Rechargeable Co3O4 porous nanoflake carbon nanotube nanocomposite lithium-ion battery anodes with enhanced energy performances†
Abstract
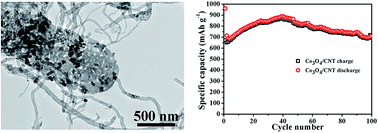
Multi-walled carbon nanotubes intertwined with porous Co3O4 nanoflakes (Co3O4/MWNT) have been hydrothermally synthesized to form a three dimensional network with superior electrical and ionic conductivity and tested as an anode material for lithium-ion batteries (LIBs). The electrochemical measurements on the Co3O4/MWNT composites demonstrated a reversible capacity of 708 mA h g−1 after 100 cycles at a current density of 100 mA g−1 and discharge capacities of 773, 805, 698, and 491 mA h g−1 at current densities of 100, 200, 400, and 800 mA g−1, respectively. The superior cycling and rate performances were attributed to the formed network structure, in which the entanglement and flexibility of MWNTs buffered the volume change of Co3O4 during the charge–discharge process and improved the electrical conductivity of the electrode.

 Please wait while we load your content...
Please wait while we load your content...