Microgels formed by enzyme-mediated polymerization in reverse micelles with tunable activity and high stability†
Song Bao,
Dongbei Wu*,
Teng Su,
Qing Wu and
Qigang Wang*
Department of Chemistry, Shanghai Key Lab of Chemical Assessment and Substainability, Advanced Research Institute, Tongji University, Shanghai 200092, PR China. E-mail: wangqg66@tongji.edu.cn; wudongbei@tongji.edu.cn
First published on 12th May 2015
Abstract
This communication describes the preparation of microgels via enzyme-triggered inverse emulsion polymerization, which provides an effective method for immobilizing enzymes with tunable catalytic performance and high stability.
Microgels are polymer particles with a hydrophilic network that serve as better carriers relative to bulk hydrogels1,2 in the fields of biocatalysis and drug delivery due to their simple substance diffusion and stimulus-responsive behaviors.3,4 Two commonly used methods for the preparation of microgels are precipitation polymerization and emulsion polymerization. Precipitation polymerization is a temperature-controlled process that prepares microgels with narrow particle size distributions and is suitable for monomers with low critical solution temperatures, such as N-isopropyl acrylamide and N-vinylcaprolactam.5,6 Emulsion polymerization is generally used to prepare microgels via free radical polymerization of monomers within reverse micelles initiated by the cleavage of persulfate ions under harsh conditions.7–9 Recently, our group developed a mild enzyme-triggered radical polymerization approach for preparing a bulk hydrogel. Our results show that gelation via HRP (horseradish peroxidase)-mediated polymerization is rapid with very little loss of the active enzyme.10 Herein, we introduce enzyme-triggered polymerization into a reverse micelle system in order to generate uniform microgels with adjustable enzymatic activities and high stabilities under mild conditions.
The facile preparation of our microgel is depicted in Fig. 1 and includes the formation of a water-in-oil (W/O) inverse emulsion, gelation via enzymatic polymerization, and the final demulsification. As a general model, we first prepared a W/O inverse emulsion by injecting an aqueous solution of N,N-dimethylacrylamide (DMAA, 7.5 wt%), N,N′-methylenebisacrylamide (MBA, 0.18 wt%) and HRP (0.056 mM) dropwise into a continuous oil phase at room temperature composed of acetylacetone (ACAC, 42 mM), Tween 20 (6 wt%) and Span 80 (12 wt%) in octane. The optimized ratio of oil to water for obtaining a suitable homogeneous W/O inverse emulsion is 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The concentrations of each component were selected according to preliminary experiments as well as our previous work.10 The polymerization of the monomer in the aqueous phase and the subsequent preparation of the microgel were then achieved by the slow addition of H2O2 (16.0 mM) into the emulsion. During this process, the catalytic generation of ACAC carbon radicals by H2O2 and HRP can initiate chain growth and gel formation from DMAA and MBA via interfacial diffusion of ACAC from the oil phase to the aqueous phase. It is worth mentioning that all experiments, including emulsion preparation and microgel polymerization, must be conducted under argon to avoid quenching of the generated radicals by oxygen. After stirring for 6 hours, the microgel particles were demulsified by ethanol, washed twice with ethanol and distilled water, and then centrifuged in order to remove any chemical residue.
1. The concentrations of each component were selected according to preliminary experiments as well as our previous work.10 The polymerization of the monomer in the aqueous phase and the subsequent preparation of the microgel were then achieved by the slow addition of H2O2 (16.0 mM) into the emulsion. During this process, the catalytic generation of ACAC carbon radicals by H2O2 and HRP can initiate chain growth and gel formation from DMAA and MBA via interfacial diffusion of ACAC from the oil phase to the aqueous phase. It is worth mentioning that all experiments, including emulsion preparation and microgel polymerization, must be conducted under argon to avoid quenching of the generated radicals by oxygen. After stirring for 6 hours, the microgel particles were demulsified by ethanol, washed twice with ethanol and distilled water, and then centrifuged in order to remove any chemical residue.
The amount of MBA present plays an important role in the preparation of the microgels and can affect various parameters of the microgel particles (Table S1, ESI†). A microgel with 0.15 wt% MBA is weak due to its low crosslinker density, which allows for the possible release of HRP during separation. In general, more crosslinking points reduces the average mesh size. The mesh size of a microgel can be theoretically deduced according to the network structure model.11 Based on this model (Fig. S1, ESI†), the average mesh size within a microgel network decreases from 5.5 nm to 3.1 nm as the MBA concentration increases from 0.15 wt% to 0.90 wt% (Table S1, ESI†).
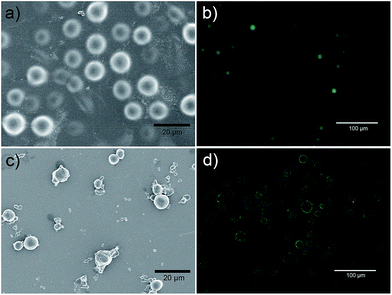
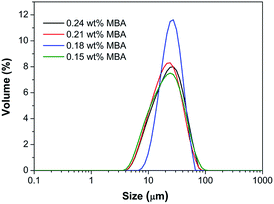
The swelling ratio of the microgels also decreases monotonically from 17.5 to 4.6 as the MBA concentration increases from 0.15 wt% to 0.90 wt%. Water absorption capacity is related to the effective chain length of the polymer, which can in turn decrease with crosslinker density.12 In addition, the reaction conversion was found to gradually increase from 27% to 62% when the concentration of MBA increased from 0.15 wt% to 0.90 wt%. The optimal concentration of MBA can be further determined by optical and SEM images of the final microgels, as shown in Fig. 2 and S2 and S3 (ESI†), which shows the difference of samples in the original situation. When the concentration of MBA was greater than 0.24 wt%, such as sample I-1 and I-2 (Fig. S2 and S3, ESI†), the morphologies of the particles were irregular due to the aggregation between microgels. Therefore, an optimized concentration of MBA between 0.15 wt% and 0.24 wt% was used for all subsequent preparations of the microgels. The average size of the vacuum-dried microgels was estimated using SEM images and found to be 5–20 μm (Fig. S2, ESI†). Moreover, the particle diameter size distribution determined using a Laser Diffraction Particle Size Analyzer (Fig. 3) was 5–80 μm, which is somewhat larger than the results found using SEM imaging due to the microgels' swelling capacities in water.
Biocatalytic evaluations of the self-immobilized HRP within the microgels are also critical for their further application due to the potential loss of activity during the gelation process. The oxidation of o-phenylenediamine (OPD) by H2O2 was selected as a model reaction for characterizing the catalytic activities of immobilized HRP (HRP(I)) and unconfined HRP (HRP(U)). For all activity tests, the concentration of HRP remained constant at 0.2 mg L−1. To compare the biocatalytic advantages of HRP(I) compared to HRP(U), kinetic parameters kcat were calculated by fitting Lineweaver–Burk plots.13 Our system using entrapped HRP possesses the unique advantage of having adjustable enzyme activity through changing the concentration of MBA. Fig. 4 shows plots of the activity value (kcat) as a function of MBA content. As MBA concentration increases, the value of kcat increases initially and then gradually decreases. When the initial amount of MBA is 0.18 wt% (sample I-5), kcat possesses a maximum value of 148 s−1, which is about 0.75 times that of unconfined HRP (Table 1). These results indicate majority of immobilized HRP's bioactivity is due to its aqueous micro-environment. Therefore, sample I-5 with a MBA concentration of 0.18 wt% was selected for further stability studies. The adjustable activity of HRP within the microgel can be attributed to the change in the microgel's average mesh size with various amounts of MBA. As shown in Table S1 (ESI†), the average mesh size of sample I-5 is about 5.2 nm, which matches well with the diameter of HRP (approximately 6 nm).14 Within the comfortable microenvironment of the microgel, HRP determines the maximum activity of sample I-5. The larger mesh size of the microgel with less than 0.18 wt% MBA can lead to a potential loss of HRP during separation. The smaller mesh size of the microgel with over 0.18 wt% MBA can affect both substrate and product diffusion within the microgel, reducing the overall biocatalytic activity of the entrapped HRP. Ultimately the catalytic activity of HRP(I) in the microgel can be adjusted by changing the crosslinker density and therefore mesh size of the microgel.
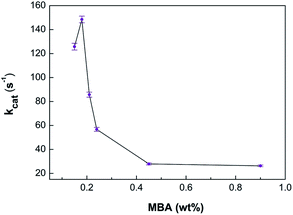 | ||
| Fig. 4 Catalytic activities in phosphate buffer solution (PBS, 50 mM, pH 7.0) at various concentrations of MBA. | ||
| Sample | Vmax (μM s−1) | Km (mM) | kcat (s−1) | kcat/Km (mM−1 s−1) |
|---|---|---|---|---|
| HRP(U) | 0.99 ± 0.01 | 2.13 | 198 ± 3 | 93.2 |
| HRP(I)-0 month | 0.74 ± 0.01 | 1.55 | 148 ± 3 | 95.6 |
| HRP(I)-3 months | 0.73 ± 0.01 | 1.51 | 146 ± 2 | 96.9 |
The microgel-entrapped HRP also exhibits enhanced performance under harsh conditions, such as incubation at higher temperatures. The microgel matrix can provide a protective water-rich microenvironment for the entrapped HRP, allowing it to achieve high thermal stability. Fig. 5 shows the thermal stability of free and immobilized HRP.15 HRP(I) remains at 70.7% of its initial catalytic activity after incubating for 5 min in PBS at 70 °C, while HRP(U) loses 73.3% of its activity after an incubation period of 5 min. After 30 min of incubation, HRP(I) and HRP(U) maintain 33.0% and 14.1% of their original activity, respectively, indicating effective protection of HRP by the microgel.
 | ||
| Fig. 5 Thermal deactivation treatment of HRP(U) and HRP(I) at 70 °C for various incubation times in PBS. | ||
In order to further test the storage stability of the immobilized HRP in the microgel, we also ran a catalytic reaction after storing the microgel at a temperature of 4 °C for 3 months. The result demonstrated that the microgel maintained 98.5% of its initial activity (Table 1), suggesting superior storage stability and excellent durability.
In summary, we have developed a mild and environmentally friendly strategy for the synthesis of microgels via inverse emulsion. Microgel-immobilized HRP with a suitable mesh size, good morphology and improved catalytic activity can be achieved by adjusting the concentration of MBA within the microgel. This microgel offers a platform for the immobilization of enzymes and therefore may have promising applications in some industrial fields. Furthermore, the excellent thermal stability and storage stability of our microgel gives it the potential to act as an enzyme-immobilized carrier for future commercial applications.
This work was supported by the National Natural Science Foundation of China (no. 21274111, 51473123 and 51402215), China Postdoctoral Science Foundation (2014M550245) and Shanghai Postdoctoral Science Foundation (14R21411200), the Recruitment Program of Global Experts, and Science & Technology Commission of Shanghai Municipality (14DZ2261100).
Notes and references
- (a) J. R. McKee, E. A. Appel, J. Seitsonen, E. Kontturi, O. A. Scherman and O. Ikkala, Adv. Funct. Mater., 2014, 24, 2706 CrossRef CAS PubMed; (b) Y. Kuang, J. F. Shi, J. Li, D. Yuan, K. A. Alberti, Q. B. Xu and B. Xu, Angew. Chem., Int. Ed., 2014, 53, 8104 CrossRef CAS PubMed; (c) A. Döring, W. Birnbaum and D. Kuckling, Chem. Soc. Rev., 2013, 42, 7391 RSC.
- (a) L. Tang, W. G. Liu and G. P. Liu, Adv. Mater., 2010, 22, 2652 CrossRef CAS PubMed; (b) X. L. Zhang, X. L. Chu, L. Wang, H. M. Wang, G. L. Liang, J. X. Zhang, J. F. Long and Z. M. Yang, Angew. Chem., Int. Ed., 2012, 51, 4388 CrossRef CAS PubMed; (c) Y. Chen and G. L. Liang, Theranostics, 2012, 2(2), 139 CrossRef CAS PubMed; (d) Q. Wu, T. Su, Y. J. Mao and Q. G. Wang, Chem. Commun., 2013, 49, 11299 RSC.
- (a) L. A. Lyon, Z. Y. Meng, N. Singh, C. D. Sorrell and A. S. John, Chem. Soc. Rev., 2009, 38, 865 RSC; (b) N. Welsch, M. Ballauff and Y. Lu, Adv. Polym. Sci., 2010, 234, 129 CrossRef CAS; (c) J. K. Oh, R. Drumright, D. J. Siegwart and K. Matyjaszewski, Prog. Polym. Sci., 2008, 33, 448 CrossRef CAS PubMed; (d) J. Zhang, F. Yang, H. Shen and D. C. Wu, ACS Macro Lett., 2012, 1, 1295 CrossRef CAS.
- (a) Y. Guan and Y. Zhang, Soft Matter, 2011, 7, 6375 RSC; (b) S. Wu, J. Dzubiella, J. Kaiser, M. Drechsler, X. Guo, M. Ballauff and Y. Lu, Angew. Chem., Int. Ed., 2012, 51, 2229 CrossRef CAS PubMed; (c) G. R. Hendrickson, M. H. Smith, A. B. South and L. A. Lyon, Adv. Funct. Mater., 2010, 20, 1697 CrossRef CAS PubMed; (d) H. Wei, R. X. Zhuo and X. Z. Zhang, Prog. Polym. Sci., 2013, 38, 503 CrossRef CAS PubMed.
- (a) J. Ramos, A. Imazb and J. Forcada, Polym. Chem., 2012, 3, 852 RSC; (b) X. L. Zhu, X. L. Gu, L. N. Zhang and X. Z. Kong, Nanoscale Res. Lett., 2012, 7(1), 519 CrossRef PubMed; (c) A. Pich and W. Richtering, in Adv Polym Sci: Chemical Design of Responsive Microgels, Springer, Verlag Berlin Heidelberg, 2010 Search PubMed.
- (a) R. H. Pelton and P. Chibante, Colloids Surf., 1986, 20, 247 CrossRef CAS; (b) R. Pelton, Adv. Colloid Interface Sci., 2000, 85, 1 CrossRef CAS; (c) I. Berndt, J. S. Pedersen, P. Lindner and W. Richtering, Langmuir, 2006, 22, 459 CrossRef CAS PubMed; (d) T. Hoare and R. Pelton, Langmuir, 2004, 20, 2123 CrossRef CAS; (e) J. Gao and B. J. Frisken, Langmuir, 2003, 19, 5212 CrossRef CAS.
- (a) K. Gawlitza, R. Georgieva, N. Tavraz, J. Keller and R. Klitzing, Langmuir, 2013, 29, 16002 CrossRef CAS PubMed; (b) M. B. Ansorge-Schumacher, in Handbook of Heterogeneous Catalysis; Immobilization of Biological Catalysts, WileyVCH, Weinheim, Germany, 2008 Search PubMed; (c) Q. Tao, A. Li, X. Liu, H. J. Gao, Z. K. Zhang, R. J. Ma, Y. L. An and L. Q. Shi, Colloids Surf., B, 2013, 111, 587 CrossRef CAS PubMed.
- (a) P. Y. Chow and L. M. Gan, in Polymer Particles: Microemulsion polymerizations and reactions, Springer, Berlin, 2005 Search PubMed; (b) H. Chen, L. H. Liu, L. S. Wang, C. B. Ching, H. W. Yu and Y. Y. Yang, Adv. Funct. Mater., 2008, 18, 95 CrossRef CAS PubMed.
- (a) M. Ciftci, M. A. Tasdelen, W. W. Li, K. Matyjaszewski and Y. Yagci, Macromolecules, 2013, 46, 9537 CrossRef CAS; (b) I. Berndt, J. S. Pedersen and W. Richtering, J. Am. Chem. Soc., 2005, 127, 9372 CrossRef CAS PubMed; (c) B. Mizrahi, S. Irusta, M. McKenna, C. Stefanescu, L. Yedidsion, M. Myint, R. Langer and D. S. Kohane, Adv. Mater., 2011, 23, H258 CrossRef CAS PubMed.
- T. Su, D. Zhang, Z. Tang, Q. Wu and Q. G. Wang, Chem. Commun., 2013, 49, 8033 RSC.
- (a) K. Haraguchi and T. Takehisa, Adv. Mater., 2002, 14, 1120 CrossRef CAS; (b) M. Wallace, D. J. Adams and J. A. Iggo, Soft Matter, 2013, 9, 5483 RSC.
- D. Takahashi, T. Hamada and T. Izumi, Polym. Bull., 2012, 68, 1777 CrossRef CAS.
- (a) Q. G. Wang, Q. M. Gao and J. L. Shi, J. Am. Chem. Soc., 2004, 126, 14346 CrossRef CAS PubMed; (b) Q. G. Wang, Z. M. Yang, L. Wang, M. L. Ma and B. Xu, Chem. Commun., 2007, 1032 RSC.
- H. G. Rennke and M. A. Venkatachalam, J. Histochem. Cytochem., 1979, 27(10), 1352 CrossRef CAS PubMed.
- S. Asad, S. F. Torabi, M. Fathi-Roudsari, N. Ghaemi and K. Khajeh, Int. J. Biol. Macromol., 2011, 48, 566 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details, supplementary figures and tables. See DOI: 10.1039/c5ra02162f |
| This journal is © The Royal Society of Chemistry 2015 |