Exceptional thermal stability of undoped anatase TiO2 photocatalysts prepared by a solvent-exchange method†
Abstract
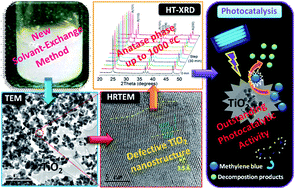
A new solvent-exchange technique to prepare anatase nanoparticles with exceptional thermal stability and photocatalytic activity is described here. The process of preparation is accomplished by using organic solvents to precipitate hydrous titania particles from a basic aqueous medium containing a titanium peroxo complex. Undoped titanium dioxide formed via a solvent exchange method has unprecedented thermal stability against transformation to the rutile phase, as opposed to TiO2 prepared by the common method of the gelation of an aqueous titanium peroxo complex. On the basis of X-ray thermodiffraction experiments, it has been established that the thermal treatment at 1000 °C of the titania prepared by ethanol precipitation contains 100% pure anatase phase. The stabilization of anatase is induced by the high defectiveness of the TiO2 nanostructure, which is evidenced from band-gap energy estimation, PXRD and HRTEM studies. The prepared TiO2 nanoparticles show an outstanding photocatalytic activity comparable to the commercial Aeroxide P25 photocatalyst in the UV-assisted decomposition of methylene blue.

 Please wait while we load your content...
Please wait while we load your content...