p electrons are excited into the 4f levels of Eu3+, and the position of the CT band is determined by the energy difference between the O2− 2p valence band and the 4f levels of Eu3+. The increased Eu–O bond length owing to lattice expansion (Fig. 4a) would lower the energy difference between the Eu3+ 4f and O2− 2p electrons, thus leading to the observed red-shifting of the CTB center.9 Besides, the electronegativities of Y3+ (1.22) and Gd3+ (1.20) are in the order Y3+ > Gd3+, and thus replacing Y3+ with Gd3+ will make charge transfer easier to occur, which would also lead to red-shifting of the CT band. Upon UV excitation into the charge transfer band of Eu3+ at ∼239 nm, the emission spectrum consists of the typical transitions of Eu3+ in a cubic Y2O3 host lattice.25,26 The strongest emission at ∼615 nm is due to the forced electronic dipole 5D0 → 7F2 transition of Eu3+. As marked in the figure, the other emission bands were observed corresponding to the 5D1 → 7F1 transition at ∼538 nm, 5D0 → 7F0 at 586 nm, the magnetic dipole 5D0 → 7F1 transition at ∼593, 597, and 604 nm (three Stark splits), the 5D0 → 7F3 transition at 656 nm, and the 5D0 → 7F4 transition at 714 nm. The relative intensity of the transition to different J levels depends on the site symmetry of Eu3+.27,28 The intensity of the 615 nm emission exhibits a clear dependence on the Gd3+ concentration, and increased emission intensity was found at a higher Gd content. When the x value increases from 0 to 0.50, the emission intensity increased by 136% (inset in Fig. 5) owing to the more efficient excitation.
The cubic Y2O3 lattice is known to have two different crystallographic positions for Eu3+ substitution: the 24d (C2) site without inversion symmetry and the 8b (S6) site with inversion symmetry.16,18 The ratio of C2 to S6 is 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and the Eu3+ activators are expected to take these two sites in a statistical way upon substitution. The Judd–Ofelt parity law predicts that the magnetic dipole transition is permitted while the electric dipole transition is forbidden, and the latter is allowed only on the condition that the Eu3+ ions occupy a site without an inversion center.27,28 The higher occupancy of Eu3+ at the non-centrosymmetric C2 sites (75%) thus yields a PL spectrum dominated by the 5D0 → 7F2 electric dipole transition at ∼615 nm, while the S6 site Eu3+ can only contribute to the 5D0 → 7F1 magnetic dipole transition.27,28 As the 5D0 → 7F2transition (monitored 615 nm emission) is hyper-sensitive to the local Eu3+ environment while 5D0 → 7F1 (monitored 593 nm emission) is not, the intensity ratio I615/I593, called asymmetry factor, has been used to effectively probe the local coordination environment of Eu3+.26 We analyzed the asymmetry factor for the whole series of oxide solid solutions and the results are plotted in Fig. 5 (see inset) as a function of the Gd content. It is clearly seen that the factor remains nearly constant at ∼4.9 up to x = 0.05 and then gradually increases to ∼8.2 at x = 0.50. The Eu3+ activator in Y2O3:Eu3+ may have 8-fold coordination for the new symmetry site of C1 distorted from S6 when the Eu–O bond is elongated at the small crystallite size.9 Since the C1 symmetry has no inversion center, its appearance might account for the increase in asymmetry factor at x > 0.05, since it would enhance the 5D0 → 7F2 electric dipole transition while suppressing the 5D0 → 7F1 magnetic dipole emission at the same time. Splitting of C1 from S6 is made possible by two reasons in this work: (1) the crystal growth was significantly confined in the ultrathin films (∼55 nm thick) to yield fine crystallites (∼12 nm), and (2) a high Gd content induces lattice expansion and thus elongated Eu–O bonds.
1 and the Eu3+ activators are expected to take these two sites in a statistical way upon substitution. The Judd–Ofelt parity law predicts that the magnetic dipole transition is permitted while the electric dipole transition is forbidden, and the latter is allowed only on the condition that the Eu3+ ions occupy a site without an inversion center.27,28 The higher occupancy of Eu3+ at the non-centrosymmetric C2 sites (75%) thus yields a PL spectrum dominated by the 5D0 → 7F2 electric dipole transition at ∼615 nm, while the S6 site Eu3+ can only contribute to the 5D0 → 7F1 magnetic dipole transition.27,28 As the 5D0 → 7F2transition (monitored 615 nm emission) is hyper-sensitive to the local Eu3+ environment while 5D0 → 7F1 (monitored 593 nm emission) is not, the intensity ratio I615/I593, called asymmetry factor, has been used to effectively probe the local coordination environment of Eu3+.26 We analyzed the asymmetry factor for the whole series of oxide solid solutions and the results are plotted in Fig. 5 (see inset) as a function of the Gd content. It is clearly seen that the factor remains nearly constant at ∼4.9 up to x = 0.05 and then gradually increases to ∼8.2 at x = 0.50. The Eu3+ activator in Y2O3:Eu3+ may have 8-fold coordination for the new symmetry site of C1 distorted from S6 when the Eu–O bond is elongated at the small crystallite size.9 Since the C1 symmetry has no inversion center, its appearance might account for the increase in asymmetry factor at x > 0.05, since it would enhance the 5D0 → 7F2 electric dipole transition while suppressing the 5D0 → 7F1 magnetic dipole emission at the same time. Splitting of C1 from S6 is made possible by two reasons in this work: (1) the crystal growth was significantly confined in the ultrathin films (∼55 nm thick) to yield fine crystallites (∼12 nm), and (2) a high Gd content induces lattice expansion and thus elongated Eu–O bonds.
We also investigated the decay kinetics of the 615 nm emission for the oxide films with various Gd contents, and the results are presented in Fig. 6 for three typical examples. No new emission peak emerges with increasing delay time, as can be seen from the time-resolved emission spectra in each case (see inset). All the fluorescence decay curves can be fitted to single exponentials. The lifetimes of the x = 0, x = 0.20 and x = 0.50 samples determined in this work are 1.62 ± 0.07, 1.65 ± 0.01, and 1.59 ± 0.09 ms, respectively, indicating that the Gd content does not substantially affect the lifetime in the range of this study. The lifetime of 1.60 ± 0.10 ms obtained in this work also falls in the range reported for Y2O3:Eu3+ red phosphors (∼1.5–2.4 ms).29–32
4. Conclusions
We have demonstrated that a high-temperature hydrothermal reaction of mixed solutions of the component nitrates in the presence of the mineralizer NH4NO3 is an effective way to obtain tens of micron-sized [(Y1−xGdx)0.95Eu0.05]2(OH)5NO3·nH2O (0 ≤ x ≤ 0.50) LRH crystals. The structural study indicated that a higher Gd content reduces the size of the LRH particles but contributes to the expansion of the (220) plane. Increasing the Gd3+ incorporation also leads to the transition of the LRH product from high-hydration to low hydration phases. With the help of hydrothermal treatment, water insoluble oleate anions have been successfully inserted into the interlayers of LRHs, which expands the interlayer distance and thus leads to an increasing thickness of the LRH crystals from ∼1 μm to several micrometers. Delamination of the oleate-intercalated LRHs in toluene has produced unilamellar nanosheets with lateral sizes close to their starting crystals and a thickness of ∼1.76 nm. Highly [111] oriented and transparent (transmittance up to 81%) films of [(Y1−xGdx)0.95Eu0.05]2O3 (0 ≤ x ≤ 0.50), with thicknesses of ∼55 nm, have been constructed through spin-coating a colloidal suspension of the exfoliated nanosheets on quartz, followed by calcination at 800 °C. Under UV excitation at 239 nm, the oxide films exhibit the strongest luminescence at 615 nm, whose intensity shows a clear dependence on the Gd3+ concentration. Raising the x value from 0 to 0.50 yielded a 136% increase in the emission intensity and a red shift of the CT band centre, both mainly due to the smaller electronegativity of Gd3+ than that of Y3+. The asymmetry factor of the luminescence, I(5D0 → 7F2)/I(5D0 → 7F1), remains nearly constant at ∼4.9 up to x = 0.05, followed by a steady increase to ∼8.2 at x = 0.50, which has been ascribed to the splitting of the C1 symmetry from the distorted S6 sites at x > 0.05. Fluorescence decay analysis yields similar lifetimes of ∼1.6 ± 0.1 ms for the 615 nm emission of all the samples, suggesting that the Gd3+ content has little influence on the fluorescence lifetime.Acknowledgements
This work was supported in part by the National Natural Science Foundation of China (Grants 51302032 and 51172038), the Fundamental Research Funds for the Central Universities (Grants N140204002), the Liaoning Province Doctor Startup Fund (Grant 20131035), and the Grant-in-Aid for Scientific Research (KAKENHI, No. 26420686).References
- S. Ida, C. Ogata, M. Eguchi, W. J. Youngblood, T. E. Mallouk and Y. Matsumoto, J. Am. Chem. Soc., 2008, 130, 7052 CrossRef CAS PubMed.
- S. Ida, D. Shiga, M. Koinuma and Y. Matsumoto, J. Am. Chem. Soc., 2008, 130, 14038 CrossRef CAS PubMed.
- S. Ida, C. Ogata, D. Shiga, K. Izawa, K. Ikeue and Y. Matsumoto, Angew. Chem., Int. Ed., 2008, 47, 2480 CrossRef CAS PubMed.
- S. Ida, C. Ogata and Y. Matsumoto, J. Phys. Chem. C, 2009, 113, 1896 CAS.
- E.-J. Oh, T. W. Kim, K. M. Lee, M.-S. Song, A.-Y. Jee, S. T. Lim, H. W. Ha, M. Lee, J.-H. Choy and S.-J. Hwang, ACS Nano, 2010, 8, 4437 CrossRef PubMed.
- Q. Zhu, J.-G. Li, C. Y. Zhi, R. Ma, T. Sasaki, J. X. Xu, C. H. Liu, X. D. Li, X. D. Sun and Y. Sakka, J. Mater. Chem., 2011, 21, 6903 RSC.
- N. Liu, F. Luo, H. X. Wu, Y. H. Liu, C. Zhang and J. Chen, Adv. Funct. Mater., 2008, 18, 1518 CrossRef CAS PubMed.
- F. Gándara, J. Perles, N. Snejko, M. Iglesias, B. Gómez-Lor, E. Gutiérrez-Puebla and M. A. Monge, Angew. Chem., Int. Ed., 2006, 45, 7998 CrossRef PubMed.
- Q. Zhu, J.-G. Li, C. Y. Zhi, X. D. Li, X. D. Sun, Y. Sakka, D. Golberg and Y. Bando, Chem. Mater., 2010, 22, 4204 CrossRef CAS.
- X. L. Wu, J.-G. Li, Q. Zhu, W. Liu, J. Li, X. D. Li, X. D. Sun and Y. Sakka, J. Phys. Chem. C, 2015, 3, 3428 CAS.
- K.-H. Lee, B. I. Lee, J. H. You and S.-H. Byeon, Chem. Commun., 2010, 46, 1461 RSC.
- S. Ida, Y. Sonoda, K. Ikeue and Y. Matsumoto, Chem. Commun., 2010, 46, 877 RSC.
- Q. Zhu, J.-G. Li, R. Ma, T. Sasaki, X. Yang, X. D. Li, X. D. Sun and Y. Sakka, J. Solid State Chem., 2012, 192, 229 CrossRef CAS PubMed.
- Q. Zhu, J.-G. Li, X. D. Li, X. D. Sun, Y. Qi, M. Y. Zhu and Y. Sakka, Sci. Technol. Adv. Mater., 2014, 15, 014203 CrossRef.
- Q. Zhu, Z. X. Xu, J.-G. Li, X. D. Li, Y. Qi and X. D. Sun, Nanoscale Res. Lett., 2015, 10, 132 CrossRef PubMed.
- G. Blasse and B. C. Grabmair, Luminescent Materials, Springer Verlag, Berlin, 1994 Search PubMed.
- M. Buijs, A. Meyerink and G. Blasse, J. Lumin., 1987, 37, 9 CrossRef CAS.
- R. G. Pappalardo and R. B. Hunt Jr., J. Electrochem. Soc., 1985, 132, 721 CrossRef CAS PubMed.
- J.-G. Li, X. D. Li, X. D. Sun and T. Ishigaki, J. Phys. Chem. C, 2008, 112, 11707 CAS.
- S. Ekambaram, K. C. Patil and M. Maaza, J. Alloys Compd., 2005, 393, 81 CrossRef CAS PubMed.
- J.-G. Li, X. D. Li, X. D. Sun, T. Ikegami and T. Ishigaki, Chem. Mater., 2008, 20, 2274 CrossRef CAS.
- Y. H. Li and G. Y. Hong, J. Lumin., 2007, 124, 297 CrossRef CAS PubMed.
- R. D. Shannon, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, 32, 751 CrossRef.
- P. Krumholz, Progress in the Science and Technology of the Rare Earth, ed. L. Eyring, Pergamon Press, New York, 1964 Search PubMed.
- Q. Zhu, J.-G. Li, X. D. Li, X. D. Sun and Y. Sakka, Sci. Technol. Adv. Mater., 2011, 12, 055001 CrossRef.
- Q. Zhu, J.-G. Li, X. D. Li and X. D. Sun, Acta Mater., 2009, 57, 5975 CrossRef CAS PubMed.
- B. R. Judd, Phys. Rev., 1962, 127, 750 CrossRef CAS.
- G. S. Ofelt, J. Chem. Phys., 1962, 37, 511 CrossRef CAS PubMed.
- H. Wang, C. K. Lin, J. Lin and M. Yu, Appl. Phys. Lett., 2005, 87, 181907 CrossRef PubMed.
- H. Wang, M. Yu, C. K. Lin and J. Lin, J. Phys. Chem. C, 2007, 111, 11223 CAS.
- H. W. Song, J. W. Wang, B. J. Chen, H. S. Peng and S. Z. Lu, Chem. Phys. Lett., 2003, 376, 1 CrossRef CAS.
- M. L. Pang, J. Lin, Z. Y. Cheng, J. Fu, R. B. Xing and S. B. Wang, Mater. Sci. Eng., B, 2003, 100, 124 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra09784c |
| This journal is © The Royal Society of Chemistry 2015 |