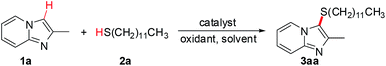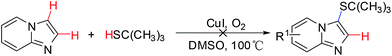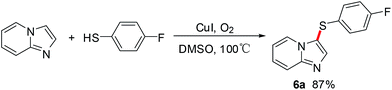Regioselective copper-catalyzed thiolation of imidazo[1,2-a]pyridines: an efficient C–H functionalization strategy for C–S bond formation†
Hua Cao*,
Longbin Chen,
Jingyun Liu,
Huiyin Cai,
Hao Deng,
Guijun Chen,
Caijuan Yan and
Ya Chen
School of Chemistry and Chemical Engineering, Guangdong Pharmaceutical University, Guangzhou 510006, P. R. China. E-mail: caohua@gdpu.edu.cn; Fax: +86 760 88207939
First published on 20th February 2015
Abstract
A highly regioselective C–H/S–H cross-coupling of imidazo[1,2-a]pyridines with thiols has been developed using molecular oxygen to form the C-3 sulfenated products in the presence of a copper catalyst. It represents a simple process for the formation of C–S bonds to prepare thioether-decorated imidazo[1,2-a]pyridines. The reaction proceeds smoothly with a broad range of substrates to give imidazo[1,2-a]pyridines in good yields.
(Hetero)aryl thioether and their derivatives have been recognized as imperative building blocks for creating molecules of natural products, pharmaceutical interest, and functional materials.1 Over the past decade, the formation of C–S bonds2 to prepare (hetero)aryl thioether has emerged as a highly attractive and powerful strategy to construct complicated compounds. The traditional methods for the construction of (hetero)aryl thioether mainly involve condensation of thiols with aryl halides3 and the reaction of aryl lithium or Grignard reagents with sulfurated electrophiles.4 However, these methods also faces some challenges: the transformation often requires harsh reaction conditions and the necessity to use prefunctionalized compounds, such as aryl halides, aryl lithium and Grignard reagents, thus adding steps and difficulties towards the formation of desired products. Hence, the development of a new process for the construction of (hetero)aryl thioether via direct C–H bond activation has drawn much attention in recent years. A number of transformation have been developed for the preparation of (hetero)aryl thioether via cross-coupling reactions using arenes,5 such as benzene,6 thiazoles,7 imidazoheterocycles,8 indoles,9 azoles,10 and 1,3,4-oxadiazoles.11 Although several transformations have proven to be highly effective in such direct functionalization processes, the development of new transformation using molecular oxygen as oxidant12 remains a significant challenge.
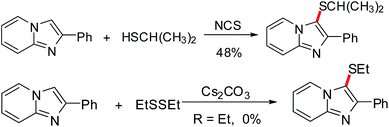
Recently, our group has reported metal-catalyzed C–H functionalization of imidazo[1,2-a]pyridine13 which are known to exhibit interesting biological activities14 and found to be key structural units in many pharmaceutically active compounds.15 In this context, we hope to develop new catalytic transformations based on selective C–H functionalization of imidazo[1,2-a]pyridines to form C–S bonds. Indeed, this kind of C–S bonds formation has already been reported by Zhang et al. and Adimurthy et al. However, what is much less explored in the previous reaction systems is the reaction scope with aliphatic thiols. As a matter of fact, only one successful attempt with aliphatic thiol as a substrate was reported; and the reaction affords corresponding thioether with only 48% yield (Scheme 1). Hence, it is important to develop a new catalytic system that has a broader substrate scope. Herein, an efficient regioselective Cu(I) catalyzed C–H/S–H cross-coupling of imidazo[1,2-a]pyridines with (hetero)aryl and alkyl thiols to construct thioether-decorated imidazo[1,2-a]pyridines has been developed using molecular oxygen as the oxidant.
The initial screening studies have been carried out using 2-methyl imidazo[1,2-a]pyridine 1a and dodecane-1-thiol 2a as model substrate to identify and optimize many different combinations of potential catalysts, oxidants, additives, solvents, and temperatures in order to achieve this goal. The key results are shown in Table 1. In the presence of 5 mol% of CuCl as a catalyst and 2 equiv. of TBHP as an oxidant, DMSO as the solvent, the reaction carried out at 100 °C afforded the desired product 3aa in 61% yield (Table 1, entry 1). Then various copper(I) salts, such as CuBr, CuI, Cu2O, and CuCN, were also examined (Table 1, entries 2–5). To our delight, the desired product 3aa was formed in 83% yield using CuI as a catalyst. Other oxidants, including O2, air, K2S2O8, oxone, and benzoquinone (BQ) were also tested (Table 1, entries 6–10). Unfortunately, K2S2O8, oxone, and benzoquinone (BQ) were ineffective for this cross-coupling of 1a with 2a. Importantly, high isolated yield of product 3aa could be obtained using molecular oxygen. Encouraged by this preliminary result, we then screened different solvents. Among then, DMSO and DMF showed good performances for this transformation while other solvents (toluene, dioxane, DCE, CH3CN) were ineffective (Table 1, entries 11–15). The reaction totally fail at room temperature (Table 1, entries 16–18). Notably, the desired product was not obtained in the absence of CuI. These results indicated that the copper catalyst should play a predominate role in this reaction. The experiments clearly demonstrated that the best way to proceed with the selective cross-coupling of 1a and 2a is by using CuI as the catalyst, O2 as oxidants, and DMSO as solvent at 100 °C for 20 h.
| Entry | Catalyst | Oxidant | Solvent | T (°C) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 1a (0.5 mmol), 2a (1.2 mmol), catalyst (5 mol%), oxidant (1.0 mmol), solvent (3.0 mL), rt-120 °C, 20 h.b GC yield.c O2 or air (500 mL).d Isolated yield.e No product.f The entry has been done without CuI. | |||||
| 1 | CuCl | TBHP | DMSO | 100 | 61 |
| 2 | CuBr | TBHP | DMSO | 100 | 64 |
| 3 | CuI | TBHP | DMSO | 100 | 83 |
| 4 | Cu2O | TBHP | DMSO | 100 | 40 |
| 5 | CuCN | TBHP | DMSO | 100 | 26 |
| 6c | CuI | O2 | DMSO | 100 | 87 (84)d |
| 7 | CuI | Air | DMSO | 100 | 53 |
| 8 | CuI | K2S2O8 | DMSO | 100 | NPe |
| 9 | CuI | Oxone | DMSO | 100 | NP |
| 10 | CuI | BQ | DMSO | 100 | NP |
| 11 | CuI | O2 | DMF | 100 | 86 |
| 12 | CuI | O2 | Toluene | 100 | NP |
| 13 | CuI | O2 | Dioxane | 100 | NP |
| 14 | CuI | O2 | DCE | 100 | NP |
| 15 | CuI | O2 | CH3CN | 100 | NP |
| 16 | CuI | O2 | DMSO | 120 | 82 |
| 17 | CuI | O2 | DMSO | rt | NP |
| 18 | CuI | O2 | DMSO | 80 | 71 |
| 19f | — | O2 | DMSO | 100 | NP |
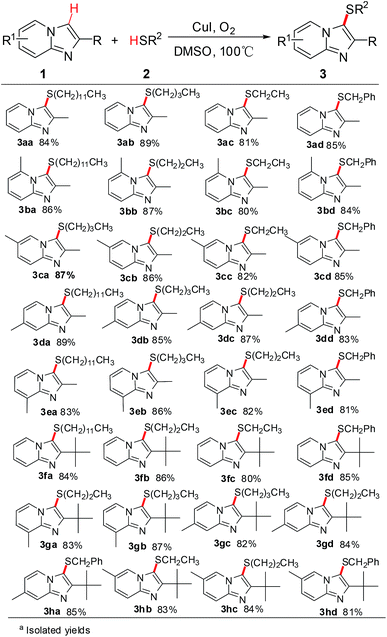
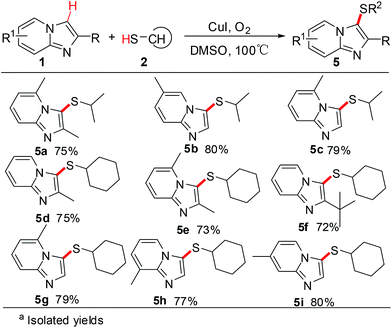
In a further set of experiments, the scope and generality of this process for the direct oxidative cross-coupling of imidazo[1,2-a]pyridines 1 and thiols 2 was investigated under the optimum reaction conditions. The results are summarized in Scheme 2. A series of primary thiols, such as dodecane-1-thiol, butane-1-thiol, propane-1-thiol, ethanethiol, and phenylmethane thiol, were examined. And the results showed that alkyl thiols can be efficiently converted to the corresponding cross-coupling products. Various substituted imidazo[1,2-a]pyridine derivatives reacted well with alkyl thiols. Different position substituted group on the pyridine or imidazole ring, having 2-CH3, 2-C(CH3)3, 5-CH3, 6-CH3, 7-CH3, 8-CH3 substitution, were smoothly participated in this transformation to provide the corresponding imidazo[1,2-a]pyridines in good yields.
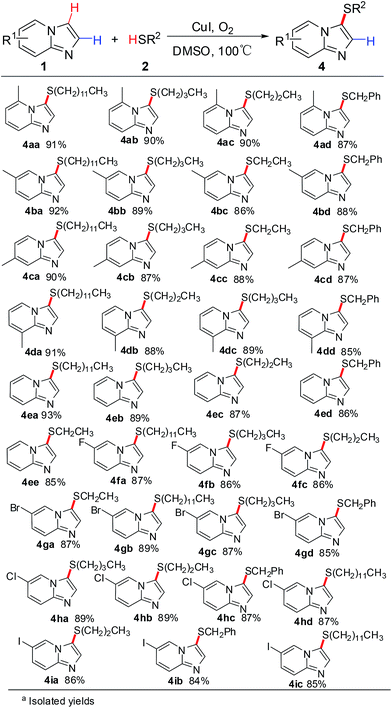
Subsequently, 2-unsubstituted imidazo[1,2-a]pyridines were also examined as the substrate with various thiols and the results are described in Scheme 3. To our delight, the reaction conditions were useful for a variety of 2-unsubstituted imidazo[1,2-a]pyridines and the selective C-3 cross-coupling products were obtained in good to excellent yields. A variety of imidazo[1,2-a]pyridines with electron-donating methyl groups at the 5-, 6-, 7-, and 8-positions were smoothly sulfenated at the 3-position with thiols. It is also worth noting that the presence of halogen groups (F, Cl, Br, and I) at 5 position of the pyridine ring provided the corresponding selective C-3 sulfenated products in good yields. The present strategy showed high functional group tolerance in different position on the pyridine ring and led to a beneficial effect on the reaction outcome.
For further investigation, commercially available aliphatic secondary thiols were employed to explore the scope of the cross-coupling reaction under the optimized conditions. And the results are outlined in Scheme 4. Aliphatic secondary thiols, such aspropane-2-thiol and cyclohexane thiol, with imidazo[1,2-a]pyridines proceeded smoothly to afford the corresponding sulfenylated products in 72–80% yields. Notably, the selective C-3 sulfenated products were successfully obtained in moderate to good yields, when the reaction carried out using 2-unsubstituted imidazo[1,2-a]pyridines as substrates with aliphatic secondary thiols. However, the desired sulfenated product was not formed using t-butylmercaptan as substrate probably due to steric hindrance of t-butyl group (Scheme 5). To our delight, the reaction of imidazo[1,2-a]pyrazine with 4-fluorobenzenethiol proceeded smoothly to afford the desired product 6a in 87% yield (Scheme 6). All the results indicated that this strategy provided a wide range of substrates to form thioether-decorated imidazo[1,2-a]pyridines in good yields, which can be used to prepare potential biologically important molecules.
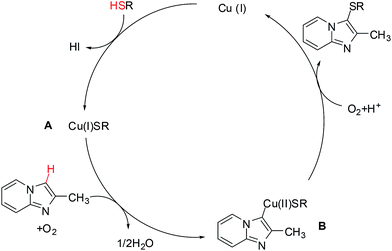
As mentioned in similar studies,7f,16 a plausible mechanism of this Cu-catalyzed direct C–H cross-coupling was depicted in Scheme 7. Initially, CuI reacts with 2 to give CuSR species A, which then would undergo a concerted metalation–deprotonation with imidazo[1,2-a]pyridines to form the intermediate B. Finally, intermediate B underwent reductive elimination to generate the cross-coupling product in the presence of molecular oxygen and released the Cu(I) catalyst.
In summary, we have developed an efficient and highly regioselective CuI-catalyzed oxidative C–H/S–H cross-coupling of imidazo[1,2-a]pyridines with thiols by using O2 as oxidant. This method provides a simple route for the synthesis of thioether-decorated imidazo[1,2-a]pyridines which are broadly applicable for the synthesis of biologically active molecules. The reaction proceeded with C-3-selectivity without the directing groups and showed a broad substrates cope in the C–H thiolation reaction. Owing to its high selectivity and broad substrate scope, this C–H thiolation reaction should be of high synthetic value. More synthetic applications of the present method are under investigation in our laboratory.
Acknowledgements
This research was financially supported by National Natural Science Foundation of China (21302023) and the Project of Department of Education of Guangdong Province (2013KJCX0111).Notes and references
- (a) F. Pinnen, I. Cacciatore, C. Cornacchia, P. Sozio, L. S. Cerasa, A. Iannitelli, C. Nasuti, F. Cantalamessa, D. Sekar, R. Gabbianelli, M. L. Falcioni and A. D. Stefano, J. Med. Chem., 2009, 52, 559–563 CrossRef CAS PubMed; (b) S. Gazal, G. Gelerman, O. Ziv, O. Karpov, P. Litman, M. Bracha, M. Afargan and C. Gilon, J. Med. Chem., 2002, 45, 1665–1671 CrossRef CAS PubMed; (c) K. A. Jacobson, L. Kiriasis, S. Barone, B. J. Bradbury, U. Kammula, J. M. Campagne, J. W. Daly, J. L. Neumeyer, W. Pfleiderer and S. Secunda, J. Med. Chem., 1989, 32, 1873–1879 CrossRef CAS; (d) S. F. Nielsen, E. O. Nielsen, G. M. Olsen, T. Liljefors and D. Peters, J. Med. Chem., 2000, 43, 2217–2226 CrossRef CAS PubMed; (e) T. D. Ocain and D. H. Rich, J. Med. Chem., 1988, 31, 2193–2199 CrossRef CAS; (f) S. Liu, C. W. Brown, K. D. Berlin, A. Dhar, S. Guruswamy, D. Brown, G. J. Gardner, M. J. Birrer and D. M. Benbrook, J. Med. Chem., 2004, 47, 999–1007 CrossRef CAS PubMed; (g) S. M. Lu and H. Alper, J. Am. Chem. Soc., 2005, 127, 14776–14784 CrossRef CAS PubMed; (h) M. A. Ciufolini, Y. C. Shen and M. J. Bishop, J. Am. Chem. Soc., 1995, 117, 12460–12469 CrossRef CAS; (i) P. Majumdar, A. Pati, M. Patra, R. K. Behera and A. K. Behera, Chem. Rev., 2014, 114, 2942–2977 CrossRef CAS PubMed; (j) Y. Cui and P. E. Floreancig, Org. Lett., 2012, 14, 1720–1723 CrossRef CAS PubMed; (k) R. Boston, K. Awaya, T. Nakayama, W. Ogasawara and S. R. Hall, RSC Adv., 2014, 4, 26824–26828 RSC; (l) M. M. Rahman, M. Younus and A. Ogawa, RSC Adv., 2014, 4, 25389–25392 RSC.
- (a) X. Tang, L. Huang, Y. Xu, J. Yang, W. Wu and H. Jiang, Angew. Chem., Int. Ed., 2014, 53, 4205–4208 CrossRef CAS PubMed; (b) L. Wang, W. He and Z. Yu, Chem. Soc. Rev., 2013, 42, 599–621 RSC; (c) D. Song, H. Liu, A. Zhang and J. Qu, RSC Adv., 2014, 48426–48432 RSC; (d) M. Arisawa, K. Suwa and M. Yamaguchi, Org. Lett., 2009, 11, 625–627 CrossRef CAS PubMed; (e) H. Cao, X. H. Liu, L. Zhao, J. H. Cen, J. X. Lin, Q. X. Zhu and M. L. Fu, Org. Lett., 2014, 16, 146–149 CrossRef CAS PubMed; (f) X. Zhang, W. Zeng, Y. Yang, H. Huang and Y. Liang, Org. Lett., 2014, 16, 876–879 CrossRef CAS PubMed; (g) X. Li, Y. Xu, W. Wu, C. Jiang, C. Qi and H. Jiang, Chem.–Eur. J., 2014, 20, 7911–7915 CrossRef CAS PubMed.
- A. V. Bierbeek and M. Gingras, Tetrahedron Lett., 1998, 39, 6283–6286 CrossRef.
- J. Ham, I. Yang and H. Kang, J. Org. Chem., 2004, 69, 3236–3239 CrossRef CAS PubMed.
- V. P. Mehta and B. Punji, RSC Adv., 2013, 3, 11957–11986 RSC.
- (a) L. D. Tran, I. Popov and O. Daugulis, J. Am. Chem. Soc., 2012, 134, 18237–18240 CrossRef CAS PubMed; (b) M. Iwasaki, M. Iyanaga, Y. Tsuchiya, Y. Nishimura, W. Li, Z. Li and Y. Nishihara, Chem.–Eur. J., 2014, 20, 2459–2462 CrossRef CAS PubMed; (c) P. Saravanan and P. Anbarasan, Org. Lett., 2014, 16, 848–851 CrossRef CAS PubMed; (d) X. Chen, X. S. Hao, C. E. Goodhue and J. Q. Yu, J. Am. Chem. Soc., 2006, 128, 6790–6791 CrossRef CAS PubMed; (e) L. Chu, X. Yue and F. L. Qing, Org. Lett., 2010, 12, 1644–1647 CrossRef CAS PubMed; (f) S. Zhang, P. Qian, M. Zhang, M. Hu and J. Cheng, J. Org. Chem., 2010, 75, 6732–6735 CrossRef CAS PubMed.
- (a) S. Ranjit, R. Lee, D. Heryadi, C. Shen, J. E. Wu, P. Zhang, K. W. Huang and X. Liu, J. Org. Chem., 2011, 76, 8999–9007 CrossRef CAS PubMed; (b) A. R. Rosario, K. K. Casola, C. E. S. Oliveira and G. Zenia, Adv. Synth. Catal., 2013, 355, 2960–2966 CrossRef CAS; (c) K. Inamoto, C. Hasegawa, K. Hiroya and T. Doi, Org. Lett., 2008, 10, 5147–5150 CrossRef CAS PubMed; (d) L. L. Joyce and R. A. Batey, Org. Lett., 2009, 11, 2792–2795 CrossRef CAS PubMed; (e) K. Inamoto, Y. Arai, K. Hiroya and T. Doi, Chem. Commun., 2008, 5529–5531 RSC; (f) C. Dai, Z. Xu, F. Huang, Z. Yu and Y. F. Gao, J. Org. Chem., 2012, 77, 4414–4419 CrossRef CAS PubMed.
- (a) W. Ge, X. Zhu and Y. Wei, Eur. J. Org. Chem., 2013, 6015–6020 CrossRef CAS; (b) C. Ravi, D. C. Mohan and S. Adimurthy, Org. Lett., 2014, 16, 2978–2981 CrossRef CAS PubMed; (c) Z. Gao, X. Zhu and R. Zhang, RSC Adv., 2014, 4, 19891–19895 RSC; (d) C. Hamdouchi, J. d Bias and J. Ezquerra, Tetrahedron, 1999, 55, 541–548 CrossRef CAS; (e) C. Hamdouchi, Synthesis, 1998, 6, 867–872 CrossRef PubMed.
- (a) X. Zhang, X. Zhou, H. Xiao and X. Li, RSC Adv., 2013, 3, 22280–22284 RSC; (b) F. L. Yang and S. K. Tian, Angew. Chem., Int. Ed., 2013, 52, 1–5 CrossRef; (c) P. Katrun, S. Hongthong, S. Hlekhlai, M. Pohmakotr, V. Reutrakul, D. Soorukram, T. Jaipetch and C. Kuhakarn, RSC Adv., 2014, 4, 18933–18938 RSC; (d) F. Xiao, H. Xie, S. Liu and G. J. Deng, Adv. Synth. Catal., 2014, 356, 364–368 CrossRef CAS; (e) W. Ge and Y. Wei, Green Chem., 2012, 14, 2066–2070 RSC.
- A. X. Zhou, X. Y. Liu, K. Yang, S. C. Zhao and Y. M. Liang, Org. Biomol. Chem., 2011, 9, 5456–5462 CAS.
- L. H. Zou, J. Reball, J. Mottweiler and C. Bolm, Chem. Commun., 2012, 48, 11307–11309 RSC.
- (a) W. Q. Wu and H. F. Jiang, Acc. Chem. Res., 2012, 45, 1736–1748 CrossRef CAS PubMed; (b) A. Z. Wang and H. F. Jiang, J. Am. Chem. Soc., 2008, 130, 5030–5031 CrossRef CAS PubMed; (c) A. Z. Wang, H. F. Jiang and H. J. Chen, J. Am. Chem. Soc., 2009, 131, 3846–3847 CrossRef CAS PubMed.
- (a) H. Cao, H. Y. Zhan, Y. G. Lin, X. L. Lin, Z. D. Du and H. F. Jiang, Org. Lett., 2012, 14, 1688–1691 CrossRef CAS PubMed; (b) H. Cao, Y. G. Lin, H. Y. Zhan, Z. D. Du, X. L. Lin, Q. M. Liang and H. Zhang, RSC Adv., 2012, 2, 5972–5975 RSC; (c) H. Zhan, L. Zhao, N. Li, L. Chen, J. Liu, J. Liao and H. Cao, RSC Adv., 2014, 4, 32013–32016 RSC; (d) H. Cao, S. Lei, N. Li, L. Chen, J. Liu, H. Cai, S. Qiua and J. Tan, Chem. Commun., 2015, 51, 1823–1825 RSC.
- (a) C. M. Marson, Chem. Soc. Rev., 2011, 40, 5514–5533 RSC; (b) G. Puerstinger, J. Paeshuyse, E. Declercq and J. Neyts, Bioorg. Med. Chem. Lett., 2007, 17, 390–393 CrossRef CAS PubMed; (c) M. A. Ismail, R. K. Arafa, T. Wenzler, R. Brun, F. A. Tanious, W. D. Wilson and D. W. Boykin, Bioorg. Med. Chem., 2008, 16, 681–685 Search PubMed.
- T. S. Harrison and G. M. Keating, CNS Drugs, 2005, 19, 65–89 CrossRef CAS PubMed.
- (a) Z. Li, J. Hong and X. Zhou, Tetrahedron, 2011, 67, 3690–3697 CrossRef CAS PubMed; (b) Y. Kondo, T. Komine and T. Sakamoto, Org. Lett., 2000, 2, 3111–3114 CrossRef CAS PubMed; (c) J. Huang, J. Chan, Y. Chen, C. J. Borths, K. D. Baucom, R. D. Larsen and M. M. Faul, J. Am. Chem. Soc., 2010, 132, 3674–3675 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra01342a |
| This journal is © The Royal Society of Chemistry 2015 |