DOI:
10.1039/C5RA07232H
(Paper)
RSC Adv., 2015,
5, 63605-63614
Impact of in vitro non-enzymatic glycation on biophysical and biochemical regimes of human serum albumin: relevance in diabetes associated complications
Received
21st April 2015
, Accepted 20th July 2015
First published on 20th July 2015
Abstract
Early glycation involves attachment of glucose on ε-NH2 of lysine residues. Human serum albumin (HSA), a lysine rich protein, is highly prone to non-enzymatic glycation. In this study, the effect of different concentrations of glucose on HSA through early glycation was assessed by various physicochemical techniques. An early glycation product Amadori HSA was estimated by colorimetric method, nitro blue tetrazolium (NBT) and thiobarbituric acid assay (TBA). The conformational changes were confirmed by UV-visible spectroscopy, tryptophan fluorescence quenching, circular dichroism (CD), and sodium dodecyl sulphate poly acrylamide gel electrophoresis (SDS-PAGE). Biochemical alterations in Amadori-HSA were analyzed by NaBH4 reduction, increased carbonyl content and decrease in free lysine, arginine and sulfhydryl groups. Most studies have reported the structural changes in protein due to AGEs or Amadori protein at a particular glucose concentration; to the best of our knowledge, no reports have been published comparing the physicochemical changes in HSA due to early glycation within a range of glucose concentrations. In the present study, the investigations confirmed the structural and biochemical alterations in Amadori-HSA which is directly proportional to glucose concentration. This might interfere with the normal function of HSA and can contribute to the progression of diabetes and its associated complications. Amadori modified HSA might be used as a biomarker for early detection of diabetes and be beneficial in preventing the diabetic complications at early stages of diabetes. In this context, this study provides additional information about biochemical and biophysical changes occurring in Amadori-HSA within the glucose range upon early glycation.
Introduction
Diabetes Mellitus (DM) is a serious metabolic disorder characterized by hyperglycemia which affects 1–2% of the population worldwide.1 The precise role of hyperglycemia in the pathogenesis of long-term complications is still unclear. However, an attractive hypothesis that has received significant interest is the role of non-enzymatic glycation of protein in diabetes. Non-enzymatic glycation is a spontaneous reaction which depends on the degree and duration of hyperglycemia, the half-life of the protein and permeability of tissues to free glucose.2 In non-enzymatic glycation, glucose molecules attached to free amine residues of the protein form a Schiff base. Through acid–base catalysis, these Schiff base labile adducts undergo rearrangements and convert into a more stable and reversible early glycated product known as an Amadori-product. Now the reaction becomes more diverse and complicated and forms an irreversible and complex advanced glycation end product (AGEs). Only a little part of Amadori-adduct undergoes further irreversible chemical reactions leading to the formation of AGEs.3 Amadori rearrangement is an early key step in non-enzymatic reaction.4 Moreover, the concentration of Amadori-glycated proteins is at least 2% of serum proteins, whereas AGEs are less than 0.01%. Amadori albumin forms five time higher rate than other serum proteins.5 Studies have shown that Amadori-adducts mediated AGEs lead to progression of various diabetic complications such as nephropathy, neuropathy, retinopathy and cardiovascular diseases.6 Glycation has potential to induce unfolding and refolding of globular proteins into cross-β structure and alters its secondary and tertiary structure and native functions. In diabetic patients, because of chronic hyperglycemia such compounds may have a direct link to the development of glucose-related complications that has evidently established by important clinical trials, such as the DCCT and UKPDS.7,8
Human serum albumin (HSA), most abundant serum protein having 59 lysine and 23 arginine residues and contains only one tryptophan residue. It has been proved that specific lysine residues in HSA are involved in glycation, in vivo.9 Non-enzymatic glycation of HSA occurs at multiple sites as lysine 199, lysine 281, lysine 439 and lysine 525 and other minor lysine and arginine residues at the N-terminal residues of polypeptides.10 As a result, the conformational and functional changes occur in HSA, which is one of the pathological conditions associated with early incidence of micro and macro-vascular complications. Earlier studies on serum albumin indicated similar reaction with glucose, yielding a stable glycated form of albumin, which is markedly elevated in diabetes than the rest of the population.11 It has been reported that Amadori albumin plays precise role in early nephropathy and retinopathy in diabetic patients.12 A number of Amadori-modified plasma proteins such as immunoglobulin heavy-chain constant regions have also been identified in type 2 diabetic patients.13 It has been shown that both Amadori product as well as Amadori-derived AGEs are involved in diabetes.14 Their remain paucity of reports on the characterization and role of Amadori products in diabetes associated complications. Oxidative stress, which is believed to play an important role in the pathogenesis of diabetes mellitus, was associated with a decrease of the HSA-thiol group (HSA-SH) content and increase in carbonyl stress.15 This effect has been used as a potential explanation for the underlying tissue damage associated with diabetic complications. Amadori-albumin is an autonomous and effective trigger of molecular mediator contributory to diabetic complications.16 McCance et. al., reported an independent association of initial Amadori adduct with diabetic nephropathy and retinopathy.17
Non-enzymatic glycation may have a variety of important physiological effects and in vitro modification of protein by glucose is regarded as an appropriate model for alterations in protein structure and function relevant to diabetes and its related complications. Reckoning with the above detailed facts; the present study was designed to characterize the glycated HSA at early stage of gylcation (Amadori-HSA). For this, HSA (15 μM) was incubated in vitro, in normal healthy subjects (5 mM), diabetic plasma (up to 25 mM) and also at high non-physiological glucose concentrations (50 mM and 75 mm) which might be present in uncontrolled diabetic plasma. This may serve as an appropriate model to analyze the effect of glucose within a range on HSA structure in diabetic patients. Amadori-HSA was determined by nitro blue titrozolium (NBT) and thiobarbituric acid (TBA) assay and authenticated by HPLC using furosine as a standard. Different concentrations of glucose induced structure and conformational changes have been studied by UV-visible and fluorescence spectroscopic studies, circular dichroism (CD) and sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE). Furthermore, Amadori modified biochemical changes have also been analyzed by estimation of free thiol group, free lysine and arginine amino acid.
Results and discussion
NBT assay for Amadori adduct
HSA (15 mM) without glucose and with 5, 25, 50 and 75 mM glucose was incubated at 37 °C for 7 days. The content of Amadori-HSA was determined by the NBT assay, a standard colorimetric method for the detection of Amadori adducts not for AGEs.18 The yield of Amadori products were quantitated as fructosamine using extinction coefficient of 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 640 cm−1 mol−1 for monoformazon19 and results were expressed in nmol mg−1. Increase fructosamine content (FC) was calculated by the following equation:
640 cm−1 mol−1 for monoformazon19 and results were expressed in nmol mg−1. Increase fructosamine content (FC) was calculated by the following equation:| |
 | (1) |
As shown in Fig. 1 fructosamine formation was started from day 0 and continued till 4 days and on 4th day, a peak was obtained that shows highest level of fructosamine content in all glycated samples. After reaching at highest level, it decays till day 7 because the fructosamine level started decreasing and early glycation products (Amadori) has been converted into AGEs. In our experimental condition, the yield of Amadori product in 5, 25, 50 and 75 mM glucose modified HSA samples measured as fructosamine was 37.7 ± 4.2, 59.3 ± 2.5, 75 ± 5.4 and 107.2 ± 2 nmol mg−1, respectively in 4 days old glycated samples (Table 1). Fourth day old HSA sample without glucose was considered as native HSA containing only 6.6 ± 1.2 nmol mg−1 fructosamine content that was considered negligible. Increase percent in fructosamine content was found to be 62.5%, 86.5%, 91.1% and 93.8% respectively compare to native sample. Higher yield of Amadori product was obtained at highest glucose concentration (75 mM) at 4th day. Level of fructosamine in glycated samples were increased respectively by increasing glucose concentrations. For further studies, 4th day old native HSA and Amadori-HSA with above concentrations samples were used.
 |
| | Fig. 1 Estimation of fructosamine content in native HSA without glucose (-□-) and its counterparts incubated with 5 mM (-♦-), 25 mM (-▲-), 50 mM (-●-) and 75 mM (-■-) glucose for 1–7 days. Each bar represents the mean ± SD of three independent assays. | |
Table 1 Biochemical characterization of 4 days old native and Amadori-HSA samples under identical experimental conditionsa
| Parameters |
Native HSA |
5 mM |
25 mM |
50 mM |
75 mM |
| 5-HMF – hydroxymethylfurfural, NaBH4 – sodium borohydride, all results are in mean ± SD, *p < 0.05 vs. native HSA. |
| Fructosamine (nmol mg−1) |
6.6 ± 1.2 |
37.7 ± 4.2* |
59.3 ± 2.5* |
75.1 ± 5.4* |
107.2 ± 2* |
| 5-HMF (nmol mg−1) |
5.98 ± 0.5 |
12.35 ± 1.9* |
21.18 ± 0.8* |
29.65 ± 2.2* |
32.42 ± 3.2* |
| NaBH4 reduction (nmol mg−1) |
5.6 ± 0.5 |
13.7 ± 0.8* |
20.5 ± 1.4* |
27.3 ± 2.4* |
41.0 ± 1.9* |
| Carbonyl content (nmol mg−1) |
2.7 ± 0.2 |
6.9 ± 0.4* |
11.4 ± 0.7* |
16.3 ± 1.3* |
17.8 ± 1.3* |
| Thiol group (nmol mg−1) |
3.03 ± 0.2 |
2.32 ± 0.1 |
2.12 ± 0.2 |
1.32 ± 0.2* |
0.68 ± 0.8* |
| Free lysine decrease (in %) |
0 |
9.3 |
15.6 |
25.1 |
34.3 |
| Free arginine decrease (in %) |
0 |
4.1 |
10.5 |
25.8 |
40.5 |
Detection of HMF by thiobarbituric acid assay
Hydroxymethylfurfural (HMF) that might have formed in all Amadori-HSA samples was determined by thiobarbituric acid assay (TBA). High yield of TBA reactive HMF was obtained at different concentrations of glucose in Amadori rich glycated samples. This observation is in agreement with an earlier finding.20 As shown in Fig. 2. HMF content in Amadori-HSA (5, 25, 50 and 75 mM) that was 12.35 ± 1.9, 21.18 ± 0.8, 29.65 ± 2.2 and 32.42 ± 3.2 nmol mg−1 respectively while for native HSA, it was 5.98 ± 0.5 nmol mg−1. TBA assay for glycated samples provides structural evidence of hexose bound to HSA.21 NBT as well as TBA assay both gave high concentration of fructosamine and HMF respectively at high concentrations of glucose at 4th day. A p value of 0.05 indicates significant difference in the fructosamine and HMF content of native and Amadori-HSA samples.
 |
| | Fig. 2 Quantitative estimation of hydroxymethylfurfural (HMF) in 4 days old native HSA without glucose and HSA modified with 5 mM, 25 mM, 50 mM and 75 mM glucose. Each bar represents the mean ± SD of three independent assays. The values are the average of three determinations. | |
NaBH4 reduction of Amadori adduct
Presence of Amadori adduct in all glycated HSA samples was confirmed by NaBH4 reduction. All Amadori-HSA samples showed marked reduction in fructosamine level when treated with NaBH4 (Table 1). In Fig. 3 comparison between the fructosamine level before and after NaBH4 reduction has been shown. The yield of fructosamine decreased after NaBH4 reduction leading to the elimination of keto group of Amadori product that causes reduction in fructosamine content. This finding confirmed that glycated samples were rich in Amadori adduct. The formation of Amadori product (fructosyl lysine) measured as furosine was further confirmed by HPLC (data not shown). Amadori-HSA with varying concentrations of glucose was characterized further by various physicochemical techniques.
 |
| | Fig. 3 Content of fructosamine in 4 days old native HSA without glucose and its counterparts 5 mM, 25 mM, 50 mM and 75 mM glucose modified HSA before (□) and after (■) NaBH4 reduction. Each bar represents the mean ± SD of three independent assays. The values are the average of three determinations. | |
Ultraviolet-visible analysis of native and Amadori-HSA
Non-enzymatic glycation induced structural changes in the Amadori-HSA samples which further evaluated by ultraviolet-visible (UV) absorption spectroscopy (Fig. 4). Upon modification of HSA, absorbance at λmax kept rising with increasing concentrations of glucose. Native HSA gave characteristic peak at 280 nm (λmax), which increased on modification by various glucose concentrations. Percent hyperchromaticity was calculated as following equation:| |
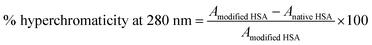 | (2) |
 |
| | Fig. 4 Ultraviolet absorption spectra of 4 days old native HSA without glucose (-□-) and HSA modified with 5 mM (-♦-), 25 mM (-▲-), 50 (-●-) and 75 mM (-■-) glucose. The spectra are the average of three determinations. | |
HSA modified with 5 mM, 25 mM, 50 mM and 75 mM glucose showed hyperchromacity of 9.3%, 23.25%, 28.57% and 34.95% respectively compare to native HSA. Highest modification reached at 75 mM of glucose, which showed a marked hyperchromicity (34.95%) as compared to native analog. Sharpness of the peak was almost same at even different concentrations of glucose. The overall structure of aromatic amino acid residues responsible for the typical absorbance of HSA at 280 nm was maintained upon early glycation. Hyperchromicity was shown with increasing glucose concentrations which may be attributed to the exposure of chromophoric aromatic amino acid residues due to the unfolding and fragmentation of protein as a result of glycation.22 To prove the absence of AGEs adducts in glycated samples, absorbance was also taken between 300–400 nm.
Fluorescence analysis of native and Amadori-HSA
Proteins exhibit intrinsic fluorescence due to the aromatic amino acids tryptophan, tyrosine and phenylalanine. The intrinsic fluorescence emission profile is an excellent parameter to monitor the polarity of tryptophan environment in the protein.23 HSA is a unique protein as it has only one tryptophan residue (Trp-214), located in domain II of HSA. To examine the conformational changes in the micro-environment around this residue upon early glycation, samples were excited at 295 nm, which is specific for tryptophan residues24 and showed a strong emission peak at 330 nm. Fig. 5 showed that quenching in tryptophan fluorescence intensities with increasing the concentration of glucose. Loss of fluorescence intensity was calculated as follows:| |
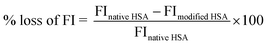 | (3) |
 |
| | Fig. 5 Fluorescence emission profile of (A): native HSA (-□-) and HSA modified with 5 mM (-♦-), 25 mM (-▲-), 50 mM (-●-) and 75 mM (-■-) glucose. The spectra are the average of three determinations. (B): a control tryptophan profile alone and with 75 mM glucose has been shown. | |
Amadori-HSA with 5, 25, 50 and 75 mM glucose showed loss of 3.85%, 11.82%, 28.62% and 66.06% in intensity compared to native HSA respectively (Table 2). Loss in the intensity of fluorescence indicates that tryptophan acquired a different micro-environment upon glycation. Early glycation occurs primarily on ε amino group of lysine residues that might play important role in altering the tryptophan-214 microenvironment.25 Quenching in tryptophan fluorescence with increase glucose concentrations were observed in all Amadori-HSA samples that might be due to the destruction of the residues and/or modification of tryptophan-214 microenvironment upon Amadori formation. Credible existence of fluorogenic AGEs in all Amadori modified samples were confirmed with AGE-specific fluorescence after excitation at 370 nm and emission at 440 nm.26
Table 2 Biophysical characterization of 4 days old native and Amadori modified HSA under identical conditionsa
| Techniques |
Amadori-HSA at different concentrations of glucose |
| Native HSA |
5 mM |
25 mM |
50 mM |
75 mM |
| FI – fluorescence intensity, λex – excitation wavelength, CD – circular dichroism, MRE value in deg cm2 dmol−1. |
| % Hyperchromacity |
— |
9.3 |
23.2 |
28.5 |
34.9 |
| % Loss in trptophan FI λex= 280 |
— |
3.8 |
11.8 |
28.6 |
66.0 |
| Far-UV CD α-helix (%) |
59.29% |
59.23% |
57.66% |
56.82% |
56.15% |
| MRE at 208 nm |
−22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 449.8 449.8 |
−22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 120.4 120.4 |
−21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 382.3 382.3 |
−21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 128.3 128.3 |
−17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 959.2 959.2 |
| MRE at 222 nm |
−22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 115.1 115.1 |
−21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 983.3 983.3 |
−20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 771.6 771.6 |
−20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 157.3 157.3 |
−16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 815.3 815.3 |
Circular dichroism analysis for native and Amadori-HSA
Secondary structure changes in Amadori-HSA with different concentrations of glucose were further confirmed by far UV-circular dichroism (CD) spectrometry. CD spectra of proteins lie in the spectral region of 200–250 nm which is associated with secondary structure.27 As shown in Fig. 6. CD spectra of native HSA exhibit two negative minima in ultraviolet region at 208 and 222 nm, a characteristic feature of α-helical structure.28 And with increase in glucose concentration, the signals have decreased. It can be stated here that the secondary structure at physiological glucose concentration (5 mM) is quite resistant and by increasing glucose concentration (25 mM and 50 mM), there is only a little change in structure but remarkable change occurred at 75 mM glucose modified HSA. The α-helix content in Amadori-HSA gradually decrease as 59.23%, 57.66%, 56.82%, 56.15% with increasing glucose concentrations as compared to native HSA (59.29%). It was calculated by K2D2 software. Mean residual ellipticity (MRE) in deg cm2 mol−1 was calculated according to the formula given below and values are shown in Table 2.| |
 | (4) |
where, θobs is the CD in milli-degree, n is the number of amino acid residues in human albumin (585 − 1 = 584), l is the path length of the cell in cm and C is the concentration of protein in moles litre−1. These changes in secondary structures showed that HSA undergoes conformational alteration upon early glycation. At higher concentration of glucose (75 mM), more reduction in α helix was obtained as compared to native and other conc. of glucose (5, 25, 50 mM). The observed decrease in fluorescence intensity together with loss of helical structure in Amadori-HSA are suggestive of unfolding of the domain II.24 Thus, all the spectroscopic findings confirm that increased glucose concentration with HSA induced increased conformational changes in the secondary structure of HSA at early stage of glycation (Table 2).
 |
| | Fig. 6 Far-UV CD spectra of 4 days old native HSA (-□-) and its counterparts at 5 mM (-♦-), 25 mM (-▲-), 50 mM (-●-) and 75 mM (-■-) glucose. The spectra are the average of three determinations. | |
Electrophoresis (SDS-PAGE)
Fig. 7 shows the migration pattern of native and all Amadori-HSA samples in 10% SDS-PAGE. Protein bands were visualized by silver staining and the band size was determined with molecular ladder, ranging from 200 kD to 29 kD. Native HSA band appeared at 67 kD. Amadori-HSA (5, 25, 50 and 75 mM) showed appreciable retardation in the electrophoretic mobility due to the increment of molecular weight by attachment of glucose molecules (Fig. 7). 75 mM glucose modified HSA showed maximum retardation in electrophoretic mobility as compare to native HSA. Moreover, advanced glycation shows cross linked dimers formation but in our experiment no dimer or polymer formation was observed, which clearly indicates the absence of any cross-linked structures. To authenticate this observation mass spectroscopy has been performed of 75 mM glucose modified HSA in comparison to native HSA.
 |
| | Fig. 7 SDS-polyacrylamide gel electrophoresis of HSA on 10% SDS-PAGE for 4 h at 80 V. Protein samples (10 μg in each lane) were loaded on well. Lanes: (1) protein molecular weight markers; (2–6) native and HSA modified with 5 mM, 25 mM, 50 mM and 75 mM glucose respectively. | |
Matrix-assisted laser desorption/ionization-mass spectrometry analysis
The matrix-assisted laser desorption/ionization-time of flight (MALDI-TOF) mass spectrometry was done to verify the molecular mass of HSA after glucose treatment (Fig. 8). A m/z mass peak was found at 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 002.36 Da. When 75 mM glucose modified HSA was analysed at mass spectroscopy at identical conditions, a m/z peak was obtained at 67
002.36 Da. When 75 mM glucose modified HSA was analysed at mass spectroscopy at identical conditions, a m/z peak was obtained at 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 972.61 Da. We observed increase in the mass of 75 mM glucose modified HSA compared to the native counterpart (ΔM = 670 Da). We propose that due to the early glycation only 6 glucose molecules have been attached in Amadori-HSA (75 mM). This result augments the changes observed in our SDS-PAGE experiment. SDS-PAGE results showed appreciable retardation in the mobility due to the increment of molecular weight by attachment of glucose molecules (Fig. 7). MALDI-TOF data of our samples authenticate this statement fully.
972.61 Da. We observed increase in the mass of 75 mM glucose modified HSA compared to the native counterpart (ΔM = 670 Da). We propose that due to the early glycation only 6 glucose molecules have been attached in Amadori-HSA (75 mM). This result augments the changes observed in our SDS-PAGE experiment. SDS-PAGE results showed appreciable retardation in the mobility due to the increment of molecular weight by attachment of glucose molecules (Fig. 7). MALDI-TOF data of our samples authenticate this statement fully.
 |
| | Fig. 8 MALDI-TOF mass spectra of (A) native HSA and (B) 75 mM glucose modified HSA with sinapinic acid as the matrix. The measured singly charged molecular ions were m/z 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 002.34 for native HSA and m/z 67 002.34 for native HSA and m/z 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 972.61 for Amadori HSA. 972.61 for Amadori HSA. | |
Carbonyl content
Amadori adducts are stable ketoamines and have a definite effect on protein structure and conformation. Ketoamine are converted to protein carbonyl compound via a protein enediol, generating superoxide radical29 which converts into the highly reactive hydroxyl radical (OH*) via Fenton reaction and create the oxidative stress.30 Protein carbonyl content is a known biomarker of oxidative stress. As shown in Fig. 9 carbonyl content in native and Amadori-HSA (5, 25, 50 and 75 mM) was found to be 2.7 ± 0.2, 6.9 ± 0.4, 11.4 ± 0.7, 16 ± 1.3 and 17.8 ± 1.3 nmol mg−1 proteins, respectively. Highest carbonyl content was found in 75 mM glucose modified HSA. This is corresponded to almost seven fold increase in carbonyl level as compared to native HSA. The oxidation of Amadori-HSA was further evident by the significant increase in its carbonyl content in comparison to native HSA.
 |
| | Fig. 9 Carbonyl content of native and HSA modified with 5 mM, 25 mM, 50 mM and 75 mM glucose. The bars show average carbonyl content (±SD) of three independent assays. | |
Thiol group estimation
Oxidative modification induced by glycation in Amadori HSA was analyzed by the estimation of free sulfhydryl group content in protein samples by Ellman's method. Anti-oxidative activity of HSA is one of the important functional property which resides in one free cysteine-derived redox-reactive thiol (–SH) group (Cys34), exists in both reduced and oxidized forms.31 The free sulfhydryl group content is a parameter of the redox state of the protein. The concentration of free thiol group was calculated by using an extinction coefficient of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 cm−1 and results were expressed in nmol mg−1. Average free sulfhydryl group content of three independent assay (mean ± SD). Native HSA contains 3.03 ± 0.2 nmol ml−1 of protein whereas a significant decrease was found in Amadori samples (5, 25, 50 and 75 mM) 2.32 ± 0.1, 2.12 ± 0.2, 1.32 ± 0.2 and 0.68 ± 0.18 nmol mg−1 respectively (Fig. 10). As expected the thiol group reactively was increased to 23.3%, 29.9%, 56.5% and 77.4% with respect to increasing concentrations of glucose as compared to native HSA. A significant oxidation of thiol group in HSA was observed due to the increasing flux of reactive dicarbonyl compounds which occurs during carbonyl stress,32 leads to the Cys side chain carbonylation and, therefore, to the decrease of the HSA-SH content.33 As a result the native functional properties of HSA may be changed due to the alteration in protein conformation.
600 M−1 cm−1 and results were expressed in nmol mg−1. Average free sulfhydryl group content of three independent assay (mean ± SD). Native HSA contains 3.03 ± 0.2 nmol ml−1 of protein whereas a significant decrease was found in Amadori samples (5, 25, 50 and 75 mM) 2.32 ± 0.1, 2.12 ± 0.2, 1.32 ± 0.2 and 0.68 ± 0.18 nmol mg−1 respectively (Fig. 10). As expected the thiol group reactively was increased to 23.3%, 29.9%, 56.5% and 77.4% with respect to increasing concentrations of glucose as compared to native HSA. A significant oxidation of thiol group in HSA was observed due to the increasing flux of reactive dicarbonyl compounds which occurs during carbonyl stress,32 leads to the Cys side chain carbonylation and, therefore, to the decrease of the HSA-SH content.33 As a result the native functional properties of HSA may be changed due to the alteration in protein conformation.
 |
| | Fig. 10 Free thiol group concentration in 4 days old native HSA and modified HSA with 5 mM, 25 mM, 50 mM and 75 mM glucose. Each bar represents the mean ± SD of three independent assays. | |
Determination of reacted lysine and arginine residues in native and Amadori-HSA
Non-enzymatic glycation occurs preferentially on lysine residue but arginine, histidine and cysteine amino acid residues are also involved.34 In this reaction, carbonyl groups interact with free ε-NH2 groups of lysine and arginine residues of proteins. A significant decrease in free lysine and arginine residues upon early glycation were observed in all Amadori samples (Fig. 11 and 12). This might be due to the involvement of several exposed negatively charged lysine and arginine residues that was neutralized by glucose. The maximum free amino acid residues decrease was observed in 75 mM glucose modified Amadori-HSA (Table 1). A percentage decrease in free lysine and arginine of Amadori-HSA was found rapidly with increasing glucose concentrations which was calculated by following formula:| |
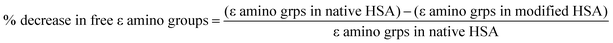 | (5) |
 |
| | Fig. 11 Free lysine group estimation in 4 days old native without glucose and modified HSA with 5 mM, 25 mM, 50 mM and 75 mM glucose samples. Each bar represents the (±SD) of three independent assays. The values are the average of three determinations. | |
 |
| | Fig. 12 Free arginine group estimation in 4 days old native without glucose and modified HSA with 5 mM, 25 mM, 50 mM and 75 mM glucose. Each bar represents the (±SD) of three independent assays. | |
The biochemical changes in native and Amadori-HSA with different glucose concentrations revealed that due to the changes in structure of HSA, native properties of HSA altered. All results are summarized in Table 1 and the p < 0.05 indicates significant difference of native and Amadori-HSA.
Experiments
Chemicals and apparatus
HSA (fatty acid free, 99%) (15 mM) was purchased from the Sigma Chemical Company (St. Louis, USA) and used without further purification. Acrylamide, N,N′-methylene bisacrylamide, 2-mercaptoethanol (MPE), ammonium persulphate, N,N,N′,N′-tetramethylethylenediamine (TEMED), sodium dodecyl sulphate (SDS), glycerol, bromophenol blue were purchased from Sigma were used as received. D-Glucose (5, 25, 50, 75 mM) and sodium borohydride (NaBH4) were obtained from Merck (Darmstadt, Germany). Nitroblue tetrazolium (NBT) was purchased from SRL Chemicals (India). Furosine-HCl was purchased from Polypeptide Laboratories, Strastbourg, France. All other chemicals and reagents used were of highest analytical grade available. HPLC grade solvents, UV transparent cuvettes and multi-well fluorescence plates were obtained from Fisher Scientific (New Lawn, NJ and USA).
UV-spectra were recorded on Shimadzu UV-1700 spectrophotometer. Fluorescence measurements were carried out on a Shimadzu (RF 5301) spectrofluorasometer (Molecular Devices, Sunnyvale, CA, USA). For excitation and emission, the slit widths were set at 5 nm. CD spectra were measured on Jasco J-815-CD spectropolarimeter calibrated with D-10-camphor sulfonic acid. Polyacrylamide gel electrophoresis assembly (Bio-Rad Laboratories, USA) and Gel-doc XR (Bio-Rad laboratories, USA) were also used. LC-MS Micromass Quattro II triple quadrupole mass spectrometer (Beverly, MA, USA) coupled with HPLC system.
Preparation of reaction mixture
The glycated HSA embodying high level of Amadori product was prepared following the published method by Cohen and Hud as detailed elsewhere.18 Stock solution of 300 mM HSA was prepared separately in 20 mM phosphate buffer saline (PBS), pH 7.4 and reactive solution 15 μM HSA, were made with different glucose concentrations 5, 25, 50 and 75 mM. 15 μM HSA without glucose was also dissolved in same buffer used as a native. Unless otherwise indicated, each reaction mixture contained 0.02% sodium azide.
Incubation of reaction mixture
To study the effect of non-enzymatic glycation on native HSA as well as different concentrations of glucose with HSA were incubated at 37 °C for one week in sterile conditions. During the incubation period, 300 μl reaction mixture from each sample was taken periodically and measured the Amadori adduct level. After the formation of Amadori product at highest level, to remove excess glucose from the samples, all samples were extensively dialyzed against PBS (pH 7.4).
Amadori detection by nitroblue tetrazolium (NBT) assay
The Amadori albumin product (fructosamine), in native HSA and glucose modified HSA was determined by NBT assay.35 Native and modified HSA samples (300 μl each) were added (in duplicate) in tubes followed by 3 ml NBT reagent (250 μl mol l−1 in 0.1 mol l−1 carbonate buffer, pH 10.35) in each tube and incubated at 37 °C. After 2 h dark purple color was appeared and it was read at 525 nm.
Detection of HMF by TBA
Formation of HMF from native and Amadori-HSA samples was estimated by thiobarbituric acid assay (TBA) as described earlier.36 Briefly, 1 ml each sample were mixed with 1 M oxalic acid and incubated at 100 °C for 2 hours and protein from the assay mixture was removed by precipitation with 40% trichloroacetic acid. 0.25 ml of thiobarbituric acid (0.05 mol l−1) was added to 0.75 ml protein-free filtrate and incubated at 40 °C for 40 minutes developed color that was read at 443 nm. The results were calculated by using molar extinction coefficient value of 4 × 104 cm−1 mol−1.
Reduction of Amadori adduct by NaBH4
Native and all glycated HSA samples were reduced with NaBH4 in 20 mM PBS at pH 7.4, as described previously.37 Slow addition of 1 N HCl was used to remove excess borohydride.
UV-visible spectroscopy
The UV absorption profile of native and Amadori-HSA samples were recorded in the wavelength range of 200–400 nm on Shimadzu UV-1700 spectrophotometer using quartz cuvette of 1 cm path length.
Intrinsic fluorescence studies
Tryptophan fluorescence profile of native and Amadori samples (5, 25 50 and 75 mM glucose) were recorded on a Shimadzu (RF 5301) spectrofluoasometer (Molecular Devices, Sunnyvale, CA, USA) at 25 ± 0.1 °C. Emission intensities spectra were taken at wavelength range 300–500 nm with excitation wavelength of 295 nm in order to excite tryptophan residues only. The excitation and emission slits were set at 5 nm.
Circular dichroism (CD) measurements
Far UV-CD profile of native and 5, 25, 50 and 75 mM glucose modified HSA were carried out with Jasco Spectropolarimeter, calibrated with D-10-camphorsulfonic acid. All CD measurements were carried out at 25 °C by placing samples in thermostatically controlled cell holder attached to a Neslab's RTE 110 water bath with temperature accuracy of ±0.1 °C. Native and modified samples were placed in 1 and 10 mm path length cuvette and recorded at 200–250 nm wavelength range with scan speed 20 nm min−1.
Sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE)
Native and Amadori-HSA were analyzed by sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) on a 10% polyacrylamide gel, as described previously.38 10 μg each of native and Amadori-HSA was loaded into the wells. Electrophoresis was performed at 80 V for 4 h at room temperature. Protein bands in the gel were visualized by silver staining.
MALDI mass spectroscopy
MALDI measurements were performed on 4700 plus mass spectrometer (AB Sciex, MA, USA) operated in the positive linear mode. Ions formed by a pulsed UV laser beam were accelerated at 30 keV. The matrix was sinapinic acid, dissolved in acetonitrile/water (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, v/v) at a concentration of about 50 mM. Samples were diluted five times in 0.1% trifluoroacetic acid aqueous solution. Equal volumes of sample and matrix solutions were added, and approximately 1–2 μl of the mixture was deposited on the stainless steel multiprobe and allowed to dry before introduction into the mass spectrometer. Mass spectra were obtained and averaged over 10 shots; three independent MALDI measurements were made for each sample to evaluate reproducibility.
50, v/v) at a concentration of about 50 mM. Samples were diluted five times in 0.1% trifluoroacetic acid aqueous solution. Equal volumes of sample and matrix solutions were added, and approximately 1–2 μl of the mixture was deposited on the stainless steel multiprobe and allowed to dry before introduction into the mass spectrometer. Mass spectra were obtained and averaged over 10 shots; three independent MALDI measurements were made for each sample to evaluate reproducibility.
Reactive carbonyl content in Amadori samples
HSA bound carbonyl groups were estimated as described earlier.39 Carbonyl contents in all samples were determined using 2,4-dinitrophenylhydrazine (DNPH). The absorbance was recorded at 360 nm against blank. Carbonyl content was determined using extinction coefficient 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 and expressed as nmol mg−1.
000 M−1 cm−1 and expressed as nmol mg−1.
Oxidative modification of thiol group
Free thiol groups in native and all Amadori-HSA samples were estimated according to Ellman's method using (5,5′-dithio-bis[2-nitrobenzoic acid)] (DTNB).40 DTNB stock and Tris dilution buffer were prepared: DTNB solution-50 mM sodium acetate, 2 mM DTNB in distilled water; Tris solution – 1 M Tris with pH 8.0. DTNB stock was diluted in order to prepare working solution by adding 100 μl Tris solution and 840 μl water. 10 μl of all native and Amadori modified samples were added to 990 μl of DTNB reagent. It was thoroughly mixed and incubated for 5 min at 37 °C. Absorbance was taken at 412 nm. The concentration of free thiol group was calculated by using an extinction coefficient of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 cm−1. Thiol groups for each sample were measured in duplicate and results were expressed in nmol mg−1.
600 M−1 cm−1. Thiol groups for each sample were measured in duplicate and results were expressed in nmol mg−1.
Lysine estimation
Free ε amino groups of lysine amino acids containing native and different concentrations of glucose modified HSA were determined by 2,4,6-trinitrobenezene sulphonic acid (TNBS) method.41 Briefly, 100 μl of 0.5% (w/v) TNBS was added to 0.5 ml of native as well as Amadori samples and incubated for 1 h at 37 °C. At the end of the incubation, the samples were solubilized in 0.25 ml of 10% SDS followed by the addition of 0.1 ml of HCl (1 N). The absorbance was read at 420 nm against a blank.
Arginine estimation
Arginine content in native and different concentrations of glucose modified HSA was determined by the method of van Pilsum et al., (1956).42 Protein samples in aqueous solution were chilled in an ice bath and made to 2.0 ml by adding required amount of 0.2 M NaOH. 5 ml of alkaline α-naphthol-thymine mixture was pipetted into tubes containing protein sample and mixed. Bleach solution (0.2 ml) was added and mixed immediately. After 1 min 0.2 ml sodium thiosulphate was added and rapidly mixed. The absorbance was recorded at 500 nm. The final color of the assay tubes was stable for several hours.
The decrease in free amino acids in HSA after glycation was evaluated using the following formula:
| |
 | (6) |
Statistical analysis
Statistical evaluation of the results was carried out by Student's t-test using Origin Software 6.1 (USA) and data are expressed as mean ± SD. A p-value of <0.05 was considered statistically significant.
Conclusion
The present study has allowed us to achieve better knowledge of glycation induced structure and functional alterations on HSA with varied glucose concentrations ranging from normal to chronic hyperglycemic range. The results of present study clearly showed the formation of Amadori-HSA upon early glycation. Fine characterization of Amadori-HSA with various biochemical and biophysical techniques revealed the devastating changes in HSA tertiary and secondary structure. Amadori-HSA is an independent and strong prompt of molecular mediators contributory to diabetes and its related complications. Reckoning with these facts, the study may be valuable to compare the effect of different concentrations of glucose on HSA structure and conformation upon early glycation. Protein carbonyl content is most commonly used biomarker of protein oxidation. Oxidation of sulfhydryl group present in Cys-34 residue in the protein may lead to the formation of disulphide bonds. In this way, carbonyl content increased and free sulfhydryl group were found to be decreased in our experimental conditions. These conditions go in the favor of oxidative stress and free radical generation that might be a platform for progression of diabetes associated complications. It could be concluded that early glycation induced modifications in HSA could potentially alter its structure, may interfere with the normal function of HSA and contribute to initiation of diabetic and its secondary complications. In this context, this study provides additional information. Amadori-HSA may be used as early diagnostic biomarker in diabetes associated complication as shown from this study. Diabetic patients, who exhibit poor plasma glucose control, are predicted to be must likely to encounter such problems. Further studies are going on in our laboratory on Amadori-HSA which may play an important role in the induction of circulating autoantibodies in diabetes.
Abbreviations
| HAS | Human serum albumin |
| NBT | Nitro blue tetrazolium |
| TBA | Thiobarbituric acid assay |
| AGEs | Advanced glycation end product |
| CD | Circular dichroism |
| SDS-PAGE | Sodium dodecyl sulphate polyacrylamide gel electrophoresis |
| DNPH | 2,4-Dinitrophenylhydrazine |
| NaBH4 | Sodium borohydride |
| HMF | Hydroxymethylfurfural |
| DTNB | 5,5′-Dithio-bis[2-nitrobenzoic acid] |
| TNBS | 2,4,6-Trinitrobenezene sulphonic acid |
| MALDI-TOF | Matrix-assisted laser desorption/ionization-time of flight |
Acknowledgements
Authors are grateful to Dr Zarina Arif, Postdoctoral Fellow (PDF) at Rajiv Gandhi Center for Diabetes and Endocrinology and Mr Mir Yasir Arafat, PhD Scholar, Department of Biochemistry, J N medical College Hospital, Faculty of Medicine, AMU, Aligarh (India) for encouragement and help during the course of work. The present work is supported by University Grant Commission, New Delhi for financial assistance in the form of fellowship.
References
- R. Bucala, H. Vlassara and A. Cerami, in Post-Translational Modifications of Proteins, ed. J. J. Harding and M. J. C. Crabbe, CRC, Boca Raton, FL, 1992, vol. 2, pp. 53–59 Search PubMed.
- J. W. Baynes and S. R. Thosrpe, Free Radical Biol. Med., 2000, 28, 1708–1716 CrossRef CAS.
- C. G. Schalkwijk and T. Miyata, Amino Acids, 2012, 42, 1193–1204 CrossRef CAS PubMed.
- J. E. Hodge, Adv. Carbohydr. Chem., 1995, 10, 169–205 Search PubMed.
- J. W. Baynes, S. R. Thorpe and M. H. Murtiashaw, Methods Enzymol., 1984, 106, 88–98 CAS.
- A. Raghav, J. Ahmad. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 2014, 8(4), 245–251 Search PubMed.
- DCCT Research Group, N. Engl. J. Med., 1993, 329, 977–986 CrossRef PubMed.
- UK Prospective Diabetes Study (UKPDS) Group, Lancet, 1998, 352, 837–853 CrossRef.
- A. Frolov, D. Singer and R. Hoffmann, J. Pept. Sci., 2006, 12, 389–395 CrossRef CAS PubMed.
- O. Ali, G. Bernett, P. Comstock, J. Estenoz, A. Gugliucci and H. V. Roohk, Glycated albumin: an intermediate glycemic index an analytical review, Corporate Information Series – 2, Epinex Diagnostics Inc., Irvine, CA, 2008 Search PubMed.
- C. E. Guthrow, M. A. Morris, J. F. Day, S. R. Thorpe and J. W. Baynes, Proc. Natl. Acad. Sci. U. S. A., 1979, 76, 4258–4261 CrossRef CAS.
- D. R. McCance, D. G. Dyer, J. A. Dunn, K. E. Bailie, S. E. Thorpe, J. W. Baynes and T. J. Lyons, J. Clin. Invest., 1993, 91, 2470–2478 CrossRef CAS PubMed.
- A. Jaleel, P. Halvatsiotis and B. Williamson, Diabetes Care, 2005, 28, 645–652 CrossRef CAS.
- V. M. Monnier, D. R. Sell, Z. Dai, I. Nemet, F. Collard and J. Zhang, Ann. N. Y. Acad. Sci., 2008, 1126, 81–88 CrossRef CAS PubMed.
- Z. Rasheed and R. Ali, Life Sci., 2006, 79, 2320–2328 CrossRef CAS PubMed.
- M. P. Cohen, E. Hud, V. Y. Wu and C. W. Shearman, Invest. Ophthalmol. Visual Sci., 2008, 566, 5089–5093 Search PubMed.
- D. R. McCance, D. G. Dyer, J. A. Dunn, K. E. Bailie, S. E. Thorpe and J. W. Baynes, J. Clin. Invest., 1993, 91, 2470–2478 CrossRef CAS PubMed.
- M. P. Cohen and E. Hud., J. Immunol. Methods, 1989, 117, 121–129 CrossRef CAS.
- R. Mironova, T. Niwa, Y. Handzhiyski, A. Sredovska and I. Ivanow, Mol. Microbiol., 2005, 546–555 Search PubMed 1801–1811.
- K. A. Ney, K. J. Colley and S. V. Pizzo, Anal. Biochem., 1981, 118, 294 CrossRef CAS.
- E. Graf, K. L. Empson and J. W. Eaton, J. Biol. Chem., 1987, 262, 11647–11650 CAS.
- N. Traverso, S. Menini, D. Cottalasso, P. Odetti, U. M. Marinari and M. A. Pronzato, Biochim. Biophys. Acta, 1997, 1336, 409–418 CrossRef CAS.
- L. Stryer, Science, 1968, 162, 526–540 CAS.
- B. Ahmad, Ankita and R. H. Khan, Arch. Biochem. Biophys., 2005, 437, 159–167 CrossRef CAS PubMed.
- D. S. Barnaby, R. L. Cerny, W. Clarke and O.
S. Hage, Clin. Chim. Acta, 2011, 412, 277–285 CrossRef PubMed.
- A. Lapolla, C. Gerhardinger, L. Baldo, D. Fedele and D. Favretto, Org. Mass Spectrom., 1992, 27, 183–187 CrossRef CAS PubMed.
- K. Rahmelow and W. Hubner, Anal. Biochem., 1996, 241, 5–13 CrossRef CAS PubMed.
- J. Tian, J. Liu, J. Xie, X. Yao, Z. Hu and X. Chen, J. Photochem. Photobiol., B, 2004, 74, 39–45 CrossRef CAS PubMed.
- K. Mazdak and L. Jennifer, Anal. Biochem., 2006, 348, 40–48 CrossRef PubMed.
- M. F. Beal, Free Radicals Biol. Med., 2002, 32, 797–803 CrossRef CAS.
- V. B. Jovanovic, A. Z. Penezic Romanjuk, I. D. Pavicevic, J. M. Ac imovic and L. M. Mandic, Anal. Biochem., 2013, 439, 17–22 CrossRef CAS PubMed.
- P. J. Thornalley, Drug Metab. Drug Interact., 2008, 23, 125–150 CAS.
- J. M. Acimovic, B. D. Stanimirovic, N. Todorovic, V. B. Jovanovic and L. M. Mandic, Chem.-Biol. Interact., 2010, 188, 21–30 CrossRef CAS PubMed.
- G. Rabbani, E. Ahmad, N. Zaidi, S. Fatima and R. H. Khan, Cell Biochem. Biophys., 2012, 62, 487–499 CrossRef CAS PubMed.
- R. Johnson, P. A. Metcalf and J. R. Baker, Clin. Chim. Acta, 1982, 127, 87–95 CrossRef.
- A. Lapolla, C. Gerhardinger, L. Baldo, D. Fedele and D. Favretto, Org. Mass Spectrom., 1992, 27, 183–187 CrossRef CAS PubMed.
- F. G. Njoroge, A. A. Fernandes and V. M. Monnier, J. Biol. Chem., 1988, 263, 10646–10652 CAS.
- N. A. Ansari, Moinuddin, K. Alam and A. Ali, Hum. Immunol., 2009, 70, 417–424 CrossRef CAS PubMed.
- R. L. Levine, J. Williams, E. R. Stadtman and E. Shacter, Methods Enzymol., 1994, 233, 346–357 CAS.
- J. Sedlak and R. H. Lindsay, Anal. Biochem., 1968, 25, 192–195 CrossRef CAS.
- R. Haynes, D. T. Osuga and R. E. Feeney, Biochemistry, 1967, 6, 541–547 CrossRef CAS.
- F. J. van Pilsum, T. R. P. Martin, E. Kito and J. Hess, J. Biol. Chem., 1956, 222, 225–236 Search PubMed.
|
| This journal is © The Royal Society of Chemistry 2015 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 640 cm−1 mol−1 for monoformazon19 and results were expressed in nmol mg−1. Increase fructosamine content (FC) was calculated by the following equation:
640 cm−1 mol−1 for monoformazon19 and results were expressed in nmol mg−1. Increase fructosamine content (FC) was calculated by the following equation:
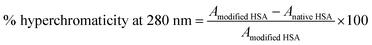
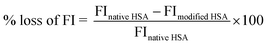
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 449.8
449.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 120.4
120.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 382.3
382.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 128.3
128.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 959.2
959.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 115.1
115.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 983.3
983.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 771.6
771.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 157.3
157.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 815.3
815.3

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 002.36 Da. When 75 mM glucose modified HSA was analysed at mass spectroscopy at identical conditions, a m/z peak was obtained at 67
002.36 Da. When 75 mM glucose modified HSA was analysed at mass spectroscopy at identical conditions, a m/z peak was obtained at 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 972.61 Da. We observed increase in the mass of 75 mM glucose modified HSA compared to the native counterpart (ΔM = 670 Da). We propose that due to the early glycation only 6 glucose molecules have been attached in Amadori-HSA (75 mM). This result augments the changes observed in our SDS-PAGE experiment. SDS-PAGE results showed appreciable retardation in the mobility due to the increment of molecular weight by attachment of glucose molecules (Fig. 7). MALDI-TOF data of our samples authenticate this statement fully.
972.61 Da. We observed increase in the mass of 75 mM glucose modified HSA compared to the native counterpart (ΔM = 670 Da). We propose that due to the early glycation only 6 glucose molecules have been attached in Amadori-HSA (75 mM). This result augments the changes observed in our SDS-PAGE experiment. SDS-PAGE results showed appreciable retardation in the mobility due to the increment of molecular weight by attachment of glucose molecules (Fig. 7). MALDI-TOF data of our samples authenticate this statement fully.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 cm−1 and results were expressed in nmol mg−1. Average free sulfhydryl group content of three independent assay (mean ± SD). Native HSA contains 3.03 ± 0.2 nmol ml−1 of protein whereas a significant decrease was found in Amadori samples (5, 25, 50 and 75 mM) 2.32 ± 0.1, 2.12 ± 0.2, 1.32 ± 0.2 and 0.68 ± 0.18 nmol mg−1 respectively (Fig. 10). As expected the thiol group reactively was increased to 23.3%, 29.9%, 56.5% and 77.4% with respect to increasing concentrations of glucose as compared to native HSA. A significant oxidation of thiol group in HSA was observed due to the increasing flux of reactive dicarbonyl compounds which occurs during carbonyl stress,32 leads to the Cys side chain carbonylation and, therefore, to the decrease of the HSA-SH content.33 As a result the native functional properties of HSA may be changed due to the alteration in protein conformation.
600 M−1 cm−1 and results were expressed in nmol mg−1. Average free sulfhydryl group content of three independent assay (mean ± SD). Native HSA contains 3.03 ± 0.2 nmol ml−1 of protein whereas a significant decrease was found in Amadori samples (5, 25, 50 and 75 mM) 2.32 ± 0.1, 2.12 ± 0.2, 1.32 ± 0.2 and 0.68 ± 0.18 nmol mg−1 respectively (Fig. 10). As expected the thiol group reactively was increased to 23.3%, 29.9%, 56.5% and 77.4% with respect to increasing concentrations of glucose as compared to native HSA. A significant oxidation of thiol group in HSA was observed due to the increasing flux of reactive dicarbonyl compounds which occurs during carbonyl stress,32 leads to the Cys side chain carbonylation and, therefore, to the decrease of the HSA-SH content.33 As a result the native functional properties of HSA may be changed due to the alteration in protein conformation.

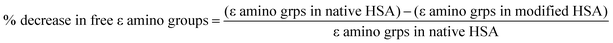

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, v/v) at a concentration of about 50 mM. Samples were diluted five times in 0.1% trifluoroacetic acid aqueous solution. Equal volumes of sample and matrix solutions were added, and approximately 1–2 μl of the mixture was deposited on the stainless steel multiprobe and allowed to dry before introduction into the mass spectrometer. Mass spectra were obtained and averaged over 10 shots; three independent MALDI measurements were made for each sample to evaluate reproducibility.
50, v/v) at a concentration of about 50 mM. Samples were diluted five times in 0.1% trifluoroacetic acid aqueous solution. Equal volumes of sample and matrix solutions were added, and approximately 1–2 μl of the mixture was deposited on the stainless steel multiprobe and allowed to dry before introduction into the mass spectrometer. Mass spectra were obtained and averaged over 10 shots; three independent MALDI measurements were made for each sample to evaluate reproducibility.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 and expressed as nmol mg−1.
000 M−1 cm−1 and expressed as nmol mg−1.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 cm−1. Thiol groups for each sample were measured in duplicate and results were expressed in nmol mg−1.
600 M−1 cm−1. Thiol groups for each sample were measured in duplicate and results were expressed in nmol mg−1.









