Investigation of the synthesis and properties of isophorone and ether units based semi-aromatic polyamides
Gang Zhang*a,
Yu-xuan Zhoub,
Yan Lia,
Xiao-Jun Wanga,
Sheng-Ru Longa and
Jie Yang*ac
aInstitute of Materials Science & Technology, Analytical & Testing Center, Sichuan University, Chengdu 610064, P. R. China. E-mail: gangzhang@scu.edu.cn; ppsf@scu.edu.cn; Fax: +86-28-8541-2866
bCollege of Chemistry, Sichuan University, Chengdu, 610064, P. R. China
cState Key Laboratory of Polymer Materials Engineering (Sichuan University), Chengdu, 610065, P. R. China
First published on 21st May 2015
Abstract
The difluoro-substituted monomer, N,N′-bis(4-fluorobenzoyl) isophorone diamine (BFID), was prepared via an interfacial reaction from isophorone diamine and 4-fluorobenzoic chloride. It was then reacted with hydroquinone (or resorcinol, 1,1-bis(4-hydroxyphenyl)-1-phenylethane (BHPPE)) to yield a series of semi-aromatic polyamides. For the synthesized semi-aromatic polyamides, differential scanning calorimetry and thermogravimetric analysis confirmed their high glass transition temperatures (Tg), which were between 217 and 239 °C, and good thermal stability with initial degradation temperatures (Td) in the range of 425–430 °C. Tensile test and dynamic mechanical analysis (DMA) results revealed the good mechanical properties of the semi-aromatic polyamides at ambient temperature and even at 200 °C. Rheological characterization displayed complex viscosities at 310 °C for the semi-aromatic polyamide in the range of 990–1350 Pa s, which suggests that they are suitable for melt processing. Better solubility properties for the resultant semi-aromatic polyamides compared to commercial ones were also found. All the results indicated the good processability of the synthesized semi-aromatic polyamides. In addition, they were found to have almost identical optical transmittance (Tran%: 83–85% at 450 nm) as polycarbonate (PC), in the UV-visible region.
Introduction
Polyamides such as nylon are a type of famous thermoplastic polymer. They have been widely used in many fields because of their excellent mechanical durability and high strength.1,2 However, aliphatic polyamides display limited thermal stability, particularly when used in the shells of automobile engines and surface mount technology (SMT). In order to improve the thermal properties of aliphatic polyamides, the incorporation of aromatic units into the polymer main chain acts as an efficient approach.3–7 As we know, aromatic polyamides such as Aramid-1414 (ref. 8) and Aramid-1313 (ref. 9) exhibit excellent mechanical properties, good heat resistance, and high chemical and solvent resistance. However, their poor solubility and high glass transition temperature result in poor processability. To solve these problems, the application of semi-aromatic polyamides is regarded as a promising alternative method. Semi-aromatic polyamides are generally used to fill the “performance gap” between high performance polyamides and nylons.10 However, only a few semi-aromatic polyamides, such as the copolymer of PA6T,11 poly(nonamethylene terephthalamide) (PA9T),12–15 poly(decamethylene terephthalamide) (PA10T), the product of Dupont, Solvay, Mitsui, Evonik and so on, are commercially available. Melt processing is impractical for semi-aromatic polyamides that contain short chain (less than 7 methylenes) diamines because their melting temperature (Tm) (as shown in Table 1) is higher than the thermal degradation temperature. To improve their processability, some effort to increase semi-aromatic solubility by the incorporation of bulky pendant groups,16,17 noncoplanar biphenylene moieties,18 triazole units,19 azo groups20,21 and hyperbranched units22,23 into the polymer main chain have been reported. On the other hand, improving melt processability (as shown in Table 1) by the introduction of naphthalene rings,24 cyclodextrin units,25 sulphone units,26 ester units,27 sulfide groups,28 dicyclohexyl methane units29 and long carbon chain diamines30,31 [such as PA-10T,32 PA-12T33 and PA-18T (Tm = 245 °C)] into the polymer backbone could be an alternative way.| Polymer | Tm (°C) | Td (°C) | Polymer | Tg (°C) | Tm (°C) | Td (°C) |
|---|---|---|---|---|---|---|
| PA4T | 430 | 350 | PA18T | — | 245 | — |
| PA6T (homopolymer) | 370 | 350 | Semi-aromatic PA (with bulky pendant groups) | 270 | — | 500 |
| PA9T | 305 | 464 | Semi-aromatic PA (with biphenylene unit) | 300 | — | 510 |
| PA10T | 315 | 472 | Semi-aromatic PA (with naphthalene ring) | — | 300 | 495 |
| PA12T | 295 | 429 | Semi-aromatic PA (with ester unit) | 110–140 | 219–260 | — |
The earlier studies on semi-aromatic polyamides were usually focused on studying the thermal properties of their aggregated structures, while there were only a few studies on their rheological and optical properties. Therefore, the aim of this work is the synthesis of new semi-aromatic polyamides with improved mechanical and thermal properties as well as processability, which is verified through the investigation of their rheological and optical properties. To achieve this aim, we introduced the isophorone unit containing stable cyclohexane and side methyl groups into the polymer chain. The ether bone (–O–) is thought to be a flexible linkage. It has already been introduced into polymer main chains to yield high performance resins such as poly(arylene ether sulfone)34 and poly(arylene ether amide). Both of them are found to have excellent processability, mechanical and thermal properties. In addition, the side group is usually expected to weaken the packing of the polymer chain and improve its optical properties. In this case, the final synthesized semi-aromatic polyamides in this work would display pronounced processability, mechanics, thermal properties, and optical performance.
In this work, we synthesized semi-aromatic polyamides through the nucleophilic substitution reaction of bisphenol and difluorobenzamide monomers containing the isophorone unit. The effects of this chemical structure on the properties of the semi-aromatic polyamides such as thermal property, mechanical property, and rheological property as well as optical properties were investigated.
Experimental
Materials
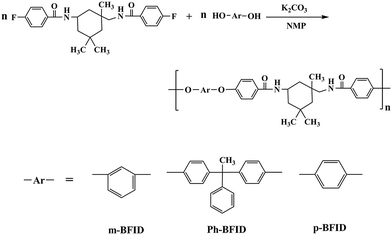
4-Fluorobenzoic chloride (4-FBC) (99.5%, Lanning Chemical Company Limited), BHPPE, which was synthesized as reported earlier by our group,35 sodium hydroxide (NaOH) (AR, SiChuan ChengDu ChangLian Chemical Reagent Company), N-methyl-2-pyrrolidone (NMP) (JiangSu NanJing JinLong Chemical Industry Company), PA6T-Dupont (DuPont Company, 18502 NC010), isophorone diamine and other reagents were obtained commercially.N,N′-Bis(4-fluorobenzoyl) isophorone diamine (BFID) (Scheme 1)
BFID was prepared as follows: deionized water (1200 mL), isophorone diamine (170 g, 1 mol), NaOH (80 g, 2 mol) and sodium dodecyl sulfate (3 g, 0.01 mol) were added into a 5000 mL three-necked flask under stirring. When the mixture was dissolved, a solution of 4-FBC (320 g, 2.02 mol) dissolved in dichloromethane (2000 mL) was added dropwise. Subsequently, 4-FBC solution was added into the flask, and the reaction mixture was stirred at ambient temperature for another 8 h. Afterwards, the mixture was filtered and washed with deionized water 3 times. Then, the crude product was recrystallized from ethanol. Finally, it was dried at 80 °C for about 24 h.Polymer synthesis
The polymerization was carried out as follows (Scheme 2). BFID (41.4 g, 0.1 mol), BHPPE (29.0 g, 0.1 mol), potassium carbonate (27.6 g, 0.2 mol), toluene (20 mL) and NMP (150 mL) were added into a 500 mL three-necked flask and then the flask was heated to 180 °C to remove the byproduct i.e., water. Afterwards, the reaction temperature was raised to 202 °C and maintained for about 6 h to yield high molecular weight polymers. The reacted solution was poured into water to precipitate fibrous polymers. The precipitate was pulverized to powder and washed with hot water 3 times. Then, the crude product was dried at 100 °C for 24 h to yield pure resins (yield: 63.1 g, 95.1%). FT-IR (cm−1): 3326 (N–H of amide group), 1640 (–CONH–), 3056 (C–H of benzene ring), 2951, 2914 (–CH3), 1596, 1539, 1493 (benzene ring skeleton), 846 (para substitution of the benzene ring). 1H-NMR [(400 MHz, deuterated DMSO-d6)/TMS, ppm]: 0.900 (s, 3H, H1), 1.027–1.049 (d, 6H, H2), 1.156–1.188 (m, 4H, H3–H4), 1.529 (s, 2H, H5), 2.112 (s, 3H, H6), 3.047 (s, 2H, H7), 4.148–4.169 (d, 1H, H8), 6.974–7.073 (m, 14H, H9–H12), 7.192–7.208 (m, 1H, H13), 7.217–7.281 (m, 2H, H14), 7.831–7.881 (m, 4H, H15), 8.104–8.123 (d, 1H, H16), 8.296 (s, 1H, H17).m-BFID and p-BFID were synthesized with a procedure similar to that of Ph-BFID.
Characterization
The intrinsic viscosities of m-BFID, Ph-BFID and p-BFID were tested at 30 ± 0.1 °C with a dilute solution of the polymers in NMP. The values were calculated with the following equation:where ηr = η/η0 and ηsp = η/η0 − 1.
The number-average molecular weight (Mn) and weight-average molecular weight (Mw) were obtained via GPC, which was performed using a Waters 1515 performance liquid chromatography pump, a Waters 2414 differential refractometer (Waters Co., Milford, MA) and a combination of Styragel HT-3 and HT-4 columns (Waters Co., Milford, MA). Their effective molecular weight ranges were 500–30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 5000–800
000 and 5000–800![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. N,N-Dimethyl formamide (DMF) was used as the eluent at a flow rate of 1.0 mL min−1 at 35 °C. Polystyrene standards were used for calibration.
000. N,N-Dimethyl formamide (DMF) was used as the eluent at a flow rate of 1.0 mL min−1 at 35 °C. Polystyrene standards were used for calibration.
The samples of monomers were measured with an elemental analyzer (EURO EA-3000). FT-IR spectroscopic measurements were performed using a Nexus670 FT-IR instrument. 1H nuclear magnetic resonance (NMR) spectra were obtained using a Bruker-400 NMR spectrometer in deuterated dimethyl sulfoxide. Differential scanning calorimetry (DSC) was performed using a Netzsch DSC 200 PC thermal analysis instrument. The heating rate for DSC measurements was 10 °C min−1. Thermogravimetric analysis (TGA) was performed using a TGA Q500 V6.4 Build 193 thermal analysis instrument with a heating rate of 10 °C min−1 in a nitrogen atmosphere. The polymer powder was processed into a sheet by a hydraulic press at 310 °C (pressure: 8 MPa) for 30 min. Then, the sheets were cut into specific shapes for the different tests. The samples with the dimensions of 50.0 mm × 3 mm × 1.0 mm (length × width × thickness) were prepared for tensile tests. The samples with the dimensions of 30.0 mm × 3.7 mm × 1.0 mm (length × width × thickness) were prepared for DMA experiments. The disks (diameter: 3 cm and thickness: 1 mm) were prepared for parallel-plate rheological measurements. An Instron Corporation 4302 instrument was used to study the stress–strain behavior of the samples. Dynamic mechanical analysis (DMA) was performed using a TA-Q800 apparatus operating in tensile mode at a frequency of 1 Hz with a heating rate of 5 °C min−1. A parallel plate rheometer (Bohlin Gemini 200, Britain) was fitted with 2.5 cm diameter stainless steel parallel plates. Temperature (temperature scanning rate: 5 °C min−1, shear strain: 2%, shear frequency: 1 Hz) and time (shear strain: 2%, shear frequency: 1 Hz) sweep tests were performed under nitrogen atmosphere. Quantitative information for the melt flow of the samples could be obtained by recording the complex viscosities versus temperature and time during processing. The optical transmittance of the polymers at 200–800 nm was characterized by UV-vis spectroscopy (U-2310II) (the samples were prepared by the casting method to form a film of thickness 10–13 μm). The solubility of the polymers in various solvents was tested at ambient temperature and temperatures with the boiling points of the solvents.
Results and discussion
Synthesis and chemical structure of semi-aromatic difluorobenzamide monomer (BFID)
The monomer BFID was synthesized with an interfacial reaction at ambient temperature without any catalyst (shown in Scheme 1). From the FT-IR spectra of the monomers (Fig. 1), we found the characteristic absorptions of –CONH–, stretching of C–H (aromatic ring), aliphatic chain, vibration of C–F and para-substituted benzene rings were near 3300, 1630 cm−1, 3070 cm−1, 2960 cm−1, 1320 cm−1 and 850 cm−1, respectively. The 1H-NMR spectrum of BFID is shown in Fig. 2. The signals at 8.210–8.230 and 8.376–8.407 ppm are ascribed to amide protons. While the signals near 7.2–8.0 ppm are characteristic protons of the aromatic moiety. The signals ranging from 0.9 to 4.2 ppm are attributed to aliphatic chain protons. Both the results (FT-IR and 1H-NMR) confirm the occurrence of the reaction between the amine and chloride units.Synthesis of m-BFID, Ph-BFID and p-BFID
The polymerization was carried out by the nucleophilic substitution reaction with K2CO3 as the base. The reaction temperature was maintained from 140 °C to 202 °C. First, the reaction temperature was maintained at 140–160 °C to remove the byproduct, i.e. water. Then it was increased to 180–202 °C to obtain polymers with high molecular weight. The ηint values of m-BFID, Ph-BFID and p-BFID, which were determined by the method of intrinsic viscosity, were in the range of 0.89–1.21 dL g−1 (Table 2), whereas their Mw values, which were measured by GPC, were in the range of 1.40–1.68 × 105. These values indicate that the synthesized polymers exhibited similar molecular weights as that of the commercially available products.| Polymers | ηint (dL g−1) | Mn (g mol−1) | Mw (g mol−1) | PDI (Mw/Mn) |
|---|---|---|---|---|
| m-BFID | 1.02 | 6.74 × 104 | 1.52 × 105 | 2.25 |
| Ph-BFID | 0.89 | 5.63 × 104 | 1.40 × 105 | 2.49 |
| p-BFID | 1.21 | 8.31 × 104 | 1.68 × 105 | 2.02 |
Chemical structure of the polymers
The chemical structures of m-BFID, Ph-BFID and p-BFID were characterized by FT-IR and 1H-NMR. As shown in Fig. 1, the characteristic absorptions of the amide group appeared near 3320 and 1640 cm−1. In contrast with the monomer BFID, the absorption of the C–F vibration near 1320 cm−1 disappeared and a new absorption of ether units near 1100 cm−1, which is attributed to the stretching of ether units, was observed. This confirms the polymerization of bisphenol and BFID. Fig. 3 shows the 1H-NMR spectra of m-BFID. The signals of the aromatic ring protons are in the range of 6.7–8.14 ppm. The signals at 8.148 and 8.320 ppm are assigned to the amide unit. The signals at 6.5–8.5 ppm belong to the aromatic protons. The signals at 0.7–4.5 ppm are characteristic protons of the aliphatic moiety. The ratio of the corresponding integral curves was 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6
6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Combined with the FT-IR and elemental analysis results, the NMR data suggest that the polymerization proceeds as described in Scheme 2. The chemical structures of Ph-BFID and p-BFID were also characterized using 1H-NMR (Fig. 4 and 5) and FT-IR spectra and the results confirm that their structures are the same as what we have designed.
1. Combined with the FT-IR and elemental analysis results, the NMR data suggest that the polymerization proceeds as described in Scheme 2. The chemical structures of Ph-BFID and p-BFID were also characterized using 1H-NMR (Fig. 4 and 5) and FT-IR spectra and the results confirm that their structures are the same as what we have designed.
Thermal properties of m-BFID, Ph-BFID and p-BFID
The thermal properties of m-BFID, Ph-BFID and p-BFID were tested by DSC (Fig. 6) and TGA (Fig. 7). As presented in Fig. 6, the glass transition temperature of m-BFID, Ph-BFID and p-BFID was 217, 224 and 239 °C, respectively (seen in Table 3). They were higher than that of PA9T (Tg = 126 °C) and the copolymers of PA6T (Tg is about 135 °C). p-BFID exhibited the highest Tg value (239 °C) in this series. From the DSC curves, no melting endothermic peak was found. The incorporation of the large bulky group reduces the movement of the polymer chain, and thus it exhibits an amorphous nature. Fig. 7 shows the thermo-degradation curves of m-BFID, Ph-BFID and p-BFID. The initial degradation temperatures (T5%) of m-BFID, Ph-BFID and p-BFID in nitrogen were 425, 438 and 430 °C, respectively. These are close to that of PA9T (Td = 464 °C) and PA10T (Td = 472 °C) and considerably higher than their glass transition temperature. The results suggest that their thermal stability is good and suitable for melt processing.| Polymers | Tg (°C) | T5% (°C) | Tensile strengths (MPa) | Young's modulus (GPa) | Elongation at break (%) | Storage modulus at 200 °C (GPa) | |
|---|---|---|---|---|---|---|---|
| a Detected by dynamic mechanical analysis (DMA). | |||||||
| m-BFID | 217 | 226a | 425 | 85 ± 5.0 | 1.7 ± 0.20 | 17.0 ± 3.0 | 1.1 |
| Ph-BFID | 224 | 235a | 438 | 73 ± 8.0 | 1.7 ± 0.27 | 16.2 ± 4.8 | 1.0 |
| p-BFID | 239 | 246a | 430 | 91 ± 6.0 | 1.9 ± 0.11 | 15.5 ± 2.9 | 1.1 |
| PA9T | 115 | — | 464 | 86 ± 4.0 | 2.7 ± 0.16 | 4.2 ± 2.6 | — |
Mechanical properties
The tensile strength of m-BFID, Ph-BFID and p-BFID were tested using a universal testing machine and the results are summarized in Table 3. The average tensile strengths of the polymers were 85, 73 and 91 MPa, respectively. The elongation at break ranged from 15.5% to 17.0%. This suggests that m-BFID, Ph-BFID and p-BFID have similar tensile strength as PA9T. The thermal mechanical properties of the resulting polyamides were characterized by DMA. As shown in Fig. 8, one evident transition peak can be observed, which is defined as the α relaxation or glass transition temperature. The α relaxation temperatures of these semi-aromatic polyamides were 226, 235 and 246 °C. These Tg values were slightly higher than those tested by DSC. The storage modulus curves of m-BFID, Ph-BFID and p-BFID are shown in Fig. 9, in which the semi-aromatic polyamides showed the high storage modulus of 1.60, 1.32 and 1.57 GPa, respectively. It was also found that the storage modulus of these semi-aromatic polyamides remained at about 1 GPa at 190 °C, which indicates the good thermal mechanical performance of the semi-aromatic polyamides.Rheological properties of polymers
Rheological measurements were carried out to investigate the resulting polyamide rheological properties. We studied the effects of testing time (0–1830 s at 330 °C) on the complex viscosities of these semi-aromatic polyamides and PC (Fig. 10). The complex viscosities of m-BFID, Ph-BFID and p-BFID exhibited a slight dependence on time. The viscosities values of the resultant semi-aromatic polyamides remained almost unchanged during the whole testing, whereas an increase in the complex viscosities of PC and particularly for PA6T-Dupont (as shown in Fig. 11) could be observed. This indicates that these polyamides had better melt stability than PC and PA6T-Dupont. We also found that the complex viscosities of m-BFID, Ph-BFID, p-BFID, PC and PA6T-Dupont at different temperatures (from 300 °C to 360 °C) were in the range of 177–4830 Pa s (Fig. 12). This gives the basic information for the melt processing parameter selection, e.g. 310 °C (990–1350 Pa s) could be used as the suitable melt processing temperature. Ph-BFID was found to have the lowest viscosity among the samples. It suggests that when a bulky side unit is incorporated into the polyamide backbone, the complex viscosity decreases. The main reason for this phenomenon is attributed to the fact that the bulky groups decrease the inter-chain interaction.Optical properties
Here, the effects of the incorporation of a side methyl group on the polymer transmittance were investigated. The optical properties of m-BFID, Ph-BFID and p-BFID films and a comparative polycarbonate (PC) sample, such as the cutoff wavelength (λcutoff) and optical transmittance at 400 nm (Tran400) and 450 nm (Tran450), are summarized in Table 4. As shown in Fig. 13, the λcutoff of the m-BFID, Ph-BFID and p-BFID films and comparative sample (PC) were 291, 292, 299 and 276 nm, respectively. The transparencies of the prepared polyamides films measured at 400 and 450 nm were 80.87–84.03% and 83.16–85.84%, respectively. This indicates that the resultant semi-aromatic polyamides had better transmittance than traditional polyamides such as PA6 and PA66. Compared with PC, this also suggests that the resultant polyamides have almost identical transmittance. These polyamides could be promising candidate materials for advanced optical devices, which need both good optical and thermal properties.| Polymers | d (μm) | λcutoff (nm) | T400 (%) | T450 (%) |
|---|---|---|---|---|
| a d: the thickness of the films; λcutoff: cutoff wavelength; Tran400, Tran450: the transmittance of films at 450 nm and 450 nm. | ||||
| m-BFID | 9.2 | 291 | 84.03 | 85.84 |
| Ph-BFID | 9.3 | 292 | 83.99 | 85.22 |
| p-BFID | 9.0 | 299 | 80.87 | 83.16 |
| PC | 9.2 | 276 | 86.57 | 87.32 |
Solubilities
The solubilities of the resultant semi-aromatic polyamides are summarized in Table 5. It was found that these materials were soluble in strong polar solvents such as DMAC, DMSO, and NMP. This suggests that they have better solubility than traditional semi-aromatic polyamides such as PA6T and PA9T. While compared with alkyl polyamides, formic acid, phosphoric acid, acetone, and the solution of NaOH (1 M) cannot dissolve these semi-aromatic polyamides. This indicates that they have better corrosion resistance than aliphatic polyamides such as PA6 and PA66.| Solvents | Polymers | |||
|---|---|---|---|---|
| PA6 | m-BFID | Ph-BFID | p-BFID | |
| a +: soluble at room temperature; ±: swelling; −: insoluble with heating. | ||||
| Concentrated sulfuric acid | + | + | + | + |
| Formic acid | + | − | − | − |
| NMP | − | + | + | + |
| CF3COOH | + | + | + | ± |
| HCl (6 mol L−1) | − | − | − | − |
| Phosphoric acid | + | − | − | − |
| NaOH (1 mol L−1) | − | − | − | − |
| Acetone | − | − | − | − |
| Chloroform | − | − | − | − |
| DMAC | − | + | + | + |
| DMSO | − | + | + | + |
| 1,4-Dioxane | − | − | − | − |
| Toluene | − | − | − | − |
| Phenol + tetrachloroethane | − | − | − | − |
Conclusions
A difluoro-benzamide monomer containing the isophorone unit was synthesized with a facile interface reaction. It was then reacted with bisphenol by nucleophilic polycondensation to synthesize a type of semi-aromatic polyamide. The resultant semi-aromatic polyamides displayed good mechanical property, high thermal stabilities (such as Tg higher than 225 °C, T5% around 430 °C), good processability (including solution processing and melt processing) and melt stability when compared with the commercial materials. In addition, these polyamides exhibited good optical performance, which the traditional semi-aromatic polyamides do not have. The cutoff wavelength and transmittance of the resultant resin films (∼9 μm thickness) were around 290 nm and 85% (450 nm), respectively. Therefore, these semi-aromatic polyamides could be potentially used as heat resistant thermoplastic materials particularly when transparency is required.Acknowledgements
This work was supported by research grants from the Youth Fund Natural Science Foundation of China (21304060).References
- M. G. José, C. Félix, F. S. García, L. José and P. de la, High-performance aromatic polyamides, Prog. Polym. Sci., 2010, 35, 623–638 CrossRef PubMed.
- L. Crespo, G. Sanclimens, M. Pons, E. Giralt, M. Royo and F. Albericio, Peptide and Amide Bond-Containing Dendrimers, Chem. Rev., 2005, 105, 1663–1681 CrossRef CAS PubMed.
- G. Zhang, D. T. Bai, D. S. Li, S. R. Long, X. J. Wang and J. Yang, Synthesis and properties of polyamides derived from 4,6-bis(4-chloroformylphenylthio) pyrimidine and 3,6-bis(4-chloroformylphenylthio) pyridazine, Polym. Int., 2013, 62, 1358–1367 CrossRef CAS PubMed.
- S. M. Du, W. B. Wang, Y. Yan, J. Zhang, M. Tian, L. Q. Zhang and X. H. Wan, A facile synthetic route to poly(p-phenylene terephthalamide) with dual functional groups, Chem. Commun., 2014, 50, 9929–9931 RSC.
- J. C. Williams, M. Meador, B. Ann, L. McCorkle, C. Mueller and N. Wilmoth, Synthesis and Properties of Step-Growth Polyamide Aerogels Crosslinked with Triacid Chlorides, Chem. Mater., 2014, 26, 4163–4171 CrossRef CAS.
- S. H. Hsiao, C. W. Chen and G. S. Liou, Novel aromatic polyamides bearing pendent diphenylamino or carbazolyl groups, J. Polym. Sci., Part A: Polym. Chem., 2004, 42, 3302–3313 CrossRef CAS PubMed.
- G. S. Liou, H. Y. Lin and H. J. Yen, Synthesis and characterization of electroactive hyperbranched aromatic polyamides based on A2B-type triphenylamine moieties, J. Mater. Chem., 2009, 19, 7666–7673 RSC.
- Y. Rao, A. J. Waddon and R. J. Farris, Structure-property relation in poly(p-phenylene terephthalamide) (PPTA) fibers, Polymer, 2001, 42, 5937–5946 CrossRef CAS.
- J. J. Ferreiro, C. J. G. dela, A. E. Lozano and A. J. de, Polyisophthalamides with heteroaromatic pendent rings: synthesis, physical properties, and water uptake, J. Polym. Sci., Part A: Polym. Chem., 2005, 43, 5300–5311 CrossRef CAS PubMed.
- S. Zulfiqar, A. Kausar, M. Rizwan and M. I. Sarwar, Probing the role of surface treated montmorillonite on the properties of semi-aromatic polyamide/clay nanocomposites, Appl. Surf. Sci., 2008, 255, 2080–2086 CrossRef CAS PubMed.
- A. Ballistreri, D. Garozzo, M. Giuffrida and P. Maravigna, Thermal decomposition processes in aliphatic-aromatic polyamides investigated by mass spectrometry, Macromolecules, 1986, 19, 2693–2699 CrossRef CAS.
- A. J. Uddin, Y. Ohkoshi, Y. Gotoh, M. Nagura and T. Hara, Influence of moisture on the viscoelastic relaxations in long aliphatic chain contained semiaromatic polyamide, (PA9-T) fiber, J. Polym. Sci., Part B: Polym. Phys., 2003, 41, 2878–2891 CrossRef CAS PubMed.
- A. J. Uddin, Y. Ohkoshi, Y. Gotoh, M. Nagura and T. Hara, Melt spinning and laser-heated drawing of a new semiaromatic polyamide, PA9-T fiber, J. Polym. Sci., Part B: Polym. Phys., 2004, 42, 433–444 CrossRef CAS PubMed.
- A. J. Uddin, Y. Gotoh, Y. Ohkoshi, M. Nagura, R. Endo and T. Hara, Hydration in a new semiaromatic polyamide observed by humidity-controlled dynamic viscoelastometry and X-ray diffraction, J. Polym. Sci., Part B: Polym. Phys., 2005, 43, 1640–1648 CrossRef CAS PubMed.
- A. J. Uddin, Y. Ohkoshi, Y. Gotoh, M. Nagura, R. Endo and T. Hara, Effects of take-up speed of melt spinning on the structure and mechanical properties of maximally laser drawn PA9-T fibers, Int. Polym. Process., 2006, 21, 263–271 CrossRef CAS.
- D. J. Liaw, P. N. Hsu, W. H. Chen and S. L. Lin, Synthesis and Properties of New Soluble Polyamides Derived from 2,2’-Dimethyl-4,4’-bis(4-carboxyphenoxy) Biphenyl, Macromolecules, 2002, 35, 4669–4676 CrossRef CAS.
- G. Zhang, Y. X. Zhou, Y. Kong, Z. M. Li, S. R. Long and J. Yang, Semiaromatic polyamides containing ether and different numbers of methylene (2-10) units: synthesis and properties, RSC Adv., 2014, 4, 63006–63015 RSC.
- D. J. Liaw, F. C. Chang, M. K. Leung, M. Y. Chou and K. Muellen, High thermal stability and rigid rod of novel organosoluble polyimides and polyamides based on bulky and noncoplanar naphthalene-biphenyldiamine, Macromolecules, 2005, 38, 4024–4029 CrossRef CAS.
- O. Ivanysenko, S. Strandman and X. X. Zhu, Triazole-linked polyamides and polyesters derived from cholic acid, Polym. Chem., 2012, 3, 1962–1965 RSC.
- H. Y. Huang, Y. T. Lee, L. C. Yeh, J. W. Jian, T. C. Huang, H. T. Liang, J. M. Yeh and Y. C. Chou, Photoactively electroactive polyamide with azo group in the main chain via oxidative coupling polymerization, Polym. Chem., 2013, 4, 343–350 RSC.
- D. Matsunaga, T. Tamaki and K. Ichimura, Azo-pendant polyamides which have the potential to photoalign chromonic lyotropic liquid crystals, J. Mater. Chem., 2003, 13, 1558–1564 RSC.
- S. Shabbir, S. Zulfiqar and M. I. Sarwar, Amine-terminated aromatic and semi-aromatic hyperbranched polyamides: synthesis and characterization, J. Polym. Res., 2011, 18, 1919–1929 CrossRef CAS PubMed.
- S. Shabbir, S. Zulfiqar, Z. Ahmad and I. S. Muhammad, Pyrimidine based carboxylic acid terminated aromatic and semiaromatic hyperbranched polyamide-esters: synthesis and characterization, Tetrahedron, 2010, 66, 7204–7212 CrossRef CAS PubMed.
- S. H. Yang, P. Fu, M. Y. Liu, Y. D. Wang, Z. P. Li and Q. X. Zhao, Synthesis, characterization and thermal decomposition of poly(decamethylene 2,6-naphthalamide), eXPRESS Polym. Lett., 2010, 4, 346–354 CrossRef CAS.
- M. Shabanian, N. J. Kang, J. W. Liu, U. Wagenknecht, G. Heinrichbd and D. Y. Wang, Bio-based semi-aromatic polyamide/functional clay nanocomposites: preparation and properties, RSC Adv., 2014, 4, 23420–23427 RSC.
- M. Shabanian, N. J. Kang, D. Y. Wang, U. Wagenknecht and G. Heinrich, Synthesis, characterization and properties of novel aliphatic–aromatic polyamide/functional carbon nanotube nanocomposites via in situ polymerization, RSC Adv., 2013, 3, 20738–20745 RSC.
- C. H. R. M. Wilsens, Y. S. Deshmukh, B. A. J. Noordover and S. Rastogi, Influence of the 2,5-Furandicarboxamide Moiety on Hydrogen Bonding in Aliphatic-Aromatic Poly(ester amide)s, Macromolecules, 2014, 47, 6196–6206 CrossRef CAS.
- G. Zhang, G. S. Huang, D. S. Li, X. J. Wang, S. R. Long and J. Yang, Facile Synthesis of Processable Semiaromatic Polyamides Containing Thioether Units, Ind. Eng. Chem. Res., 2011, 50, 7056–7064 CrossRef CAS.
- S. Moisa, G. Landsberg, D. Rittel and J. L. Halary, Hysteretic thermal behavior of amorphous semi-aromatic polyamides, Polymer, 2005, 46, 11870–11875 CrossRef CAS PubMed.
- S. H. Yang, P. Fu, M. Y. Liu, Y. D. Wang, Y. C. Zhang and Q. X. Zhao, Synthesis, characterization of polytridecamethylene 2,6-naphthalamide as semiaromatic polyamide containing naphthalene-ring, eXPRESS Polym. Lett., 2010, 4, 442–449 CrossRef CAS.
- W. Z. Wang, X. W. Wang, R. X. Li, B. Y. Liu, E. G. Wang and Y. H. Zhang, Environment-Friendly Synthesis of Long Chain Semiaromatic Polyamides with High Heat Resistance, J. Appl. Polym. Sci., 2009, 114, 2036–2042 CrossRef CAS PubMed.
- T. F. Novitsky, C. A. Lange, L. J. Mathias, S. Osborn, R. Ayotte and S. Manning, Eutectic melting behavior of polyamide 10,T-co-6,T and 12,T-co-6,T copolyterephthalamides, Polymer, 2010, 51, 2417–2425 CrossRef CAS PubMed.
- M. Y. Liu, K. F. Li, S. H. Yang, P. Fu, Y. D. Wang and Q. X. Zhao, Synthesis and Thermal Decomposition of Poly(dodecamethylene terephthalamide), J. Appl. Polym. Sci., 2011, 122, 3369–3376 CrossRef CAS PubMed.
- Y. Song, J. Y. Wang, G. H. Li, Q. M. Sun, X. G. Jian, J. Teng and H. B. Zhang, Synthesis, characterization and optical properties of cross-linkable poly(phthalazinone ether ketone sulfone), Polymer, 2008, 49, 724–731 CrossRef CAS PubMed.
- G. Zhang, X. J. Xing, D. S. Li, X. J. Wang and J. Yang, Effects of thioether content on the solubility and thermal properties of aromatic polyesters, Ind. Eng. Chem. Res., 2013, 52, 16577–16584 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |