Synthesis of nitrogen doped mesoporous carbons for carbon dioxide capture†
Chitrakshi Goel ,
Haripada Bhunia and
Pramod K. Bajpai*
,
Haripada Bhunia and
Pramod K. Bajpai*
Department of Chemical Engineering, Thapar University, Patiala-147004, Punjab, India. E-mail: chitrakshigoel@yahoo.co.in; hbhunia@thapar.edu; pkbajpai@thapar.edu; Fax: +91-175-2393005; Tel: +91-175-2393438
First published on 19th May 2015
Abstract
Nitrogen doped mesoporous carbons were prepared by nanocasting method at varying carbonization temperatures followed by characterization in terms of their structural, textural and chemical properties. Melamine-formaldehyde resin and mesoporous silica were used as the polymeric precursor and hard template respectively. Meso-structural ordering of the template was retained by the prepared carbons as suggested by the structural analysis. Evolution of nitrogen and oxygen functionalities along with textural properties of nitrogen doped carbons were regulated by the carbonization temperature. The prepared carbons obtained by carbonization at 700 °C exhibited a maximum surface area of 266 m2 g−1 along with a nitrogen content up to 21 weight%. CO2 adsorption was studied in a fixed-bed column at several temperatures (30 to 100 °C) and CO2 concentrations (5 to 12.5%). Adsorbent reusability was examined by carrying out multiple adsorption–desorption cycles. MF-700 showed the highest CO2 adsorption capacity of 0.83 mmol g−1 at 30 °C. CO2 adsorption kinetics were investigated by fitting experimental CO2 uptake data to different adsorption kinetic models, out of which the fractional order model was found to fit over the complete adsorption range with the error% between experimental and model predicted data within the range of 5%. In addition, the isosteric heat of adsorption was estimated to be around 17 kJ mol−1, confirming the occurrence of the physiosorption process.
1. Introduction
Carbon dioxide (CO2) emissions, resulting from fossil fuel combustion for power generation, have lead to growing concerns on global warming and climate change. On account of our strong dependence on fossil fuels and the inability of other energy sources, CO2 non-emitting sources like solar energy, nuclear energy etc., to cope up with the energy requirements, there is an urgent requirement in the development of effective CO2 capture techniques to reduce these CO2 emissions.1–3 Technologies proposed to capture CO2 include absorption, adsorption, membrane separation and cryogenic separation.4 Absorption of CO2 with alkanolamine solvents like mono-ethanol amine, di-ethanol amine etc. is used industrially but suffers from several drawbacks like high energy demand, corrosive nature and oxidative degradation of solvents.5,6 Separation technology based on adsorption process is associated with low energy requirement, ease of regeneration etc. and hence is regarded as an attractive option for CO2 capture.Various adsorbents such as zeolites,7,8 carbon based adsorbents,9–11 metal organic frameworks,12,13 and amine-modified silicas14–17 have been studied. Among these available adsorbents, carbon materials are comprised of desired features for CO2 capture like high surface area, fast kinetics, tailored pore structure, hydrophobic character, low energy for regeneration and high stability.18 In addition carbon adsorbents can be prepared from variety of raw materials and various techniques namely sol–gel process, nanocasting and pyrolysis of polymeric precursor. Among these nanocasting is an effective method for the fabrication of mesoporous carbons with controlled pore structure that are otherwise difficult to be prepared by the conventional methods. It consists of three steps (a) infiltration of carbon precursor into the pores of the template, (b) heat treatment in controlled atmosphere and (c) template removal.19,20 The affinity of carbon adsorbents towards CO2 can be increased by surface modification with amines or incorporation of nitrogen groups into the carbon matrix.21–23 Nitrogen functional groups increase the surface polarity of carbon materials and basicity which enhances the interaction between acidic CO2 molecules and carbon surface. Amine modified carbons have high CO2 adsorption capacities but they suffer from amine degradation during regeneration. Nitrogen incorporated carbons can be prepared by pyrolysis of nitrogen containing polymers like melamine, acetonitrile, etc. with various templates such as zeolite, silica etc.24–26 Hao et al.27 reported the synthesis of porous carbon materials from resorcinol and formaldehyde with lysine as catalyst which exhibited static CO2 adsorption capacity up to 3.13 mmol g−1 at 25 °C. Zhao et al.28 prepared nitrogen-doped carbons from IBN-9 mesoporous silica hard template by nanocasting technique and chemical activation. These materials showed equilibrium CO2 uptake of 4.5 mmol g−1 at 25 °C and 1 bar. Porous carbon materials, obtained from melamine-formaldehyde resin by nanocasting route, showed CO2 capacity of 2.25 mmol g−1 at 25 °C and 0.86 mmol g−1 at 75 °C under 100% CO2 flow.24 Nitrogen enriched carbon material was prepared from urea-formaldehyde resin by carbonization followed by chemical activation with KOH and showed adsorption capacity of 3.21 mmol g−1 at 25 °C.29
Most of the carbon materials obtained from nanocasting technique are given post-synthesis treatment (physical or chemical activation) that are energy intensive and time consuming processes. Furthermore these materials are evaluated for CO2 capture under static conditions at room temperature which are not pertinent to flue gas application. In the present study, nitrogen doped mesoporous carbons were prepared from nanocasting technique with the aim to evaluate them for CO2 capture from flue gas. Nitrogen doping was employed in order to improve the selectivity of the adsorbents. They were characterized with X-ray diffraction (XRD), transmission electron microscopy (TEM), scanning electron microscopy (SEM), nitrogen adsorption–desorption, elemental analysis (CHN), Fourier transform infrared (FTIR) spectroscopy and X-ray photoelectron spectroscopy (XPS) for their physico-chemical properties. Fixed-bed column adsorption studies under simulated flue gas conditions have been carried out to evaluate their CO2 adsorption capacity, regenerability and kinetics. Adsorption capacity at high temperatures like 75 °C and 100 °C was also evaluated keeping in view of their application to CO2 capture from flue gas.
2. Experimental
All the reagents and solvents were procured from M/s S. D. Fine Chemicals India Ltd. Mesoporous silica (MCM-41 type) with specific surface area of 450 m2 g−1 and average pore diameter of 3.5 nm was used as template and was purchased from M/s Tianjin Chemist Scientific Ltd., Tianjin, China. Dry helium, nitrogen and carbon dioxide gases of grade-1 purity (99.999%) were obtained from M/s Sigma Gases and Services, India.2.1 Preparation of nitrogen doped mesoporous carbons
Mesoporous carbons were prepared by nanocasting technique using melamine-formaldehyde resin as polymeric precursor and mesoporous silica as template. It included templated resin synthesis, followed by carbonization and then template removal in the last. To a mixture of 200 ml methanol (5 weight%) and melamine (46.6 g), heated to 70 °C, 200 ml of formaldehyde solution (37% w/v) was slowly added over a period of 3 h to form hexamethylomelamine. Potassium carbonate was added to adjust the pH in the range of 8–9. Advanced polymerization was carried out by raising the reaction temperature to 75 °C and maintaining the solution pH to 7–8, by the addition of sodium hydroxide (N/10) and borax (0.4 g), for further 2 h. This was followed by cooling down of the reaction mixture to room temperature and addition of 6 ml of H2SO4 solution (48%). 15.0 g of mesoporous silica (template) was added and mixed thoroughly to this reaction mixture. After this, curing of templated resin was carried out at 60 °C for 2 h and then left at room temperature until complete solidification had occurred.Templated resin samples were carbonized in a quartz tubular furnace under a nitrogen flow by heating at 10 °C min−1 from room temperature up to 400–700 °C for 1 h. The mesoporous carbons were obtained after dissolution of the silica framework in 40 weight% NaOH solution followed by washing with water and then drying at 100 °C. Prepared materials were marked as MF-x, where x denotes the carbonization temperature (400–700 °C).
2.2 Characterization of nitrogen doped mesoporous carbons
The powder X-ray diffraction (XRD) patterns of the prepared carbons were collected using a PANalytical X'Pert Pro diffractometer using Cu–Kα radiation at 45 kV and 40 mA. The low angle XRD patterns were recorded over the scanning range (2θ) of 0.5 to 10° while the wide angle patterns were collected over the scanning range (2θ) of 10 to 80°. Transmission electron microscopy of carbon materials was carried out on a Philips CM200 transmission electron microscope at an accelerating voltage of 200 kV. Specimen preparation included the dispersion of carbon sample in toluene on carbon-coated copper grid followed by evaporation of toluene at room temperature. Scanning electron micrographs were recorded on a JEOL JSM-7600F field emission gun-scanning electron microscope (FEG-SEM) in the back scattering mode with an accelerating voltage of 10.0 kV. The samples were coated with a gold film of 50 μm thickness in an automatic sputter coater (Polaron) in order to avoid charging under electron beam.Nitrogen adsorption–desorption isotherms were measured on a Micromeritics ASAP 2010 sorption analyzer at −196 °C. Prior to adsorption measurements, the samples were outgassed at 220 °C for 6 h under vacuum. The Brunauer–Emmett–Teller (BET) method was used to calculate the specific surface area. The pore size distribution and pore volume were obtained from the adsorption branch using the Barrett–Joyner–Halenda (BJH) model.
Elemental (CHN) analysis was done using a Thermo Finnigan Flash EA 1112 Series elemental analyzer. Oxygen content was determined by difference. Fourier transform infrared (FTIR) spectroscopic studies of prepared carbons were carried out using a Perkin Elmer Spectrum 100 FTIR spectrometer (Perkin Elmer, USA) in the range of 4000–625 cm−1 with a resolution of 4 cm−1. Surface chemistry of the carbons was analyzed by carrying out X-ray photoelectron spectroscopy (XPS) on a SPECS system equipped with a hemispherical electron analyzer using Mg Kα X-ray source (energy 1253.6 eV) operating at an emission current of 10 mA and anode potential of 15 kV. In the analysis chamber, the pressure was maintained below 2 × 10−9 torr. The pass energies for low and high resolution scans were 50 and 20 eV respectively. XPS peak 4.1 software was used to analyze the data and all the core level spectra were peak fitted to a mixed Gaussian–Lorentzian convoluted function (80/20) after Shirley background subtraction.
2.3 Dynamic adsorption–desorption measurements
The experimental setup used for dynamic CO2 adsorption–desorption experiments is represented schematically in Fig. S1.† The adsorption column is made up of stainless steel with a length of 300 mm and an internal diameter of 9.39 mm. The adsorption column is filled with 2 g of dry adsorbent mixed with dry inert glass beads and placed in a temperature-controlled oven. Both the ends of the column are filled with temperature resistant glass wool to minimize the adsorbent loss. A K-type thermocouple, placed in the middle of the bed, monitors the adsorbent bed temperature. CO2 and N2 are mixed in a gas mixing section, made up of stainless steel, prior to adsorption column. Feed concentration is modulated by regulating the flow rates of pure CO2 and N2 with the aid of two mass flow controllers (Bronkhorst, Netherlands). The concentration of these gases is measured by a gas chromatograph (GC 7890A, Agilent Technologies, US), equipped with a thermal conductivity detector. Fused silica capillary column (HP Plot Q, Agilent Technologies, USA) having a length of 30 m, inner diameter of 0.32 mm and film thickness of 20 μm is used for the analysis.In a typical experiment, the adsorbent is treated under a N2 flow at 200 °C for 2 h to remove pre-adsorbed gases or moisture. The temperature is then lowered to the adsorption temperature (30 to 100 °C) and the nitrogen flow is switched to simulated flue gas at a flow rate of 80 ml min−1. CO2 concentration in the simulated flue gas was varied from 5 to 12.5 volume%. CO2 concentration at the bed exit is continuously monitored until the saturation is reached. After adsorption, the simulated flue gas is switched back to pure N2 and temperature is increased to 200 °C to carry out the desorption study. Four adsorption–desorption cycles were carried out to check the cyclic stability of the adsorbents.
The CO2 adsorption capacity of the adsorbent is calculated using the following equation:
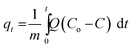 | (1) |
2.4 Equilibrium CO2 sorption measurements
CO2 adsorption and desorption experiments on prepared carbon (MF-700) was carried out on a Micromeritics ASAP 2010 volumetric analyzer at four different temperatures 30 °C, 50 °C, 75 °C and 100 °C and at CO2 pressures from 0 to 1 bar. Before adsorption measurements, samples were degassed at 200 °C for 12 h under vacuum.2.5 Adsorption kinetic models
Fast kinetics is one of the essential parameters expected in a good adsorbent. In a dynamic process like adsorption in a fixed-bed system, kinetic study of the adsorption process provides a base to determine the adsorbent's efficiency and its ability to withstand high adsorbate flow. Typically the experimental data is fitted with a series of the established models and the best fitting model is selected. In the present work, pseudo-first order, pseudo-second order and fractional order adsorption kinetic models have been used to study the adsorption process of the synthesized adsorbents.The Lagergren's first order equation is one of the most widely used model describing adsorption kinetics based on adsorption capacity. The pseudo-first order model can be written as:30
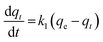 | (2) |
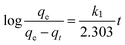 | (3) |
| qt = qe(1 − e−k1t) | (4) |
The pseudo-second order model is represented as:31
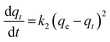 | (5) |
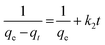 | (6) |
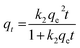 | (7) |
The fractional order kinetic model assumes adsorption rate to be influenced by nth power of driving force and mth power of the adsorption time32,33 and is represented as eqn (8):
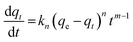 | (8) |
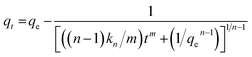 | (9) |
Linear regression analysis of pseudo-first order and pseudo-second order kinetic models has been widely used to determine the best fitted adsorption kinetic model. Pseudo-second order kinetic model equation can be presented in four different linear forms, each resulting in different model parameters. This is because of the change in the error structure upon linearization of the non-linear form of the kinetic model. Hence nonlinear regression is the appropriate way to predict adsorption kinetics.34–37 Thus parameters of the above adsorption kinetic models were calculated by nonlinear regression on the experimental data with the help of OriginPro 8 software.
The adequacy of each model is determined by calculating an error function based on the normal standard deviation. The consistency between the experimental values and the model predicted values are expressed by this error function.
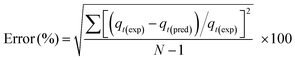 | (10) |
2.6 Isosteric heat of adsorption
The isosteric heat of adsorption is defined as the difference between the activation energy required for adsorption and desorption process and it represents the strength between the adsorbate and the adsorbent.38 It was calculated from the experimental adsorption data by using Clausius–Clapeyron equation:
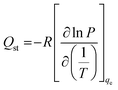 | (11) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P against 1/T at a constant amount of CO2 adsorbed (qe) which yields a straight line with a slope equal to −Qst/R.
P against 1/T at a constant amount of CO2 adsorbed (qe) which yields a straight line with a slope equal to −Qst/R.
3. Results and discussion
3.1 Characterization of nitrogen doped mesoporous carbons
Fig. 1a shows the low angle powder XRD patterns of the silica template and the prepared carbons. The typical structure of silica exhibits two well resolved peaks at 2θ = 0.87° and 1.99° corresponding to (100) and (110) reflections respectively of two dimensional hexagonal mesostructure with lattice constant ao = 11.7 nm. All the prepared carbons showed similar XRD patterns implying that the prepared materials exhibit mesostructural ordering. | ||
| Fig. 1 (a) Low angle XRD patterns of silica template and prepared mesoporous carbons, and (b) wide angle XRD patterns of prepared carbons. | ||
The wide angle XRD patterns of the prepared carbons are presented in Fig. 1b. A broad diffraction peak is obtained at 2θ = 26–27° corresponding to the (002) diffraction planes of graphitic carbon having hexagonal structure (JCPDS X-ray Powder Diffraction Database no. 75-1621) with a d-spacing of 0.34 nm and is very similar to that of ideal graphite.39 Also the resulting carbon materials are not completely crystalline as suggested by the absence of strong diffraction patterns. The peak intensity decreased with increasing carbonization temperature suggesting an increase in irregularity of layer structures. Line broadening of the most intense peak indicates very small crystallite size suggesting the prepared carbons to be nanostructured.
Transmission electron micrographs (Fig. S2a and S2b†) of carbon prepared at 700 °C show the formation of nanostructured materials with particle size in the range of 30 to 50 nm. Heterogeneous types of pores can be clearly seen in SEM images (Fig. S2c and S2d†). External pores between the particles are also present which help in the gas diffusion from bulk phase to adsorbent surface.
Textural parameters of the synthesized carbons and mesoporous silica are obtained by nitrogen sorption measurements. Fig. 2 presents the N2 adsorption–desorption isotherms of mesoporous silica and MF-x materials prepared at different carbonization temperatures. Mesoporous silica and carbons other than MF-700 show a typical type IV isotherm with a hysteresis loop caused by capillary condensation which is characteristic of mesoporous materials. While MF-700 displays a combination of I and IV type profile, demonstrating the presence of both micropores and mesopores. Mesoporosity of the carbons is obtained on the dissolution of the silica template while small microporosity in MF-700 is generated during the pyrolysis of polymeric precursor of its own accord. Nitrogen adsorption increases with increase in carbonization temperature and is highest for 700 °C sample. Carbonization up to 600 °C is found to be not sufficient enough for the porosity development as indicated by small surface area values. MF-700 exhibits surface area of 266 m2 g−1 and pore volume of 0.25 cm3 g−1 with very small micropore volume of 0.03 cm3 g−1. Width of the hysteresis loop is more pronounced for parent silica template as compared to the porous carbons. Additionally large difference in pore diameters of silica template and prepared carbons is observed which could be ascribed to occurrence of hysteresis loop for silica at lower relative pressure as compared to that of the replicated carbons. Moreover, it is worth mentioning that the pore size of the prepared carbons is larger than the wall thickness of the silica template (ca. 8.4 nm) and this could be due to either partial filling of polymeric precursor in the template's pores or shrinkage of polymeric precursor filled in the pores of the template during high thermal treatments. Porous properties of parent silica and obtained porous carbons are congruous with already reported literature.40–42 Hence employment of nanocasting technique for the development of carbon materials produces tailor made porous structure by the silica template having felicitous pore size and wall thickness.
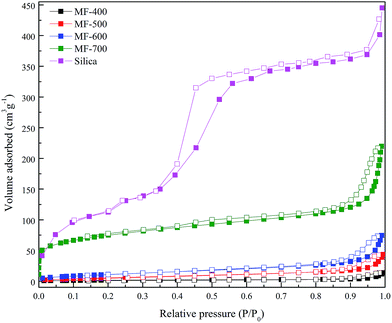 | ||
| Fig. 2 N2 adsorption (closed symbols) and desorption (open symbols) isotherms of prepared carbons and mesoporous silica. | ||
Pore size distribution of carbon materials from adsorption branch of BJH method is shown in Fig. S3† and all the samples show average pore diameter in the mesopore region i.e. 2 to 50 nm. Textural properties of these carbons and silica are given in Table 1.
| Sample | SBET (m2 g−1) | VBJH (cm3 g−1) | dp (nm) |
|---|---|---|---|
| a SBET: Surface area obtained from BET method; VBJH: Pore volume obtained from BJH method using adsorption branch; dp: Average pore size. | |||
| MF-400 | 6 | 0.022 | 14.80 |
| MF-500 | 23 | 0.067 | 9.89 |
| MF-600 | 42 | 0.117 | 9.41 |
| MF-700 | 266 | 0.249 | 10.04 |
| Silica | 450 | 0.697 | 3.5 |
Fig. S4† depicts the elemental composition of the prepared carbon materials. Nitrogen content of the prepared adsorbents decreases continuously from 35 weight% to 21 weight% with increase in carbonization temperature from 400 to 700 °C. This reduction in nitrogen amount at higher carbonization temperatures results in increase in carbon amount of the prepared materials from 41 weight% to 52 weight%.
Investigation of the chemical functionalities developed in carbon adsorbents from melamine-formaldehyde resin is important in order to understand the adsorption process of these materials. It is difficult to determine the exact structure of the melamine-formaldehyde resin because of its complex structure. Moreover the exact structure of carbonized MF resin based carbon adsorbents is also difficult to be determined on account of development of wider functional groups during the carbonization process. Possible reaction equations for the polymerization of melamine and formaldehyde are demonstrated in Fig. 3. Hexamethylomelamine is formed during the addition of formaldehyde to melamine. Advanced polymerization step involves the formation of two kinds of bridges between the hexamethylomelamine molecules namely ether linkage (–N–CH2–O–CH2–N) and methylene linkage (–N–CH2–N–).
FTIR spectra of the mesoporous carbon adsorbents show the development of similar functional groups in the materials (Fig. S5†). A small peak at 1059 cm−1 is observed indicating the presence of C–O linkage of ether, alcohol or ester group. A peak at 1246 cm−1 is observed attributing to the stretching vibration of aromatic C–N. A band around 1604 cm−1 can be assigned to the in-plane deformation vibrations of N–H band and/or stretching vibration of aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds suggesting the presence of aromatic rings. Peak at 1770 cm−1 corresponds to stretching vibration of C
C bonds suggesting the presence of aromatic rings. Peak at 1770 cm−1 corresponds to stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds of esters, carbonyls, anhydrides. A small peak around 3000 cm−1 corresponds to stretching vibration of C–H bond in –CH2–NH–CH2– or –CH2–NH–CH3 groups. The weak bands ca. 3400 cm−1 is credited to symmetric stretching vibration of N–H group.27,43
O bonds of esters, carbonyls, anhydrides. A small peak around 3000 cm−1 corresponds to stretching vibration of C–H bond in –CH2–NH–CH2– or –CH2–NH–CH3 groups. The weak bands ca. 3400 cm−1 is credited to symmetric stretching vibration of N–H group.27,43
The nature of the functional groups present on the surface of melamine-formaldehyde based carbons was evaluated by X-ray photoelectron spectroscopy (XPS). In the survey spectra (Fig. S6†) of the carbon materials, three distinct peaks ca. 285, 400, 532 eV were observed attributing to carbon (C1s), nitrogen (N1s) and oxygen (O1s) respectively. No peak attributing to Si (2s or 2p) was detected demonstrating the complete removal of silica template from the mesoporous carbons by dissolution in sodium hydroxide solution. High resolution spectra were analysed to determine the chemical binding states in the prepared carbons.
The high resolution XPS spectra of the C1s region of MF-based carbons are shown in Fig. 4. The deconvolution of each C1s core level spectrum resulted in five well resolved peaks with binding energies around 284.9, 285.7, 286.8, 288.6 and 290.0 eV labelled as C1, C2, C3, C4 and C5 respectively. C1 peak corresponds to sp2 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) and sp3 (C–C) hybridized graphitic carbon while C2 ascribes to C–O bond of phenol, alcohol or ether and/or C
C) and sp3 (C–C) hybridized graphitic carbon while C2 ascribes to C–O bond of phenol, alcohol or ether and/or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond. C3 peak is assigned for carbonyl or quinone groups and/or C–N linkage, C4 for carboxyl or ester linkages, and C5 for shake up satellite peaks because of π–π* transitions in aromatic rings.44 The corresponding binding energy (B. E.), full width at half maximum (FWHM) and relative area contribution (A%) of each deconvoluted peak are reported in Table 2. Relative area percentage of C1 peak increased from ca. 3.7% to 23.4% on increasing the carbonization temperature from 400 to 700 °C suggesting enhanced graphitization at higher temperatures. MF-400 and MF-500 display similar envelopes for C1s region with carbonyl or quinone and/or C–N linkages being the dominant components. Carbonization at or above 600 °C resulted in a significant change in the surface functional groups as seen in Fig. 4. There is a considerable increase in ether, phenol, alcohol and/or C
N bond. C3 peak is assigned for carbonyl or quinone groups and/or C–N linkage, C4 for carboxyl or ester linkages, and C5 for shake up satellite peaks because of π–π* transitions in aromatic rings.44 The corresponding binding energy (B. E.), full width at half maximum (FWHM) and relative area contribution (A%) of each deconvoluted peak are reported in Table 2. Relative area percentage of C1 peak increased from ca. 3.7% to 23.4% on increasing the carbonization temperature from 400 to 700 °C suggesting enhanced graphitization at higher temperatures. MF-400 and MF-500 display similar envelopes for C1s region with carbonyl or quinone and/or C–N linkages being the dominant components. Carbonization at or above 600 °C resulted in a significant change in the surface functional groups as seen in Fig. 4. There is a considerable increase in ether, phenol, alcohol and/or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N linkages for MF-600 and MF-700 as compared to MF-400 and MF-500. Hence with increase in carbonization temperature from 400 to 700 °C, the relative area percentages of graphitic carbon, carbon–nitrogen structures and carbon–oxygen structures have varied appreciably suggesting the cleavage of C–N, C–O and C
N linkages for MF-600 and MF-700 as compared to MF-400 and MF-500. Hence with increase in carbonization temperature from 400 to 700 °C, the relative area percentages of graphitic carbon, carbon–nitrogen structures and carbon–oxygen structures have varied appreciably suggesting the cleavage of C–N, C–O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O structures and also the formation of more stabilised structures.
O structures and also the formation of more stabilised structures.
| Sample | C1 | C2 | C3 | C4 | C5 | |
|---|---|---|---|---|---|---|
| a BE: Binding energy (eV); FWHM: Full width at half maximum (eV); A%: relative area percentage. | ||||||
| MF-400 | BE | 284.93 | 285.89 | 286.71 | 288.58 | 289.81 |
| FWHM | 1.02 | 1.29 | 1.87 | 1.92 | 1.91 | |
| A% | 3.72 | 21.76 | 47.50 | 16.59 | 10.43 | |
| MF-500 | BE | 284.98 | 285.89 | 286.75 | 288.58 | 289.86 |
| FWHM | 1.29 | 1.33 | 1.96 | 1.86 | 2.23 | |
| A% | 5.80 | 23.68 | 42.46 | 15.54 | 12.52 | |
| MF-600 | BE | 284.84 | 285.58 | 286.87 | 288.65 | 290.22 |
| FWHM | 0.95 | 1.36 | 2.27 | 1.83 | 1.93 | |
| A% | 21.233 | 39.23 | 29.80 | 6.65 | 3.09 | |
| MF-700 | BE | 284.84 | 285.54 | 286.87 | 288.63 | 290.22 |
| FWHM | 0.99 | 1.71 | 2.29 | 1.98 | 2.37 | |
| A% | 23.42 | 48.93 | 19.70 | 4.75 | 3.20 | |
High resolution XPS spectra of N1s region (Fig. 5) was deconvoluted into four different peaks with binding energies around 398.8, 400.0, 401.1, and 403.0 eV referred as N1, N2, N3, and N4 respectively with corresponding B. E., FWHM and relative area contributions listed in Table 3. Peak N1 is attributed to the pyridinic form of nitrogen and is reported to be stable up to even 1000 °C. Peak N2 corresponds for pyrrolic and/or pyridonic nitrogen. Pyridonic nitrogen is not found at same binding energy as pyridinic nitrogen though it contains nitrogen in a six membered ring structure. As a consequence of tautomeric equilibrium in pyridone, nitrogen in pyridone and pyrrole exhibits similar chemical environment and it contributes two p-electrons to the aromatic π-system. In contrast, pyridinic nitrogen contributes only one p-electron to the π-system. Thus within the precision of XPS measurements, pyridonic-N and pyrrolic-N cannot be distinguished. Peak N3 ascribes to quaternary nitrogen and peak N4 to pyridinic nitrogen oxides. Several structures can be possible for quaternary nitrogen however only nitrogen atoms substituting carbon in aromatic graphene structure at center or valley positions are stable during carbonization process.44–47 Binding energy of N2 peak increased from 399.84 to 400.14 eV on increasing the carbonization temperature suggesting the presence of other nitrogen structures like amine, amide or imide in addition to pyrrolic and pyridonic nitrogen. Amide groups are found around binding energy of 399.6–399.9 eV while imides are reported to be found around 399.7 eV and hence it becomes difficult to separate these peaks.48
| Sample | N1 | N2 | N3 | N4 | |
|---|---|---|---|---|---|
| a BE: Binding energy (eV); FWHM: Full width at half maximum (eV); A%: relative area percentage. | |||||
| MF-400 | BE | 398.80 | 399.86 | 401.16 | 403.05 |
| FWHM | 1.65 | 1.73 | 1.56 | 4.45 | |
| A% | 10.76 | 58.70 | 24.89 | 5.66 | |
| MF-500 | BE | 398.83 | 399.84 | 401.14 | 403.04 |
| FWHM | 0.98 | 1.76 | 1.57 | 4.41 | |
| A% | 8.25 | 63.04 | 23.38 | 5.33 | |
| MF-600 | BE | 398.73 | 400.09 | 401.07 | 403.13 |
| FWHM | 1.54 | 1.71 | 1.44 | 4.52 | |
| A% | 53.22 | 28.85 | 12.70 | 5.23 | |
| MF-700 | BE | 398.73 | 400.14 | 401.14 | 403.12 |
| FWHM | 1.55 | 1.64 | 1.33 | 4.48 | |
| A% | 46.66 | 30.19 | 15.84 | 7.31 | |
Up to 500 °C, nitrogen functionalities are present mainly in the form of pyrrolic and/or pyridonic nitrogen in addition to amines, amides and imides. On increasing the temperature from 500 to 600 °C, a considerable increase in area percentage of N1 peak and decrease in area percentage of N2 peak is observed. This could be attributed to conversion of pyrrolic nitrogen to pyridinic nitrogen under severe carbonization conditions and also to loss of primary amines that are present in melamine-formaldehyde resin and are reported to decompose at 600 °C. Pyridinic nitrogen oxides are present as minor component in all the prepared carbon adsorbents. Pyridinic nitrogen and quaternary nitrogen are reported to be the most stable forms of nitrogen formed during the carbonization process. In the present study, contribution of these two functionalities increased from ca. 35.6% to 62.5% with increase in temperature from 400 to 700 °C. Moreover, as most of the nitrogen functionality in the prepared carbons is present in the form pyridinic and pyrrolic or pyridonic nitrogen, which are reported to have electron donating capability and hence exhibit Lewis basicity, they may serve as active sites for adsorption of CO2.
Fig. 6 shows the high resolution O1s core level spectra of the prepared carbons and their deconvolution lead to four separate peaks with binding energies around 531.3, 532.4, 533.6, and 535.0 eV labelled as O1, O2, O3 and O4 respectively (Table 4). Peak O1 is ascribed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O of carbonyl or ketone groups, O2 to carbonyl oxygen of esters, anhydrides, amides, and oxygen atom of phenol, alcohol or ether groups, O3 to oxygen of carboxylic groups, and O4 to oxygen in water.44 In MF-400, peak O3 has the highest relative area contribution and hence oxygen is mainly present in the form of carboxylic group. But with increase in carbonization temperature, relative peak area of O1 and O2 peaks have increased appreciably at the expense of carboxylic groups. Development of oxygen functionalities with carbonization temperature as predicted by O1s spectra are in accordance with that obtained from C1s core level spectra of the carbons.
O of carbonyl or ketone groups, O2 to carbonyl oxygen of esters, anhydrides, amides, and oxygen atom of phenol, alcohol or ether groups, O3 to oxygen of carboxylic groups, and O4 to oxygen in water.44 In MF-400, peak O3 has the highest relative area contribution and hence oxygen is mainly present in the form of carboxylic group. But with increase in carbonization temperature, relative peak area of O1 and O2 peaks have increased appreciably at the expense of carboxylic groups. Development of oxygen functionalities with carbonization temperature as predicted by O1s spectra are in accordance with that obtained from C1s core level spectra of the carbons.
| Sample | O1 | O2 | O3 | O4 | |
|---|---|---|---|---|---|
| a BE: Binding energy (eV); FWHM: full width at half maximum (eV); A%: relative area percentage. | |||||
| MF-400 | BE | 531.35 | 532.49 | 533.64 | 535.01 |
| FWHM | 1.05 | 1.33 | 2.41 | 1.27 | |
| A% | 3.46 | 13.92 | 70.85 | 11.77 | |
| MF-500 | BE | 531.36 | 532.47 | 533.68 | 535.02 |
| FWHM | 1.30 | 1.98 | 2.29 | 1.30 | |
| A% | 6.62 | 40.18 | 46.90 | 6.31 | |
| MF-600 | BE | 531.30 | 532.45 | 533.65 | 535.06 |
| FWHM | 1.69 | 1.73 | 1.77 | 2.59 | |
| A% | 36.98 | 33.18 | 25.00 | 4.85 | |
| MF-700 | BE | 531.26 | 532.44 | 533.67 | 535.16 |
| FWHM | 1.78 | 1.73 | 1.75 | 3.11 | |
| A% | 39.84 | 26.40 | 21.03 | 12.73 | |
3.2 CO2 adsorption measurements
Highest adsorption capacity of 0.78 mmol g−1 is achieved under 10% CO2 feed gas by the adsorbent prepared at 700 °C. This could be attributed to the improvement of textural properties like surface area and pore volume in addition to increase in basic functional groups on the adsorbent surface. As observed from the nitrogen sorption isotherms at 77 K, there is significant improvement in the textural properties of the prepared carbons with development of some micropores in MF-700 with increase in the carbonization temperature. Basicity of carbons is due to presence of heteroatoms like nitrogen and oxygen. As observed from the XPS analysis of prepared carbons, nitrogen moieties present on MF-400 and MF-500 are mainly pyrrolic/pyridonic nitrogen followed by quaternary nitrogen. Pyridinic nitrogen content is very small on these carbons. On the other hand, most of the nitrogen functionality present on carbons obtained at 600 °C and 700 °C is in the form of pyridinic nitrogen followed by pyrrolic/pyridonic nitrogen. The relative percentage of quaternary nitrogen decreased in these carbons. Both pyridinic and pyrrolic/pyridonic nitrogen groups have lone pair of electrons and exhibit Lewis basicity thereby providing active sites for acidic CO2 adsorption. However pyridinic nitrogen is found to have higher affinity for CO2 molecules than pyrrolic nitrogen attributable to its more basic nature.43,49 In contrast quaternary nitrogen is found to have Lewis acidity.50,51 Hence MF-600 and MF-700 samples have higher basic character than MF-400 and MF-500. It is also important to note that CO2 adsorption capacity is not directly associated with total nitrogen content of the carbon materials but is dependent on the kind of nitrogen moieties. Consequently CO2 uptake of MF-600 and MF-700 is higher than that of MF-400 and MF-500 although there is decrease in total nitrogen content with carbonization temperature. Kind of oxygen functionalities present also affect the surface chemistry of the carbon material. Carbonyl, pyrones and ether groups are responsible for basicity of the carbon surface while carboxyl, lactones or lactols and phenol groups contribute towards surface acidity. With increase in carbonization temperature, there is an increase in relative percentage of carbonyl and pyrone groups and a decrease in carboxyl and lactone/lactol groups as suggested by O1s core level spectra. But the contribution of ether and phenol groups cannot be distinguished from XPS spectra as both appear around 532.4 eV binding energy. Enhancement in surface basicity of the porous carbons is observed with rise in carbonization temperature. Consequently, both the textural properties and surface chemistry of carbon materials play important role in CO2 adsorption performance. As MF-700 demonstrated best textural properties and high basic character along with highest CO2 uptake, this adsorbent is selected for further studies.
In addition to adsorption capacity of the adsorbents, selectivity of CO2 over N2 is an important parameter to be studied. Breakthrough curves of N2 and CO2 on MF-600 and MF-700 for adsorption at 30 °C under 5% inlet CO2 concentration are depicted in Fig. 7b. Both the porous carbons exhibit very low N2 adsorption capacity as suggested by presence of nitrogen gas right away in the exit gas stream. Alternatively, CO2 gas concentration gradually increased for MF-600 but for MF-700 CO2 gas concentration at the column exit was zero for around 3.5 minutes.
 | ||
| Fig. 8 CO2 adsorption capacity of MF-700 at different CO2 partial pressures as a function of adsorption temperature. | ||
Activated carbon obtained from carbonization of coal tar pitch and furfural followed by activation with steam showed CO2 adsorption capacity of 0.61 mmol g−1 under 15% CO2 rest N2 flow at 30 °C. The capacity of this material reduced to 0.30 and 0.15 mmol g−1 at 50 °C and 80 °C respectively.52 Equilibrium CO2 capacity of MF-700 at 50 °C is ∼2 times more than this synthetic activated carbon. Carbon from soybean by chemical activation with ZnCl2 and physical activation with CO2 exhibited equilibrium capacity of 0.93 and 0.51 mmol g−1 at 30 °C and 75 °C respectively under 15.4% CO2 atmosphere. But for adsorption at 75 °C, this carbon material could not be regenerated completely in the second cycle resulting in zero capacity in the next adsorption cycle.53 Fly ash derived activated carbons exhibited CO2 uptake of 0.93 and 0.42 mmol g−1 at 30 °C and 75 °C respectively.54 Hence the CO2 adsorption capacity achieved for the prepared carbons are in good agreement with the values reported in literature.
CO2 adsorption capacity of MF-700 at different temperatures under 12.5% CO2 atmosphere during repeated cycles is depicted in Fig. 9b. CO2 adsorption capacities for four runs were nearly identical implying adsorption process to be completely reversible, being assigned to weak van der Waal forces acting between the adsorption sites and CO2 molecules. This confirms the stability of CO2 adsorption performance of MF-700 for long-term cyclic operations.
CO2 uptake of 1.4 mmol g−1 at 25 °C and 1 bar was demonstrated by carbon nitride materials obtained from polymerization of ethylenediamine and carbon tetrachloride in the pore of mesoporous silica FDU-12 and carbonization at 600 °C for 5 h.42 Direct pyrolysis of resorcinol, formaldehyde and lysine at 500C (RFL-500) produced carbon that exhibited very high CO2 uptake of 3.13 mmol g−1 at 25 °C as compared to our material.27 But the CO2 uptake at higher temperatures are comparable with 0.62 mmol g−1 at 120 °C for RFL-500 and 0.52 mmol g−1 at 100 °C for MF-700. Adsorption capacities at higher temperatures are more relevant with respect to CO2 capture from flue gas.
 | ||
| Fig. 11 Experimental CO2 uptake on MF-700 and corresponding fit to kinetic models at 10% CO2 and different adsorption temperatures. | ||
| Kinetic model | Parameter | Temperature (°C) | |||
|---|---|---|---|---|---|
| 30 | 50 | 75 | 100 | ||
| Pseudo-first order | k1 (min−1) | 0.244 | 0.203 | 0.158 | 0.155 |
| qe (mmol g−1) | 0.773 | 0.589 | 0.484 | 0.263 | |
| R2 | 0.996 | 0.993 | 0.979 | 0.961 | |
| Error% | 4.480 | 4.026 | 7.791 | 10.919 | |
| Pseudo-second order | k2 (g mmol−1 min−1) | 0.383 | 0.398 | 0.344 | 0.640 |
| qe (mmol g−1) | 0.871 | 0.675 | 0.569 | 0.308 | |
| R2 | 0.976 | 0.996 | 0.995 | 0.985 | |
| Error% | 10.414 | 2.480 | 2.997 | 6.571 | |
| Fractional order | kn (mmol1−m gm−1 min−n) | 0.307 | 0.272 | 0.111 | 0.072 |
| qe (mmol g−1) | 0.783 | 0.615 | 0.493 | 0.269 | |
| n | 1.755 | 1.302 | 0.521 | 0.445 | |
| m | 1.614 | 0.943 | 0.565 | 0.503 | |
| R2 | 0.999 | 0.998 | 0.996 | 0.996 | |
| Error% | 0.749 | 1.579 | 2.223 | 2.594 | |
Pseudo-first order and pseudo-second order kinetic models underestimated the CO2 uptake at the initial adsorption phase for adsorption at all temperatures except 30 °C. Pseudo-second order model also overestimated the equilibrium CO2 capacity of the adsorbent while pseudo-first order model was found to be in good accord with the experimental data at the final adsorption phase. Also higher values of error%, based on normal standard deviation, for these two kinetic models suggest that these models are not strictly followed by the experimental CO2 uptake for mesoporous carbon.
On the other hand, adsorption behavior as predicted by fractional order model was in good agreement with the experimental data over the entire adsorption region with maximum error of 2.6%. Lower values of error% from fractional order model compared to pseudo-first and pseudo-second order models, within the range of adsorption temperature and CO2 feed concentration, recommend its applicability for mesoporous carbon adsorbent.
As CO2 adsorption is an exothermic process and is favored at lower temperatures, there was a decrease in adsorption rate constant kn with increase in adsorption temperature. Parameters n and m of fractional order model indicate the effect of the driving force and diffusion resistance respectively. Increase in adsorption temperature led to a decrease in both of these parameters. Pseudo-order with respect to driving force i.e. n had the maximum value at 30 °C indicating the large driving force at this temperature. The system under study followed kinetics with n around 1.3–1.8 at low temperatures (30 and 50 °C) but for adsorption at higher temperature of 100 °C, the order reduced to 0.45 with respect to driving force. Predicted equilibrium adsorption capacity was also found to be in good agreement with the experimental value.
Fig. 12 shows the experimental and fractional order model predicted uptake of CO2 vs. time as a function of CO2 feed concentration at 30 °C. Fractional order model was found to predict the CO2 adsorption behavior of mesoporous carbon adsorbent, MF-700, appropriately for all the CO2 concentrations. CO2 uptake is smaller at low partial pressure of CO2 because of the smaller driving force.
4. Conclusions
Nitrogen doped mesoporous carbons, prepared from melamine-formaldehyde resin by nanocasting technique, have been characterized for their structural, textural and chemical properties. Carbonization temperature is found to have significant effect on their physico–chemical properties. Elemental analysis, FTIR and XPS analysis confirmed the development of nitrogen doped mesoporous carbons with nitrogen species being present majorly in the form of pyridinic and pyrrolic/pyridonic nitrogen. CO2 adsorption capacity of MF-700 decreased with increase in adsorption temperature but increased with CO2 feed concentration. Decline in capacity with temperature is attributed to exothermic adsorption process. Furthermore these prepared carbons exhibited complete regeneration and stability for long-term use. Kinetic analysis of CO2 adsorption on nitrogen doped mesoporous carbons was carried out by validating different adsorption kinetic models with experimental data of CO2 adsorption measurements at various CO2 feed concentrations. Fractional order kinetic model successfully predicted the dynamic adsorption of CO2 as signified by error% values between experimental and predicted data within the range of 5%.Acknowledgements
The financial support from Science and Engineering Research Board under Department of Science and Technology (DST), and All India Council of Technical Education (AICTE), New Delhi, is acknowledged. Chitrakshi Goel acknowledges the financial support from DST-INSPIRE under its Assured Opportunity for Research Careers (AORC) scheme.References
- B. P. Spigarelli and S. K. Kawatra, J. CO2 Util., 2013, 1, 69 CrossRef CAS
.
- Q. Wang, J. Luo, Z. Zhong and A. Borgna, Energy Environ. Sci., 2011, 4, 42 CAS
.
- K. M. K. Yu, I. Curcic, J. Gabriel and S. C. E. Tsang, ChemSusChem, 2008, 1, 893 CrossRef CAS PubMed
.
- R. Thiruvenkatachari, S. Su, H. An and X. X. Yu, Prog. Energy Combust. Sci., 2009, 35, 438 CrossRef CAS
.
- D. Aaron and C. Tsouris, Sep. Sci. Technol., 2005, 40, 321 CrossRef CAS
.
- F.-Y. Chang, K.-J. Chao, H.-H. Cheng and C.-S. Tan, Sep. Purif. Technol., 2009, 70, 87 CrossRef CAS
.
- J. Merel, M. Clausse and F. Meunier, Ind. Eng. Chem. Res., 2008, 47, 209 CrossRef CAS
.
- R. V. Siriwardane, M.-S. Shen and E. P. Fisher, Energy Fuels, 2005, 19, 1153 CrossRef CAS
.
- A. Wahby, J. M. Ramos-Fernández, M. Martínez-Escandell, A. Sepúlveda-Escribano, J. Silvestre-Albero and F. Rodríguez-Reinoso, ChemSusChem, 2010, 3, 974 CrossRef CAS PubMed
.
- N. Sun, C. Sun, H. Liu, J. Liu, L. Stevens, T. Drage, C. E. Snape, K. Li, W. Wei and Y. Sun, Fuel, 2013, 113, 854 CrossRef CAS
.
- S. Himeno, T. Komatsu and S. Fujita, J. Chem. Eng. Data, 2005, 50, 369 CrossRef CAS
.
- A. R. Millward and O. M. Yaghi, J. Am. Chem. Soc., 2005, 127, 17998 CrossRef CAS PubMed
.
- Q. Yang, C. Xue, C. Zhong and J.-F. Chen, AIChE J., 2007, 53, 2832 CrossRef CAS
.
- K. S. N. Kamarudin and N. Alias, Fuel Process. Technol., 2013, 106, 332 CrossRef CAS
.
- M. R. Melloa, D. Phanon, G. Q. Silveira, P. L. Llewellyn and C. M. Ronconi, Microporous Mesoporous Mater., 2011, 143, 174 CrossRef
.
- X. Wang, X. Ma, C. Song, D. R. Locke, S. Siefert, R. E. Winans, J. Möllmer, M. Lange, A. Möller and R. Gläser, Microporous Mesoporous Mater., 2013, 169, 103 CrossRef CAS
.
- Y. Belmabkhout, R. Serna-Guerrero and A. Sayari, Ind. Eng. Chem. Res., 2010, 49, 359 CrossRef CAS
.
- M. Sevilla and A. B. Fuertes, J. Colloid Interface Sci., 2012, 366, 147 CrossRef CAS PubMed
.
- T. Valdés-Solís and A. B. Fuertes, Mater. Res. Bull., 2006, 41, 2187 CrossRef
.
- C. Zhao, W. Wang, Z. Yu, H. Zhang, A. Wang and Y. Yang, J. Mater. Chem., 2010, 20, 976 RSC
.
- M. L. Gray, Y. Soong, K. J. Champagne, J. Baltrus, R. W. Stevens, P. Toochinda and S. S. C. Chuang, Sep. Purif. Technol., 2004, 35, 31 CrossRef CAS
.
- M. G. Plaza, C. Pevida, A. Arenillas, F. Rubiera and J. J. Pis, Fuel, 2007, 86, 2204 CrossRef CAS
.
- D. Bezerra, R. Oliveira, R. Vieira, C. Cavalcante Jr and D. S. Azevedo, Adsorption, 2011, 17, 235 CrossRef CAS
.
- C. Pevida, T. C. Drage and C. E. Snape, Carbon, 2008, 46, 1464 CrossRef CAS
.
- Y. Huang, F. Yang, Z. Xu and J. Shen, J. Colloid Interface Sci., 2011, 363, 193 CrossRef CAS PubMed
.
- A. Lu, A. Kiefer, W. Schmidt and F. Schüth, Chem. Mater., 2004, 16, 100 CrossRef CAS
.
- G.-P. Hao, W.-C. Li, D. Qian and A.-H. Lu, Adv. Mater., 2010, 22, 853 CrossRef CAS PubMed
.
- Y. Zhao, L. Zhao, K. X. Yao, Y. Yang, Q. Zhang and Y. Han, J. Mater. Chem., 2012, 22, 19726 RSC
.
- Z. Liu, Z. Du, H. Song, C. Wang, F. Subhan, W. Xing and Z. Yan, J. Colloid Interface Sci., 2014, 416, 124 CrossRef CAS PubMed
.
- S. Lagergren, Ksver. Veterskapsakad. Handl., 1898, 24, 1 Search PubMed
.
- Y. S. Ho and G. McKay, Process Biochem., 1999, 34, 451 CrossRef CAS
.
- A. Zhao, A. Samanta, P. Sarkar and R. Gupta, Ind. Eng. Chem. Res., 2013, 52, 6480 CrossRef CAS
.
- A. Heydari-Gorji and A. Sayari, Chem. Eng. J., 2011, 173, 72 CrossRef CAS
.
- K. V. Kumar and S. Sivanesan, J. Hazard. Mater., 2006, 134, 277 CrossRef CAS PubMed
.
- Y.-S. Ho, Water Res., 2006, 40, 119 CrossRef CAS PubMed
.
- Y.-S. Ho and A. E. Ofomaja, J. Hazard. Mater., 2006, 137, 1796 CrossRef CAS PubMed
.
- K. V. Kumar and S. Sivanesan, J. Hazard. Mater., 2005, 123, 288 CrossRef CAS PubMed
.
- B. Guo, L. Chang and K. Xie, J. Nat. Gas Chem., 2006, 15, 223 CrossRef CAS
.
- Y. Xia and R. Mokaya, Chem. Mater., 2005, 17, 1553 CrossRef CAS
.
- A. Vinu, K. Ariga, T. Mori, T. Nakanishi, S. Hishita, D. Golberg and Y. Bando, Adv. Mater., 2005, 17, 1648 CrossRef CAS
.
- A. Vinu, S. Anandan, C. Anand, P. Srinivasu, K. Ariga and T. Mori, Microporous Mesoporous Mater., 2008, 109, 398 CrossRef CAS
.
- K. S. Lakhi, W. S. Cha, S. Joseph, B. J. Wood, S. S. Aldeyab, G. Lawrence, J.-H. Choy and A. Vinu, Catal. Today, 2015, 243, 209 CrossRef CAS
.
- C. Chen, J. Kim and W.-S. Ahn, Fuel, 2012, 95, 360 CrossRef CAS
.
- K. László, E. Tombácz and K. Josepovits, Carbon, 2001, 39, 1217 CrossRef
.
- L. Wang and R. T. Yang, J. Phys. Chem. C, 2012, 116, 1099 CAS
.
- J. R. Pels, F. Kapteijn, J. A. Moulijn, Q. Zhu and K. M. Thomas, Carbon, 1995, 33, 1641 CrossRef CAS
.
- A. Olejniczak, M. Lezanska, J. Wloch, A. Kucinska and J. P. Lukaszewicz, J. Mater. Chem. A, 2013, 1, 8961 CAS
.
- C. Pevida, M. G. Plaza, B. Arias, J. Fermoso, F. Rubiera and J. J. Pis, Appl. Surf. Sci., 2008, 254, 7165 CrossRef CAS
.
- B. Zhu, K. Li, J. Liu, H. Liu, C. Sun, C. E. Snape and Z. Guo, J. Mater. Chem. A, 2014, 2, 5481 CAS
.
- M. Sevilla, P. Valle-Vigón and A. B. Fuertes, Adv. Funct. Mater., 2011, 21, 2781 CrossRef CAS
.
- J. Zhou, W. Li, Z. Zhang, W. Xing and S. Zhuo, RSC Adv., 2012, 2, 161 RSC
.
- M. Balsamo, T. Budinova, A. Erto, A. Lancia, B. Petrova, N. Petrov and B. Tsyntsarski, Sep. Purif. Technol., 2013, 116, 214 CrossRef CAS
.
- J. A. Thote, K. S. Iyer, R. Chatti, N. K. Labhsetwar, R. B. Biniwale and S. S. Rayalu, Carbon, 2010, 48, 396 CrossRef CAS
.
- M. M. Maroto-Valer, Z. Lu, Y. Zhang and Z. Tang, Waste Manag., 2008, 28, 2320 CrossRef PubMed
.
- I. A. A. C. Esteves, M. S. S. Lopes, P. M. C. Nunes and J. P. B. Mota, Sep. Purif. Technol., 2008, 62, 281 CrossRef CAS
.
- M. D. Hornbostel, J. Bao, G. Krishnan, A. Nagar, I. Jayaweera, T. Kobayashi, A. Sanjurjo, J. Sweeney, D. Carruthers, M. A. Petruska and L. Dubois, Carbon, 2013, 56, 77 CrossRef CAS
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra05684e |
| This journal is © The Royal Society of Chemistry 2015 |









