Effect of maleimide-functionalized gold nanoparticles on hybrid biohydrogels properties†
Abstract
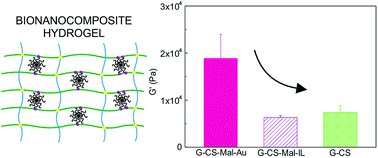
The role of well-dispersed gold nanoparticles as cross-linking agents in nanocomposite hydrogel formation was studied. Maleimide-coated gold nanoparticles were synthesized and used for Diels–Alder cycloaddition with furan modified gelatin. Hydrogel formation was aided by additional amide coupling of the modified gelatin with chondroitin sulfate. The cross-linking ability of the functionalized nanoparticles was evaluated and the final hydrogel properties were compared to those of a hybrid hydrogel containing inert-linker coated gold nanoparticles. The storage modulus of the nanoparticle cross-linked nanocomposites was 2.5–3 times compared to the controls. The presence of nanoparticles also affected the swelling properties, resulting in lower swelling ratios due to the formation of the more cross-linked structures. Conducted drug delivery experiments and the study of the light irradiation on the drug release behavior revealed promising features for the applications of nanocomposite polymer for drug delivery.

 Please wait while we load your content...
Please wait while we load your content...