A comprehensive review on biodiesel cold flow properties and oxidation stability along with their improvement processes
Abstract
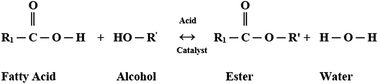
Biodiesel, which comprises fatty acid esters, is derived from different sources, such as vegetable oils from palm, sunflower, soybean, canola, Jatropha, and cottonseed sources, animal fats, and waste cooking oil. Biodiesel is considered as an alternative fuel for diesel engines. However, biodiesel has poor cold flow behavior (i.e., high cloud point & pour point) and oxidation stability compared with petroleum diesel because of the presence of saturated and unsaturated fatty acid esters. Consequently, the performance of biodiesel during cold weather is affected. When biodiesel is oxidized, the subsequent dregs can adversely affect the performance of the fuel system as well as clog the fuel filter, fuel lines, and injector. This phenomenon results in start-up and operability problems. Cold flow behavior is usually assessed through the pour point (PP), cloud point (CP), and cold filter plugging point (CFPP). Earlier studies on cold flow focused on reducing the devastating effect of poor cold flow problems, such as lowering the PP, CP, and CFPP of biodiesel. This present paper provides an overview of the cold flow behavior and oxidation stability of biodiesel, as well as their effect on the engine operation system. The improvements on the behavior of cold flow of biodiesel are also discussed.

 Please wait while we load your content...
Please wait while we load your content...