Very efficient and broad-in-scope palladium-catalyzed Hiyama cross-coupling. The role of water and copper(I) salts†
Carla I. Traficante,
Ernesto G. Mata* and
Carina M. L. Delpiccolo*
Instituto de Química Rosario (CONICET-UNR), Facultad de Ciencias Bioquímicas y Farmacéuticas, Universidad Nacional de Rosario, Suipacha 531, 2000 Rosario, Argentina. E-mail: mata@iquir-conicet.gov.ar; Fax: +54 341 4370477; Tel: +54 341 4370477
First published on 9th March 2015
Abstract
A very high-yielding Pd-catalyzed cross-coupling between aryl halides and aryl(trialkoxy)silanes is achieved in the presence of Cu(I) and a measured amount of water. This novel methodology is useful for the generation of a wide range of biaryls, particularly non-para substituted derivatives, which are usually less reported.
Metal-catalyzed carbon–carbon bond forming reactions have become crucial synthetic tools for preparing complex molecules from simple precursors.1 Among them, the Hiyama reaction is drawing increasing attention as a very attractive methodology for obtaining biaryl and alkyl and vinyl aromatic derivatives. Hiyama coupling is achieved by treating aryl or alkenyl halides or pseudo halides with organosilicon compounds under palladium catalysis and activation by a fluoride ion or a base.2
Particularly, the synthesis of unsymmetrical biaryl moieties have wide applications as polymers, agrochemicals, and pharmaceutical intermediates.3 While Stille4 and Suzuki5 reactions have been widely recognized for generating aryl–aryl coupling products, the value of Hiyama reaction lays on the use of organosilanes as coupling partners, due to their ease of preparation and handling, stability toward air/moisture, and low toxicity compared to tin and boron reagents. Besides, Hiyama coupling is interesting from the environmental point of view since silicon waste is easily converted to innocuous silicon dioxide by incineration.6
The importance of this methodology was established by Hiyama and Hatanaka when they demonstrated that the high affinity between silicon and fluoride ions can accelerate the rate-determining transmetalation step (Scheme 1).7
Better results were later obtained when fluorophilicity of the silanes was increased by introducing F–Si bounds.2a However, Hiyama coupling did not achieve great success in its early days. The reason was probably the intrinsic resistance of the organosilicon compounds to undergo cross-coupling reactions in the absence of a significant polarity at the C–Si bond. After the introduction by Tamao et al.8 of silicon species containing oxygen atoms, Hiyama coupling has expanded its use to a wide range of substrates by improving reactivity and selectivity since oxygen facilitate coordination to palladium atom, apart from increasing polarity at the C–Si bond. Since then, an extensive research has been carried out within this field, establishing different and very useful variants, such as those introduced by DeShong,9 and Denmark.10
As part of our research dealing with the generation of libraries of biologically promising compounds,11 we were particularly interested in carboxy-substituted biaryl derivatives, which have demonstrated attractive antimitotic properties as modulators of tubulin dynamics.12 We notice that, despite of the success of Hiyama coupling, most of the examples involving synthetically accessible aryl(trialkoxy)silanes9b are referring to the preparation of 4′-substituted 1,1′-biphenyl derivatives. Reaction of ortho and meta-substituted aryl iodide with (3-substituted-phenyl)triethoxysilanes is less described and, in many cases, achieved in low to moderate yields.6,9a,13
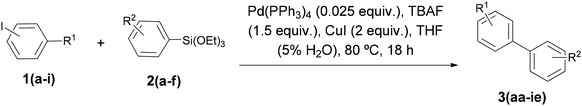
Hence, we decided to study the synthesis of methyl 3′-methoxybiphenyl-4-carboxylate (3aa) as model compound, starting from methyl 4-iodobenzoate (1a) and the corresponding aryl(triethoxy)silane (2a) in the presence of palladium catalyst and TBAF (Table 1). Using standard conditions, 1.5 equivalents of 2a and TBAF, catalytic tetrakis(triphenylphosphine)–palladium(0) (0.025 equiv.), at 80 °C in dry THF, for 18 h, the expected biaryl 3aa was obtained in only 19% isolated yield (entry 1). Interestingly, the ethyl ester analogue 4 was the major product (33%). Formation of 4 could be explained by a Pd-catalyzed transesterification with an ethoxy group which can be generated by a nucleophilic cleavage of one Si–O bond by the fluoride coming from the TBAF.14 Yield of 3aa increased from 19 to 45% without evidence of ethyl ester formation, under the same conditions but using non-anhydrous THF (entry 2). Later, by adding 2 equivalents of CuI, isolated yield of 3aa was improved to 85% (entry 3). The addition of stoichiometric copper(I) salts has been suggested to improve efficiency in Pd-catalyzed cross-coupling reactions,15 including Hiyama coupling.16 CuI has also proven to have a beneficial effect by preventing homocoupling of the aryl halide.17
| Entry | Catalyst (equiv.) | Co-catalyst (equiv.) | Solvent | Producta (%) |
|---|---|---|---|---|
| a Yield calculated after purification by column chromatography. NR = no reaction.b No TBAF was added.c CsF was added instead of TBAF.d Determined from the 1H NMR spectrum of the crude reaction mixture. | ||||
| 1 | Pd(PPh3)4 (0.025) | — | THF (anhydrous) | 3aa (19)/4 (33) |
| 2 | Pd(PPh3)4 (0.025) | — | THF (no anhydrous) | 3aa (44) |
| 3 | Pd(PPh3)4 (0.025) | CuI (2) | THF (no anhydrous) | 3aa (85) |
| 4 | Pd(PPh3)4 (0.025) | CuI (2) | THF (5% H2O) | 3aa (93) |
| 5 | — | CuI (2) | THF (5% H2O) | NR |
| 6 | Pd(PPh3)4 (0.025) | CuI (2) | THF (5% H2O) | NRb |
| 7 | Pd(PPh3)4 (0.025) | CuI (2) | THF (5% H2O) | 3aa (88)c |
| 8 | Pd(PPh3)4 (0.025) | CuI (2) | THF (50% H2O) | 1a/3aa/5d |
| 9 | Pd(OAc)2 (0.025) | CuI (2) | THF (5% H2O) | 3aa/5d |
Recently, the role of water in Hiyama reaction has began to be considered. Denmark noticed that hydration level of TBAF was crucial for the success of this cross-coupling during the synthesis of natural product isodomoic acid H,18 while Sajiki found that the addition of a measured amount of water significantly increase the yield of Hiyama cross-coupling between aryl halides and aryltriethoxysilanes.19 For this reason we decided to add 5% of water to the best conditions obtained until that moment. To our delight, methyl 3′-methoxybiphenyl-4-carboxylate (3aa) was isolated by column chromatography in 93% yield (entry 4). Based on entry 5, we could assume that CuI is only a co-catalyst, since no reaction was observed in the absence of Pd. Furthermore, TBAF is absolutely necessary since no reaction occurred in its absence (entry 6). Nevertheless, TBAF is not the only fluoride source that can be used, CsF was equally efficient, giving similar yield of the coupling product 3aa (entry 7). The addition increasing amount of water (entry 8), produced a mixture of 3aa, the starting material and homocoupling product 5. Such result was in agreement with a report by Sajiki and co-workers,20 where they assumed that the addition of a large excess of water might speed up the formation of an inactive silanol polymer which decreases the reactivity of the silane. Regarding palladium source, Pd(OAc)2 was not as efficient as Pd(PPh3)4 giving a mixture of 3aa and the homocoupling product 5 (entry 9).
To evaluate the scope and efficiency of the optimized methodology, we have synthesized a range of biaryl derivatives, achieving the same excellent results (Table 2). Complete conversion was obtained, in all cases with very high isolated yields. Both para and meta-substituted methyl iodobenzoates (1a–b) reacted smoothly with various para and meta-substituted aryltriethoxysilanes carrying an electron-donating or an electron-withdrawing group (entries 1–13). Moreover, in the case of the ortho-substituted methyl iodobenzoate (1c), coupling with triethoxy(3-methoxyphenyl)silane (2a) and triethoxy(p-tolyl)silane (2c) gave also the corresponding 3′ and 4′-substituted biphenyl-2-carboxylates (3ca and 3cc) in very high isolated yield (82 and 88%), albeit longer reaction times were required (entries 14 and 16). Apart from carboxy-substituted iodides, other aryl iodides were tested (entries 17–30). They have proven to be very effective substrates for our Hiyama coupling conditions, including those bearing strong electron-donating groups (entries 27–30). Following our aim to apply the Hiyama reaction to compounds of greater complexity and interest, we decided to use the optimized conditions on β-lactam derivatives. Thus, biaryl-contained β-lactams (3id and 3ie) where obtained in very high isolated yield (entries 31 and 32, Table 2).
| Entry | Aryl iodide | Silane | Product | Yielda (%) |
|---|---|---|---|---|
| a Yield calculated after purification by column chromatography.b Reaction carried out using 0.5 equiv. of CuI.c Reaction time: 40 h.d Yield calculated by 1H NMR from an inseparable mixture of cross-coupling product 3ee and homocoupling product from the silane 2e. | ||||
| 1 | 4-I, R1 = COOMe (1a) | R2 = 3-OMe (2a) | 3aa | 93 |
| 2 | 4-I, R1 = COOMe (1a) | R2 = –H (2b) | 3ab | 84 |
| 3 | 4-I, R1 = COOMe (1a) | R2 = 4-Me (2c) | 3ac | 98 |
| 4 | 4-I, R1 = COOMe (1a) | R2 = 4-OMe (2d) | 3ad | 86 |
| 5 | 4-I, R1 = COOMe (1a) | R2 = 4-Cl (2e) | 3ae | 93 |
| 6 | 4-I, R1 = COOMe (1a) | R2 = 4-CF3 (2f) | 3af | 77 |
| 7 | 4-I, R1 = COOMe (1a) | R2 = 3-OMe (2a) | 3aa | 90b |
| 8 | 3-I, R1 = COOMe (1b) | R2 = 3-OMe (2a) | 3ba | 93 |
| 9 | 3-I, R1 = COOMe (1b) | R2 = –H (2b) | 3bb | 78 |
| 10 | 3-I, R1 = COOMe (1b) | R2 = –H (2b) | 3bb | 78b |
| 11 | 3-I, R1 = COOMe (1b) | R2 = 4-Me (2c) | 3bc | 76 |
| 12 | 3-I, R1 = COOMe (1b) | R2 = 4-OMe (2d) | 3bd | 92 |
| 13 | 3-I, R1 = COOMe (1b) | R2 = 4-Cl (2e) | 3be | 85 |
| 14 | 2-I, R1 = COOMe (1c) | R2 = 3-OMe (2a) | 3ca | 82c |
| 15 | 2-I, R1 = COOMe (1c) | R2 = –H (2b) | 3cb | 90c |
| 16 | 2-I, R1 = COOMe (1c) | R2 = 4-Me (2c) | 3cc | 88c |
| 17 | 4-I, R1 = COMe (1d) | R2 = 3-OMe (2a) | 3da | 91 |
| 18 | 4-I, R1 = COMe (1d) | R2 = –H (2b) | 3db | 88 |
| 19 | 4-I, R1 = COMe (1d) | R2 = 4-Me (2c) | 3dc | 93 |
| 20 | 4-I, R1 = COMe (1d) | R2 = 4-OMe (2d) | 3dd | 85 |
| 21 | 4-I, R1 = COMe (1d) | R2 = 4-Cl (2e) | 3de | 91 |
| 22 | 4-I, R1 = Me (1e) | R2 = 3-OMe (2a) | 3ea | 76 |
| 23 | 4-I, R1 = Me (1e) | R2 = 4-OMe (2d) | 3ed | 93 |
| 24 | 4-I, R1 = Me (1e) | R2 = 4-Cl (2e) | 3ee | 62d |
| 25 | I, R1 = H (1f) | R2 = 3-OMe (2a) | 3fa | 88 |
| 26 | I, R1 = H (1f) | R2 = 4-OMe (2d) | 3fd | 93 |
| 27 | 4-I, R1 = NEt2 (1g) | R2 = –H (2b) | 3gb | 65 |
| 28 | 4-I, R1 = NEt2 (1g) | R2 = 4-OMe (2d) | 3gd | 71 |
| 29 | 4-I, R1 = OMe (1h) | R2 = –H (2b) | 3fd | 95 |
| 30 | 4-I, R1 = OMe (1h) | R2 = 4-Me (2c) | 3ed | 92 |
| 31 |  |
R2 = 4-OMe (2d) | 3id | 68 |
| 32 |  |
R2 = 4-Cl (2e) | 3ie | 90 |
Since biaryl derivatives substituted with heteroatoms are present in biologically active structures,21 we have extended the scope to the reaction with heteroaryl iodides and silanes. The 3-iodopyridine (1j), 2-iodothiophene (1k) and triethoxy(thiophen-2-yl)silane (2g) successfully underwent the coupling reaction to give the desired products in very high yields (Scheme 2).
 | ||
| Scheme 2 Hiyama reaction using 3-iodopyridine (1j), 2-iodothiophene (1k) and triethoxy(thiophen-2-yl)silane (2g) as substrates. | ||
A tentative mechanism for this undescribed version of the aryl(trialkoxy)silane-type Hiyama coupling, involving the combination of adding copper salts and a measured amount of water, is outlined in Scheme 3. It was theorized that a small amount of water could lead to a partial hydrolysis of the arylalkoxysilanes (A) giving a mixture of the corresponding arylsilanol derivatives (B).20 The electrophilic character of the silicon atom is strengthened by the formation of silanols B, facilitating the attack of the fluoride from TBAF to give the pentacoordinate arylsilicate anion C.22 After the active complex C is formed, a transmetalation occurs in the presence of CuI to give the organocuprate intermediate D,17 which, in turn, is subjected to a further transmetalation to give the organopalladium species which finally undergoes a reductive elimination to give the desired biaryl compound and regenerate the palladium(0) catalyst.23
 | ||
| Scheme 3 Plausible metal catalyzed mechanism of the Hiyama cross-coupling with copper iodide and measured amount of water. | ||
Conclusions
In summary, we have developed a novel version of the Hiyama reaction in which the role of Cu(I) and a small amount of water is crucial for achieving very high yields, after purification by column chromatography. The reaction conditions were found to be applicable to the preparation of unsymmetrical biaryl compounds. Particularly outstanding is the generation of non-linearly substituted biphenyl derivatives, such as, for instance, 3,3′, 3,4′ and 4,3′-disubstituted biphenyl derivatives, which syntheses are scarcely reported and often achieved in low to moderate yields. From the best of our knowledge, the combination of adding copper salts and a measured amount of water has not been yet applied to Hiyama coupling involving synthetically accessible aryl(trialkoxy)silanes. Further studies are in progress to extend of use of this protocol to the generation of libraries of biologically promising compounds.Acknowledgements
Support from CONICET, ANPCyT, Fundación Prats and Universidad Nacional de Rosario from Argentina is gratefully acknowledged. C.I.T. thanks CONICET for fellowship.Notes and references
- J. F. Hartwig, Organotransition metal chemistry: from bonding to catalysis, University Science Books, Sausalito, CA, 2010 Search PubMed.
- (a) Y. Hatanaka and T. Hiyama, J. Org. Chem., 1989, 54, 268 CrossRef CAS; (b) Y. Hatanaka, S. Fukushima and T. Hiyama, Chem. Lett., 1989, 18, 1711 CrossRef; (c) Y. Nakao and T. Hiyama, Chem. Soc. Rev., 2011, 40, 4893 RSC; (d) H. F. Sore, W. R. J. D. Galloway and D. R. Spring, Chem. Soc. Rev., 2012, 41, 1845 RSC.
- (a) Transition Metals for Organic Synthesis: Building Blocks and Fine Chemicals, ed. M. Beller and C. Bolm, Wiley-VCH, Weinheim, 2nd edn, 2004 Search PubMed; (b) Handbook of Organopalladium Chemistry for Organic Synthesis, ed. E.-I. Negishi and A. de Meijere, John Wiley, New York, 2002 Search PubMed.
- (a) J. W. Labadie and J. K. Stille, J. Am. Chem. Soc., 1983, 105, 669 CrossRef CAS; (b) M. M. Heravi, E. Hashemi and F. Azimian, Tetrahedron, 2014, 70, 7 CrossRef CAS PubMed.
- N. Miyaura and A. Suzuki, Chem. Rev., 1995, 95, 2457 CrossRef CAS.
- T. Yanase, Y. Monguchi and H. Sajiki, RSC Adv., 2012, 2, 590 RSC.
- Y. Hatanaka and T. Hiyama, J. Org. Chem., 1988, 53, 918 CrossRef CAS.
- K. Tamao, K. Kobayashi and Y. Ito, Tetrahedron Lett., 1989, 30, 6051 CrossRef CAS.
- (a) M. E. Mowery and P. DeShong, Org. Lett., 1999, 1, 2137 CrossRef CAS; (b) A. S. Manoso, C. Ahn, A. Soheili, C. J. Handy, R. Correia, W. M. Seganish and P. DeShong, J. Org. Chem., 2004, 69, 8305 CrossRef CAS PubMed; (c) W. Seganish and P. DeSong, Org. Lett., 2004, 6, 4379 CrossRef CAS PubMed; (d) K. H. Shukla and P. DeShong, J. Org. Chem., 2008, 73, 6283 CrossRef CAS PubMed.
- (a) S. E. Denmark and C. S. Regens, Acc. Chem. Res., 2008, 41, 1486 CrossRef CAS PubMed; (b) W.-T. T. Chang, R. C. Smith, C. S. Regens, A. D. Bailey, N. S. Werner and S. E. Denmark, Org. React., 2011, 75, 213 CAS.
- (a) S. A. Testero and E. G. Mata, Org. Lett., 2006, 8, 4783 CrossRef CAS PubMed; (b) L. Méndez, S. A. Testero and E. G. Mata, J. Comb. Chem., 2007, 9, 189 CrossRef PubMed; (c) A. A. Poeylaut-Palena and E. G. Mata, J. Comb. Chem., 2009, 11, 791 CrossRef CAS PubMed; (d) L. Méndez and E. G. Mata, J. Comb. Chem., 2010, 12, 810 CrossRef PubMed; (e) A. A. Poeylaut-Palena, S. A. Testero and E. G. Mata, Chem. Commun., 2011, 47, 1565 RSC; (f) E. Cortés, L. Méndez, E. G. Mata, R. Abonia, J. Quiroga and B. Insuasty, Mol. Diversity, 2012, 16, 839 CrossRef PubMed; (g) P. G. Cornier, D. B. Boggián, E. G. Mata and C. M. L. Delpiccolo, Tetrahedron Lett., 2012, 53, 632 CrossRef CAS PubMed; (h) A. La-Venia, S. A. Testero, M. P. Mischne and E. G. Mata, Org. Biomol. Chem., 2012, 10, 2514 RSC; (i) P. G. Cornier, C. M. L. Delpiccolo, D. B. Boggián and E. G. Mata, Tetrahedron Lett., 2013, 54, 4742 CrossRef CAS PubMed.
- L. Laraia, J. Stokes, A. Emery, G. J. McKenzie, A. R. Venkitaraman and D. R. Spring, ACS Med. Chem. Lett., 2014, 5, 598 CrossRef CAS PubMed.
- C. I. Traficante, C. M. L. Delpiccolo and E. G. Mata, ACS Comb. Sci., 2014, 16, 211 CrossRef CAS PubMed.
- C. M. So, H. W. Lee, C. P. Lau and F. Y. Kwong, Org. Lett., 2009, 11, 317 CrossRef CAS PubMed.
- J. Z. Deng, D. V. Paone, A. T. Ginnetti, H. Kurihara, S. D. Dreher, S. A. Weissman, S. R. Stauffer and C. S. Burgey, Org. Lett., 2009, 11, 345 CrossRef CAS PubMed.
- F. Louërat and P. C. Gros, Tetrahedron Lett., 2010, 51, 3558 CrossRef PubMed.
- P. Pierrat, P. Gros and Y. Fort, Org. Lett., 2005, 7, 697 CrossRef CAS PubMed.
- S. E. Denmark, J. H.-C. Liu and J. M. Muhuhi, J. Am. Chem. Soc., 2009, 131, 14188 CrossRef CAS PubMed.
- Y. Monguchi, T. Yanase, S. Mori and H. Sajiki, Synthesis, 2013, 45, 40 CrossRef CAS PubMed.
- T. Yanase, S. Mori, Y. Monguchi and H. Sajiki, Chem. Lett., 2011, 40, 910 CrossRef CAS.
- (a) K. L. Billingsley and S. L. Buchwald, Angew. Chem., 2008, 120, 4773 CrossRef; (b) E. Tyrell and P. Brookes, Synthesis, 2004, 4, 469 Search PubMed.
- C. J. Handy, A. S. Manoso, W. T. McElroy, W. M. Seganish and P. DeShong, Tetrahedron, 2005, 61, 12201 CrossRef CAS PubMed.
- Since we proposed that Cu(I) may be regenerated after the transmetalation process, a couple of test reactions were performed using 0.5 equiv. of CuI, obtaining similar yields to those using 2 equiv. of the Cu salt (Table 2, entries 7 and 10).
Footnote |
| † Electronic supplementary information (ESI) available: See DOI: 10.1039/c5ra03732h |
| This journal is © The Royal Society of Chemistry 2015 |