Quinoline benzimidazole-conjugate for the highly selective detection of Zn(ii) by dual colorimetric and fluorescent turn-on responses†
Abstract
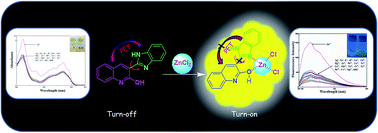
A quinoline benzimidazole-conjugate (QBC) has been synthesized for the highly selective detection of Zn(II) both by colorimetry and fluorimetry. Probe QBC senses Zn2+ over other cations as fluorescence ‘off–on’ behaviour in HEPES-buffered CH3CN–H2O (1 : 1, v/v, pH = 7.0) solution. A possible mechanism is proposed based on the inhibition of PET and intramolecular restricted torsional rotation through the C–C single bond between the quinoline benzimidazole-conjugate. The Chemosensor is utilized to detect Zn2+ in much real sample analysis.

 Please wait while we load your content...
Please wait while we load your content...