Controlled growth of hexagonal Zn2GeO4 nanorods on carbon fibers for photocatalytic oxidation of p-toluidine†
Abstract
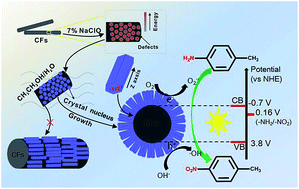
In this paper, hexagonal Zn2GeO4 nanorods grown on the surface of carbon fibers (CFs) that were pre-activated by sodium hypochlorite were prepared by a facile solvothermal method. The possible growth mechanism has been investigated by theoretical calculations and a simulation experiment by growth of Zn2GeO4 on a 2D flat surface of graphene. The (113) crystal facet of Zn2GeO4 nanorods attached to the CFs surface was the most stable structure. Due to the synergistic effect between the photocatalytic activity of Zn2GeO4 and excellent adsorption capacity of CFs, the resultant Zn2GeO4/CFs composites exhibited excellent photocatalytic activity for oxidation of p-toluidine.

 Please wait while we load your content...
Please wait while we load your content...