Synthesis, crystal structure and photoluminescence properties of new blue-green Ba1−x(PO3)2:Eux2+ (0 < x ≤ 0.040) phosphors for near ultraviolet based white light-emitting diodes
Zhan-Chao Wu*a,
Hong-Hong Fua,
Jie Liua,
Shao-Ping Kuanga,
Ming-Mei Wu*b,
Jun-Gu Xubc and
Xiao-Jun Kuangc
aKey Laboratory of Sensor Analysis of Tumor Marker, Ministry of Education, Laboratory of Inorganic Synthesis and Applied Chemistry, College of Chemistry and Molecular Engineering, Qingdao University of Science and Technology, Qingdao 266042, P. R. China. E-mail: wuzhan_chao@163.com; Tel: +86 15806594068
bSchool of Chemistry and Chemical Engineering, Sun Yat-Sen University, Guangzhou 510275, P. R. China. E-mail: ceswmm@sysu.edu.cn
cCollege of Materials Science and Engineering, Guilin University of Technology, Guilin 541004, P. R. China
First published on 29th April 2015
Abstract
A series of new blue-green Ba1−x(PO3)2:Eux2+ (x = 0.005, 0.010, 0.020, 0.030, 0.035, 0.040) phosphors were synthesized by a solid-state reaction. The phase purities and crystal structures of the series were characterized by X-ray diffraction (XRD) and Rietveld refinements. Their photoluminescence properties were investigated by means of excitation, emission spectra and Commission International de L'Eclairage (CIE) chromaticity coordinates. The concentration quenching mechanism of Eu2+ emission in Ba(PO3)2 hosts was studied. Thermal stability was evaluated by temperature-dependent luminescence of Ba0.970(PO3)2:Eu0.0302+ phosphor. Moreover, the activation energy (Ea) for thermal quenching was calculated. The photoluminescence of Ba0.970(PO3)2:Eu0.0302+ phosphor shows higher thermal stability than that of the commercial blue phosphor BaMgAl10O17:Eu2+ (BAM). All the results show that Ba(PO3)2:Eu2+ phosphor is a potential blue-green phosphor for near ultraviolet-based white light-emitting diodes (WLED).
1. Introduction
White light-emitting diodes (WLEDs) have begun to replace conventional incandescent and fluorescent lamps in recent years due to their superior energy efficiencies, long lifetimes and environmental friendliness.1–3 Presently, white light is frequently created by combining a Y3Al5O12:Ce3+ phosphor with a blue InGaN chip.4,5 However, this method suffers a low color rendering index (Ra < 80) and high correlated color temperature (Tc > 4500 K) because it lacks a red component.6 To obtain white light with high Ra and suitable Tc, one approach is to use near-ultraviolet (NUV, 350–420 nm) LED chips with red, blue and green multi-phased phosphors.7 In this case, it is essential to develop new phosphors that can be effectively excited by NUV chips.As it is well known, rare earth ions with rich energy levels are ideal for designing new luminescent materials.8 In particular, Eu2+ is one of the most excellent activators due to its unique electronic configurations. Eu2+ has a 4f7 electronic configuration in the ground state and a 4f65d1 electronic configuration in the excited state. Since the transition between 4f7 and 4f65d1 states is spin and parity allowed, the absorption and emission of Eu2+ ions are broadband. In addition, the emission of Eu2+ ions strongly depends on the crystal field, which is influenced by the nature of the host and ligand–activator interactions. When Eu2+ is incorporated into different compounds, different types of emission centers can be formed. Therefore, the color of Eu2+ emission can vary from ultraviolet to red depending on the host materials.9,10
The phosphors in phosphor-conversion (pc) luminescent materials applied in WLEDs are generally required to have high fluorescence conversion efficiency for NUV light, appropriate excitation and emission bands, as well as high chemical stability. In addition, they should provide excellent thermally stable luminescence in order to withstand the high temperatures generated by LEDs, which generally operate about 120 °C. The thermal stability of the phosphors greatly influences the light output of pc-LEDs and the lifetimes of LED devices. Moreover, optimal blue phosphors are expected to be prepared at low temperatures from cheap raw materials. It can be noted that the high sintering temperature (over 1400 °C) of the commercial blue phosphor BaMgAl10O17:Eu2+ (BAM) requires specialized production equipment that consumes huge amounts of electric energy. Phosphors based on phosphates show many advantages, such as low preparation temperatures, high luminescence efficiencies, cheap raw materials and relative physical and chemical stabilities, compared with other host systems such as aluminates (SrAl2O4:Eu2+),11 silicates (β-Ca3SiO4Cl2:Eu2+),12 and nitrides ((Sr, Ca)2Si5N8:Eu2+).13
In 1968, Costas C. Lagos reported on the UV excitation of the divalent europium-activated Ba(PO3)2, which showed a weak violet emission band peak at 375 nm under 253.7 nm excitation.14,15 However, the structure of Ba(PO3)2:Eu2+ phosphor was not reported. Obviously, the Ba(PO3)2:Eu2+ phosphor reported by Lagos cannot be applied in WLEDs. Furthermore, there were few reports about it.
In this study, a series of new Eu2+-activated blue-green phosphate phosphors Ba1−x(PO3)2:Eux2+ (x = 0.005, 0.010, 0.020, 0.030, 0.035, 0.040) were prepared by a solid-state method at a lower temperature of 600 °C. The crystal structures, luminescence properties, and thermal stabilities of the phosphors were investigated in detail. Finally, the activation energy (Ea) for thermal quenching was calculated from the temperature-dependent luminescence of Ba0.970(PO3)2:Eu0.0302+ phosphor.
2. Experimental section
2.1. Materials and synthesis
The Ba1−x(PO3)2:Eux2+ samples (x = 0.005, 0.010, 0.020, 0.030, 0.035, 0.040) were synthesized by a solid-state reaction. The raw materials BaCO3 (AR grade), NH4H2PO4 (AR grade) and Eu2O3 (99.99% purity) were stoichiometrically weighed and thoroughly mixed by grinding in an agate mortar. Then, they were placed into an alundum crucible and heated in a muff furnace at 600 °C for 6 h in a CO atmosphere. Finally, the synthesized samples were allowed to cool to room temperature before further experiments.2.2. Measurements and characterization
Crystal phase identification was carried out on an X-ray diffractometer (XRD, D-MAX2500/PC, RIGAKU Corporation, Japan) using 40 kV, 20 mA and Cu Kα radiation (λ = 1.5418 Å). Microtopography and composition of the calcined particles were observed by scanning electron microscopy (SEM, JSM-6700F, JEOL, Japan) and energy disperse spectroscopy (EDS, INCA Energy, Oxford). Room temperature excitation and emission spectra of Ba(PO3)2:Eu2+ were analyzed by a fluorescence spectrophotometer (F-2700, HITACHI High-Technologies Corporation, Japan) with a photomultiplier tube operating at 400 V, and a 150 W Xe lamp was used as the excitation source. Temperature-dependent luminescence was measured by a FluoroMax-4 spectrofluorometer (HORIBA, JOBIN YVON, France) and the excitation sources used include a 150 W xenon lamp.3. Results and discussion
3.1. Crystal structure and microtopography
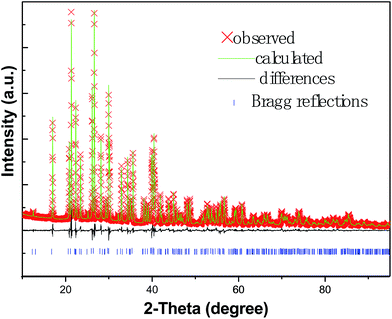
The XRD patterns of Ba1−x(PO3)2:Eux2+ samples (x = 0.005, 0.010, 0.020, 0.030, 0.035, 0.040) are shown in Fig. 1 and the standard data for Ba(PO3)2 (PDF card no. 43-0518) are also shown as a reference. All the diffraction peaks of the samples can be indexed to the standard data of PDF#43-0518, which has been refined to be orthorhombic with a space group of P212121 and cell parameters of a = 4.499 Å, b = 13.394 Å, c = 8.340 Å, Z = 4 and V = 502.53 Å3. The results indicate that the prepared phosphors are isostructural with Ba(PO3)2, and the doping of a small amount of Eu2+ has no obvious influence on the host structure. Moreover, no impurities are detected in the prepared products, suggesting that Eu2+ ions have been successfully incorporated in Ba(PO3)2 host lattice due to the similarities in ionic radii and valences between Ba2+ and Eu2+.To further study the structure of the obtained samples, Rietveld structure refinement of the Ba0.980(PO3)2:Eu0.0202+ phosphor was performed using Topas Academic software.16 Fig. 2 shows the observed, calculated and difference synchrotron XRD profiles for the Rietveld refinement of the Ba0.980(PO3)2:Eu0.0202+ phosphor. The results of the Rietveld refinement further indicate that Ba2+ was substituted by Eu2+ ion in the Ba(PO3)2 host and the incorporated Eu2+ ions caused no obvious impurity or secondary phases. The lattice parameters of the Ba0.980(PO3)2:Eu0.0202+ phosphor are a = 4.50128(5) Å, b = 13.4005(1) Å, c = 8.34468(8) Å and V = 503.350(9) Å3, which are slightly different because a small amount of Eu2+ replaces Ba2+. The final refined structural parameters are listed in Table 1. The refinement finally converged to Rp (profile R value) = 3.60%, Rwp (weighted-profile R value) = 4.82% and RB (Bragg-intensity R value) = 2.07%, indicating that all these factors satisfy well with the reflection condition. Fig. 3 and 4 show the refined unit cell structure of the samples. The Ba/Eu atoms, oxygen atoms and PO4 tetrahedra are plotted in blue, red and green, respectively. The black numbers in Fig. 4 denote the interatomic distances between Ba/Eu and coordinated O atoms. In the Ba(PO3)2 host, there is only one type of Ba2+ site and Ba is defined as being 10-coordinated. From the refinement results, the average length of Ba–O was calculated to be about 2.89 Å.
| Atoms | x | y | z | Occup. | Biso (Å2) |
|---|---|---|---|---|---|
| a a = 4.501 Å, b = 13.401 Å, c = 8.344 Å, V = 503.350 Å3, α = 90°, β = 90°, γ = 90°, Z = 4 with space group P212121, Rp = 3.60%, Rwp = 4.82%, RB = 2.07%. | |||||
| Ba1 | 0.25899 | 0.12586 | 0.19855 | 0.98120 | 0.93568 |
| Eu1 | 0.25899 | 0.12586 | 0.19855 | 0.01880 | 0.93568 |
| P1 | 0.70993 | 0.35414 | 0.21916 | 1 | 1.00402 |
| P2 | 0.21378 | 0.40271 | 0.42374 | 1 | 1.00402 |
| O1 | 0.37794 | 0.33489 | 0.28201 | 1 | 0.47143 |
| O2 | 0.88652 | 0.36844 | 0.39288 | 1 | 0.47143 |
| O3 | 0.68331 | 0.44888 | 0.13245 | 1 | 0.47143 |
| O4 | 0.79082 | 0.25747 | 0.12960 | 1 | 0.47143 |
| O5 | 0.78578 | 0.01036 | 0.12345 | 1 | 0.47143 |
| O6 | 0.79746 | 0.13454 | 0.39819 | 1 | 0.47143 |
The morphology of Ba0.970(PO3)2:Eu0.0302+ phosphor was observed by SEM (Fig. 5a). The microstructure of the phosphor consists of irregular fine grains with diameters ranging from 200 to 300 nm. It seems that the surface of the particles is not smooth and every particle is composed of many tiny particles. Therefore, the average crystalline size of the particles was estimated by the Debye–Scherrer equation:17
D = 0.89λ/β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos(θ) cos(θ)
| (1) |
3.2. Photoluminescence properties
Fig. 6 depicts the excitation and emission spectra of Ba1−x(PO3)2:Eux2+ (x = 0.005, 0.010, 0.020, 0.030, 0.035, 0.040) phosphors at room temperature. Monitored at 445 nm, the samples show a broad intense excitation band from 220 to 400 nm, which matches the emission of NUV chips. The excitation band consists mainly of three peaks located at 268, 323 and 350 nm. Since absorption bands of phosphate hosts are located in the vacuum ultraviolet region (100–200 nm),18 the excitation bands in the range of 220–400 nm are attributed to the electronic transition from the 4f7 ground state to the 4f65d1 excited state of the Eu2+ ions.19 The profile of the excitation spectrum is assigned to the crystal field splitting of the 5d level of Eu2+ ions. The excited 5d levels are not shielded from the crystal field, giving rise to a marked splitting of the excited level, which means the excitation spectrum is composed of several excitation peaks. For Ba(PO3)2:Eu2+ phosphor, the excitation peak at 323 nm is the strongest.The emission spectra of Ba1−x (PO3)2:Eux2+ (x = 0.005, 0.010, 0.020, 0.030, 0.035, 0.040) when excited by 323 nm light are shown in Fig. 6b. Upon excitation with NUV light at 323 nm, the sample shows a broad asymmetric intense blue-green emission band with the strongest peak around 445 nm, which is assigned to the electron transition from the 4f65d1 excited state to the 4f7 ground state of Eu2+.20 The emission intensity increases with the increase in Eu2+ concentration until a maximum value is reached, and then it decreases due to concentration quenching effects. The inset of Fig. 6b shows the dependence of the emission intensity on the concentration of Eu2+, which suggests that the optimum doping amount of Eu2+ is x = 0.030. In general, the concentration quenching of luminescence is due to energy transfer between the activator ions at high concentrations. In the energy transfer process, the energy is lost at a killer (or quenching) site, resulting in the decrease of luminescence intensity.21 It is also observed that the emission peaks shift gradually to longer wavelengths (from 438 to 450 nm) with increasing concentration of Eu2+, which is consistent with previous studies.19,22 The inter-atomic distance between two Eu2+ ions is expected to become shorter due to the increase of Eu2+-doping concentration, and the interaction is enhanced. The crystal field strength surrounding the Eu2+ ions is thus increased, leading to the red-shifting of the emission peak.
In addition, the excitation and emission spectra of Ba0.970(PO3)2:Eu0.0302+ and the commercial blue-emitting phosphor BAM are presented in Fig. 7 for comparison. In the range of 310–380 nm, Ba0.970(PO3)2:Eu0.0302+ shows a higher excitation intensity than BAM, which is favorable for application in NUV chip based LEDs. Similarly, Ba0.970(PO3)2:Eu0.0302+ exhibits a stronger and broader emission band than BAM upon NUV light excitation.
3.3. Temperature-dependent luminescence properties
The temperature-dependent luminescence of phosphors is an essential parameter, which has great influence on the light output of phosphors used in WLEDs. Fig. 8 shows temperature-dependent photoluminescence of Ba0.970(PO3)2:Eu0.0302+ phosphor excited at 323 nm in the temperature range of 298–423 K. It can be seen that the shape, position and intensity of the emission spectra gradually changed when the temperature was increased from 298 to 423 K. The emission integrated intensity of Ba0.970(PO3)2:Eu0.0302+ decreases gradually to 66.5% of the initial value as the temperature increases from 298 to 423 K. It can be noted that the emission integrated intensity of Ba0.970(PO3)2:Eu0.0302+ only decreased to 83.6% of the initial value when the temperature increases to 398 K, which is close to the working temperature of LEDs (about 120 °C). As a comparison, the temperature-dependent luminescence of commercial blue phosphor BAM is also shown in the inset of Fig. 8. The emission integrated intensity of Ba0.970(PO3)2:Eu0.0302+ decreases more slowly than that of BAM. These results suggest that Ba0.970(PO3)2:Eu0.0302+ has a higher thermal stability than BAM.The temperature-dependent emission intensity of Ba0.970(PO3)2:Eu0.0302+ phosphor can be described by a modified Arrhenius equation23 as:
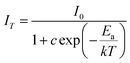 | (2) |
 | ||
| Fig. 9 The Arrhenius fitting of the emission intensity of Ba0.970(PO3)2:Eu0.0302+ phosphor and the calculated activation energy (Ea) for thermal quenching. | ||
The decrease in emission intensity with increasing temperature can be described by thermal quenching in a configuration coordinate diagram (shown in Fig. 10). Through phonon interactions, the excited luminescent center is thermally activated and then thermally released through the crossing point between the excited state and the ground state. This nonradiative transition probability is greatly dependent on the temperature, which leads to the decrease of emission intensity with increasing temperature. The main mechanism for the temperature quenching of Eu2+ luminescence in solids is the thermal excitation of the 5d electron to the conduction band states. The 5d electron probably remains bonded in a Eu2+ trapped excited state, from which it returns nonradiatively to the Eu2+ ground state. The activation energy referred to is the energy required to raise the electron from the relaxed excited level into the host lattice conduction band.25 The high quenching temperature and Ea for Eu2+ emission in Ba(PO3)2 indicate that the lowest energy level of the Eu2+ 4f65d1 electronic configuration is well isolated from the host lattice conduction band.
In addition, the maximum emission wavelength of Ba0.970(PO3)2:Eu0.0302+ shows a blue shift from 453 to 447 nm with increasing temperature. This phenomenon can be ascribed to thermal back-transfer over the barrier from the excited states of the low energy emission band to the excited states of the high energy emission band of Eu2+ by the assistance of thermally active phonons (Fig. 11). At low temperature, low-energy emission is dominant. As temperature increases, thermally activated phonon-assisted tunneling from the excited states of the low-energy emission band to the excited states of the high-energy emission band (see Fig. 11) occurs and the electron population of Eu2+ in the upper vibration level of the excited state, from which radiative transition takes place, becomes dominating under phonon-assistant. Thus, a blue shift is observed with increasing temperature. Therefore, the profiles of the emission spectra at high temperature become more symmetric gradually (as shown in Fig. 8).
3.4. CIE chromaticity coordinates
Fig. 12 shows the Commission International de L'Eclairage (CIE) chromaticity coordinates calculated from the emission spectrum of Ba1−x(PO3)2:Eux2+ (x = 0.005, 0.010, 0.020, 0.030, 0.035) under 323 nm light excitation. As shown in Fig. 12 and Table 2, the CIE chromaticity coordinates shift from (0.162, 0.174) to (0.165, 0.198) as Eu2+ concentrations increase from 0.005 to 0.035. The results indicate that the CIE chromaticity can be varied within a certain range by adjusting the content of Eu2+. A digital image of the Ba0.970(PO3)2:Eu0.0302+ phosphor under NUV light about 365 nm irradiation is also shown in inset of Fig. 12. It can be seen that the sample shows bright blue-green emission.| No. | Composition | CIE (x, y) |
|---|---|---|
| 1 | Ba0.995(PO3)2:Eu0.0052+ | (0.162, 0.174) |
| 2 | Ba0.990(PO3)2:Eu0.0102+ | (0.163, 0.183) |
| 3 | Ba0.980(PO3)2:Eu0.0202+ | (0.164, 0.188) |
| 4 | Ba0.970(PO3)2:Eu0.0302+ | (0.164, 0.196) |
| 5 | Ba0.965(PO3)2:Eu0.0352+ | (0.165, 0.198) |
4. Conclusion
In conclusion, a series of blue-green emitting phosphors Ba1−x(PO3)2:Eux2+ (x = 0.005, 0.010, 0.020, 0.030, 0.035) were synthesized by solid-state methods at a low temperature of 600 °C. The results of XRD and Rietveld refinement indicate that there is only one type of Ba2+ site and Ba2+ is substituted by Eu2+ in the Ba(PO3)2 host. The photoluminescence excitation spectra are very broad, extending from 200 to 400 nm. Under NUV light excitation, Ba(PO3)2:Eu2+ exhibits a strong blue-green emission band peaking at 445 nm. The optimal concentration of Eu2+ in a Ba(PO3)2 host is 3.0 mol%. The CIE chromaticity can be varied within a certain range by adjusting the Eu2+ content. The temperature-dependent luminescence spectra show that Ba0.970(PO3)2:Eu0.0302+ has excellent thermal stability. All these results indicate that Ba(PO3)2:Eu2+ phosphor is a promising blue-green emitting phosphor for application in NUV WLEDs.Acknowledgements
This study was financially supported by the National Natural Science Foundation of the People's Republic of China (no. 51472132), the Natural Science Foundation of Shandong Province (no. ZR2012BQ017), the Qingdao Project of Science and Technology (no. 13-1-4-114-jch), the Joint Funds of the National Natural Science Foundation of China and Guangdong Province (no. U1301242), the Research Fund for the Doctoral Program of Higher Education of China (RFDP) (no. 20130171130001) and the Guangdong Province project for industrial applications of rare earth materials (2012B09000026).References
- M. Krings, G. Montana, R. Dronskowski and C. Wickleder, Chem. Mater., 2011, 23, 1694–1699 CrossRef CAS.
- R. J. Xie, N. Hirosaki, M. Mitomo, Y. Yamamoto and T. Suehiro, J. Phys. Chem. B, 2004, 108, 12027–12031 CrossRef CAS.
- M. F. Zhang, Y. J. Liang, R. Tang, D. Y. Yu, M. H. Tong, Q. Wang, Y. L. Zhu, X. Y. Wu and G. G. Li, RSC Adv., 2014, 4, 40626–40637 RSC.
- V. Bachmann, C. Ronda and A. Meijerink, Chem. Mater., 2009, 21, 2077–2084 CrossRef CAS.
- W. Chen and A. Zhou, J. Phys. Chem. C, 2012, 116, 24748–24751 CAS.
- (a) W. R. Liu, C. H. Huang, C. W. Yeh, J. C. Tsai, Y. C. Chiu, Y. T. Yeh and R. S. Liu, Inorg. Chem., 2012, 51, 9636–9641 CrossRef CAS PubMed; (b) C. H. Huang, D. Y. Wang, Y. C. Chiu, Y. T. Yeh and T. M. Chen, RSC Adv., 2012, 2, 9130–9134 RSC.
- L. Wu, Y. Zhang, M. Y. Gui, P. Z. Lu, L. X. Zhao, S. Tian, Y. F. Kong and J. J. Xu, J. Mater. Chem., 2012, 22, 6463–6470 RSC.
- X. Chen, P. P. Dai, X. T. Zhang, C. Li, S. Lu, X. L. Wang, Y. Jia and Y. C. Liu, Inorg. Chem., 2014, 53, 3441–3448 CrossRef CAS PubMed.
- S. J. Gwak, P. Arunkumar and W. B. Im, J. Phys. Chem. C, 2014, 118, 2686–2692 CAS.
- X. M. Zhang, F. G. Meng, W. L. Li and H. J. Seo, Ceram. Int., 2013, 39, 8975–8978 CrossRef CAS PubMed.
- T. Z. Zhan, C. N. Xu, H. Yamada, Y. Terasawa, H. Iwase, M. Kawaid and L. Zhang, RSC Adv., 2012, 2, 328–332 RSC.
- T. G. Kim, T. Y. Kim, J. Y. Kim, S. J. Kim and S. J. Im, J. Phys. Chem. C, 2014, 118, 12428–12435 CAS.
- T. Suehiro, R. J. Xie and N. Hirosakiet, Ind. Eng. Chem. Res., 2013, 52, 7453–7456 CrossRef CAS.
- C. C. Lagos, ECS J. Solid State Sci. Technol., 1968, 115, 1271–1274 CAS.
- P. Dorenbos, J. Lumin., 2003, 104, 239–260 CrossRef CAS.
- A. A. Coelho, TOPAS Academic Version 4.1, Coelho Software, Brisbane, Australia, 2007 Search PubMed.
- B. D. Cullity, Element of X-ray Diffraction, Addison wesley, Reading, MA, 2nd edn, 1978 Search PubMed.
- C. C. Wu, K. B. Chen, C. S. Lee, T. M. Chen and B. M. Cheng, Chem. Mater., 2007, 19, 3278–3285 CrossRef CAS.
- H. P. Ji, Z. H. Huang, Z. G. Xia, M. S. Molokeev, V. V. Atuchin, M. H. Fang and S. F. Huang, Inorg. Chem., 2014, 53, 5129–5135 CrossRef CAS PubMed.
- C. H. Huang, D. Y. Wang, Y. C. Chiu, Y. T. Yeh and T. M. Chen, RSC Adv., 2012, 2, 9130–9134 RSC.
- C. C. Lin, R. S. Liu, Y. S. Tang and S. F. Huc, J. Electrochem. Soc., 2008, 155, J248 CrossRef CAS PubMed.
- Y. Zhang, Z. G. Xia and W. W. Wu, J. Am. Ceram. Soc., 2013, 96, 1043–1046 CrossRef CAS PubMed.
- I. Baginskiy and R. S. Liu, J. Electrochem. Soc., 2009, 156, G29–G32 CrossRef CAS PubMed.
- C. H. Huang and T. M. Chen, Inorg. Chem., 2011, 50, 5725–5730 CrossRef CAS PubMed.
- P. Dorenbos, J. Phys.: Condens. Matter, 2005, 17, 8103–8111 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |