Essential oil encapsulations: uses, procedures, and trends
Abstract
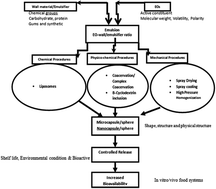
Recently there has been an increased interest towards the biological activities of essential oils (EOs). However, EOs are unstable and susceptible to degradation when exposed to environmental stresses like oxygen, temperature, and light. Therefore, attempts have been made to preserve them through encapsulation in various colloidal systems such as microcapsules, nanospheres, nanoemulsions, liposomes, and molecular inclusion complexes. This review focuses on various techniques used for the encapsulation of EOs, potential applications in food, and their behaviours/trends after encapsulation. The encapsulation efficiency, particle size, and physical stability of EOs encapsulated in colloidal systems is dependent on the kind of technique and the type and concentration/ratio of emulsifier/wall material used. Moreover, the benefits associated after encapsulation, namely bioavailability, controlled release, and protection of EOs against environmental stresses are discussed. The applications of encapsulated EOs are also summarized in this review. Encapsulated EOs are promising agents that can be used to increase the anti-microbial, antifungal, antiviral, and pesticidal activities of EOs in real food systems, to study their action mechanism, and to provide nonlethal therapeutic agents to treat several diseases.

 Please wait while we load your content...
Please wait while we load your content...