Self-assembly of thiolated graphene oxide onto a gold surface and in the supramolecular order of discotic liquid crystals†
Abstract
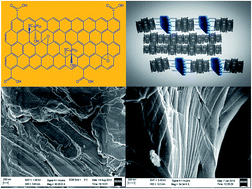
Graphene oxide can be covalently functionalized with thiol molecules to produce thiolated graphene oxide which shows interesting behaviour of self-assembly on gold surfaces and in the supramolecular structures of discotic liquid crystals. The formation of a self-assembled monolayer on the gold surface was confirmed by electrochemical, XPS, SEM, and grazing angle IR studies. Supramolecular nanocomposites of anthraquinone based discotic liquid crystals with thiolated graphene oxide were characterized using SAXS, POM, DSC and Cryo-SEM. Such self-assembling supramolecular nanocomposites open up new and interesting possibilities for fundamental studies as well as for optoelectronic applications.

 Please wait while we load your content...
Please wait while we load your content...