Albumin stabilized silver nanoparticles–clotrimazole β-cyclodextrin hybrid nanocomposite for enriched anti-fungal activity in normal and drug resistant Candida cells
Abstract
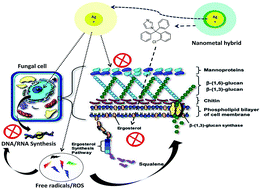
Nanotechnology unlocked distinctive platforms to move inside a hybrid therapeutic zone. Currently, nano-metal technology is the targeted field with exceptional advantages. Exceptionally small size and dominance of the surface properties such as high surface charge has raised a great deal of interest. This work is designed to exploit an interesting mechanistic feature i.e. multiple therapeutic targets possessed by metal nanoparticles. In this study we selected silver nanoparticles (AgNPs), which possess well documented anti-fungal activity and a standard antifungal molecule i.e. “clotrimazole”. A hybrid of AgNPs and clotrimazole was aimed to tackle clotrimazole resistance. Clotrimazole was firstly included into a β-cyclodextrin cavity to render it water soluble; subsequently the drug loaded dextrin moiety is functionalized on the surface of bovine serum albumin (BSA) stabilized silver nanoparticles using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide/N-hydroxysulfosuccinimide (EDC/NHS) chemistry. BSA stabilisation was essential to shield the physical interaction of AgNPs with the clotrimazole β-cyclodextrin complex that is otherwise experienced. Spectral and morphological characterization of the complex assures the synthesis of a hybrid metal complex. A cellular toxicity assay was performed to determine the toxic nature of the hybrid. This hybrid was then evaluated for its fungicidal activity on normal and clotrimazole resistant Candida cells. The toxicity and efficacy outcomes revealed a potent profile with a handy therapeutic window. Mechanistic explanations for this hybrid nature were supported by aggravated apoptotic cell percentage and reactive oxygen species production in both resistant and non-resistant cells. Cell cycle arrest studies further revealed G2/M phase cell cycle arrest, directing towards compromised cell membrane and DNA synthesis process equivalently in clotrimazole resistant cells.

 Please wait while we load your content...
Please wait while we load your content...