The molecular mechanism of conformational changes of the triplet prion fibrils for pH†
Abstract
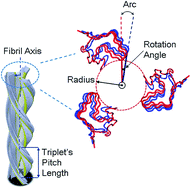
The HET-s prion fibril, which is found in the filamentous fungus Podospora anserina, exhibits conformational changes due to variations in pH. Here, we explain the effects of changing pH on the conformational changes of fibrils through the fundamental eigenmodes of the fibrils, in particular the torsional and bending modes, using a parameter free elastic network model. In particular, the motion resulting from these fundamental eigenmodes is found to be very similar to the conformational changes stimulated by pH variations as shown in previous experimental results. Finally, we calculated the mechanical properties of the triplet prion fibrils to elucidate its variations in the infectious state.

 Please wait while we load your content...
Please wait while we load your content...