Synthesis of rattle-type magnetic mesoporous Fe3O4@mSiO2@BiOBr hierarchical photocatalyst and investigation of its photoactivity in the degradation of methylene blue†
Abstract
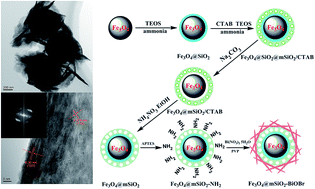
A rattle-type magnetic mesoporous Fe3O4@mSiO2@BiOBr hierarchical photocatalyst was successfully synthesized by a facile solvothermal method under the orientation of the surface amino-groups of rattle-type magnetic mesoporous Fe3O4@mSiO2 microspheres. Then, this photocatalyst was characterized via X-ray diffraction, transmission electron microscopy, field-emitting scanning electron microscopy, Fourier transform-infrared spectroscopy, X-ray photoelectron spectroscopy and vibrating sample magnetometry. Due to the presence of an inner cavity and orderly mesoporous opening structure, this novel photocatalyst exhibits superior adsorption and transfer performance to organic contaminants in aqueous systems. In particular, the complex between BiOBr and SiO2 had significantly increased absorption ability to visible-light due to the some extent of the direct contact of the interfaces of the two materials. Studies show that the assembly capacity of BiOBr nanosheets plays an important role in enhancing the photoactivity. Even though methylene blue is a relatively stable organic contaminant, it can still be decomposed completely by this novel photocatalyst in a very short amount of time (about 120 min). Encouragingly, the photoactivity of this novel photocatalyst is far higher (about 2.6 times) than that of pure BiOBr photocatalyst for its unique structure. According to the radical trapping experiments, the photogenerated holes (h+) and superoxide radicals (O2˙−) are considered to be the main active species that drive the photodegradation under visible-light irradiation. Due to the unique structures and fast interfacial charge transfer, this novel photocatalyst is absolutely a superior alternative visible-light-driven photocatalyst.

 Please wait while we load your content...
Please wait while we load your content...