High strength chitosan rod prepared via LiOH/urea solvent through centrifugation induced orientation processing
Jingyi Nie†
ab,
Zhengke Wang†*ab,
Jiazhen Zhangab,
Ling Yangab,
Yichuan Pangab and
Qiaoling Hu*ab
aMOE Key Laboratory of Macromolecular Synthesis and Functionalization, Department of Polymer Science and Engineering, Zhejiang University, Hangzhou 310027, China. E-mail: wangzk@zju.edu.cn; huql@zju.edu.cn
bJoint Laboratory for Adsorption and Separation Materials of Zhejiang University-Zhejiang Tobacco Industry Co. Ltd, Zhejiang University, Hangzhou 310027, China
First published on 4th August 2015
Abstract
Chitosan material is a promising candidate for bioabsorbable internal fixation devices, owing to its biocompatibility, biodegradability and versatility in orthopedic treatment. However, mechanical strength of existing chitosan rod materials is still unsatisfactory. In this study, chitosan rods with excellent mechanical performance had been prepared via a novel solvent, i.e. LiOH/urea solvent. The bending strength of chitosan rod prepared via LiOH/urea solvent could reach 450.2 MPa, which is over 300% higher than chitosan rods prepared via acidic solvent. Reasons behind the high bending strength of chitosan rods could be summarized in two aspects. Firstly, the gelation process of chitosan LiOH/urea solution is distinct from that of traditional acidic chitosan solution, which endows the material with homogeneous network structure. Secondly, due to the state of macromolecules in the solution, centrifugation processing can generate flow orientation in the material. Resulted from unique characteristics of chitosan LiOH/urea solution, the improvement of strength had made the novel chitosan rod a promising candidate of biomedical device for bone fracture internal fixation.
Introduction
Bioabsorbable internal fixation devices are favourable in applications where only a temporary presence of the implant is needed. Because of their degradability, there is no need for a removal operation after healing of tissues, which is preferred financially and physically.1–3 Although various biocompatible and biodegradable polymers are now available, bioabsorbable devices are essentially made of polylactic acid (PLA) and polyglycolic acid (PLGA).4,5 But problems like acidic degradation and aseptic inflammation remain to be solved.6–9 So it is significant to develop strong and safe bioabsorbable material for internal fixation.Chitosan (CS), a polysaccharide obtained from the deacetylation of chitin, had received considerable attention for its intrinsic properties: biocompatibility, bacteriostatic effects and abundance in nature.10–14 CS was also considered as biodegradable since it was metabolized by certain human enzymes, e.g. lysozyme. Moreover, CS materials were suitable for cell ingrowth and osteoconduction, and had been proved a versatile biopolymer in orthopedic treatment.15–19 These properties made CS a potential internal fixation material.
Three-dimensional CS rods which could be used for bone fracture internal fixation had been prepared by in situ precipitation method. Moreover, biodegradation and biocompatibility of these materials have been explored.20–25 Apart from properties mentioned above, adequate initial mechanical strength was also required. The bending strength of pure CS rods made by in situ precipitation method was approximately 92.4 MPa, which was lower than the quota for internal fixation of bone fracture in clinical application.26 In order to improve the mechanical performance of CS rods, various strategies had been employed, including 60Co irradiation,27 modification by reinforcements, etc.17,26,28 However, the introduction of crosslink bonds or auxiliary substance may jeopardize the biocompatibility and biodegradability of CS rods. Thus, it is challenging to prepare high strength CS rods without aforesaid disadvantages.
Pure CS cannot be manufactured by screw extruder, since the multiple hydrogen-bonds between macromolecular chains made melt extrusion unsuitable. Thus, CS solution was very important for the production of CS materials. So far, solvent used in the preparation of CS rods was mainly dilute acetic acid aqueous solution.20,22,29 LiOH/urea aqueous solution was a non-toxic solvent system, which had been applied in the preparation of cellulose and chitin materials. The dissolve mechanism had been explored: inclusion complexes (ICs) of macromolecules LiOH and urea were formed by self-assembly in the solution, which was induced by hydrogen bonds at low temperature.30
CS can also be dissolved in LiOH/urea solvent.31,32 Hydrogel prepared via this system showed high strength, which was over ten times higher than hydrogel prepared via acidic solvent.33 This highlighted the possibility of preparing novel high strength CS rods via LiOH/urea solvent. Moreover, no crosslinker was needed in the preparation, which was favoured in biomedical applications.34 In this work, three-dimensional high strength CS rods were prepared via LiOH/urea solvent. And the mechanism of strength improvement was also explored in the following section.
Experimental section
1. Materials
CS was prepared in our laboratory by heterogeneous N-deacetylation from commercial chitin (Zhejiang Gold Shell Pharmaceutical Co., Ltd). The average viscosity molecular weight (Mη) of CS was 1.12 × 106 Da and degree of deacetylation (DD) was 94.4%. Fluorescein iso-thiocyanate (FITC) was purchased from Sigma Chemical Co. (USA). FITC–CS was prepared following the process reported in ref. 35. Lithium hydroxide, urea and acetic acid were purchased from Sinopharm Chemical Reagent Co., Ltd.2. Preparation of CS rods via LiOH/urea solvent
LiOH·H2O and urea were dissolved in deionized water to form a transparent solution with weight percentage of 4.8 wt% and 8.0 wt% respectively. The solution was used as solvent in the preparation of CS material (referred to as the LiOH/urea solvent). A certain amount of dried CS powder was mixed in the solvent. The mixture was treated by a freeze (−40 °C)-thawing (20 °C) process three times to get transparent CS LiOH/urea solution.A polypropylene centrifuge tube (35 mL) was used as mold in the preparation of CS rod. The solution was treated by centrifugation for 8 min at 4 °C, with a rotation speed of 8000 rpm. Then the solution was held for 2 h at 35 °C and turned to CS hydrogel rod containing LiOH and urea. The rod was fully washed in deionized water to remove LiOH and urea after the mold was unloaded. The detailed washing procedure of chitosan hydrogel rod was described as follows. The gel rod was washed with substantial water. The water in the bath was replaced when the pH of washing bath reached 14. The washing procedure was monitored by a pH meter (PHS-3C, INESA Scientific Instrument Co., Ltd). The procedure ended when the pH of washing bath reached the pH of deionized water and did not increase anymore. The hydrogel rod was then moved into fresh deionized water and immersed for 3 days. The hydrogel rod was then air-dried in oven at 60 °C. The centrifuge used in this study was Biofuge stratos from Thermo Fisher Scientific Inc., and the angle of rotor was 45°.
For samples without centrifugation were prepared as followed. The solution was transferred into an identical mold after centrifugation. The transformation eliminated the influence on solution caused by centrifugation. Then the solution was let to stand for sufficient time at 4 °C. The deaeration process ensured no air bubbles were trapped in the viscous solution. The rest of the preparation was the same as samples with centrifugation.
3. Mechanical properties
Three-point bending tests were performed to determine the mechanical properties of CS rods. The span length was 40 mm and loading rate was 2 mm min−1. The ultimate bending strength (σb) was calculated according to eqn (1)
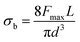 | (1) |
4. Morphology of CS gel and dry rods
The fracture surface of CS rod was observed by scanning electron microscopy (SEM). Samples were air-dried in oven at 60 °C for 6 h to remove the remaining moisture, and then gold-sprayed for conductance. HITACHI S-4800 SEM was used in this study.Hydrogel samples were prepared using FITC–CS for the fluorescent observation. Hydrogel sample via acetic acid aqueous solvent was prepared following the process reported in ref. 36. While hydrogel samples via LiOH/urea solvent was prepared according to Section 2 (with centrifugation process). Samples were studied by confocal laser scanning microscope (CLSM, Leica TCS SP5).
The effect of centrifugation was observed from the gel state of CS rod by CLSM. The samples were observed immediately after gelation.
5. Study of aggregates in the CS LiOH/urea solution
For the exploration of aggregates in the CS LiOH/urea solution, 220 nm microporous membranes were used to treat CS LiOH/urea solution and LiOH/urea solvent. c(CS) (the CS concentration of solution) was 2 wt%. CS solution was transparent and well dissolved. The membranes were then dried at ambient temperature. The top side of membrane was observed by SEM.Results and discussion
1. Mechanical properties of CS rods
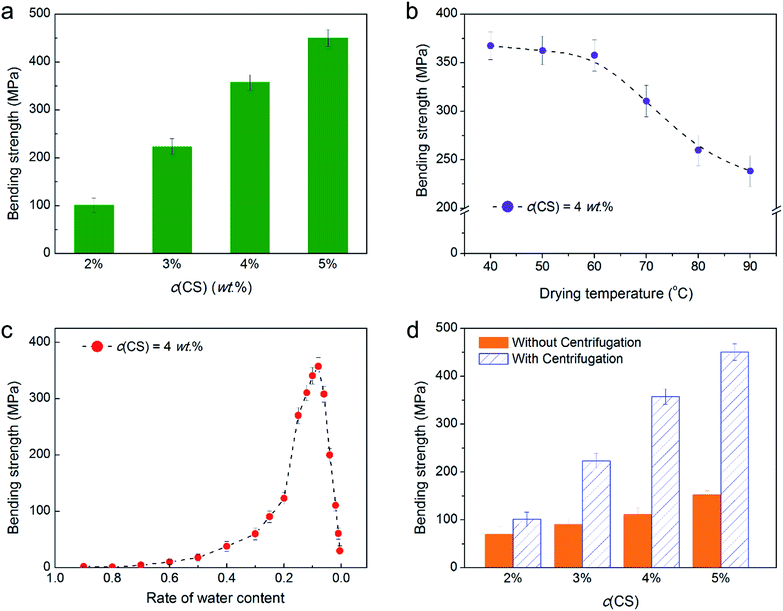
Fig. 1 showed the CS hydrogel rod and dried rod prepared via LiOH/urea solvent. Mechanical tests showed that the bending strength of rod was over 100 MPa for c(CS) from 2 wt% to 5 wt% (Fig. 2a). For CS rod prepared by conventional method, high c(CS) was required due to the preparation procedure. When c(CS) was 4 wt%, the bending strength of conventional CS rod was 80.1 MPa.37 While for CS rod prepared via LiOH/urea solvent, the bending strength was 357.6 MPa. When c(CS) was 5 wt%, the bending strength of corresponding rod was 450.2 MPa, which was over 300% higher than CS rods prepared via acidic solvent (92.4 MPa). This demonstrated convincingly it was possible to prepare high performance CS rod via LiOH/urea solvent. | ||
| Fig. 1 CS rod prepared via LiOH/urea solvent system: (a) hydrogel rod, c(CS) was 4 wt%; (b) dried rod, rate of water content was 0.08. | ||
Relationships between bending strength and preparation conditions had been explored to optimize the quality of CS rods. When c(CS) increased from 2 wt% to 5 wt%, the bending strength of CS rod increased from 101.1 MPa to 450.2 MPa (Fig. 2a). This indicated higher c(CS) was favoured in the view of strength. However, when c(CS) was over 6 wt%, the solution was too viscous to process. So for CS material with Mη of 106, c(CS) of solution should be in the range of 2–5 wt%.
The bending strength of CS rod was also affected by the drying temperature as shown in Fig. 2b. When the drying temperature increased from 40 °C to 90 °C, the bending strength decreased monotonically. The decrease appeared to be more apparent at higher temperature range (70 °C to 90 °C). This could be explained by solvent diffusion in the drying process. Accompanied by desolvation, swollen macromolecules started to shrink. At high drying temperature, this process firstly happened at the surface of CS hydrogel rod, quickly forming a collapsed compact skin layer. The surface skin formation prompted the accumulation of internal hydrodynamic pressure, because outflux of solvent was blocked. If the stress inside the hydrogel was too high, polymer networks physically tear upon shrinking, leading to cracks.38 So a mild temperature range was required, although higher drying temperature guaranteed shorter drying time.
In the drying process, the bending strength of CS rod increased with the reduction of water content, and reached a maximum when the rate of water content is 0.08 (Fig. 2c). The bending strength showed a dramatic decrease when the rate of water content continued to decrease. This was because water molecules could bond to polysaccharide macromolecules, and interactions between water and CS resulted in the strengthening of the hydrogen bond network. When water content in CS rod was too low, inter-/intra-molecular hydrogen bonds are affected by the loss of bond water.39 On the other hand, intermediate water in the material acted as a plasticizer. The loss of water could increase the brittleness of material. As a result, the rate of water content should be 0.08 to 0.1 to provide optimized initial mechanical strength.
Fig. 2d showed that CS rods with centrifugation process had higher bending strength than those without centrifugation. Since defects induced by air bubbles had been excluded, this indicated that centrifugation contributed largely to the high performance of CS rod. Moreover, this contribution became more important with the rise of c(CS). For material that underwent centrifugation, fracture surface of CS rod showed striated pattern (Fig. 3a), which became more distinct with the rise of c(CS) (Fig. 3b). However, CS rods without centrifugation showed no organized pattern (Fig. 3c and d). This indicated that centrifugation indeed exerted influence on the structure of CS rod.
2. Mechanism of the high bending strength
CS rods prepared via LiOH/urea solvent showed remarkable improvement in mechanical property than traditional CS rods. This was closely related to the bulk structure of the material, which was fundamentally determined by the bulk structure of CS hydrogel. CS hydrogels prepared from acetic acid aqueous solution and LiOH/urea solvent showed distinctions in bulk structure, which were pore size and homogeneity of the hydrogel structure (Fig. 4). Firstly, pore size of hydrogel via LiOH/urea system was about 1 μm (Fig. 4d), while that in hydrogel via acidic solution was over 10 μm (Fig. 4b). Secondly, hydrogel via LiOH/urea system exhibited uniform network structure, while larger voids randomly located in the hydrogel via acidic solution. Homogeneity of structure led to uniform distribution of loading. Besides, uniform network reduced structural defects in the hydrogel. In conclusion, resulted from the novel LiOH/urea solvent system, the homogenous network structure laid the foundation of high mechanical performance of CS rod. | ||
| Fig. 4 CLSM images of FITC–CS hydrogel: (a and b) prepared via 2 vol% acetic acid aqueous solution; (c and d) prepared via LiOH/urea solvent. | ||
Apart from bulk structure of the material, it had been demonstrated that the centrifugal field contributed to the high bending strength. This could be explained by the unique state of macromolecules in the solution. In the solution, LiOH and urea acted as protective covers around macromolecules, and prevented their self-association. Macromolecules existed as single IC and aggregation of ICs.30 In dilute cellulose LiOH/urea solution, single ICs co-exist and interact with aggregations of ICs. The distribution of particle size in the solution can be altered by changing polymer concentration and temperature. If the solution become more concentrated or the temperature becomes higher, volume and portion of aggregations become larger.30
Due to the structural resemblance between cellulose and CS macromolecules,40 one could speculated similar state of solute in CS LiOH/urea solution. Although aggregations in cellulose solution had been observed,30,41 the existence of CS aggregations had not been explored sufficiently. Dynamic light scattering had been employed in the study of aggregates in dilute and semi-dilute CS LiOH/urea solution.42 However, for the concentrated CS solution for the preparation of CS rods, light scattering was not effective due to the complexity of solution. So another approach was adapted in the exploration of aggregates.
Filter membrane with pore size of 220 nm was used to intercept aggregates in the solution. Flower-like micro-structures were observed on the top side of membrane, and the micro-structures were isolated from each other (Fig. 5b–e). One flower-like micro-structure consisted of crystals (LiOH and urea) and filamentous polymer component (CS) (Fig. 5c). The formation of micro-structures could be attributed to the disassembly of aggregates.30,41 Aggregates were intercepted on the membrane, while single ICs were not. Then the aggregates disassembled in the drying process. LiOH and urea formed crystals respectively, which corresponded to the “petals” of flower-like micro-structure. CS in the aggregates attached to the crystals after disassembly. To verify this hypothesis, three control experiments were performed. (1) Filtration of solvent (Fig. 5f). After filtration of LiOH/urea solvent, the surface of membrane was covered with crystals (Fig. 5g). However, no separated flower-like micro-structures were observed. This proved that flower-like micro-structures were not formed by simple stacking of crystals. (2) Selectivity for aggregates (Fig. 5h). When the membrane was coated with CS LiOH/urea solution, the surface of membrane was covered with crystals and clumps of polymer component. In this case, one CS aggregate was surrounded by single ICs and other aggregates. So in the drying process, the tendency of self-association between CS macromolecules became even stronger,30 and consequently created continuous layers on the surface (Fig. 5i). This result indicated that the flower-like micro-structures were not formed because of simple drying process of CS solution. (3) Number density of flower-like micro-structures. The number density of flower-like micro-structures after filtered 2 mL solution was approximately twice of that after filtered 1 mL (Fig. 5d and e). This indicated that the flower-like micro-structures were not formed due to residual solution on the membrane, otherwise the number density would be approximately the same. In fact, the value was proportional to the volume of filtered solution, which revealed the correlation between micro-structures on the membrane and CS aggregations in the solution. In summary, results discussed above indicated the existence of aggregates in the CS LiOH/urea solution.
Based on the unique state of macromolecules in the LiOH/urea solution, the influence of centrifugal field could be explained as following. During the process of centrifugation, centrifugal acceleration far outweighed gravitational acceleration, so the latter was negligible. For a particle (density of ρ2) in liquid (density of ρ1), if ρ2 > ρ1, the particle would move along the direction of centrifugal field. In the measurement of molecular weight of polymer by ultra-centrifugal sedimentation method, the centrifugal force is 3.5 × 105 times of gravity force, which ensures the sedimentation of macromolecules. While in the present work, the centrifugal force was about 2 × 104 times of gravity force, which would not separate single IC from solution. But considering the structural difference, there existed disparity in density between single IC and aggregation. As a result, relative motion could take place between aggregations and solution.
The influence of relative motion was observed by the gel state of CS rod after centrifugation and gelation. Hydrogel samples with different c(CS) all showed flow marks along centrifugal field. In extreme case, c(CS) was too high and CS could not be perfectly dissolved. Gel particles appeared in the system (Fig. 6e), which also deformed along the same direction. Flow marks in hydrogel became more distinct with the rise of c(CS). This was in great accordance with the results on mechanical properties (Fig. 2d) and microstructure of CS rods (Fig. 3a and b). The amount and volume of aggregations increased with the rise of c(CS), thus enhanced the influence of centrifugation. Results discussed above confirmed that orientation could be generated by centrifugation and preserved in CS rods.
A strengthening mechanism was subsequently proposed based on the unique characteristic of CS LiOH/urea solution (Scheme 1). In the solution, macromolecules interacted with aggregates (Scheme 1a). So when relative motion of aggregates happened during centrifugation, surrounding macromolecules were compelled to flow along the same direction. Meanwhile, viscous force resisted the tendency to flow, generating flow orientation (Scheme 1b). Orientation of material consequently improved the bending strength of CS rod. At higher c(CS), the amount and volume of aggregates were larger, leading to higher degree of orientation and bending strength.
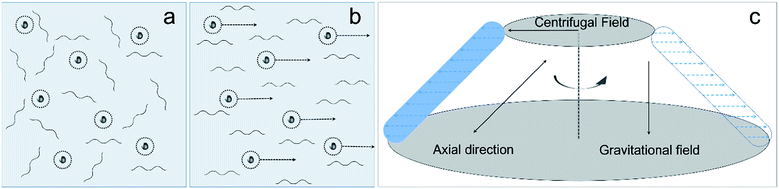 | ||
| Scheme 1 (a and b) molecular arrangement in CS LiOH/urea solution: (a) random state without centrifugation, (b) oriented state after centrifugation, (c) centrifugation process. | ||
Conclusion
In this study, CS rods had been prepared via a novel solvent, i.e. LiOH/urea solvent. Compared to CS rods made via acetic acid aqueous solution, the new CS material showed significant improvement in mechanical performance. Reasons behind the high bending strength of CS rods could be summarized in two aspects. Firstly, the gelation process of CS LiOH/urea solution was distinct from that of traditional acidic CS solution, which endowed the material with homogeneous network structure. Secondly, due to the state of macromolecules in the solution, centrifugation in the processing can generate flow orientation in the material. Resulted from unique characteristics of CS LiOH/urea solution, the improvement of strength had made the novel CS rod a promising candidate for bone fracture internal fixation.Acknowledgements
This work was financially supported by the Key Science Technology Innovation Team of Zhejiang Province (No. 2013TD02), the National Natural Science Foundation of China (Nos 21104067, 21274127, 21374099 and 51473144), and the Key Basic Research Development Plan (973 Program) of China (Nos 2009CB930104 and 2011CB606203).Notes and references
- P. Tormala, T. Pohjonen and P. Rokkanen, P. I. Mech. Eng. H, 1998, 212, 101–111 CrossRef CAS.
- P. U. Rokkanen, O. Bostman, E. Hirvensalo, E. A. Makela, E. K. Partio, H. Patiala, S. Vainionpaa, K. Vihtonen and P. Tormala, Biomaterials, 2000, 21, 2607–2613 CrossRef CAS.
- D. L. Cheng, X. D. Cao, H. C. Gao and Y. J. Wang, RSC Adv., 2013, 3, 6871–6878 RSC.
- E. Waris, Y. T. Konttinen, N. Ashammakhi, R. Suuronen and S. Santavirta, Expert Rev. Med. Devices, 2004, 1, 229–240 CrossRef PubMed.
- Y. H. An, S. K. Woolf and R. J. Friedman, Biomaterials, 2000, 21, 2635–2652 CrossRef CAS.
- H. Peltoniemi, N. Ashammakhi, R. Kontio, T. Waris, A. Salo, C. Lindqvist, K. Gratz and R. Suuronen, Oral Surg., Oral Med., Oral Pathol., 2002, 94, 5–14 CrossRef.
- O. M. Bostman, J. Bone Jt. Surg., Am. Vol., 1991, 73, 148–153 CAS.
- W. D. Hovis, B. W. Kaiser, J. T. Watson and R. W. Bucholz, J. Bone Jt. Surg., Am. Vol., 2002, 84, 26–31 Search PubMed.
- A. Werner, A. Wild, A. Ilg and R. Krauspe, Knee Surg. Sports Traumatol. Arthrosc., 2002, 10, 30–32 CrossRef CAS PubMed.
- M. Rinaudo, Prog. Polym. Sci., 2006, 31, 603–632 CrossRef CAS PubMed.
- A. M. Das, R. Khan, M. P. Hazarika, D. Baruah and P. D. Bhuyan, RSC Adv., 2015, 5, 10065–10071 RSC.
- P. Mukhopadhyay, K. Sarkar, S. Bhattacharya, R. Mishra and P. P. Kundu, RSC Adv., 2014, 4, 43890–43902 RSC.
- X. Huang, J. Jia, Z. Wang and Q. Hu, Chin. J. Polym. Sci., 2015, 33, 284–290 CrossRef CAS.
- H. Lu, W. Wang and A. Wang, RSC Adv., 2015, 5, 17775–17781 RSC.
- A. di Martino, M. Sittinger and M. V. Risbud, Biomaterials, 2005, 26, 5983–5990 CrossRef CAS PubMed.
- G. C. Wang, L. Zheng, H. S. Zhao, J. Y. Miao, C. H. Sun, H. Liu, Z. Huang, X. Q. Yu, J. Y. Wang and X. T. Tao, ACS Appl. Mater. Interfaces, 2011, 3, 1692–1701 CAS.
- Y. Li, P. Zhuang, Y. Zhang, Z. Wang and Q. Hu, Mater. Lett., 2012, 84, 73–76 CrossRef CAS PubMed.
- J. Zhang, J. Nie, Q. Zhang, Y. Li, Z. Wang and Q. Hu, J. Biomater. Sci., Polym. Ed., 2014, 25, 61–74 CrossRef CAS PubMed.
- K. Zhang, M. Zhao, L. Cai, Z. Wang, Y. Sun and Q. Hu, Chin. J. Polym. Sci., 2010, 28, 555–561 CrossRef CAS.
- Q. L. Hu, B. Q. Li, M. Wang and J. C. Shen, Biomaterials, 2004, 25, 779–785 CrossRef CAS.
- Z. K. Wang, Q. L. Hu and Y. X. Wang, Sci. China: Chem., 2011, 54, 380–384 CrossRef CAS.
- X. M. Pu, K. Wei and Q. Q. Zhang, Mater. Lett., 2013, 94, 169–171 CrossRef CAS PubMed.
- X. M. Pu, Q. Q. Yao, Y. Yang, Z. Z. Sun and Q. Q. Zhang, Int. J. Biol. Macromol., 2012, 51, 868–873 CrossRef CAS PubMed.
- Z. K. Wang and Q. L. Hu, Biomed. Mater., 2010, 5, 045007 CrossRef PubMed.
- J. Nie, Z. Wang, K. Zhang and Q. Hu, RSC Adv., 2015, 5, 37346–37352 RSC.
- Z. K. Wang, H. Zhao, L. Fan, J. Lin, P. Y. Zhuang, W. Z. Yuan, Q. L. Hu, J. Z. Sun and B. Z. Tang, Carbohydr. Polym., 2011, 84, 1126–1132 CrossRef CAS PubMed.
- K. Shen, Q. L. Hu, Z. K. Wang and J. Qu, Mater. Sci. Eng., C, 2011, 31, 866–872 CrossRef CAS PubMed.
- X. Wang, Y. Gao, H. Zhao, X. Liu, Z. Wang, A. Qin, Q. Hu, J. Sun and B. Tang, Polym. Chem., 2014, 6216–6224 RSC.
- Z. K. Wang, Q. L. Hu, X. G. Dai, H. Wu, Y. X. Wang and J. C. Shen, Polym. Compos., 2009, 30, 1517–1522 CrossRef CAS PubMed.
- A. Lue, Y. T. Liu, L. Zhang and A. Potthas, Polymer, 2011, 52, 3857–3864 CrossRef CAS PubMed.
- M. Fan and Q. L. Hu, Carbohydr. Res., 2009, 344, 944–947 CrossRef CAS PubMed.
- M. Fan, Q. L. Hu and K. Shen, Carbohydr. Polym., 2009, 78, 66–71 CrossRef CAS PubMed.
- Y. F. Sun, Y. L. Li, J. Y. Nie, Z. K. Wang and Q. L. Hu, Chem. Lett., 2013, 42, 838–840 CrossRef CAS.
- J. Berger, M. Reist, J. M. Mayer, O. Felt, N. A. Peppas and R. Gurny, Eur. J. Pharm. Biopharm., 2004, 57, 19–34 CrossRef CAS.
- Z. K. Wang, S. J. Chen, J. W. Y. Lam, W. Qin, R. T. K. Kwok, N. Xie, Q. L. Hu and B. Z. Tang, J. Am. Chem. Soc., 2013, 135, 8238–8245 CrossRef CAS PubMed.
- J. Nie, W. Lu, J. Ma, L. Yang, Z. Wang, A. Qin and Q. Hu, Sci. Rep., 2015, 5, 7635 CrossRef CAS PubMed.
- B. Q. Li, Q. L. Hu, X. Z. Qian, Z. P. Fang and J. C. Shen, Acta Polym. Sin., 2002, 828–833 CAS.
- Y. Kaneko, R. Yoshida, K. Sakai, Y. Sakurai and T. Okano, J. Membr. Sci., 1995, 101, 13–22 CrossRef CAS.
- J. Lu, Q. L. Hu and J. C. Shen, Acta Polym. Sin., 2006, 1019–1023 CAS.
- C. K. S. Pillai, W. Paul and C. P. Sharma, Prog. Polym. Sci., 2009, 34, 641–678 CrossRef CAS PubMed.
- A. Lue, L. Zhang and D. Ruan, Macromol. Chem. Phys., 2007, 208, 2359–2366 CrossRef CAS PubMed.
- Y. L. Li, PhD thesis, Zhejiang University, 2012.
Footnote |
| † J. Y. Nie and Z. K. Wang contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2015 |