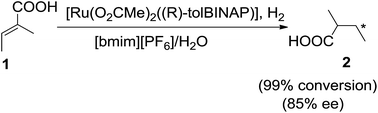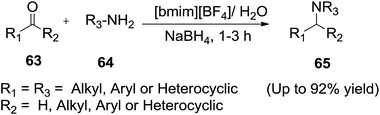Ionic liquid/water mixture promoted organic transformations
Pedavenkatagari Narayana Reddy
*a,
Pannala Padmaja
*b,
Basireddy V. Subba Reddyc and
Gundla Rambabua
aDepartment of Chemistry, Gitam School of Technology, Gitam University, Hyderabad, T.S, India 502 102. E-mail: pnreddy@gitam.in
bDepartment of Chemistry, JNTUH College of Engineering, Kukatpally, Hyderabad, T.S, India 500 085. E-mail: padduiict@gmail.com
cCSIR Indian Institute of Chemical Technology, Hyderabad, India 500 007
First published on 2nd June 2015
Abstract
Ionic liquids (ILs) have been used in numerous applications in chemistry. Wet ILs constitute a new class of solvents with their own new and interesting properties. The properties of ILs are significantly influenced by the addition of water and also affect reaction rates and selectivity. IL/water mixing makes it easy to control the properties of the solution and the formation of these ionic liquid mixtures increases synthetic flexibility. In this review, mixtures of IL/water solvent system promoted organic reactions have been described and the results are compared with other solvent systems. In many cases IL/water combinations were superior compared to conventional organic solvents and biphasic IL/organic co-solvent media with respect to catalytic performance as well as to catalyst separation and recycling.
1 Introduction
Ionic liquids (ILs) have attracted intense interest in recent years due to their wide applications in synthesis, catalysis and chemical separations.1,2 They possess a large array of fascinating properties, such as a negligible vapor pressure, a high chemical and thermal stability, a large temperature range and widely tunable thermophysical properties.3–8 Because of these properties, they can serve as a green recyclable alternative to the volatile organic compounds that are traditionally used as industrial solvents.9,10 In fact, ILs have been successfully used as green solvents in many applications including organic and inorganic synthesis,11–13 polymer synthesis,14,15 chemical separations16–24 and electrochemistry.25,26 The range of research studies on ILs have increased from the past decades and are continually expanding.27–41Multiphase processes employing binary mixtures of immiscible, or partly miscible, aqueous and organic phases are widely used in organic synthesis. The immiscibility of ionic liquids with traditional solvents such as water, ether, hexane and benzene allowed for the formation of bi- and triphasic reaction systems which significantly facilitated purification and extraction of the desired products.42 Furthermore, the mixing of ILs with other molecular liquids is much easier for tuning their properties than the structural design of pure ILs. In this strategy, water is now a very important partner for ILs because water also has a wide variety of properties that pure ILs does not possess.43
When these unique properties of both ILs and water are suitably exploited by mixing them, the resulting mixtures should provide numerous kinds of applications for many fields, especially for bioscience. Generally, hydrophobic ILs are immiscible with water and easily forms liquid/liquid biphasic systems. On the other hand, ILs with hydrophilic ions are mostly miscible with water. Cation species also affect the miscibility of the ILs with water. In particular, the introduction of long alkyl chains into the cations dramatically lowers the miscibility of the ILs with water. On the other hand, ILs having longer alkyl chains are phase separated with water.44
The properties of IL/water mixtures depend strongly on the ion species.45 The component ions of ILs interact strongly with water via ion-dipole interactions. Even a small amount of water can dramatically influence the liquid properties of ILs such as diffusion coefficient, viscosity, polarity, and surface tension.46–49 This may have an effect on reaction rates due to the coordinating ability with a catalyst and also on the solubility of other substances in ILs. There have been several reports that studied the effect of water when IL/water mixtures used as solvent.50–54 The other use of IL/water mixtures involves liquid/liquid phase-separated systems. In particular, separated biphasic liquid mixtures are expected to be suitable media for separation, condensation, and extraction.55 Employing water as the added solvent to ILs enhanced the catalytic activities significantly, ascribed to the creation of a well mixed emulsion-like system. An insight at the molecular level into the state of water dissolved in ILs is needed for further understanding of ILs as media for chemical synthesis and separation. Recently attempts have been made to take this idea further by using mixtures of IL/water.56–58 The objective of this review is to draw attention to the recent trends of mixtures of IL/water solvent system promoted organic reactions such as hydrogenation, oxidation, reduction, substitution, halogenations, three component coupling reactions and other organic reactions.
2 Ionic liquid/water mixtures promoted reactions
2.1 Hydrogenation
Ionic liquids have been successfully used as reaction media for a wide variety of substrates for hydrogenation reactions. Ionic liquids provide a stabilizing medium for catalysts and facilitate their immobilization, thereby facilitating recycling procedures. Biphasic hydrogenations have also demonstrated recyclability, the substrates and products residing in a separate phase to the IL and catalyst.In 2003, Jessop and his group59 reported the asymmetric hydrogenation of tiglic acid (1) catalyzed by Ru(O2CMe)2((R)-tolBINAP) in mixture of 1-butyl-3-methylimidazolium hexafluorophosphate ([bmim][PF6]) and water (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) to afford 2-methylbutanoic acid (2) with high enantioselectivity (85%) and conversion (99%) (Scheme 1). The product was extracted from the IL by supercritical CO2. The enantioselectivity was shown to be hydrogen pressure dependent in wet [bmim][PF6]. At low pressure, the amount of water had no effect on the enantioselectivity but at higher pressure adding water enhanced the enantiomeric excess.
2) to afford 2-methylbutanoic acid (2) with high enantioselectivity (85%) and conversion (99%) (Scheme 1). The product was extracted from the IL by supercritical CO2. The enantioselectivity was shown to be hydrogen pressure dependent in wet [bmim][PF6]. At low pressure, the amount of water had no effect on the enantioselectivity but at higher pressure adding water enhanced the enantiomeric excess.
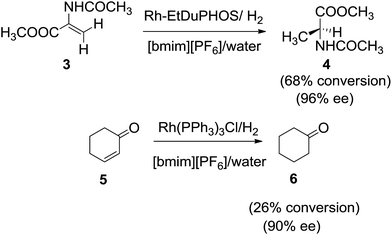
Afterwards, Wolfson et al.60 in 2005 examined the asymmetric hydrogenation of methyl 2-acetamidoacrylate (3) with Rh-EtDuPHOS and the hydrogenation of 2-cyclohexen-1-one (5) with Wilkinson's catalyst in [bmim][PF6] in the presence of water (Scheme 2).
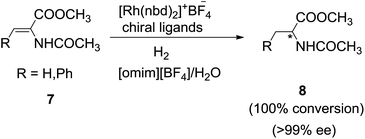
Employing water as second solvent the reaction rate in the biphasic system yielded much higher reaction rates than with the other solvents. The authors attributed this clear beneficial effect of water was due to the creation of a well mixed emulsion-like system. Similarly, Feng et al.61 focused on enantioselective version of asymmetric hydrogenation of enamides in IL/water system (Scheme 3). The hydrogenation of methyl α-acetamidoacrylate (7) with rhodium-ferrocenyl diphosphine complexes afford the product (8) with better conversion and ee in 1-methyl-3-octylimidazolium tetrafluoroborate ([omim][BF4])/H2O and 1-butyl-3-methylimidazolium bis (trifluoromethylsulfonyl)imide ([bmim][Tf2N])/H2O than in conventional organic solvents and ionic liquid/organic systems. It was shown that IL/water combination allow repeated catalyst recycling without significant loss of activity and that industrially relevant turnover numbers of >10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 can be obtained.
000 can be obtained.
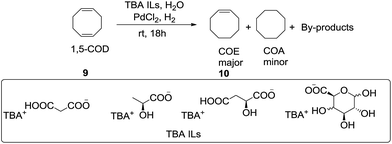
More recently, Bouquillon et al.62 investigated the tetrabutylammonium ionic liquids (TBA ILs) derived from with natural organic anions and tetrabutylammonium hydroxide in catalytic selective hydrogenation of 1,5-cyclooctadiene (9) into cyclooctene (10) at room temperature under atmospheric H2 pressure in water (Scheme 4). They are more suitable for selective hydrogenation of 1,5-COD into COE under milder conditions than commercial 1-butyl-3-methylimidazolium ionic liquids (BMIM ILs).
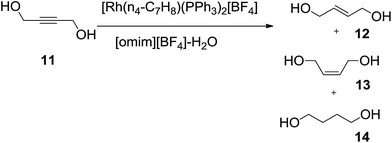
Welton and co-workers63 employed the concept of biphasic systems of IL/water in the hydrogenation of 2-butyne-1,4-diol (11) (Scheme 5). At room temperature, the ionic liquid [omim][BF4], containing [Rh(η4-C7H8) (PPh3)2][BF4] catalyst, forms a separate layer to water containing 2-butyne-1,4-diol. In a stirred autoclave the mixture was pressurized with hydrogen to 60 atm and heated to 80 °C giving a homogeneous single phase solution. On cooling to room temperature, two phases reform, with the ionic liquid phase containing the catalyst and the aqueous phase containing a mixture of 2-butene-1,4-diol (12, 13) and butane-1,4-diol (14) products that can be simply removed without catalyst contamination.
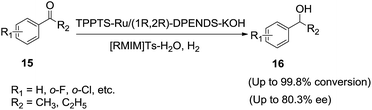
Chen et al.64 prepared the TPPTS-stabilized Ru catalyst and applied successfully for the asymmetric hydrogenation of aromatic ketones (15) in (1-alkyl-3-methylimidazolium p-methylphenylsulfonates) ([RMIM]Ts)/water mixed solvent, which gave the corresponding chiral alcohols (16) with good enantioselectivity (Scheme 6).
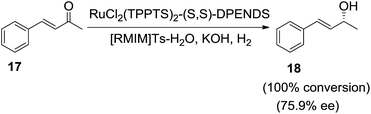
The authors observed that the addition of appropriate amounts of water can increase the ee value from 45.1% to 80.3%. The increase in the ee values can be attributed to the enhancement of the solubility of the water-soluble catalyst in ionic liquid. However, a lower activity and enantioselectivity are observed when only water was used as a solvent. The same research group65 further examined the asymmetric hydrogenation of α,β-unsaturated ketones catalyzed by the achiral ruthenium monophosphine complex RuCl2(TPPTS)2–(S,S)-DPENDS–KOH in [RMIM]Ts/water mixture (Scheme 7). Under the optimized conditions benzalacetone (17) afford the chiral alcohol (18) with 100% chemoselectivity and 75.9% ee. The addition of appropriate amount of water was not only beneficial to exerting the catalytic activity and chemoselectivity of the catalyst, but also helps improve the enantioselectivity, because water enhances the solubility of catalyst in ionic liquids. However, lower activity and chemoselectivity are observed when only water was used as solvent.
2.2 Oxidation
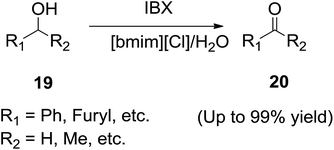
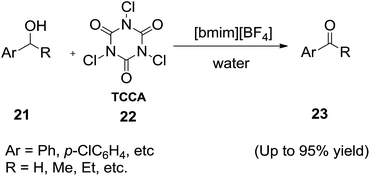
Ionic liquids are highly stable and have been evaluated as media for oxidation reactions.66 In 2003, Chen and co-workers67 reported a mild, efficient and eco-friendly procedure for the oxidation of alcohols with IBX in 1-butyl-3-methylimidazolium chloride ([bmim][Cl]) and water (Scheme 8). Simply stirring of a solution of the alcohol (19) and IBX in [bmim][Cl]/H2O at room temperature followed by extraction with ether or ethyl acetate and removal of the solvent gave the corresponding carbonyl compounds (20). Compared with the classical procedure in DMSO, this procedure in [bmim][Cl] and water shares the advantage of mild reaction conditions, homogeneous solution and facile recovery of the oxidant and recycling of the ionic liquid.Recently, Lee and his group68 demonstrated the oxidation reaction of benzylic alcohols (21) with trichloroisocyanuric acid (22) and 1-butyl-3-methylimidazolium tetrafluoroborate ([bmim][BF4]) in water at room temperature to afford the corresponding carbonyl compounds (23) in good yields (Scheme 9). The use of pure water as reaction medium, mildness of reaction conditions and operational simplicity made this method an important alternative to the other methods previously reported.
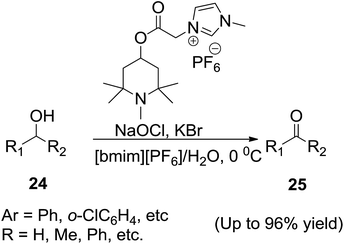
In 2005, Gao et al.69 reported the synthesis of a 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) radical bearing an ionic liquid and its use as catalyst for the metal-free, chemoselective oxidation of primary and secondary alcohols (24) to aldehydes and ketones (25) in aqueous-[bmim][PF6] biphasic conditions respectively (Scheme 10). The new IL-supported radical proved to be an efficient, selective and recoverable catalyst for the selective oxidation of alcohols and showed catalyst properties similar to non-supported TEMPO in terms of activity and selectivity.
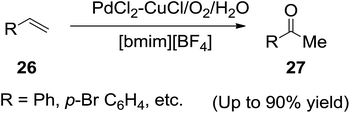
Ansari and co-workers70 in 2005, found that a combination of the ionic liquid ([bmim][BF4]) and water was an excellent solvent system for the Wacker oxidation of different types of olefins (26) to corresponding ketones (27) using the classical Pd/Cu catalysts under an oxygen atmosphere (Scheme 11). The ionic liquid [bmim][BF4] was selected for this study since it has an excellent miscibility with H2O. The best yields were observed when the IL/water ratio was 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 by volume. The Authors showed that under these conditions it was not necessary to add any acid, contrary to the classical Wacker process due to the hydrolysis of BF4− liberating HF during the reaction, which may act as the catalyst.
1 by volume. The Authors showed that under these conditions it was not necessary to add any acid, contrary to the classical Wacker process due to the hydrolysis of BF4− liberating HF during the reaction, which may act as the catalyst.
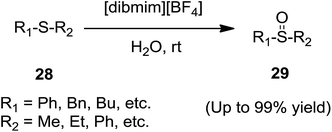
Qian and co-workers71 disclosed the chemoselective oxidation of sulfides (28) to sulfoxides (29) using 1-(4-diacetoxyiodobenzyl)-3-methylimidazolium tetrafluoroborate ([dibmim][BF4]) in water under mild conditions (Scheme 12). The addition of small amount of water to ionic liquid shortened the reaction time. Aliphatic and aromatic sulfides are selectively oxidized to the corresponding sulfoxides at room temperature in excellent yields.
2.3 Suzuki-cross coupling
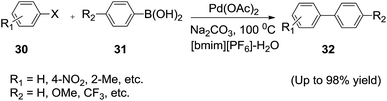
The Suzuki reaction is one of the most widely used catalytic carbon–carbon bond forming tools in organic synthesis. Palladium-catalyzed Suzuki cross-coupling reactions in ionic liquids have been reported. However water was found to have a remarkable rate accelerating effect on the Suzuki reaction in ionic liquids.In 2005, Zhang et al.72 successfully performed the Suzuki reaction in a mixture of IL/water by using palladium(II) acetate (Pd(OAc)2) as the catalyst in air (Scheme 13). The reactions of arylhalides (30) with arylboronic acids (31) using Pd(OAc)2 as the catalyst in mixture of IL/water provided the desired coupling products (32) in good yields. The addition of the water dramatically improves the reactivity of the Suzuki reaction in ILs. The best yield was obtained when the mass ratio of water and [bmim][PF6] was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and when the mass ratio of the [bmim][BF4] and water was 1
3 and when the mass ratio of the [bmim][BF4] and water was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The Pd(OAc)2–[bmim][PF6]–water system can be reused seven times only with a small deactivation of reactivity.
1. The Pd(OAc)2–[bmim][PF6]–water system can be reused seven times only with a small deactivation of reactivity.
 | ||
| Scheme 13 Suzuki cross-coupling reaction of aryl halides with phenylboronic acid in the mixture of [bmim][PF6]/water. | ||
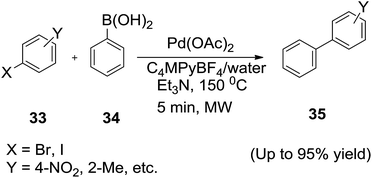
Recently, De Souza and co-workers73 reported the ionic liquid 1-butyl-4-methylpyridinium tetrafluoroborate (C4MPyBF4), derived from 4-picoline was a very efficient solvent when mixed with water for the Suzuki–Miyaura cross coupling promoted by microwave irradiation (Scheme 14).
The resulting phosphine-free catalytic system was able to activate both iodo and bromoarenes toward reaction conditions. Reactions between haloarenes (33) with phenylboronic acids (34) catalyzed by Pd(OAc)2 in the presence of C4MPyBF4/H2O system and triethylamine as base under microwave activation resulted the desired cross coupling products (35). The presence of the ionic liquid seems to be crucial because a drop in the catalytic activity was verified when water was used as the unique solvent. High performances obtained in C4MPyBF4/H2O system may stem from an increase in the fluidity of the reaction media allowing energy and mass transference between organic (substrates) and aqueous-ionic (catalytic system) and easy the work-up of the product.
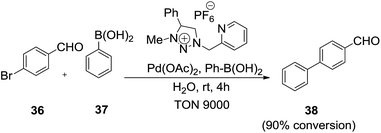
Later on, Kosmrlj et al.74 effectively used 1,4-disubstituted-3-methyl-1,2,3-triazolium salts as ionic liquids in palladium-catalyzed Suzuki–Miyaura catalysis in the environmentally benign solvent water (Scheme 15). The reaction of 4-bromo-benzaldehyde (36) and phenyl boronic acid (37) with catalyst Pd(OAc)2 and 1,4-disubstituted-3-methyl-1,2,3-triazolium salt in water at room temperature afford 4-phenylbenzaldehyde (38).
2.4 Diels–Alder reaction
Diels–Alder reactions conducted in the presence of an ionic liquid are known and quite effective.75–81 In 2011, Nino et al.82 investigated the MacMillan iminium catalyst for asymmetric Diels–Alder cycloadditions in methylpyridinium triflate ([mpy][OTf])/water homogeneous phase (Scheme 16).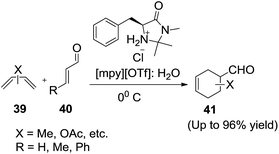 | ||
| Scheme 16 Diels–Alder reaction of different dienes and dienophiles using the chiral iminium salt in IL/water. | ||
Diels–Alder reactions between dienes (39) and dienophiles (40) were carried out at 0 °C, using MacMillan iminium in mixture of [mpy][OTf]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O to afford the corresponding products (41). Water plays a fundamental role in this reaction both as the co-solvent, allowing a homogeneous phase for transformations carried out in [mpy][OTf] and for mechanistic reasons in facilitating the iminium ion hydrolysis. The protocol provided good yields, selectivity and shorter reaction times in comparison with classical organic solvents.
H2O to afford the corresponding products (41). Water plays a fundamental role in this reaction both as the co-solvent, allowing a homogeneous phase for transformations carried out in [mpy][OTf] and for mechanistic reasons in facilitating the iminium ion hydrolysis. The protocol provided good yields, selectivity and shorter reaction times in comparison with classical organic solvents.
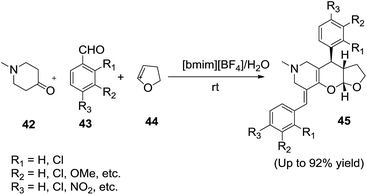
Recently, Siddiqui and co-workers83 developed the multicomponent one pot synthesis of furopyranopyridines (45) in IL/water mixture (Scheme 17). The synthetic strategy involves a reaction between N-methylpiperidinone (42), an aromatic aldehydes (43) and 2,3-dihydrofuran (44) using the ionic liquid [bmim][BF4] and water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) can afford the products (45) in good yields. The ionic liquid plays the dual role of solvent and catalyst in this transformation. When the reaction was carried out in water the reaction does not proceed at all. But when the reaction was performed in [bmim][BF4]/water in a 1
1) can afford the products (45) in good yields. The ionic liquid plays the dual role of solvent and catalyst in this transformation. When the reaction was carried out in water the reaction does not proceed at all. But when the reaction was performed in [bmim][BF4]/water in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio the best yield was obtained.
1 ratio the best yield was obtained.
 | ||
| Scheme 17 Multicomponent synthesis of furopyranopyridine using ionic liquid [bmim][BF4] and water in one-pot. | ||
2.5 Substitution reaction
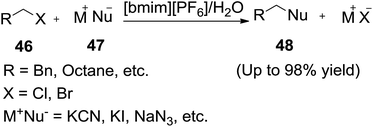
The nucleophilic substitution reaction is one of the important method for inserting functional groups into a carbon skeleton. Many nucleophilic substitution reactions have been found with enhanced reactivity and selectivity in ionic liquid.84 In 2003, Afonso and his co-workers85 demonstrated the nucleophilic substitution reactions of alkyl halide (46) with various nucleophiles (47) under aqueous-RTIL phase transfer conditions at room temperature (Scheme 18).The use of this biphasic aqueous-RTIL procedure, in opposition to the reported method seems particularly appealing, since it avoids the use of less friendly solvent DMSO.
Chi and co-workers86 reported a facile hydroxylation of haloalkanes (49) at the primary and secondary aliphatic and benzylic positions using water as a nucleophile in the presence of ionic liquids (Scheme 19).
The use of ionic liquid as an alternative reaction medium not only enhanced the nucleophilicity of water but also reduced the formation of elimination products predominantly formed under the conventional basic reaction conditions.
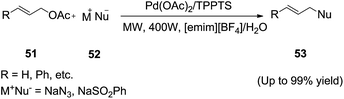
Later on, Liang et al.87 developed an efficient microwave assisted Tsuji–Trost reaction for allylic substitution with various carbon and heteronucleophiles catalyzed by Pd(OAc)2/TPPTS in an ethylmethylimidazolium tetrafluoroborate ([emim][BF4])/H2O system (Scheme 20). The reactions of allylic acetates (51) with various nucleophiles (52) catalyzed by palladium(0)-TPPTS in an IL/water medium via microwave irradiation are proceeded well to afford the corresponding products (53). The water plays presumably an important role in the process because of good water solubility to Pd(OAc)2 and TPPTS. The catalyst system was very easy to separate from the reaction product, so it can be used repeatedly.
 | ||
| Scheme 20 Tsuji–Trost reaction of allylic substrates with various nucleophiles in a mixture of IL/water. | ||
2.6 Reduction
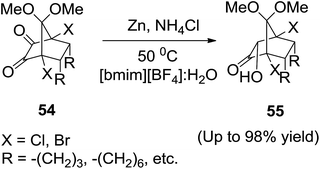
The most studied ionic liquids in reduction reactions have been simple salts of N,N′-dialkylimidazolium tetrafluoroborates or hexafluorophosphates, usually hydrophobic and immiscible with some organic solvents such as alkanes, isopropyl alcohol, and toluene. Khan and co-workers88 have investigated the efficient reduction of mono- and disubstituted norbornyl α-diketones (54) with zinc in [bmim][BF4]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (10
H2O (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to afford the corresponding acyloins (55) in excellent yields and diastereoselectivities (Scheme 21).
1) to afford the corresponding acyloins (55) in excellent yields and diastereoselectivities (Scheme 21).
In case of the monosubstituted norbornyl α-diketones, very high regioselectivities ranging from 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 to 100
10 to 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, in favor of diastereomer possessing endo-hydroxyl diagonal to endo-substituent were observed. It was necessary that water be used as a co-solvent as the reactions did not proceed in the absence of water. Between the two ionic liquids used, [bmim][BF4] was a better solvent as the reaction in [bmim][PF6] was sluggish be because [bmim][PF6] and water are immiscible.
0, in favor of diastereomer possessing endo-hydroxyl diagonal to endo-substituent were observed. It was necessary that water be used as a co-solvent as the reactions did not proceed in the absence of water. Between the two ionic liquids used, [bmim][BF4] was a better solvent as the reaction in [bmim][PF6] was sluggish be because [bmim][PF6] and water are immiscible.
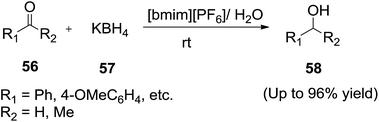
Li and co-workers reported89 the reduction reactions of a series of aldehydes and ketones (56) by potassium borohydride (57) in an IL/water ([bmim][PF6]/H2O) biphasic system to afford the corresponding alcohols (58) with high purity in excellent yields (Scheme 22). The IL/water biphasic system could promote the chemoselectivity and the substituents such as nitro group and chlorine remained intact. Aromatic ketones were not as active as aromatic aldhydes and cyclic ketones owing to their higher steric hindrance. The reduction in the IL/water biphasic system starts from the transport of the anion BH4− from the water phase by the onium cation of the ionic liquid either between the interface of the ionic liquid phase and water phase or slightly amount of ionic liquid dissolved in water.
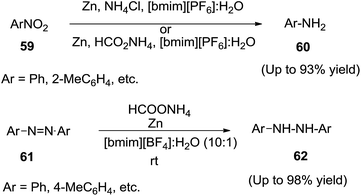
Khan and co-workers reported90 the chemoselective reduction of nitroarenes (59) to the corresponding amines (60) using zinc and aqueous ammonium salts in IL/water mixture as a safe and recyclable reaction medium (Scheme 23). The combination of Zn/NH4Cl in [bmim][PF6] or Zn/HCO2NH4 in [bmim][BF4] were the suitable conditions for the reduction of nitroarenes. Azobenzenes (61) were also smoothly reduced to hydrazobenzenes (62) with Zn/HCO2NH4 in recyclable [bmim][BF4] without any over reduction to the corresponding anilines. Use of 4 equivalents of Zn, 3 equivalents of ammonium salt in 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ionic liquid and water gave a clean reaction profile.
1 ionic liquid and water gave a clean reaction profile.
 | ||
| Scheme 23 Zinc-mediated reduction of aromatic nitro compounds and azobenzenes in mixture of IL/water. | ||
Nagaiah et al.91 demonstrated that [bmim][BF4]/water (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) system was an efficient reaction medium for one-pot reductive amination of carbonyl compounds (63) with sodium borohydride at ambient temperature (Scheme 24). Several aldimines obtained by the condensation of carbonyl compounds (63) with primary amines (64) yielded the desired products (65) in excellent yields. The role of water can be explained by the solubility of NaBH4 in the ionic medium. Furthermore, the reduction process also requires protic solvents such as alcohol or water.
1) system was an efficient reaction medium for one-pot reductive amination of carbonyl compounds (63) with sodium borohydride at ambient temperature (Scheme 24). Several aldimines obtained by the condensation of carbonyl compounds (63) with primary amines (64) yielded the desired products (65) in excellent yields. The role of water can be explained by the solubility of NaBH4 in the ionic medium. Furthermore, the reduction process also requires protic solvents such as alcohol or water.
2.7 Michael addition
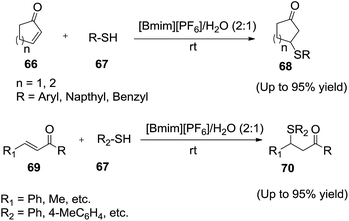
The Michael addition reaction is a one of the most important classes of new carbon–carbon bond-forming reactions for the preparation of organic target products in synthetic organic chemistry. Recently, room-temperature ionic liquids have been used as alternate catalysts to carry out Michael addition.In 2003, Yadav and co-workers reported92 the conjugate addition α,β-unsaturated ketones (66, 69) with thiols (67) rapidly in a hydrophobic ionic liquid [bmim][PF6]/H2O solvent system (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) in the absence of any acid catalyst to afford the corresponding Michael adducts (68, 70) in high to quantitative yields with excellent 1,4-selectivity under mild and neutral conditions (Scheme 25). Authors observed that, the combination of IL/water was found to be the most effective solvent system for this conversion. In this reaction, the efficiency of ionic liquid was strongly influenced by the nature of the anion.
1) in the absence of any acid catalyst to afford the corresponding Michael adducts (68, 70) in high to quantitative yields with excellent 1,4-selectivity under mild and neutral conditions (Scheme 25). Authors observed that, the combination of IL/water was found to be the most effective solvent system for this conversion. In this reaction, the efficiency of ionic liquid was strongly influenced by the nature of the anion.
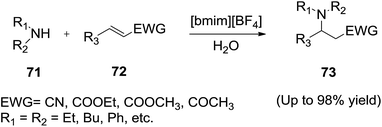
Thereafter, Xia et al.93 reported the efficient conjugate addition of aliphatic amines (71) to α,β-unsaturated compounds (72) catalyzed by simple quaternary ammonium salts and ionic liquids in the green solvent, water to afford the corresponding products (73) (Scheme 26). The feasibility of recovery and reuse of the catalyst in the water was also examined through a series of sequential Michael reactions and the recovered [bmim][BF4] catalyst was reused five times without loss of activity.
 | ||
| Scheme 26 Ionic liquid catalyzed aza-Michael reaction of aliphatic amines with α,β-unsaturated compounds in water. | ||
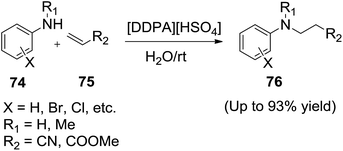
Recently, Lu et al.94 employed a functionalized ionic liquid, 3-(N,N-dimethyldodecylammonium) propanesulfonic acid hydrogen sulphate ([DDPA][HSO4]) as catalyst in water for the aza-Michael reactions of aromatic amines (74) with α,β-unsaturated compounds (75) at room temperature to produce β-amino compounds (76) in good yields (Scheme 27). The catalyst can be reused for several times without obvious loss of the catalytic activity.
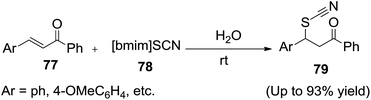
Yadav and co-workers reported95 the hydrothiocyanation of chalcones (77) with the task-specific ionic liquid (TSIL), 1-n-butyl-3-methylimidazolium thiocyanate ([bmim][SCN]) (78) in water to afford the corresponding products (79) (Scheme 28).
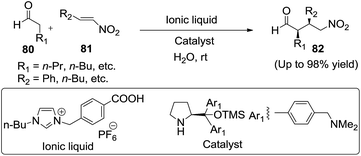
A highly efficient organocatalyst in combination with ILs-benzoic acid as a catalytic system for Michael additions of aldehydes (80) to nitroolefins (81) in water has been developed by Ni and his group96 (Scheme 29). A notable feature of this organocatalytic system was that the catalyst can be recycled more than 12 times without significant loss of enantioselectivity (Scheme 29).
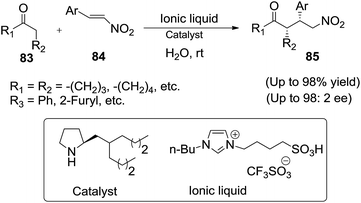
The same research group97 further examined another highly efficient asymmetric Michael addition of ketones and aldehyde (83) to nitroolefins (84) catalyzed by a water-compatible chiral pyrrolidine-based organocatalyst, in combination with ILs sulfonic acid in water (Scheme 30). The hydrophobicity of the catalyst forces it into close contact with hydrophobic reactants in water and excludes the reaction transition state from water.
2.8 Aldol reaction
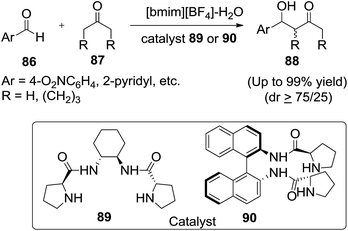
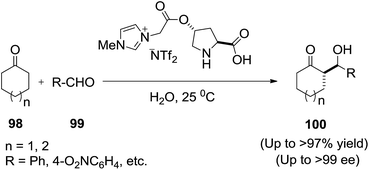
Asymmetric aldol condensation of unmodified aldehydes with ketones is used for the preparation of β-hydroxy carbonyl compounds, valuable synthons in organic synthesis. Asymmetric aldol reactions in the presence of water, is the most cost-effective and environment-friendly reaction medium.Kucherenko et al.98 reported the asymmetric aldol condensation of selected aldehydes (86) with ketones (87) catalyzed by 1(R),2(R)-bis((S)-prolinamido)cyclohexane (89) or (Rax)-2,2′-bis((S)-prolinamido)-1,1′-binaphtyl (90) in an IL/water system (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) proceeds with high reaction rate and diastereoselectivity (Scheme 31). The dependence of ee of the dominating anti-diastereomer of aldol on the percentage of water has a maximum at 50 vol%. Addition of water allows decreasing the consumption of expensive IL twofold and in some cases increasing the reaction rate.
1, v/v) proceeds with high reaction rate and diastereoselectivity (Scheme 31). The dependence of ee of the dominating anti-diastereomer of aldol on the percentage of water has a maximum at 50 vol%. Addition of water allows decreasing the consumption of expensive IL twofold and in some cases increasing the reaction rate.
In 2008, Zlotin and co-workers99 designed and synthesized a novel chiral (S)-proline-modified task-specific ionic liquid as an efficient recoverable organocatalyst for the direct asymmetric aldol reaction between cycloalkanones (91) and aromatic aldehydes (92) in the presence of water to afford aldol products (93) (Scheme 32). The catalyst retains its activity and selectivity over at least five reaction cycles.
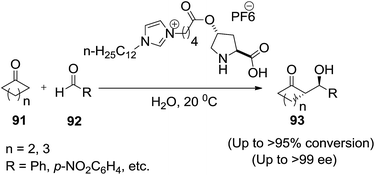 | ||
| Scheme 32 The catalytic asymmetric aldol reaction in the presence of water catalyzed by amphiphilic organocatalyst. | ||
The same research group100 further studied chiral ionic liquids bearing a prolinamide motif as organocatalysts in asymmetric aldol reactions in water (Scheme 33).
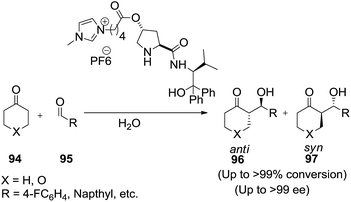 | ||
| Scheme 33 Aldol reactions between cycloalkanones and aromatic aldehydes in the presence of prolinamide derivatives in water. | ||
In the presence of a hydrophobic organocatalyst, cyclic ketones and methylketones (94) react with aromatic aldehydes (95) in the aqueous medium affording respective aldols (96, 97) in high yields and with excellent regio-, diastereo- and enantioselectivities. The reactions were run in excess water (100 equivalents relative to aldehyde) at 3–25 °C.
In 2008 Lombardo et al.101 reported imidazolium-tagged trans-4-hydroxy-L-proline, as an efficient catalyst for the asymmetric cross-aldol reaction in water. Reaction of cyclohexanones (98) and aromatic aldehydes (99) catalyzed by imidazolium-tagged trans-4-hydroxy-L-proline under aqueous biphasic conditions afford the aldols (100) with anti/syn ratios up to 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and ee (anti) up to 99% (Scheme 34).
2 and ee (anti) up to 99% (Scheme 34).
Han and co-workers102 employed a new functional IL (2-hydroxyethyl)-trimethyl-ammonium (S)-2-pyrrolidinecarboxylic acid salt ([Choline][Pro]) to catalyze direct aldol reactions between a variety of ketones (101) and aromatic aldehydes (102) efficiently in water to afford aldols (103) (Scheme 35).
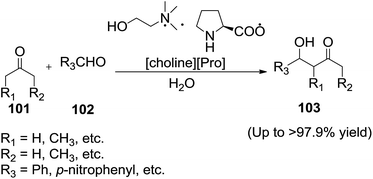 | ||
| Scheme 35 Direct aldol reactions of aromatic aldehydes and ketones catalyzed by [Choline][Pro] in water. | ||
The reaction mixtures separate into an aqueous phase and an organic phase after reaction. After simple separation and extraction, the IL-containing aqueous phase can be reused without any obvious decrease in activity. Therefore, using water as the solvent made the separation easier. The aldol reactions catalyzed by this IL can be finished in a very short time with good yields and there was no dehydrated product produced in most cases.
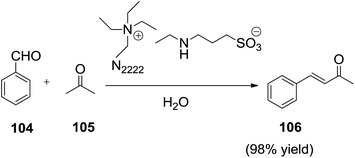
Recently, a green and efficient functionalized IL catalytic system was developed by Gao et al.103 to facilitate the aldol condensation reactions through the use of microwave heating technology (Scheme 36). Aldol reaction of benzaldehyde (104) and acetone (105) in the amine-functionalized tetraethylammonium ethylamino-propanesulfonic ionic liquid ([N2222][EtNHC3SO3]) offered the desired 4-phenylbut-3-en-2-one (106) in yield of 28% (Scheme 36). In contrast, when 50 wt% of H2O was added to the reaction system, aldol product was obtained in a conversion of more than 99% with high selectivity. Hence, all aldol condensation reactions in this work were carried out in a solution of 50 wt% IL and 50 wt% water. In addition, this catalytic system can be successfully extended to the Henry reactions.
2.9 Biocatalysis
Enzymatic reactions in ILs comprise another important category of environmentally benign processes.104–107 Recently, the use of ionic liquids as reaction media has been extended to biocatalytic processes.Lye and coworkers108 examined the use of a biphasic [bmim][PF6]/H2O medium for the conversion of 1,3-dicyanobenzene to 3-cyanobenzamide and 3-cyanobenzoic acid catalyzed by a nitrile hydratase contained in a whole cells of Rhodococcus R312. Stephens and co-workers109 conducted the hydrogenation of caffeate with the whole cells of Sporomusa termitida in a biphasic system 0.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 [bmim][PF6]/H2O. The yields were very low, suggesting that [bmim][PF6] became toxic for the cells. The feasibility of using isolated enzymes in ionic liquid media has also been demonstrated. Thus, Erbeldinger et al.110 reported the thermolysin-catalysed synthesis of Z-aspartame (109) from L-phenylalanine methyl ester hydrochloride (107) and carbobenzoxy-L-aspartate (108) in [bmim][PF6]/H2O (95/5, v/v) as shown in Scheme 37. To fully dissolve the reactants, it was essential to add the water to a suspension of ionic liquid.
1 [bmim][PF6]/H2O. The yields were very low, suggesting that [bmim][PF6] became toxic for the cells. The feasibility of using isolated enzymes in ionic liquid media has also been demonstrated. Thus, Erbeldinger et al.110 reported the thermolysin-catalysed synthesis of Z-aspartame (109) from L-phenylalanine methyl ester hydrochloride (107) and carbobenzoxy-L-aspartate (108) in [bmim][PF6]/H2O (95/5, v/v) as shown in Scheme 37. To fully dissolve the reactants, it was essential to add the water to a suspension of ionic liquid.
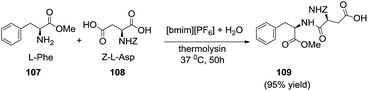 | ||
| Scheme 37 Thermolysin-catalyzed reaction of carbobenzoxy-L-aspartate and L-phenylalanine methyl ester hydrochloride in [bmim][PF6]/H2O. | ||
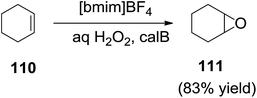
Bjorkling et al.111 developed the epoxidation of cyclohexene (110) by peroctanoic acid, generated in situ by Novozym 435-catalyzed reaction of octanoic acid with commercially available 60% aqueous hydrogen peroxide in [bmim][BF4], afforded cyclohexene oxide (111) in 83% yield in 24 h (Scheme 38). For comparison, a yield of 93% was observed in 24 h in acetonitrile.
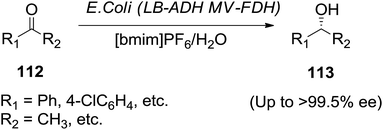
Brautigam and co-workers112 demonstrated the ionic liquids such as [bmim][PF6] and 1-butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ([bmim][NTf]) are good alternatives to organic solvents in biphasic biotransformation (Scheme 39). Asymmetric reductions of several prochiral ketones (112) were performed with the use of a recombinant Escherichia coli as biocatalyst in biphasic IL/water system to offered chiral alcohols (113).
2.10 Halogenation
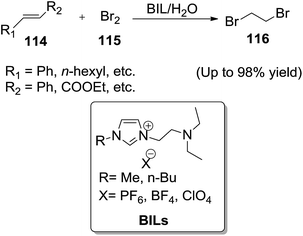
Primerano and co-workers113 developed a novel one-pot method for chemo- and diastereo-selective dibromination of electron-rich and electron-poor alkenes (114) with bromine (115) in water in the presence of amino-functionalized basic ionic liquids (BILs) to afford dibromo derivatives (116) (Scheme 40). Addition of water (ratio BIL/water = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) improved the reaction yield, likely making more homogeneous and less dense the reaction mixture. It was noteworthy that the water does not interfere on the final product, rather it accelerates the progress of the reaction.
3) improved the reaction yield, likely making more homogeneous and less dense the reaction mixture. It was noteworthy that the water does not interfere on the final product, rather it accelerates the progress of the reaction.
2.11 Halohydroxylation
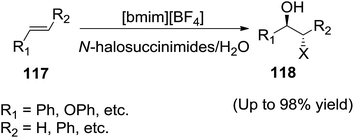
In 2005, Yadav and co-workers reported114 the use of IL/water system for bromo- and iodohydroxylation of olefins (117) with N-bromo- and N-iodosuccinimides, to produce the corresponding halohydrins (118) respectively in high to quantitative yields (Scheme 41). N-Halosuccinimides showed enhanced reactivity in ionic liquids thereby reducing the reaction times and improving the yields considerably. The ionic liquid plays a dual role as solvent and activator of the N-halosuccinimides.2.12 Dihydroxylation
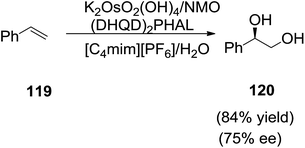
Afonso and co-workers reported the use of room-temperature ionic liquids (RTILs) in the sharpless catalytic asymmetric dihydroxylation (AD) as a cosolvent (Scheme 42).115 The asymmetric dihydroxylation of styrene (119) using cooxidant NMO and ligand (DHQD)2PHAL with catalyst K2OsO2(OH)4 under IL/water biphasic conditions afford the dihydroxy derivative (120). The AD reaction was faster in 1-n-butyl-3-methylimidazolium hexafluorophosphate ([C4mim][PF6]) as a co-solvent with higher yields and enantiomeric excess (ee) compared to the conventional solvent system. Due to high affinity of the catalytic osmium/quiral ligand system to the ionic liquid, the use of IL/water (biphasic) solvent systems provides a recoverable, reusable, robust, efficient, and simple system for the AD reaction.2.13 Resolution
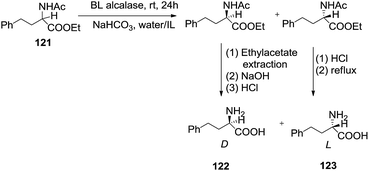
Zhao et al.116 developed the enzymatic resolution of amino acid ester (121) to obtain enantiomeric amino acid homophenylalanine (122, 123) (Scheme 43). The use of mixed solvents such as IL/water led to lower enantioselectivity than water alone. It was assumed that high concentration of ionic liquid in water denatures the enzyme and thus decreases the enantioselectivity.2.14 Epoxidation
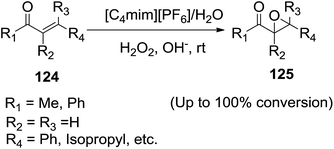
In 2003, Yang et al.117 conducted the epoxidation reactions of α,β-unsaturated carbonyl compounds (124) in [C4mim][PF6]/H2O biphasic system using hydrogen peroxide as an oxidant at room temperature (Scheme 44). Under optimized reaction conditions, unsaturated carbonyl compounds such as mesityl oxide, benzylideneacetone and chalcone have been almost completely converted to the corresponding epoxyketones (125). Compared with traditional organic solvent/H2O phase transfer catalyst (PTC) systems, [C4mim][PF6]/H2O biphasic catalytic system has exhibited excellent selectivity to epoxyketones.2.15 Allylation
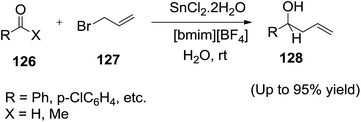
Li and co-workers118 disclosed an efficient SnCl2-mediated carbonyl allylation of allyl bromide (127) to aldehydes and ketones (126) in IL/water mixture (Scheme 45).By applying ionic liquid, some previously reported serious operational problems associated with the SnCl2-mediated allylation reaction are avoided. The small amount of water seemed to cause the hydrolysis of the Sn(IV)–Cl bonds to activate the allylic tin intermediate. The authors observed that ketones, which are less reactive than aldehydes, can also be allylated in high yields with this system. Furthermore, when H2O was employed as solvent instead of ionic liquid, no allylation for ketones occurred.
2.16 1,2,3 Triazole synthesis
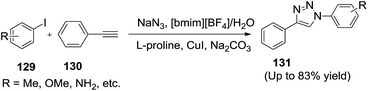
Liang et al.119 disclosed the IL/H2O was a good reaction medium for the one-pot synthesis of 1,4-disubstituted 1,2,3-triazoles using either halides at sp3-hybridized carbon atoms or halides at sp2-hybridized carbon atoms in good yields (Scheme 46). Reaction of various halides (129), sodium azide with terminal alkynes (130) in [bmim][BF4]/H2O afforded corresponding triazoles (131) in good to excellent yields. Better performance was obtained than that of conventional reaction condition. Authors observed that, using [bmim][BF4]/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) instead of [bmim]BF4, the product was obtained in excellent yield due to good water solubility of sodium azide.
1) instead of [bmim]BF4, the product was obtained in excellent yield due to good water solubility of sodium azide.
2.17 Prins reaction
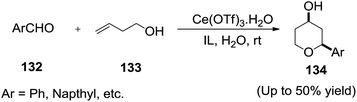
Li and co-workers120 reported a facile and direct formation of tetrahydropyranols (134) by the reaction of an aldehyde (132) and homoallyl alcohol (133) in the presence of a catalytic amount of cerium triflate, in IL/water media (Scheme 47).2.18 Pinacol coupling
Manchanayakage et al.121 investigated the electrochemical pinacol coupling reactions of aromatic carbonyl compounds (135) using an 80% [bmim][BF4]/H2O mixture as the electrolytic medium (Scheme 48). The corresponding diols (136, 137) were obtained in good to excellent yields with moderate diastereoselectivity. The authors observed that the combination of the ionic liquid ([bmim][BF4]) with the co-solvent water, resulted in the best yields. The conductivity and viscosity of RTILs can be changed by adding a co-solvent H2O. The viscosity for the 80% [bmim][BF4]/H2O mixture which gave the best yields was determined to be substantially lower than that of the pure ionic liquid. The conductivity for the 80% [bmim]BF4/H2O mixture was measured to be about ten times higher than the conductivity of pure ionic liquid.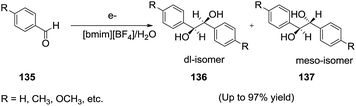 | ||
| Scheme 48 Electrochemical Pinacol coupling of aromatic carbonyl compounds in a [bmim][BF4]/H2O mixture. | ||
2.19 Beckmann rearrangement
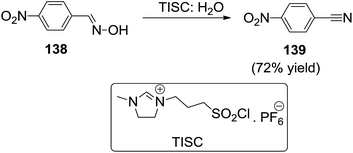
Sun et al.122 performed the Beckmann rearrangement of ketoximes (138) to the corresponding amine or cyanide (139) in a novel task-specific ionic liquid consisting sulfonyl chloride (TISC) (Scheme 49). Especially for the conversion of cyclohexanone oxime to ε-caprolactam, ε-caprolactam has good solubility in water while the task-specific ionic liquid was immiscible with water, therefore, ε-caprolactam could be easily separated from the reaction system by water extraction.2.20 Mannich reaction
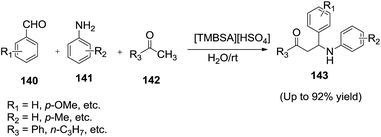
Zu-liang and co-workers123 developed an acyclic Bronsted acidic task-specific ionic liquid N,N,N-trimethyl-N-butanesulfonic acid ammonium hydrogen sulphate ([TMBSA][HSO4]) was an efficient catalyst for one-pot three-component Mannich reaction of aromatic aldehydes (140), aromatic amines (141) and ketones (142) at room temperature in water to afford β-amino carbonyl compounds (143) in good yields (Scheme 50).The products could simply be separated from the catalyst/water, and the catalyst could be reused at least 7 times without noticeably decreasing the catalytic activity.
2.21 Three component coupling reactions
The IL-three component coupling reactions are extremely useful for the rapid entry to highly functionalized heterocyclic molecules of potentials use in medicianl chemistry. Thus, Su et al.124 reported the three-component reactions of 4-hydroxycoumarin (144), aldehydes (145) and cyclic 1,3-dicarbonyl compounds (146) prompted by novel sulfonic acid functionalized ionic liquids 1,3-dimethyl-2-oxo-1,3-bis(4-sulfobutyl)imidazolidine-1,3-diium hydrogen sulfate ([DMDBSI]·2HSO4) in water at reflux temperature to provide a novel series of 10,11-dihydrochromeno[4,3-b]chromene-6,8(7H,9H)-dione derivatives (147) in high yields (Scheme 51).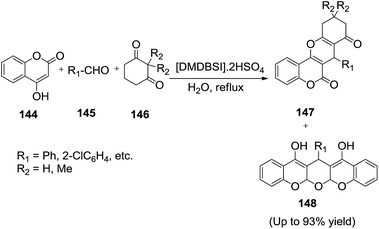 | ||
| Scheme 51 [DMDBSI]·2HSO4 promoted synthesis of 10,11-dihydrochromeno[4,3-b]chromene-6,8(7H,9H)-dione derivatives in H2O. | ||
Liu and co-workers125 synthesized the halogen-free acyclic task-specific ionic liquids (TSILs) such as N,N,N-triethyl-N-butanesulfonic acid ammonium hydrogen sulfate ([TEBSA][HSO4]), N,N,N-tributyl-N-propanesulfonic acid ammonium hydrogen sulfate ([TBPSA][HSO4]) and used an efficient catalysts for synthesis of 5-oxo-5,6,7,8-tetrahydro-4H-benzo[b]pyrans (152) by one-pot three-component condensation of aromatic aldehyde (149), malononitrile (or ethyl cyanoacetate) (150), and dimedone (or 1,3-cyclohexanedione) (151) in water (Scheme 52). After the reaction, the products could simply be separated from the catalyst/water and the catalyst could be reused at least 10 times without noticeably decreasing the catalytic activity.
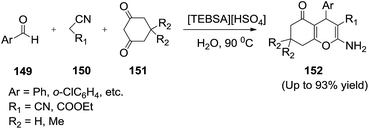 | ||
| Scheme 52 Synthesis of 5-oxo-5,6,7,8-tetrahydro-4H-benzo[b]pyrans catalyzed by acidic ionic liquids. | ||
Guo et al.126 described the ionic liquid 2-(hydroxyl)ethylammonium acetate (HEAA) catalyzed a three-component, one-pot condensation of quinone or ninhydrin (153), malononitrile/ethyl cyanoacetate (154) and 1,3-dicarbonyl compound/enol (155) in water to afford a spiropyran derivative (156). This method has advantages of mild reaction conditions, short reaction time and environmental friendliness (Scheme 53).
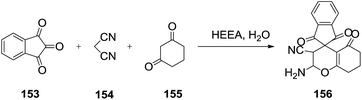 | ||
| Scheme 53 Ionic liquid catalyzed three-component synthesis of spiropyran derivatives reaction in water. | ||
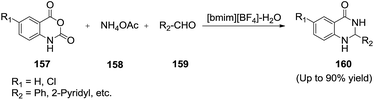
Su and co-workers127 synthesized a series of 2,3-dihydroquinazolin-4(1H)-ones (160) in high to excellent yields through one-pot three-component cyclocondensation of isatoic anhydrides (157), ammonium acetate (158) and aldehydes (159) in IL/water solvent system without the use of any additional catalyst (Scheme 54). The volume ratio of IL/water was examined and the best results were obtained by carrying out the reaction in [bmim][BF4]/H2O with a ratio of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (v/v). The desired product was obtained in good yield in IL/water solvent system instead of ionic liquid. The water presumably plays an important role in the process due to the good water solubility of ammonium acetate.
2 (v/v). The desired product was obtained in good yield in IL/water solvent system instead of ionic liquid. The water presumably plays an important role in the process due to the good water solubility of ammonium acetate.
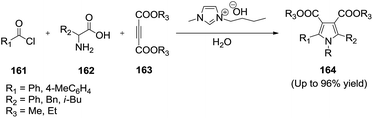
Yavari and co-workers128,129 demonstrated a basic functionalized ionic liquid, 1-butyl-3-methylimidazolium hydroxide ([bmim][OH]), catalyzed the three-component condensation reaction of acid chlorides (161), amino acids (162), and dialkyl acetylenedicarboxylates (163) in water to afford functionalized pyrroles (164) in high yields (Scheme 55). The [bmim][OH]/H2O system was found to be a much better catalytic medium for this reaction and the catalyst can be recycled for subsequent reactions without appreciable loss of efficiency.
 | ||
| Scheme 55 One-pot synthesis of pyrroles in aqueous media catalyzed by task-specific basic ionic liquid. | ||
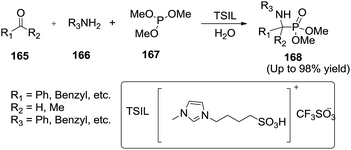
Heydari et al.130 developed a sulfonic acid functionalized ionic liquid (TSIL) catalyzed one-pot, three-component synthesis of α-aminophosphonates (168) from aldehydes or ketones (165), amines (166) and trimethyl phosphate (167) at room temperature in water (Scheme 56). This homogeneous catalytic procedure is simple and efficient and the catalyst can be reused at least six times without any noticeable decrease in catalytic activity. The authors found that the reaction in the absence of water did not take place, but in the presence of water the product formed rapidly.
2.22 Other organic reactions
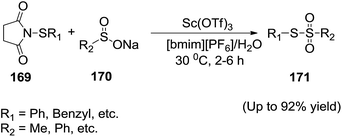
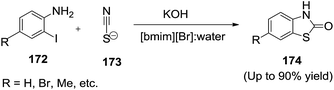
In 2012, Chen and co-workers131 developed the first example of Sc(OTf)3-catalyzed sulfenylation of sodium sulfinates (170) with N-(organothio)succinimides (169) in IL/water solvent system to achieve the thiosulfonates (171) in moderate to excellent yields (Scheme 57). They recovered Sc(OTf)3/IL easily after the reactions and reused without a significant loss in the catalytic activity. The water plays an important role in the reaction due to the good water solubility of sodium sulfinates.Siddiqui et al.132 achieved an expeditious, facile, ionic liquid catalyzed, water accelerated synthesis of substituted benzothiazole-2(3H)-one derivatives (174) from readily available 2-iodoanilines (172) and potassium thiocayanate (173) under basic conditions (Scheme 58). The strategy involves nucleophilic substitution forming N–C and S–C bond resulting the desired heterocyclic scaffold. The reactions proceeded smoothly at an ambient condition with high yield, using water a green reaction medium and basic 1-butyl-3-methylimidazolium bromide ([bmim][Br]) as recyclable catalyst system.
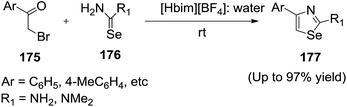
Srinivasan and co-workers133 established a highly efficient synthesis of 4-aryl-2-amino-1,3-selenazole (177) by the condensation of various phenacyl bromide (175) with selenourea (176) in 1-n-butylimidazolium tetrafluoroborate ([Hbim][BF4])/H2O solvent system (Scheme 59). The role of IL may be postulated in terms of Lewis/Brønsted acidity, thereby promoting the reaction. The addition of water to IL may presumably play an important role in the process due to the good water solubility of the selenoureas.
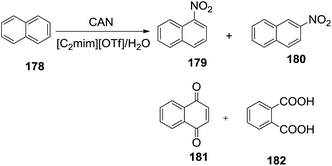
Parac-Vogt and co-workers134 reported the reaction of ceric ammonium nitrate ((NH4)2[Ce(NO3)6 or CAN), with naphthalene (178) in the ionic liquid 1-ethyl-3-methylimidazolium triflate ([C2mim][OTf]). The reaction products 1-nitronaphthalene (179), 2-nitronaphthalene (180), 1,4-naphthoquinone (181) and phthalic acid (182) are strongly dependent on the water content of the ionic liquid and that cerium(IV) in the ionic liquid can electrochemically be regenerated (Scheme 60).
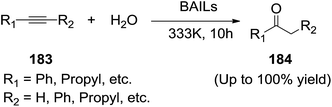
Srivastava and co-workers135 disclosed an efficient, non-toxic, noble metal free, and Bronsted acidic ionic liquid (BAIL) based economical route for the hydration reaction of alkynes (183) to afford ketones (184). N-SO3H functionalized BAIL ([SO3Hmim][Cl]) was found to be more active than the N-R-SO3H (R = alkyl/benzyl) functionalized BAILs. These reactions are easy to perform and the purification protocol was simple (Scheme 61).
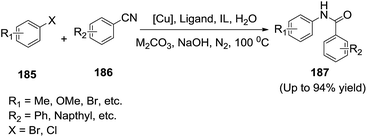
Recently, Pan et al.136 employed an efficient copper catalyzed tandem reaction for the synthesis of benzanilide (187) in water. Ionic liquid was used as a phase transfer catalyst and this method could be applied in the N-arylation between benzonitriles (186) and aryl halides (185) (or alkenyl bromides) (Scheme 62). Advantages of this reaction include the use of water as the environmental friendly solvent, short reaction time and the tolerance of various functional groups.
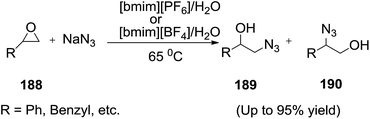
In 2005, Yadav et al.137 proved an IL/water solvent system was an effective reaction medium for the synthesis of 2-azidoalcohols (189, 190) from epoxides (188) by playing the dual role of solvent as well as promoter. The epoxides showed a significant increase in reactivity thereby reducing the reaction times and improving the yields substantially (Scheme 63). The combination of IL/water as solvent system (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was found to be an effective reaction media for this conversion.
1) was found to be an effective reaction media for this conversion.
Recently same group138 examined the use of [bmim][PF6]/H2O or [bmim][BF4]/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
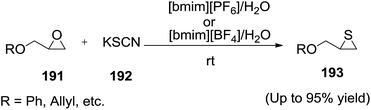
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent system as an alternative reaction medium for the synthesis of episulfides (193) from epoxides (191) with potassium thiocyanate (192) (Scheme 64). The ionic liquid plays a dual role of solvent as well as catalyst. The use of ionic liquids for this transformation avoids the use of heavy metal halides as promoters and chlorinated hydrocarbons as solvents. The ionic liquid was recycled in five to six subsequent runs with gradual decrease in activity.
1) solvent system as an alternative reaction medium for the synthesis of episulfides (193) from epoxides (191) with potassium thiocyanate (192) (Scheme 64). The ionic liquid plays a dual role of solvent as well as catalyst. The use of ionic liquids for this transformation avoids the use of heavy metal halides as promoters and chlorinated hydrocarbons as solvents. The ionic liquid was recycled in five to six subsequent runs with gradual decrease in activity.
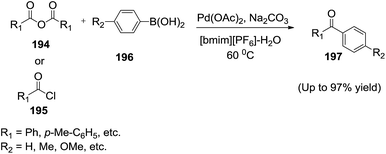
Zhang and co-workers139 developed the palladium acetate-catalyzed coupling reaction of carboxylic anhydride (194) or acyl chloride (195) with aryl boronic acid (196) in water in the presence of [bmim][PF6] to gave high yields of ketones (197) without the use of phosphine ligands. The Pd(OAc)2–H2O–[bmim][PF6] catalytic system can be recovered and reused eight times with high efficiency for both carboxylic anhydride and acyl chloride (Scheme 65). The ratio of water and [bmim]PF6 acted on the reactivity and best yield was obtained when the ratio of [bmim][PF6] and water achieved 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5.
2.5.
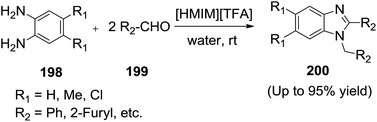
Dabiri et al.140 employed a new procedure for the rapid and selective synthesis of 2-aryl-1-arylmethyl-1H-1,3-benzimidazoles (200) from ortho phenylenediamines (198) and aldehydes (199) in the presence of 1-methylimidazoluim triflouroacetate ([HMIM][TFA]) as a Bronsted acidic ionic liquid (BAIL) at room temperature (Scheme 66). The authors observed that in the presence of water an inconceivable decrease in the reaction time was observed.
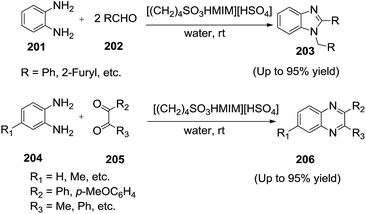
Beheshtiha and co-workers141 reported the ionic liquid 1-(4-sulfonic acid)butyl-3-methylimidazolium hydrogen sulphate ([(CH2)4SO3HMIM][HSO4]) catalyzed reaction of o-phenylenediamine (201) with different aromatic aldehydes (202) and also the reaction of 1,2-diaminoarene derivatives (204) with 1,2-diketones (205) to synthesis 1,2-disubstituted benzimidazoles (203) and quinoxalines (206) in water, respectively (Scheme 67).
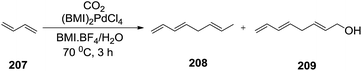
In 1998, Dupont et al.142 investigated the hydrodimerization of 1,3-butadiene (207) catalyzed by bis(1-butyl-3-methylimidazolium) tetrachloropalladate ([BMI]2PdCl4) in 1-n-butyl-3-methylimidazolium tetrafluoroborate (BMI·BF4) and water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). In addition to the dimer, 1,3,6-octatriene (208), 2,7-octadienol (209) was also produced (Scheme 68). The products selectivity and catalytic activity depend on the reaction conditions. 1,3-Butadiene conversion up to 28%, a turnover frequency (TOF) of 118 h−1, and a selectivity of 94% on telomer were achieved with (BMI)2PdCl4 dissolved in BMI·BF4. The 1,3-butadiene conversion and TOF were significantly increased to 49% and 204 h−1, respectively, by a 5 atm pressure of carbon dioxide. The recovered ionic catalyst solution can be reused several times without any significant changes in the catalytic performance.
1). In addition to the dimer, 1,3,6-octatriene (208), 2,7-octadienol (209) was also produced (Scheme 68). The products selectivity and catalytic activity depend on the reaction conditions. 1,3-Butadiene conversion up to 28%, a turnover frequency (TOF) of 118 h−1, and a selectivity of 94% on telomer were achieved with (BMI)2PdCl4 dissolved in BMI·BF4. The 1,3-butadiene conversion and TOF were significantly increased to 49% and 204 h−1, respectively, by a 5 atm pressure of carbon dioxide. The recovered ionic catalyst solution can be reused several times without any significant changes in the catalytic performance.
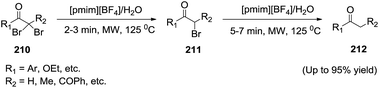
Chattopadhyay and co-workers143 developed novel application of ionic liquid for selective debromination of gem-α-dibromoketones (210) to monobromo (211) and debromoketones (212) by proper control of reaction time (Scheme 69). The use of 1-methyl-3-pentylimidazolium tetrafluoroborate ([pmim][BF4]) in water was also found suitable for the dehalogenation of α-halo ketones and esters and of vic-bromoacetals.
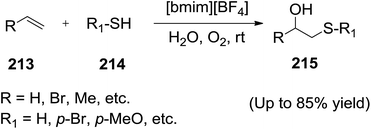
Kamal et al.144 demonstrated the direct one-pot synthesis of β-hydroxysulfides (215) from alkenes (213) and thiophenols (214) in the presence of aerial oxygen using a mixture of ionic liquid [bmim][BF4] and water under neutral conditions (Scheme 70). The thiophenol attacks the alkene in an anti-Markonikov manner resulting in the formation of β-hydroxysulfides in a mixture of IL/water in presence of oxygen. This protocol tolerates a wide variety of functional groups or substrates and does not require the use of either acid or base catalysts. Ionic liquid can be recovered and reused for a number of runs with negligible loss of its activity.
 | ||
| Scheme 70 One-pot synthesis of β-hydroxysulfides from terminal olefins in a mixture of [bmim][BF4] and water. | ||
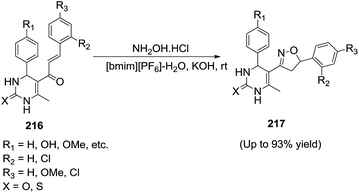
Rahatgaonkar et al.145 accomplished the convenient synthesis of a library of pyrimidine–isoxazoline hybrids (217) in [bmim][PF6]–water/potassium hydroxide at ambient temperature (Scheme 71). The IL/water biphasic system proved to be effective at enhancing the efficiency of the reaction. The ionic liquid [bmim][PF6], immiscible in water, can be easily recycled for reuse after separation of the products without any noticeable diminution in its activity. The ionic liquid [bmim][PF6] stabilized the hydrophobic reactant, and water stabilized hydroxylamine hydrochloride. The reaction presumably occurs at the junction of the two immiscible phases.
 | ||
| Scheme 71 Convenient synthesis of a library of pyrimidine–isoxazoline hybrids in [bmim][PF6]/water biphasic system. | ||
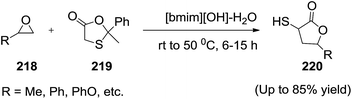
Yadav and his group146 reported the task-specific ionic liquid 1-butyl-3-methyl imidazolium hydroxide ([bmim][OH]) promoted one-pot synthesis of α-mercapto-γ-lactones (Scheme 72). The present protocol involves regioselective epoxide ring opening and intramolecular translactonisation cascade. A variety of epoxides (218) undergo this ring-opening-ring-closing cascade with 2-methyl-2-phenyl-1,3-oxathiolan-5-one (219) to afford α-mercapto-γ-lactones (220) diastereoselectively in good to excellent yields. After isolation of the product, the ionic liquid [bmim][OH] could be easily recovered and reused without any loss of efficiency.
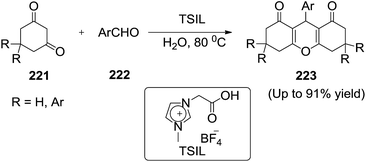
Salunkhe et al.147 prepared 1,8-dioxo-octahydroxanthenes (223) efficiently from 1,3-cyclohexanediones (221) and aldehydes (222) by a recyclable carboxyl-functionalized 1-carboxymethyl 3-methylimidazolium tetrafluoroborate ([cmmim][BF4]) task specific ionic liquid [TSIL] in aqueous media in high yield. The ionic liquid can be recycled at least 6 times without significant loss of activity (Scheme 73). The best results were obtained in presence of 20 mol% TSIL and water as solvent.
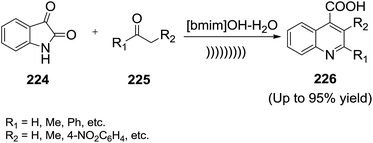
Kowsari and co-workers148 reported the basic ionic liquid (BIL) catalyzed condensation reaction of isatin (224) with ketones (225) by ultrasonic irradiation in aqueous media for quinoline (226) synthesis (Scheme 74). The reaction was favoured in IL/water medium compared other solvent mixtures. When two different α-protons are available in a ketone, a mixture of two quinolines was obtained.
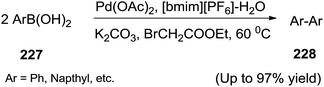
Zhang et al.149 developed the homocoupling reaction of the arylboronic acids (227) in a mixture of IL/water in the presence of ethyl bromoacetate ester using Pd(OAc)2 as catalyst to afford biaryls (228) in high yield (Scheme 75). The separation of desired product was easily performed by extraction with diethyl ether and Pd(OAc)2–[bmim][PF6] can be reused eight times accompanied with only a slight decrease in activity. It was observed that the addition of water clearly enhanced the reactivity of the homocoupling reaction and an optimum yield was obtained when the ratio of water and [bmim][PF6] was 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.
3.
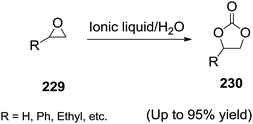
Sun et al.150 reported a novel method for the synthesis of cyclic carbonates in IL/water. By tuning the amount of water, cycloaddition of CO2 to epoxides (229) in aqueous medium leads to cyclic carbonates (230) with moderate to excellent yields and high selectivities (Scheme 76). The presence of water could remarkably improve the activity of ionic liquids by which the turnover frequency of the reaction is about 4–5 times higher in the presence than in the absence of water. In this reaction, water played a similar function like a Lewis acid on the ring-opening of epoxide, by which the Lewis base could show excellent activity in the absence of Lewis acid.
3 Conclusions
The analysis of the available literature presented above demonstrates that mixtures of IL/water have shown a remarkable degree of predictability in many organic reactions. The formation of mixtures of IL/water makes it easy to control the properties of the solution. The challenge of separation of the reaction products from ILs can be overcome with the use of IL/water biphasic system. In addition, IL/water solvent system requires less amount of ionic liquid compared with their pure form which may affect the economic viability. We anticipate that this novel reaction media will be of broad interest and opened new vistas in the field of organic chemistry.Acknowledgements
Narayana Reddy thanks to DST-SERB, India for a Start-Up Research Grant., Padmaja thankful to DST, India for Inspire Faculty Fellowship.References
- T. Welton, Chem. Rev., 1999, 99, 2071–2084 CrossRef CAS PubMed.
- A. E. Visser, R. P. Swatloski and R. D. Rogers, Green Chem., 2001, 2, 1–4 RSC.
- R. Sheldon, Chem. Commun., 2001, 2399–2407 RSC.
- N. Meine, F. Benedito and R. Rinaldi, Green Chem., 2010, 12, 1711–1714 RSC.
- S. Ahrens, A. Peritz and T. Strassner, Angew. Chem., Int. Ed., 2009, 48, 7908–7910 CrossRef CAS PubMed.
- T. Welton and P. Wasserscheid, Ionic Liquids in Synthesis, Wiley-VCH, Weinheim, 2008 Search PubMed.
- R. Ludwig, Angew. Chem., Int. Ed., 2011, 40, 1808–1827 CrossRef.
- M. Freemantle, An Introduction to Ionic Liquids, RSC Publishing, UK, 2010 Search PubMed.
- J. S. Wilkes and M. J. Zaworotko, Chem. Commun., 1992, 965–967 RSC.
- J. G. Huddleston, H. D. Willauer, R. P. Swatloski and R. D. Rogers, Chem. Commun., 1998, 1765–1766 RSC.
- R. C. Buijsman, E. V. Vuuren and J. G. Serrenburg, Org. Lett., 2001, 3, 3785–3787 CrossRef CAS PubMed.
- J. M. Fraile, J. I. Garcia, C. I. Herrerias, J. A. Mayoral and M. Carrie, Tetrahedron: Asymmetry, 2002, 12, 1891–1894 CrossRef.
- M. M. Anna, V. Gallo, P. Mastrorilli, C. F. Nobile, G. Romanazzi and G. P. Suranna, Chem. Commun., 2002, 434–435 Search PubMed.
- P. Kubisa, Prog. Polym. Sci., 2004, 29, 3–12 CrossRef CAS PubMed.
- P. Kubisa, J. Polym. Sci., Part A: Polym. Chem., 2005, 43, 4675–4683 CrossRef CAS PubMed.
- M. L. Dietz and J. A. Dzielawa, Chem. Commun., 2001, 2124–2125 RSC.
- L. A. Blanchard, D. Hancu, E. J. Beckman and J. F. Brennecke, Nature, 1999, 399, 28–29 CrossRef.
- L. A. Blanchard and J. F. Brennecke, Ind. Eng. Chem. Res., 2001, 40, 287–292 CrossRef CAS.
- S. G. Kazarian, B. J. Briscoe and T. Welton, Chem. Commun., 2000, 2047–2048 RSC.
- D. W. Armstrong, L. K. Zhang, L. He and M. L. Gross, Anal. Chem., 2001, 73, 3679–3686 CrossRef CAS.
- D. W. Armstrong, L. He and Y. S. Liu, Anal. Chem., 1999, 71, 3873–3876 CrossRef CAS.
- A. Berthod, H. He and D. W. Armstrong, Chromatographia, 2001, 53, 63–68 CAS.
- M. Vaher, M. Koel and M. Kaljurand, Electrophoresis, 2002, 23, 426–430 CrossRef CAS.
- E. G. Yanes, S. R. Gratz, M. J. Baldwin, S. E. Robinson and A. M. Stalcup, Anal. Chem., 2001, 73, 3838–3844 CrossRef CAS.
- D. L. Compton and J. A. Laszlo, J. Electroanal. Chem., 2002, 520, 71–78 CrossRef CAS.
- B. M. Quinn, Z. Ding, R. Moulton and A. J. Bard, Langmuir, 2002, 18, 1734–1742 CrossRef CAS.
- P. Wasserscheid, Nature, 2006, 439, 797–798 CrossRef CAS PubMed.
- M. Freemantle, Chem. Eng. News, 2002, 80, 38–39 Search PubMed.
- B. C. Ranu and S. Banerjee, J. Org. Chem., 2005, 70, 4517–4519 CrossRef CAS PubMed.
- Ionic liquids in Synthesis, ed. P. Wasserscheid and P. Welton, Wiley-VCH, West Sussex, UK, 2003 Search PubMed.
- Ionic Liquids: Applications and Perspectives, ed. A. Kokorin, InTech-ope, New York, 2011 Search PubMed.
- Ionic Liquids: Industrial Applications for Green Chemistry, ed. R. D. Roger and K. R. Seddon, ACS Symposia, ACS publications, Washington DC, 2002 Search PubMed.
- Ionic Liquids in Biotransformations and Organocatalysis, ed. P. Dominguez de Maria, Wiley-VCH, West Sussex, UK, 2012 Search PubMed.
- N. Isambert, M. Duque, J. Plaquevent, Y. Genisson, J. Rodriguez and T. Constantieux, Chem. Soc. Rev., 2011, 40, 1347–1357 RSC.
- A. R. Hajipoura and F. Rafiee, J. Iran. Chem. Soc., 2009, 6, 647–678 CrossRef.
- Y. H. Moon, S. M. Lee, S. H. Ha and Y. Koo, Korean J. Chem. Eng., 2006, 23, 247–263 CrossRef CAS.
- R. Singh, M. Sharma, R. Mamgain and D. S. Rawat, J. Braz. Chem. Soc., 2008, 19, 357–379 CrossRef CAS PubMed.
- H. Zhao and S. V. Malhotra, Aldrichimica Acta, 2002, 35, 75–83 CrossRef CAS.
- C. Baudequin, D. Bregeon, J. Levillain, F. Guillen, J. Plaqueventb and A. Gaumont, Tetrahedron: Asymmetry, 2005, 16, 3921–3945 CrossRef CAS PubMed.
- R. Martínez-Palou, J. Mex. Chem. Soc., 2007, 51, 252–264 Search PubMed.
- A. D. Headley and B. Ni, Aldrichimica Acta, 2007, 40, 107–117 Search PubMed.
- A. J. Carmichael, M. J. Earle, J. D. Holbrey, P. B. Mccormac and K. R. Seddon, Org. Lett., 1999, 1, 977–979 CrossRef.
- R. Ludwig, Angew. Chem., Int. Ed., 2011, 40, 1808–1827 CrossRef.
- J. D. Holbrey and K. R. Seddon, J. Chem. Soc., Dalton Trans., 1999, 2133–2140 RSC.
- K. Seddon, A. Stark and M. Torres, Pure Appl. Chem., 2000, 12, 2275–2287 Search PubMed.
- U. Schroder, J. D. Wadhawan, R. G. Compton, F. Marken, P. A. Z. Suarez, C. S. Consorti, R. F. D. Souza and J. Dupont, New J. Chem., 2000, 24, 1009–1015 RSC.
- K. R. Seddon, A. Stark and M. J. Torres, Pure Appl. Chem., 2000, 72, 2275–2287 CrossRef CAS.
- K. A. Fletcher and S. Pandey, J. Phys. Chem. B, 2003, 107, 13532–13539 CrossRef CAS.
- M. G. Freire, P. J. Carvalho, A. M. Fernandes, I. M. Marrucho, A. J. Queimada and J. A. P. Coutinho, J. Colloid Interface Sci., 2007, 314, 621–630 CrossRef CAS PubMed.
- R. A. Brown, P. Pollet, E. Mckoon, Ch. A. Eckert, Ch. L. Liotta and P. G. Jessop, J. Am. Chem. Soc., 2001, 123, 1254–1255 CrossRef CAS.
- B. Pugin, M. Studer, E. Kuesters, G. Sedelmeier and X. Feng, Adv. Synth. Catal., 2004, 346, 1481–1486 CrossRef CAS PubMed.
- A. Wolfson, I. F. J. Vankelecom and P. A. Jacobs, Tetrahedron Lett., 2005, 46, 2513–2516 CrossRef CAS PubMed.
- M. Berthod, J. M. Joerger, G. Mignani, M. Vaultier and M. Lemaire, Tetrahedron: Asymmetry, 2004, 15, 2219–2221 CrossRef CAS PubMed.
- C. Baudequin, J. Baudoux, J. Levillain, D. Cahard, A. C. Gaumont and J. C. Plaquevent, Tetrahedron: Asymmetry, 2003, 14, 3081–3093 CrossRef CAS.
- P. A. Schweitzer, Handbook of Separation Techniques for Chemical Engineers, McGraw-Hill, New York, 1996 Search PubMed.
- H. Niedermeyer, J. P. Hallett, I. J. Villar-Garcia, P. A. Hunt and T. Welton, Chem. Soc. Rev., 2012, 41, 7780–7802 RSC.
- C. D. Tran, S. H. D. Paolilacerda and D. Oliveira, Appl. Spectrosc., 2003, 57, 152–157 CrossRef CAS PubMed.
- Y. Kohno and H. Ohno, Chem. Commun., 2012, 48, 7119–7130 RSC.
- P. G. Jessop, R. R. Stanley, R. A. Brown, C. A. Eckert, C. L. Liotta, T. T. Ngo and P. Pollet, Green Chem., 2003, 5, 123–128 RSC.
- A. Wolfson, I. F. J. Vankelecomb and P. A. Jacobs, Tetrahedron Lett., 2005, 46, 2513–2516 CrossRef CAS PubMed.
- B. Pugin, M. Studer, E. Kuesters, G. Sedelmeier and X. Feng, Adv. Synth. Catal., 2004, 346, 1481–1486 CrossRef CAS PubMed.
- N. Ferlin, M. Courty, S. Gatard, M. Spulak, B. Quilty, I. Beadham, M. Ghavre, A. Hai, K. Kummerer, N. Gathergood and S. Bouquillon, Tetrahedron, 2013, 69, 6150–6161 CrossRef CAS PubMed.
- P. J. Dyson, D. J. Ellis and T. Welton, Can. J. Chem., 2001, 79, 705–708 CrossRef CAS.
- J. Wang, J. Feng, R. Qin, H. Fu, M. Yuan, H. Chen and X. Li, Tetrahedron: Asymmetry, 2007, 18, 1643–1647 CrossRef CAS PubMed.
- J. Wang, R. Qin, H. Fu, J. Chen, J. Feng, H. Chen and X. Li, Tetrahedron: Asymmetry, 2007, 18, 847–851 CrossRef CAS PubMed.
- P. Bonhote, A. P. Dias, N. Papageorgiou, K. Kalyanasundaram and M. Gratzel, Inorg. Chem., 1996, 35, 1168–1178 CrossRef CAS PubMed.
- Z. Liu, Z. C. Chen and Q. Zheng, Org. Lett., 2003, 5, 3321–3323 CrossRef CAS PubMed.
- J. C. Lee, J. Kim, S. B. Lee, S. Chang and Y. J. Jeong, Synth. Commun., 2011, 41, 1947–1951 CrossRef CAS PubMed.
- X. E. Wu, L. Ma, M. Ding and L. Gao, Synlett, 2005, 607–610 CAS.
- I. A. Ansari, S. J. M. K. Gupta, J. S. Yadav and R. Greeb, Tetrahedron Lett., 2005, 46, 7507–7510 CrossRef CAS PubMed.
- W. Qian and L. Pei, Synlett, 2006, 709–712 CrossRef CAS PubMed.
- B. Xin, Y. Zhang, L. Liu and Y. Wang, Synlett, 2005, 3083–3086 CAS.
- K. L. Castro, P. G. Lima, L. S. M. Miranda and R. O. M. A. De Souza, Tetrahedron Lett., 2011, 52, 4168–4171 CrossRef PubMed.
- A. Bolje and J. Kosmrlj, Org. Lett., 2013, 15, 5084–5087 CrossRef CAS PubMed.
- A. G. Avent, P. A. Chaloner, M. P. Day, K. R. Seddon and T. Welton, J. Chem. Soc., Dalton Trans., 1994, 3405–3413 RSC.
- H. Joshua, H. Keith, F. Darren and M. Paul, Tetrahedron Lett., 1997, 38, 3097–3100 CrossRef.
- A. J. Arduengo III, R. L. Harlow and M. Kline, J. Am. Chem. Soc., 1991, 113, 361–363 CrossRef.
- A. J. Arduengo III, H. V. R. Dias, R. L. Harlow and K. Michael, J. Am. Chem. Soc., 1992, 114, 5530–5534 CrossRef.
- D. V. Jarikote, S. A. Siddiqui, R. Rajagopal, T. Daniel, R. J. Lahoti and K. V. Srinivasan, Tetrahedron Lett., 2003, 44, 1835–1838 CrossRef CAS.
- R. Rajagopal, D. V. Jarikote, R. J. Lahoti, T. Daniel and K. V. Srinivasan, Tetrahedron Lett., 2003, 44, 1815–1817 CrossRef CAS.
- A. R. Gholap, K. Venkatesan, T. Daniel, R. J. Lahoti and K. V. Srinivasan, Green Chem., 2003, 5, 693–696 RSC.
- A. D. Nino, O. Bortolini, L. Maiuolo, A. Garofalo, B. Russo and G. Sindona, Tetrahedron Lett., 2011, 52, 1415–1417 CrossRef PubMed.
- I. R. Siddiqui, S. Shamim, M. A. Waseem, A. Srivastava and Rahila, RSC Adv., 2013, 3, 14423–14427 RSC.
- Y. R. Jorapur and D. Y. Chi, Bull. Korean Chem. Soc., 2006, 27, 345–354 CrossRef CAS.
- M. T. Nuno, O. Lourenc and C. A. M. Afonso, Tetrahedron, 2003, 59, 789–794 CrossRef.
- D. W. Kim, D. J. Hong, J. W. Seo, H. S. Kim, H. K. Kim, C. E. Song and D. Y. Chi, J. Org. Chem., 2004, 69, 3186–3189 CrossRef CAS PubMed.
- M. Liao, X. Duan and Y. Liang, Tetrahedron Lett., 2005, 46, 3469–3472 CrossRef CAS PubMed.
- F. A. Khan and Ch. Sudheer, ARKIVOC, 2009, 222–228 CrossRef CAS.
- H. Luo and Y. Li, Chin. J. Chem., 2005, 23, 345–348 CrossRef CAS PubMed.
- F. A. Khan, J. Dash, C. H. Sudheer and R. K. Gupta, Tetrahedron Lett., 2003, 44, 7783–7787 CrossRef CAS PubMed.
- K. Nagaiah, V. N. Kumar, R. S. Rao, B. V. S. Reddy, A. V. Narsaiah and J. S. Yadav, Synth. Commun., 2006, 36, 3345–3352 CrossRef CAS PubMed.
- J. S. Yadav, B. V. S. Reddy and G. Baishya, J. Org. Chem., 2003, 68, 7098–7100 CrossRef CAS PubMed.
- L. Xu, J. Li, S. Zhou and C. Xia, New J. Chem., 2004, 28, 183–184 RSC.
- X. Liu, M. Lu, T. Lu and G. Gu, J. Chin. Chem. Soc., 2010, 57, 1221–1226 CrossRef CAS PubMed.
- L. D. S. Yadav, R. Patel, V. K. Rai and V. P. Srivastava, Tetrahedron Lett., 2007, 48, 7793–7795 CrossRef CAS PubMed.
- D. Sarkar, R. Bhattarai, A. D. Headley and B. Ni, Synthesis, 2011, 1993–1997 CAS.
- Q. Chen, Y. Qiao and B. Ni, Synlett, 2013, 839–842 Search PubMed.
- A. S. Kucherenko, D. E. Syutkin and S. G. Zlotin, Russ. Chem. Bull., 2008, 57, 591–594 CrossRef CAS.
- D. E. Siyutkin, A. S. Kucherenko, M. I. Struchkova and S. G. Zlotin, Tetrahedron Lett., 2008, 49, 1212–1216 CrossRef CAS PubMed.
- D. E. Siyutkin, A. S. Kucherenko and S. G. Zlotin, Tetrahedron, 2010, 66, 513–518 CrossRef CAS PubMed.
- M. Lombardo, F. Pasi, S. Easwar and C. Trombini, Synlett, 2008, 2471–2474 CrossRef CAS PubMed.
- S. Hu, T. Jiang, Z. Zhang, A. Zhu, B. Han, J. Song, Y. Xie and W. Li, Tetrahedron Lett., 2007, 48, 5613–5617 CrossRef CAS PubMed.
- C. Wang, J. Liu, W. Leng and Y. Gao, Int. J. Mol. Sci., 2014, 15, 1284–1299 CrossRef PubMed.
- U. Kragl, M. Eckstein and N. Kaftzik, Curr. Opin. Biotechnol., 2002, 13, 565–571 CrossRef CAS.
- F. V. Rantwijk and R. A. Sheldon, Chem. Rev., 2007, 107, 2757–2785 CrossRef PubMed.
- C. Roosen, P. Muller and L. Greiner, Appl. Microbiol. Biotechnol., 2008, 81, 607–614 CrossRef CAS PubMed.
- R. A. Sheldon, R. M. Lau, M. J. Sorgedrager, F. V. Rantwijk and K. R. Seddon, Green Chem., 2002, 4, 147–151 RSC.
- S. G. Cull, J. D. Holbrey, V. Vargas, K. R. Seddon and G. J. Lye, Biotechnol. Bioeng., 2000, 69, 227–233 CrossRef CAS.
- A. Lenourry, J. M. Gardiner and G. Stephens, Biotechnol. Lett., 2005, 27, 161–165 CrossRef CAS PubMed.
- M. Erbeldinger, M. Mesiano and A. J. Russell, Biotechnol. Prog., 2000, 16, 1129–1131 CrossRef CAS PubMed.
- F. Bjorkling, H. Frykman, S. E. Godtfredsen and O. Kirk, Tetrahedron, 1992, 48, 4587–4592 CrossRef CAS.
- S. Brautigam, S. Bringer and D. Weuster, Tetrahedron: Asymmetry, 2007, 18, 1883–1887 CrossRef PubMed.
- P. Primerano, M. Cordaro and A. Scala, Tetrahedron Lett., 2013, 54, 4061–4063 CrossRef CAS PubMed.
- J. S. Yadav, B. V. S. Reddy, G. Baishya, S. J. Harshavardhan, C. H. J. Chary and M. K. Gupta, Tetrahedron Lett., 2005, 46, 3569–3572 CrossRef CAS PubMed.
- L. C. Branco and C. A. M. Afonso, J. Org. Chem., 2004, 69, 4381–4389 CrossRef CAS PubMed.
- H. Zhao, R. G. Luo and S. V. Malhotra, Biotechnol. Prog., 2003, 19, 1016–1018 CrossRef CAS PubMed.
- B. Wang, Y. Kang, L. Yang and J. Suo, J. Mol. Catal. A: Chem., 2003, 203, 29–36 CrossRef CAS.
- L. Tang, L. Ding, W. Chang and J. Li, Tetrahedron Lett., 2006, 47, 303–306 CrossRef CAS PubMed.
- Y. Zhao, Z. Yan and Y. Liang, Tetrahedron Lett., 2006, 47, 1545–1549 CrossRef CAS PubMed.
- C. C. K. Keh, V. V. Namboodiri, R. S. Varma and C. Li, Tetrahedron Lett., 2002, 43, 4993–4996 CrossRef CAS.
- H. Kronenwetter, J. Husek, B. Etz, A. Jones and R. Manchanayakage, Green Chem., 2014, 16, 1489–1495 RSC.
- J. Gui, Y. Deng, Z. Hua and Z. Sun, Tetrahedron Lett., 2004, 45, 2681–2683 CrossRef CAS PubMed.
- F. Dong, L. Jun, Z. Li and L. Zu-Liang, Catal. Lett., 2007, 116, 76–80 CrossRef.
- Z. Chen, Q. Zhu and W. Su, Tetrahedron Lett., 2011, 52, 2601–2604 CrossRef CAS PubMed.
- D. Fang, H. Zhang and Z. Liu, J. Heterocycl. Chem., 2010, 47, 63–67 CAS.
- S. Jin, M. Ding and H. Y. Guo, Heterocycl. Commun., 2013, 19, 139–143 CrossRef CAS.
- J. Chen, W. Su, H. Wu, M. Liub and C. Jin, Green Chem., 2007, 9, 972–975 RSC.
- I. Yvari and E. Kowsari, Synlett, 2008, 897–899 CrossRef PubMed.
- I. Yavari and E. Kowsari, Mol. Diversity, 2009, 13, 519–528 CrossRef CAS PubMed.
- J. Akbari and A. Heydari, Tetrahedron Lett., 2009, 50, 4236–4238 CrossRef CAS PubMed.
- G. Liang, J. Chen, J. Chen, W. Li, J. Chen and H. Wu, Tetrahedron Lett., 2012, 53, 6768–6770 CrossRef CAS PubMed.
- M. A. Waseem, Shireen, A. Srivastava, A. Srivastava and R. I. R. Siddiqui, J. Saudi Chem. Soc., 2014 Search PubMed , in press, corrected proof, DOI: 10.1016/j.jscs.2014.05.006.
- T. M. Potewar, S. A. Ingale and K. V. Srinivasan, ARKIVOC, 2008, 117–125 CrossRef CAS.
- K. Deleersnyder, S. Schaltin, J. Fransaer, K. Binnemans and T. N. Parac-Vogt, Tetrahedron Lett., 2009, 50, 4582–4586 CrossRef CAS PubMed.
- R. Kore and R. Srivastava, Tetrahedron Lett., 2012, 53, 3245–3249 CrossRef CAS PubMed.
- J. Wang, X. Yin, J. Wu, D. Wu and Y. Pan, Tetrahedron, 2013, 69, 10463–10469 CrossRef CAS PubMed.
- J. S. Yadav, B. V. S. Reddy, B. Jyothirmai and M. S. R. Murty, Tetrahedron Lett., 2005, 46, 6559–6562 CrossRef CAS PubMed.
- J. S. Yadav, B. V. S. Reddy, C. H. Srinivas Reddy and K. Rajasekhar, J. Org. Chem., 2003, 68, 2525–2527 CrossRef CAS PubMed.
- B. Xin, Y. Zhang and K. Cheng, J. Org. Chem., 2006, 71, 5725–5731 CrossRef CAS PubMed.
- M. Dabiri, P. Salehi, M. Baghbanzadeh and M. S. Nikcheh, Synth. Commun., 2008, 38, 4272–4281 CrossRef CAS PubMed.
- Y. S. Beheshtiha, M. M. Heravi, M. Saeedi, N. Karimi, M. Zakeri and N. Hossieni, Synth. Commun., 2010, 40, 1216–1223 CrossRef CAS PubMed.
- J. E. L. Dullius, P. A. Z. Suarez, S. Einloft, R. F. Souza, J. Dupont, J. Fischer and A. D. Cian, Organometallics, 1998, 17, 815–819 CrossRef CAS.
- B. C. Ranu, K. Chattopadhyay and R. Jana, Tetrahedron, 2007, 63, 155–159 CrossRef CAS PubMed.
- A. Kamal, D. R. Reddy and Rajendar, J. Mol. Catal. A: Chem., 2007, 272, 26–30 CrossRef CAS PubMed.
- K. Lanjewar, A. Rahatgaonkar, M. Chorghade and B. Saraf, Synthesis, 2011, 2644–2650 CAS.
- R. Patel, V. P. Srivastava and L. D. S. Yadav, Synlett, 2010, 1797–1802 CAS.
- A. Kumbhar, S. Kamble, G. Rashinkar, K. Mote and R. Salunkhe, Arch. Appl. Sci. Res., 2010, 2, 235–239 CAS.
- E. Kowsari and M. Mallakmohammadi, Ultrason. Sonochem., 2011, 18, 447–454 CrossRef CAS PubMed.
- K. Cheng, B. Xin and Y. Zhang, J. Mol. Catal. A: Chem., 2007, 273, 240–243 CrossRef CAS PubMed.
- J. Sun, J. Ren, S. Zhang and W. Cheng, Tetrahedron Lett., 2009, 50, 423–426 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |