A tunable ratiometric pH sensor based on phenanthro[9,10-d]imidazole covalently linked with vinylpyridine†
Arockiam Jesin Benetoa,
Viruthachalam Thiagarajanb and
Ayyanar Siva*a
aSchool of Chemistry, Madurai Kamaraj University, Madurai-21, Tamilnadu, India. E-mail: drasiva@gmail.com; ptcsiva@yahoo.co.in; Tel: +91-451-2458471
bSchool of Chemistry, Bharathidasan University, Tiruchirapalli, Tamilnadu, India
First published on 29th July 2015
Abstract
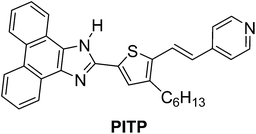
A rational design of the novel phenanthro[9,10-d]imidazole based pH sensing fluorophore PITP was developed, in which phenanthroimidazole acts as a donor and pyridine acts as an acceptor. Various spectral studies confirm that the protonation occurs at two different nitrogen centres, each of which produces an observable color change depending on the pH of the buffer solution. DFT calculations also support the experimental results. We show that PITP can be used as a ratiometric and colorimetric pH sensor in the acidic region.
The design and synthesis of novel fluorophores are of great interest because of the wide range of applications for these compounds in clinical diagnostics and environmental monitoring.1–4 Fluorophores can be used as sensors for the detection of biomolecules, cations, anions, and biological and ecological pH.5,6 pH sensors that work under acidic conditions usually have a single protonation site. Upon protonation on such a site, there will be a physical or chemical change that occurs either through photoinduced electron transfer (PET) or intramolecular charge transfer (ICT) and that is in turn used to precisely measure the pH of the medium. pH sensors that deal with a single protonation site can only detect a single pH and are widely known.7–9 To the best of our knowledge, pH sensor molecules having multiple protonation sites that can sense different solution pHs have not been described to date.
We have designed a molecule in such a way that it can accommodate multiple functional groups having different pKa values, which can alter the ICT depending on the pH of the medium and hence can sense different pHs under different reaction conditions. For that purpose, we chose phenanthro[9,10-d]imidazole in combination with thiophene tethered pyridine as our target molecule (PITP). Under different pH conditions, either the imidazolyl nitrogen or the pyridinyl nitrogen is protonated and significantly red shifted by more than 115 nm, which is very useful to detect the pH of the analyte in the visible region. It is important to note that the PITP molecule can act both as a ratiometric as well as a colorimetric pH sensor under the reaction conditions used.
In general, phenanthro[9,10-d]imidazole based molecules act as metal ion sensors for Hg2+ and Fe3+ (ref. 10–12); to date, only very few reports are available for pH sensors that detect within a very narrow pH region.13,14 In contrast PITP can act as a sensor molecule for the pH region between 2.0 to 7.5. The presence of extended π-conjugation in PITP results in a large Stokes shift, which in turn becomes an essential requirement to detect the wide range of pH of the analyte. Herein, we report the novel synthesis of the phenanthro[9,10-d]imidazole based fluorophore PITP containing extended π-conjugation in the form of 3-hexyl-thiophene having two protonation sites. We found that it acts as an efficient ratiometric and colorimetric pH sensor in a range of pHs. The sensing ability of PITP was investigated using UV-vis, fluorescence, cyclic voltammetry studies and DFT calculations to further support the experimental results. Fluorophore PITP was synthesized from 2-bromo-3-hexylthiophene as depicted in Scheme 1. Compound 2 was initially obtained by the Vilsmeier–Haack reaction of 2-bromo-3-hexylthiophene and was further treated with 9,10-phenanthridinone in the presence of acetic acid and ammonium acetate at 120 °C to get PIT. Fluorophore PITP was obtained by the Heck coupling reaction of vinyl pyridine with PIT under the described reaction conditions.
 | ||
| Scheme 1 (a) POCl3, DMF, EDC, 80 °C; (b) 9,10-phenanthroquinone, NH4OAc, AcOH, 120 °C; (c) 4-vinylpyridine, Pd(OAc)2, DMF, 120 °C, TBAB, K2CO3. | ||
The absorption and emission spectral studies of PITP were carried out in a series of protic and aprotic solvents and we infer that the spectral behaviours are very similar in all solvents studied. Strong absorption and emission bands are present around 400 and 500 nm, respectively, which occurs due to the charge transfer from the phenanthro[9,10-d]imidazole donor to the pyridine acceptor within the PITP fluorophore. The optical response of PITP at different pHs was studied in a 0.2 M phosphate buffer (containing 60% methanol as a co-solvent). When the pH of the solution is decreased from 7.51 to 4.12, the absorbance at around 400 nm decreases with the simultaneous appearance of a new peak at around 450 nm with an isosbestic point at 420 nm, as shown in Fig. 1a. As the pH of the solution decreased further from 4, the newly formed absorption peak at around 450 nm was blue shifted (420 nm) along with an isosbestic point at 450 nm. The absorbance at around 400 nm for PITP is due to the neutral form and the new peaks at around 450 nm and 420 nm with the decrease in pH are due to the successive protonations at the pyridine nitrogen (acceptor) and imidazole nitrogen (donor) centers. It can be anticipated that protonation of the donor or acceptor moiety would change the photophysical properties of the fluorophore because it would affect the efficiency of the intramolecular charge transfer within PITP. The first protonation at the pyridine nitrogen increases the electron accepting character of the acceptor group. This increase in charge transfer results in a red shift in the absorption spectrum. The second protonation at the imidazole nitrogen decreases the electron donating character of the donor group and results in a blue shift15,16 in the absorption spectrum (Fig. 1b). Similar studies were carried out using PIT (PITP without the pyridine moiety); upon decreasing the pH of the solution from 7.10 to 1.88, a red shift in the absorption spectrum is observed from 355 nm to 361 nm, due to the sole protonation of the imidazole nitrogen, and this shift happens only at a pH below 4.0 (Fig. S-1†). This confirms that the second protonation occurring in PITP below pH 4.0 is that of the imidazole nitrogen (donor moiety).
Similar to the absorption spectra, the emission spectra of PITP is red shifted by 116 nm with a clear isoemissive point at 620 nm upon decreasing the pH from 7.51 to 4.12 (Fig. 2). As the pH decreases, the intensity of the new emission peak at around 646 nm enhances with a simultaneous decrease in the intensity of the 528 nm peak upon excitation at the isosbestic point, as shown in Fig. 2. The emission at around 500 nm for PITP is due to the neutral form. The appearance of the new red shifted emission is caused by protonation at the pyridine moiety, which results in an increase in the electron withdrawing ability of the acceptor moiety.
 | ||
| Fig. 2 Fluorescence spectra of PITP (10 μM) in a 0.2 M phosphate and KCl–HCl buffer solution (containing 60% methanol as a co-solvent) at pH 7.51 to 4.12, excited at 450 nm. | ||
The large red shift makes this molecule suitable for ratiometric pH sensing in the acidic region. Upon decreasing the pH from 7.51 to 4.12, the emission intensity ratio (I528/I646) changes from 8.16 to 0.33 and the pKa (5.47) value was calculated from the emission ratio as a function of pH (Fig. S-4a†).
In addition to the absorption and emission spectral changes, the color of the PITP solution changed from colorless to orange-red upon decreasing the pH from 7.5 to 4.0, then changed from orange-red to pale yellow upon decreasing the pH from 3.5 to 2.0, and the color changes are visible to the naked eye (Fig. 3a). Fig. 3b shows the emission color changes of the PITP solution at three different pH values (the photo was taken under illumination from a UV lamp). The color and spectral changes are reversible, and the reaction is reversed upon increasing the pH by using aqueous ammonia. Hence, PITP can be used as a pH sensor within three different pH regions: 7.2/6.2/5.2, 4.2, and 3.2/2.2.
 | ||
| Fig. 3 Change in color of PITP at pH 7.5, 4.2 and 2.0 respectively (a) under visible light and (b) under UV light (365 nm). | ||
To establish the optical response of sensor PITP observed at different pHs, we carried out density functional theory (DFT) calculations using the DFT-B3LYP-6-31G level in the Gaussian 03 package.17 The DFT study shows the charge transfer state originating from the highest occupied molecular orbital (HOMO) to the lowest unoccupied molecular orbital (LUMO) upon excitation. The HOMO is localized on phenanthro[9,10-d]imidazole (donor), and the LUMO is localized on the thiophene and vinyl pyridine moieties (acceptors).
Upon mono- and bis-protonation, the electronic distributions in the HOMO are similar to that in PITP (only on phenanthro[9,10-d]-imidazole). The electronic distributions in the LUMO of the mono-protonated form are located on pyridine only, while in the bis-protonated form it is located on pyridine as well as on the imidazole ring; this results in the blue shift of the CT state (Fig. 4). The energy differences between the HOMOs and LUMOs of the PITP, PITP–H+ and PITP–2H+ forms are 3.23 eV, 2.77 eV and 3.06 eV, respectively. These results are in concordance with our experimental results.
 | ||
| Fig. 4 Molecular orbital plots showing the HOMO–LUMO energy gaps of PITP, and the mono and bis-protonated forms of PITP. | ||
In addition to the UV-visible studies, the cyclic voltammetric analysis of PITP was also studied at pH 2.0 and 4.2 (Fig. S-2†). The result is as follows: Eox = 0.961 V, Ered = −0.066 V; as ΔE > 1000 mV, it is an irreversible redox couple. There is a shift in the oxidation peak from 0.50 to 0.64 V when we change the pH from 2 to 4.2, and the corresponding ΔE values are 0.27 V and 0.59 V. We infer from these values that a double protonation occurs (2e− transfer) at pH 2.0 while a single protonation (1e− transfer) occurs at pH 4.2 (Fig. 5).18,19
 | ||
| Fig. 5 A schematic representation of the single and double protonations of PITP at the pyridine and imidazole nitrogen centres. | ||
To confirm the protonation site, we recorded the 1H NMR spectra of PITP with and without the passing of HCl vapour (Fig. 6b). There, we observe that the protons adjacent to the pyridine nitrogen, i.e. Ha and Hb, are shifted from δ 8.20–8.30 to 8.60–8.70 ppm and from δ 8.04–8.10 to 8.40–8.50 ppm, respectively. Also, the imidazole –NH proton at 13.9 ppm disappears. This is due to the possible bis-protonation of the pyridine as well as the phenanthro[9,10-d]imidazole N atoms, which in turn leads to a fast exchange of protons between the imidazole nitrogens (Fig. 6a). The He–h protons are also shifted downfield to obtain a well-resolved 1H NMR spectrum of PITP.
 | ||
| Fig. 6 (a) Schematic representation of the tautomerisation of the protonated imidazole and (b) the 1H NMR spectrum of PITP with and without passing HCl vapour. | ||
In order to show that PITP can act as a pH sensor, we did the following experiment: first, the PITP fluorophore was dissolved in methanol and a few drops of it were placed over Whatman paper and dried in air for a few minutes. It was then dipped in different acidic buffer solutions (Fig. 7). As illustrated in Fig. 7, the change in colors is in accordance with the spectral results, suggesting that the designed fluorophore could act as a pH sensor within the neutral and acidic regions.
 | ||
| Fig. 7 Change in color of PITP absorbed on paper upon adding buffer solutions (containing 60% methanol) at pH 7.5, 4.2 and 2.0 (left to right). | ||
In summary, we have successfully synthesized a novel phenanthro[9,10-d]imidazole based fluorophore having an extended π-conjugation. We observed multiple protonations at two different nitrogen centres of PITP by conducting UV-vis, fluorescence, CV and 1H NMR studies. DFT studies of PITP support our experimental results. In conclusion, we demonstrated the utility of PITP as a ratiometric and calorimetric pH sensor in the acidic region.
Acknowledgements
We acknowledge the financial support of the Department of Science and Technology, New Delhi, India (Grant No. SR/F/1584/2012-13) and the Council of Scientific and Industrial Research, New Delhi, India (Grant No. 01(2540)/11/EMR-II), the University Grants Commission, New Delhi, India (Grant No. UGC No. 41-215/2012 (SR)); VT acknowledges the UGC-FRP (Grant no. F.4-5(24-FRP)/2013(BSR)), New Delhi, India for its financial support.Notes and references
- X. Chen, T. Pradhan, F. Wang, J. S. Kim and J. Yoon, Chem. Rev., 2012, 112, 1910–1956 CrossRef CAS PubMed.
- A. P. de Silva, H. Q. N. Gunaratne, T. Gunnlaugsson, A. J. M. Huxley, C. P. McCoy, J. T. Rademacher and T. E. Rice, Chem. Rev., 1997, 97, 1515–1566 CrossRef CAS PubMed.
- R. M. Duke, E. B. Veale, F. M. Pfeffer, P. E. Krugerc and T. Gunnlaugsson, Chem. Soc. Rev., 2010, 39, 3936–3953 RSC.
- J. P. Byrne, J. A. Kitchen and T. Gunnlaugsson, Chem. Soc. Rev., 2014, 43, 5302–5325 RSC.
- A. Loudet and K. Burgess, Chem. Rev., 2007, 107, 4891–4932 CrossRef CAS PubMed.
- D. Wencel, T. Abel and C. McDonagh, Anal. Chem., 2014, 86, 15–29 CrossRef CAS PubMed.
- D. Cui, X. Qian, F. Liu and R. Zhang, Org. Lett., 2004, 6, 2757–2760 CrossRef CAS PubMed.
- C. Zhou, Y. Li, Y. Zhao, J. Zhang, W. Yang and Y. Li, Org. Lett., 2011, 13, 292–295 CrossRef CAS PubMed.
- L. Long, X. Li, D. Zhang, S. Meng, J. Zhang, X. Sun, C. Zhang, L. Zhou and L. Wang, RSC Adv., 2013, 3, 12204–12209 RSC.
- K. Benelhadj, J. Massue, P. Retailleau, G. Ulrich and R. Ziessel, Org. Lett., 2013, 15, 2918–2921 CrossRef CAS PubMed.
- B. Venkatachalapathy, P. Ramamurthy and V. T. Ramakrishnan, J. Photochem. Photobiol., A, 1997, 111, 163–169 CrossRef CAS.
- M. Swaminathan and S. K. Dogra, J. Chem. Soc., Perkin Trans. 2, 1983, 1641–1644 RSC.
- W. Lin, L. Long, L. Yuan, Z. Cao and J. Feng, Anal. Chim. Acta, 2009, 634, 262–266 CrossRef CAS PubMed.
- Y. Yan, R. Zhang, X. Yu and H. Xu, ChemPlusChem, 2014, 79, 1676–1680 CAS.
- A. J. Zucchero, J. Tolosa, L. M. Tolbert and U. H. F. Bunz, Chem. – Eur. J., 2009, 15, 13075–13081 CrossRef CAS PubMed.
- V. Schmitt, S. Moschel and H. Detert, Eur. J. Org. Chem., 2013, 25, 5655–5669 CrossRef PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci and G. A. Petersson, Gaussian 09, revision C.01, Gaussian, Inc., Wallingford, CT, 2010 Search PubMed.
- P. L. Runnels, D. J. Joseph, M. J. Logman and M. R. Wightman, Anal. Chem., 1999, 71, 2782–2789 CrossRef CAS.
- R. Gulaboski, I. Bogeski, V. Mirčeski, S. Saul, B. Pasieka, H. H. Haeri, M. Stefova, J. P. Stanoeva, S. Mitrev, M. Hoth and R. Kappl, Sci. Rep., 2013, 3, 1865, DOI:10.1038/srep 01865.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental procedures, spectral data, copies of 1H & 13C NMR and ESI-MS. See DOI: 10.1039/c5ra09536k |
| This journal is © The Royal Society of Chemistry 2015 |