Regioselective three-component reactions of enaminones, 2-aminopyridines and enals for the synthesis of 1,2-dihydropyridines†
Shuo Caoa,
Luoting Xina,
Yunyun Liua,
Jie-Ping Wan*a and
Chengping Wen*b
aKey Laboratory of Functional Small Organic Molecules, Ministry of Education, and College of Chemistry and Chemical Engineering, Jiangxi Normal University, Nanchang 330022, P R China. E-mail: wanjieping@gmail.com; wanjieping@jxnu.edu.cn
bCollege of Basic Medical Sciences, Zhejiang Chinese Medical University, Hangzhou, 310053, P R China. E-mail: cpwen.zcmu@yahoo.com
First published on 10th March 2015
Abstract
A three-component synthetic method involving the assembly of enals, N,N-disubstituted enaminones and 2-aminopyridines has been designed, which leads to the facile and regioselective synthesis of 1,2-dihydropyridines (1,2-DHPs) with broad scope and generally good yields in the presence of simple acid catalysts.
As the most frequently occurring dihydropyridine isomers, 1,4- and 1,2-DHPs are known as important heterocyclic scaffolds due to their very broad applications in providing biologically functional compounds and participating in the construction of valuable organic products.1 Thanks to the pioneering work of Hantzsch and the continuous efforts of the chemical community, the synthesis of 1,4-DHPs has received spectacular success, as indicated by the occurrence of numerous new and diverse synthetic methodologies.2 These advances have also significantly contributed to the discovery of many useful biologically active compounds containing this privileged heterocycle. However, as the isomeric equivalents of 1,4-DHPs, the 1,2-DHPs have achieved far fewer advances, either in the field of organic synthesis or related biological investigation.3 The main reason is that the available synthetic methods, especially diversity-oriented synthetic methods towards 1,2-DHPs, are much fewer in number than the equivalent synthetic methods for 1,4-DHPs. Previously, the synthesis of 1,2-DHPs relied mainly on reduction or nucleophilic addition to pyridines/pyridinium salts4 and pericyclic ring formation.5 More recently, an array of different alternative strategies towards 1,2-DHPs has been reported, of which multicomponent reactions6 and domino reactions based on prior designed substrates7 comprise the major content. However, diversity-oriented synthetic methods using simple and readily available starting materials are still scarce. In this regard, developing new synthetic approaches such as a multicomponent synthesis for 1,2-DHPs with easily accessible reactants is currently an issue of urgency.
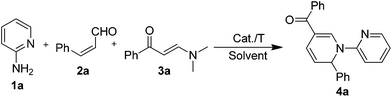
As a class of versatile synthetic building blocks with easy availability, dialkylamino functionalized enaminones have exhibited tremendous applications in organic synthesis, especially in the area of heterocycle synthesis.8 Notably, a typical transformation of these enaminones is the transamination of the dimethylamino group with a primary amine (or similar primary amino containing substrates, such as ammonium, etc.) component, which has been found to be useful in the synthesis of various N-containing heterocycles.9 By making use of such an enaminone as the main building block, we report herein the first three-component method for the synthesis of 1,2-DHPs containing an N-pyridinyl fragment by using α,β-unsaturated aldehydes, dimethylamino functionalized enaminones and 2-aminopyridines.
Initially, a model reaction using 2-aminopyridine 1a, cinnamaldehyde 2a and enaminone 3a was tentatively run in toluene in the presence of p-toluenesulfonic acid (PTSA) by heating at 90 °C, and it was found that 1,2-DHP 4a could be obtained in 22% yield (entry 1, Table 1). Based on this result in forming the 1,2-DHP 4a, optimization experiments were then conducted. Firstly, a survey of different solvents, such as toluene, MeCN, DMF, DMSO and THF, indicated that THF was a better solvent than the other tested candidates (entries 2–5, Table 1). Next, experiments using different acidic catalysts were also performed, wherein TMSCl turned out to be the best one out of the reactions using a single catalyst (entries 5–8, Table 1). However, the yield of the target product was not yet acceptable, even though the loading was varied and a strong acid, i.e. triflic acid, was tested (entries 9–11, Table 1). Considering this fact, the employment of combined acidic catalysts was attempted, and the reaction employing the AcOH/p-TSA system was found to give an evidently improved yield at a loading of 1 eq. (entries 12 and 13, Table 1). Reducing the amount of the acids led to a decrease in the product yield (entry 14, Table 1). The requirement of high acid loading might result from the basicity of the 2-aminopyridine reactant, which neutralized some of the acids.
| Entrya | Solvent | Catalyst | T (°C) | Yieldb (%) |
|---|---|---|---|---|
| a General conditions: 2-aminopyridine 1a (0.3 mmol), cinnamaldehyde 2a (0.3 mmol), enaminone 3a (0.3 mmol) and acid catalyst (0.15 mmol) in 2 mL of solvent, stirred at 90 °C for 12 h.b Yield of isolated products.c The external heating temperature was 90 °C.d 0.3 mmol catalyst.e 0.45 mmol TMSCl.f AcOH (0.3 mmol) and TMSCl (0.3 mmol).g AcOH (0.3 mmol) and p-TSA (0.3 mmol).h AcOH (0.15 mmol) and PTSA (0.15 mmol). | ||||
| 1 | Toluene | PTSA | 90 | 22 |
| 2 | MeCN | PTSA | Reflux | 20 |
| 3 | DMF | PTSA | 90 | 17 |
| 4 | DMSO | PTSA | 90 | Trace |
| 5c | THF | PTSA | Reflux | 25 |
| 6 | THF | TMSCl | Reflux | 31 |
| 7 | THF | TFA | Reflux | Trace |
| 8 | THF | Benzoic acid | Reflux | 25 |
| 9d | THF | TMSCl | Reflux | 40 |
| 10d | THF | Triflic acid | Reflux | 40 |
| 11e | THF | TMSCl | Reflux | 39 |
| 12f | THF | AcOH/TMSCl | 90 | 42 |
| 13g | THF | AcOH/p-TSA | 90 | 62 |
| 14h | THF | AcOH/p-TSA | 90 | 55 |
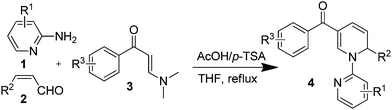
After obtaining the proper reaction parameters, the scope of this three-component reaction for synthesizing different 1,2-DHPs was examined by employing substrates containing various substituents on each of the components. As shown by the results listed in Table 2, the present synthetic protocol was well tolerated for the synthesis of 1,2-DHPs containing various functional groups, such as alkyl, alkoxy, halogen, nitro and fluorinated fragments. Generally, substituted functional groups with electron withdrawing properties had a positive impact on the yield of the related products (4e–4i, 4p and 4q, Table 2). On the other hand, the properties of the functional groups on the enal and enaminone components exhibited no clear pattern in their impact on the reaction results. While all 1,2-DHPs were acquired in moderate to good yields, the product diversity and the simple operation by way of multicomponent synthesis were notable advantages. All the products have been fully characterized, and the dd coupled peak of the vinyl proton around 5.7 ppm in the 1H NMR was the characteristic signal defining the presence of the 1,2-DHP ring.6g
| R1 | R2 | R3 | Product | Yield (%)b |
|---|---|---|---|---|
| a General conditions: 2-aminopyridine 1 (0.3 mmol), cinnamaldehyde 2 (0.3 mmol), enaminone 3 (0.3 mmol), AcOH–p-TSA (0.3 mmol/0.3 mmol) in 2 mL of solvent, stirred at reflux for 12 h.b Yield of isolated products. | ||||
| H | Ph | H | 4a | 62 |
| 4-CH3 | Ph | H | 4b | 43 |
| 6-CH3 | Ph | H | 4c | 40 |
| 4-Cl | Ph | H | 4d | 61 |
| 5-Cl | Ph | H | 4e | 64 |
| 5-Cl | Ph | 4-CF3 | 4f | 63 |
| 5-Cl | 2-ClC6H4 | 4-CH3O | 4g | 64 |
| 5-Cl | 2-ClC6H4 | 4-Cl | 4h | 74 |
| 5-Cl | 2-ClC6H4 | 4-NO2 | 4i | 60 |
| 4-Cl | 4-CH3OC6H4 | 4-CH3 | 4j | 44 |
| H | 3-CH3C6H4 | 4-NO2 | 4k | 44 |
| 5-Br | 4-NO2C6H4 | 4-Br | 4l | 44 |
| 4-Cl | 4-CH3OC6H4 | 2-CH3 | 4m | 40 |
| 4-CH3 | 4-NO2C6H4 | 4-Br | 4n | 41 |
| 4-CH3 | 4-NO2C6H4 | 3,4-Cl2 | 4o | 32 |
| 5-Cl | 4-NO2C6H4 | 4-CH3 | 4p | 70 |
| 4-Cl | 4-NO2C6H4 | 3,4-Cl2 | 4q | 63 |
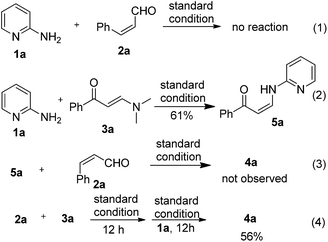
In order to probe the potential reaction mechanism, we conducted a series of control experiments as outlined in eqn (1)–(4). Under the standard reaction conditions, no reaction took place between 1a and 2a. The reaction of enaminone 3a and aminopyridine 1a led to the formation of NH-containing enaminone 5a through transamination; however, no target product 4a was observed when 5a was reacted with enal 2a under the standard conditions. On the other hand, an evident reaction took place by subjecting enal 2a and enaminone 3a to the reaction conditions. Although the isolation of the corresponding pure intermediate was not successful at this stage because of its sensitivity, simply adding 2-aminopyridine 1a to the reaction residue after the reaction of 2a and 3a was able to provide the 1,2-DHP 4a in reasonable yield (eqn (4)).
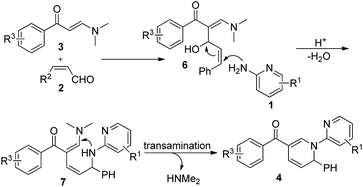
Based on the clues acquired from the control experiments, we proposed the reaction mechanism shown in Scheme 1. At the beginning, the Henry-type reaction in the presence of the enal, initiated by the nucleophilic α-carbon of the enaminone, leads to the occurrence of intermediate 6. Subsequently, the dehydration of 6 facilitates the addition of 2-aminopyridine to the double bond and provides another intermediate 7. Finally, the intramolecular transamination of 7 produces 1,2-DHP 4.
Conclusions
In conclusion, we have developed a new three-component synthetic method toward the selective, diversity-oriented synthesis of 1,2-DHPs via the assembly of 2-aminopyridines, enals and enaminones. The synthetic reaction is advantageous due to the use of cheap AcOH and p-TSA as catalysts and the broad synthetic tolerance. Due to the relatively rare availability of multicomponent synthetic methods for 1,2-DHPs, the present method will be useful in complementing the syntheses 1,2-DHPs.Acknowledgements
This work is financially supported by a grant from the Open Project Program from the College of Basic Medical Sciences, Zhejiang Chinese Medical University, a grant from the Open Project Program of the Key Laboratory of Functional Small Organic Molecules, Ministry of Education, Jiangxi Normal University (No. KLFS-KF-201418) and the Innovative Program of Graduate Students of Jiangxi Province (YC2014-S158).Notes and references
- (a) P. Ioan, E. Carosati, M. Micucci, G. Gruciani, F. Broccatelli, B. S. Zhorov, A. Chiarini and R. Budriesi, Curr. Med. Chem., 2011, 18, 4901 CrossRef CAS; (b) E. Carosati, P. Ioan, M. Micucci, F. Broccatelli, G. Gruciani, B. S. Zhorov, A. Chiarini and R. Budriesi, Curr. Med. Chem., 2012, 19, 4306 CrossRef CAS; (c) A. H. Li, S. Moro, N. Forsyth, N. Melman, X.-D. Ji and K. A. Jacobson, J. Med. Chem., 1999, 42, 706 CrossRef CAS PubMed; (d) R. M. Martin, R. G. Bergman and J. A. Ellman, Org. Lett., 2013, 15, 444 CrossRef CAS PubMed; (e) N. Nakano, K. Osone, M. Takeshita, E. Kwon, C. Seki, H. Matsuyama, N. Takano and Y. Kohari, Chem. Commun., 2010, 4827 RSC; (f) H. Nakano, N. Tsugawa, K. Takahashi, Y. Okuyama and R. Fujita, Tetrahedron, 2006, 62, 10879 CrossRef CAS PubMed; (g) F. Berti, V. D. Bussolo and M. Pineschi, J. Org. Chem., 2013, 78, 7324 CrossRef CAS PubMed.
- (a) A. Hantzsch Justus, Liebigs Ann. Chem., 1882, 215, 1 CrossRef; (b) U. Eisner and J. Kuthan, Chem. Rev., 1972, 72, 1 CrossRef CAS; (c) D. M. Stout and A. I. Meyers, Chem. Rev., 1982, 82, 223 CrossRef CAS; (d) J.-P. Wan and Y. Liu, RSC Adv., 2012, 2, 9763 RSC.
- For a review on the chemistry of 1,2-DHPs, see: E. M. P. Silva, P. A. M. M. Varandas and A. M. S. Silva, Synthesis, 2013, 3053 CrossRef CAS PubMed.
- (a) F. W. Foowler, J. Org. Chem., 1972, 37, 1321 CrossRef CAS; (b) R. J. Sundberg, G. Hamilton and C. Trindle, J. Org. Chem., 1986, 51, 3672 CrossRef CAS; (c) D. L. Comins, S. P. Joseph and R. R. Goehring, J. Am. Chem. Soc., 1994, 116, 4719 CrossRef CAS; (d) A. B. Charette, M. Grenon, A. Lemire, M. Pourashraf and J. Martel, J. Am. Chem. Soc., 2001, 123, 11829 CrossRef CAS.
- (a) K. Tanaka, H. Mori, M. Yamamoto and S. Katsumura, J. Org. Chem., 2001, 66, 3099 CrossRef CAS PubMed; (b) H. M. Sklenicka, R. P. Hsung, L.-L. Wei, M. J. McLaughlin, A. L. Gerasyuto and S. J. Degen, Org. Lett., 2000, 2, 1161 CrossRef CAS.
- (a) S. Yamada, T. Misonoo, C. Morita and N. Nunami, Tetrahedron Lett., 2003, 44, 7365 CrossRef CAS; (b) J.-P. Wan, S.-F. Gan, G.-L. Sun and Y.-J. Pan, J. Org. Chem., 2009, 74, 2862 CrossRef CAS PubMed; (c) J. Sun, Y. Sun, E.-Y. Xia and C.-G. Yan, ACS Comb. Sci., 2011, 13, 436 CrossRef CAS PubMed; (d) C. Challa, M. John and R. S. Lankalapalli, Tetrahedron Lett., 2013, 54, 3810 CrossRef CAS PubMed; (e) T. Harschneck and S. F. Kirsch, J. Org. Chem., 2011, 76, 2145 CrossRef CAS PubMed; (f) H. Wei, Y. Wang, B. Yue and P.-F. Xu, Adv. Synth. Catal., 2010, 352, 2450 CrossRef CAS; (g) J.-P. Wan, Y. Lin, Y. Jing, M. Xu and Y. Liu, Tetrahedron, 2014, 70, 7874 CrossRef CAS PubMed; (h) I. Yavari, M. J. Bayat, M. Sirouspour and S. Souri, Tetrahedron, 2010, 66, 7995 CrossRef CAS PubMed.
- (a) D. Tejedor, L. Cotos, G. Méndez-Abt and F. García-Tellado, J. Org. Chem., 2014, 79, 10655 CrossRef CAS PubMed; (b) D. I. S. P. Resende, S. Guieu, C. G. Oliva and A. M. S. Silva, Tetrahedron Lett., 2014, 55, 585 CrossRef PubMed; (c) Y. Hoshimoto, T. Ohata, M. Ohashi and S. Ogoshi, Chem.–Eur. J., 2014, 20, 4105 CrossRef CAS PubMed; (d) X. Xin, D. Wang, F. Wu, X. Li and B. Wan, J. Org. Chem., 2013, 78, 4065 CrossRef CAS PubMed; (e) Y. Shao, K. Zhu, Z. Qin, E. Li and Y. Li, J. Org. Chem., 2013, 78, 5731 CrossRef CAS PubMed; (f) T. Harschneck and S. F. Kirsch, J. Org. Chem., 2011, 76, 2145 CrossRef CAS PubMed; (g) M. R. Fructos, E. Álvarez, M. M. Díaz-Requejo and P. J. Pérez, J. Am. Chem. Soc., 2010, 132, 4600 CrossRef CAS PubMed; (h) D. Tejedor, G. Méndez-Abt and F. García-Tellado, Chem.–Eur. J., 2010, 16, 428 CrossRef CAS PubMed; (i) H. Liu, Q. Zhang, L. Wang and X. Tong, Chem. Commun., 2010, 46, 312 RSC.
- For reviews on enaminone chemistry, see: (a) A.-Z. A. Elassar and A. A. El-Khair, Tetrahedron, 2003, 59, 8463 CrossRef CAS; (b) G. Negri, C. Kascheres and A. J. Kascheres, J. Heterocycl. Chem., 2004, 41, 461 CrossRef CAS.
- For references on the transamination of dimethylamino functionalized enaminones and related syntheses, see: (a) Y. Liu, R. Zhou and J.-P. Wan, Synth. Commun., 2013, 43, 2475 CrossRef CAS; (b) S. Cao, Y. Jing, Y. Liu and J. Wan, Chin. J. Chem., 2014, 34, 876 CrossRef CAS; (c) J.-P. Wan, Y. Zhou and S. Cao, J. Org. Chem., 2014, 79, 9872 CrossRef CAS PubMed; (d) J.-P. Wan, Y. Zhou, K. Jiang and H. Ye, Synthesis, 2014, 3256 CrossRef CAS PubMed; (e) J.-P. Wan, R. Zhou, Y. Liu and M. Cai, RSC Adv., 2013, 3, 2477 RSC; (f) J.-P. Wan, C. Wang and Y. Pan, Tetrahedron, 2011, 67, 922 CrossRef CAS PubMed; (g) J.-P. Wan and Y.-J. Pan, Chem. Commun., 2009, 2768 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: General experimental information, procedure for the synthesis of 1,2-DHPs 4, characterization data, and 1H/13C NMR spectra of all products. See DOI: 10.1039/c5ra01901j |
| This journal is © The Royal Society of Chemistry 2015 |