A colorimetric chemosensor for the sequential detection of copper ion and amino acids (cysteine and histidine) in aqueous solution†
Yong Sung Kim,
Gyeong Jin Park,
Seul Ah Lee and
Cheal Kim*
Department of Fine Chemistry and Department of Interdisciplinary Bio IT Materials, Seoul National University of Science and Technology, Seoul 139-743, Korea. E-mail: chealkim@seoultech.ac.kr; Fax: +82-2-973-9149; Tel: +82-2-970-6693
First published on 24th March 2015
Abstract
A simple and selective colorimetric chemosensor 1 was reported for the sequential detection of Cu2+, cysteine (Cys) and histidine (His) in aqueous solution. The presence of Cu2+ led to a distinct naked-eye color change from pale yellow to orange. Also, the receptor enabled analysis of Cu2+ ions with a sensitivity limit of 0.37 μM, which is far below the WHO acceptable limit (31.5 μM) in drinking water. 1 could be also used as a practical, visible colorimetric test strip for Cu2+ (down to 10 μM) in aqueous media. Moreover, the resulting 1–Cu2+ complex sensed cysteine and histidine with an absorption change via UV-visible or a naked-eye color change, although His and Cys were indistinguishable with the method proposed herein.
Introduction
Copper is one of most vital metal ions in biological systems, because it functions as an important cofactor by taking an active part in a large variety of enzymes, including superoxide dismutase, cytochrome c oxidase and tyrosinase.1,2 However, with excessive loading, copper ion can cause several diseases including Alzheimer's, Parkinson's and Wilson diseases.3,4 Consequently, considerable efforts have been devoted to the development of colorimetric or fluorescent Cu2+-selectivity chemosensors.5–24Among many naturally existing amino acids, cysteine (Cys) and histidine (His) have attracted much attention because of their important biological functions. Cys has been proven to act as a potential neurotoxin, a biomarker for various medical conditions, and a disease-associated physiological regulator.25,26 Cys deficiency would cause many troubles including retarded growth in children, hair depigmentation, hematopoiesis decrease, skin lesions and psoriasis.27,28 Hence, it is of great importance to selectively detect and discriminate Cys as a disease-associated biomarker in biological systems. So far, various conventional detection processes such as high performance liquid chromatography (HPLC).29,30 optical detections and capillary electrophoresis31 have been exploited for the determination of Cys. Among these various methods, optical detection techniques have been proven to be the most convenient.32
His is essential for human growth and plays vital roles in the biological system such as control of transmission of metal and as repair of tissue.33 Recent studies have shown that a deficiency of His in plasma may lead to an impaired nutritional state in patients with chronic kidney disease.34 Therefore, the developments of sensory probes for an amino acid such as His are still highly demanded. Among them, the metal complex-based colorimetric compounds to selectively detect an amino acid have attracted much attention.35–37
In recent years, there has been a great emergence of interests in the development of sensors for the sequential detection of various cations and amino acids.38–43 Among the different types of chemosensors, the probes based on colorimetric determination of the cations and amino acids have many advantages because of the simplicity, low cost, and rapid tracking of analytes.44 Therefore, our group has been interested in the colorimetric sequential recognition of metal ions and amino acids.
Herein, we describe a new compound 1 based on a julolidine moiety, which was designed and synthesized as a colorimetric receptor of Cu2+ with the color change from pale yellow to orange in aqueous solution. Moreover, the resulting 1–Cu2+ complex showed the colorimetric recognition of Cys and His.
Experimental section
General information
All solvents and reagents (analytical grade and spectroscopic grade) were obtained from Sigma-Aldrich and used as received. Both 1H NMR and 13C NMR were performed on a Varian 400 MHz and 100 MHz spectrometer and chemical shifts are recorded in ppm. Electrospray ionization mass spectra (ESI-MS) were collected on a Thermo Finnigan (San Jose, CA, USA) LCQTM Advantage MAX quadruple ion trap instrument. Elemental analysis for carbon, nitrogen, and hydrogen was carried out by using a Flash EA 1112 elemental analyzer (thermo) in Organic Chemistry Research Center of Sogang University, Korea. Absorption spectra were recorded at room temperature using a Perkin Elmer model Lambda 2S UV-vis spectrometer.Synthesis of 1
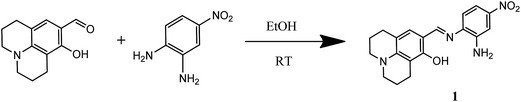
8-Hydroxyjulolidine-9-carboxaldehyde (0.68 g, 3 mmol) and 4-nitro-1,2-phenylenediamine (0.50 g, 3.2 mmol) were dissolved in 20 mL of absolute ethanol. Then, three drops of phosphoric acid were added into the reaction mixture, which was stirred for 4 h at room temperature. The orange powder was produced, which was collected by filtration, washed with diethyl ether and air-dried. Yield: 0.814 g (77.0%). 1H NMR (DMSO-d6, 400 MHz): δ 12.76 (s, 1H), 8.59 (s, 1H), 7.87 (d, 1H), 7.82 (d, 1H), 6.99 (s, 1H), 6.76 (d, 1H), 6.49 (d, 2H), 3.24 (m, 4H), 2.62 (m, 4H), 1.86 (m, 4H). 13C NMR (DMSO-d6, 100 MHz): δ 163.50, 157.93, 150.25, 147.53, 137.04, 135.07, 131.30, 123.69, 114.47, 113.46, 113.37, 108.85, 105.75, 50.08, 49.64, 27.32, 22.06, 21.13, 20.64 ppm. ESI-MS: m/z calcd for C19H20N4O3 − H+ ([M − H+]), 351.146; found, 351.563. Anal. calc. for C19H20N4O3: C, 64.76; H, 5.72; N, 15.90; found: C, 64.82; H, 5.96; N, 15.58%.A colorimetric chemosensor
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (2.4 mg, 0.01 mmol) was dissolved in DMF (2 mL). 0.9–12.6 μL of the Cu2+ solution (5 mM) were transferred to each receptor solution (15 μM) prepared above. After mixing them for a few seconds, UV-vis absorption spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (2.4 mg, 0.01 mmol) was dissolved in DMF (2 mL). 0.9–12.6 μL of the Cu2+ solution (5 mM) were transferred to each receptor solution (15 μM) prepared above. After mixing them for a few seconds, UV-vis absorption spectra were taken at room temperature.For Cys, receptor 1 (2.1 mg, 0.003 mmol) was dissolved in DMF (2 mL) and 15 μL of 1 (3 mM) were diluted with 2.985 mL of DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF (2 mL) and 3.2 μL of this Cu2+ solution (20 mM) were transferred to each receptor solution (15 μM) to give 1.4 equiv. Then, Cys (4.8 mg, 0.04 mmol) was dissolved in bis-tris buffer (2 mL) and 0–13.5 μL of this Cys solution (20 mM) were transferred to a mixture of 1 and Cu2+ (15 μM) to give 6 equiv. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF (2 mL) and 3.2 μL of this Cu2+ solution (20 mM) were transferred to each receptor solution (15 μM) to give 1.4 equiv. Then, Cys (4.8 mg, 0.04 mmol) was dissolved in bis-tris buffer (2 mL) and 0–13.5 μL of this Cys solution (20 mM) were transferred to a mixture of 1 and Cu2+ (15 μM) to give 6 equiv. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
For His, receptor 1 (2.1 mg, 0.003 mmol) was dissolved in DMF (2 mL) and 15 μL of 1 (3 mM) were diluted with 2.985 mL of DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF and 3.2 μL of this Cu2+ solution (20 mM) were transferred to each receptor solution (15 μM) to give 1.4 equiv. Then, His (6.2 mg, 0.04 mmol) was dissolved in bis-tris buffer (2 mL) and 0–16.9 μL of this His solution (20 mM) were transferred to a mixture of 1 and Cu2+ (15 μM) to give 7.5 equiv. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF and 3.2 μL of this Cu2+ solution (20 mM) were transferred to each receptor solution (15 μM) to give 1.4 equiv. Then, His (6.2 mg, 0.04 mmol) was dissolved in bis-tris buffer (2 mL) and 0–16.9 μL of this His solution (20 mM) were transferred to a mixture of 1 and Cu2+ (15 μM) to give 7.5 equiv. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make a total volume of 2.985 mL. Cu(NO3)2·2.5H2O (1.4 mg, 0.006 mmol) was dissolved in DMF (2 mL). 0, 1.5, 3, 4.5, 6, 7.5, 9, 10.5, 12, 13.5, and 15 μL of Cu(NO3)2·2.5H2O solution (3 mM) were added to each diluted receptor solution. Each vial had a total volume of 3 mL. After shaking the vials for a few minutes, UV-vis absorption spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make a total volume of 2.985 mL. Cu(NO3)2·2.5H2O (1.4 mg, 0.006 mmol) was dissolved in DMF (2 mL). 0, 1.5, 3, 4.5, 6, 7.5, 9, 10.5, 12, 13.5, and 15 μL of Cu(NO3)2·2.5H2O solution (3 mM) were added to each diluted receptor solution. Each vial had a total volume of 3 mL. After shaking the vials for a few minutes, UV-vis absorption spectra were taken at room temperature.For Cys, 1 (2.1 mg, 0.003 mmol) and Cu(NO3)2·2.5H2O (1.4 mg, 0.006 mmol) were dissolved in DMF (2 mL), respectively. The two solutions were mixed to make 1–Cu2+ complex. 15, 13.5, 12, 10.5, 9, 7.5, 6, 4.5, 3, 1.5, and 0 μL of the 1–Cu2+ complex solution (3 mM) were taken and transferred to vials. Each vial was diluted with DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make a total volume of 2.985 mL. Cys (0.7 mg, 0.006 mmol) was dissolved in bis-tris buffer (2 mL). 0, 1.5, 3, 4.5, 6, 7.5, 9, 10.5, 12, 13.5, and 15 μL of the Cys solution were added to each diluted 1–Cu2+ solution. Each vial had a total volume of 3 mL. After reacting them for a few seconds, UV-vis spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make a total volume of 2.985 mL. Cys (0.7 mg, 0.006 mmol) was dissolved in bis-tris buffer (2 mL). 0, 1.5, 3, 4.5, 6, 7.5, 9, 10.5, 12, 13.5, and 15 μL of the Cys solution were added to each diluted 1–Cu2+ solution. Each vial had a total volume of 3 mL. After reacting them for a few seconds, UV-vis spectra were taken at room temperature.
For His, 1 (2.1 mg, 0.003 mmol) and Cu(NO3)2·2.5H2O (1.4 mg, 0.006 mmol) were dissolved in DMF (2 mL), respectively. The two solutions were mixed to make 1–Cu2+ complex. 15, 13.5, 12, 10.5, 9, 7.5, 6, 4.5, 3, 1.5, and 0 μL of the 1–Cu2+ complex solution (3 mM) were taken and transferred to vials. Each vial was diluted with DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make a total volume of 2.985 mL. His (0.9 mg, 0.006 mmol) was dissolved in bis-tris buffer (2 mL). 0, 1.5, 3, 4.5, 6, 7.5, 9, 10.5, 12, 13.5, and 15 μL of the His solution were added to each diluted 1–Cu2+ solution. Each vial had a total volume of 3 mL. After reacting them for a few seconds, UV-vis spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make a total volume of 2.985 mL. His (0.9 mg, 0.006 mmol) was dissolved in bis-tris buffer (2 mL). 0, 1.5, 3, 4.5, 6, 7.5, 9, 10.5, 12, 13.5, and 15 μL of the His solution were added to each diluted 1–Cu2+ solution. Each vial had a total volume of 3 mL. After reacting them for a few seconds, UV-vis spectra were taken at room temperature.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. MNO3 (M = Na, K; 0.02 mmol), M(NO3)2 (M = Mn, Co, Ni, Cu, Zn, Cd, Mg, Ca, Pb; 0.02 mmol), M(ClO3)2 (M = Fe; 0.02 mmol) or M(NO3)3 (M = Al, Fe, Cr, Ga, In; 0.02 mmol) were dissolved in DMF (1 mL), respectively. 3.2 μL of each metal solution (20 mM) were taken and added into 3 mL of each 1 solution (15 μM) prepared above to make 1.4 equiv. Then, 3.2 μL of the Cu(NO3)2·2.5H2O solution (20 mM) were added into the mixed solution of each metal ion and 1 to make 1.4 equiv. After mixing them for a minute, UV-vis absorption spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. MNO3 (M = Na, K; 0.02 mmol), M(NO3)2 (M = Mn, Co, Ni, Cu, Zn, Cd, Mg, Ca, Pb; 0.02 mmol), M(ClO3)2 (M = Fe; 0.02 mmol) or M(NO3)3 (M = Al, Fe, Cr, Ga, In; 0.02 mmol) were dissolved in DMF (1 mL), respectively. 3.2 μL of each metal solution (20 mM) were taken and added into 3 mL of each 1 solution (15 μM) prepared above to make 1.4 equiv. Then, 3.2 μL of the Cu(NO3)2·2.5H2O solution (20 mM) were added into the mixed solution of each metal ion and 1 to make 1.4 equiv. After mixing them for a minute, UV-vis absorption spectra were taken at room temperature.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.For His, 1 (2.1 mg, 0.003 mmol) and Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) were dissolved in DMF (2 mL), respectively. The two solutions were mixed to make 1–Cu2+ complex (3 mM). Various amino acids and peptide such as Gly, Ala, Ser, Thr, Val, Leu, Ile, Met, Pro, Phe, Trp, Asp, Glu, Asn, Gln, His, Lys, Arg or GSH (0.02 mmol) were dissolved in 10 mM bis-tris (1 mL). 16.9 μL of each amino acid and GSH solution (20 mM) were taken and added into 3 mL of the solution of His (112.5 μM) to give 7.5 equiv. of amino acids or GSH. Then, 15 μL of 1–Cu2+ solution (3 mM) were added into the mixed solution of each amino acid or GSH and His to make 1 equiv. After the procedure was carried out, the mixed solutions were diluted with 2.985 mL of DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make a final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF (2 mL) and 3.2 μL of the Cu2+ ion solution (20 mM) were added to the solution of 1 (15 μM) prepared above. After mixing it for a few seconds, UV-vis spectrum was taken at room temperature. Ethylenediaminetetraacetic acid disodium salt dehydrate (EDTA, 0.04 mmol) was dissolved in buffer solution (2 mL) and 3.2 μL of the EDTA solution (20 mM) were added to the solution of 1–Cu2+ complex (15 μM) prepared above. After mixing it for a minute, UV-vis spectrum was taken. For the reversibility study, another 3.2 μL of the Cu2+ ion solution (20 mM) was added to the above solution. After mixing it for a minute, UV-vis spectrum was taken at room temperature. The same experimental procedure was repeated one more time.
5, v/v, 10 mM, bis-tris, pH 7.0) to make a final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF (2 mL) and 3.2 μL of the Cu2+ ion solution (20 mM) were added to the solution of 1 (15 μM) prepared above. After mixing it for a few seconds, UV-vis spectrum was taken at room temperature. Ethylenediaminetetraacetic acid disodium salt dehydrate (EDTA, 0.04 mmol) was dissolved in buffer solution (2 mL) and 3.2 μL of the EDTA solution (20 mM) were added to the solution of 1–Cu2+ complex (15 μM) prepared above. After mixing it for a minute, UV-vis spectrum was taken. For the reversibility study, another 3.2 μL of the Cu2+ ion solution (20 mM) was added to the above solution. After mixing it for a minute, UV-vis spectrum was taken at room temperature. The same experimental procedure was repeated one more time.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF (2 mL). 3.2 μL of the Cu2+ solution (20 mM) were transferred to each receptor solution (15 μM) prepared above. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.
5, v/v, 10 mM, bis-tris, pH 7.0) to make the final concentration of 15 μM. Cu(NO3)2·2.5H2O (9.6 mg, 0.04 mmol) was dissolved in DMF (2 mL). 3.2 μL of the Cu2+ solution (20 mM) were transferred to each receptor solution (15 μM) prepared above. After mixing them for a few seconds, UV-vis spectra were taken at room temperature.Results and discussion
Synthesis of 1
The receptor 1 was obtained by the condensation reaction of 4-nitro-1,2-phenylenediamine and 8-hydroxyjulolidine-9-carboxaldehyde with a 77% yield in ethanol (Scheme 1), and characterized by 1H NMR, 13C NMR, ESI-mass spectrometry and elemental analysis.Absorption spectroscopic studies of 1 toward Cu2+ ion
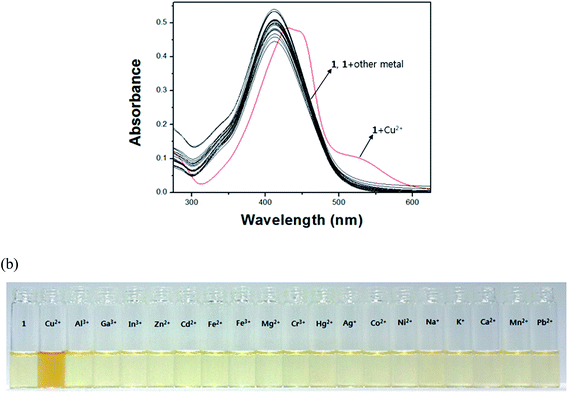
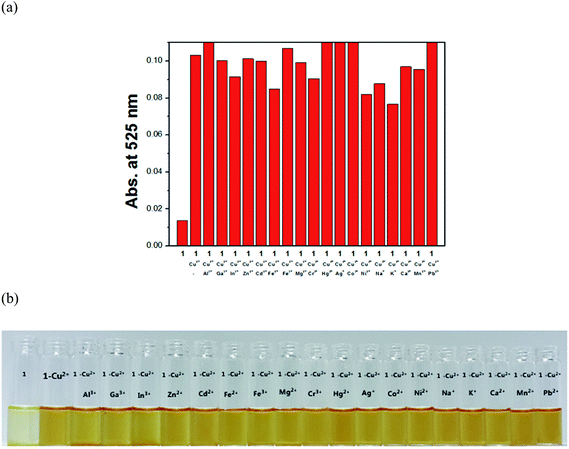
The chromogenic sensing ability of receptor 1 was studied in the presence of nineteen different cations such as Cu2+, Al3+, Ga3+, In3+, Zn2+, Cd2+, Fe2+, Fe3+, Mg2+, Cr3+, Hg2+, Ag+, Co2+, Ni2+, Na+, K+, Ca2+, Mn2+ and Pb2+ in DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0). Upon the addition of 1.4 equiv. of each metal ion, only Cu2+ induced a distinct spectral change while other metal ions showed either no or small changes in the absorption spectra relative to the free 1 (Fig. 1a). Consistent with the change of the UV-vis spectrum, the solution color of 1 changed from pale yellow to orange with copper ion (Fig. 1b), indicating that the 1 could serve as a potential candidate of colorimetric chemosensor for Cu2+.
5, v/v, 10 mM, bis-tris, pH 7.0). Upon the addition of 1.4 equiv. of each metal ion, only Cu2+ induced a distinct spectral change while other metal ions showed either no or small changes in the absorption spectra relative to the free 1 (Fig. 1a). Consistent with the change of the UV-vis spectrum, the solution color of 1 changed from pale yellow to orange with copper ion (Fig. 1b), indicating that the 1 could serve as a potential candidate of colorimetric chemosensor for Cu2+.
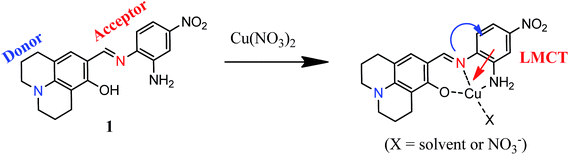
The sensing mechanism of 1 to Cu2+ might be explained by intramolecular charge transfer (ICT) and ligand-to-metal charge-transfer (LMCT) mechanisms. Kaur et al. suggested that ICT mechanism referred to the push–pull effect of the electron-donating and electron-withdrawing groups.45 That is, the red shift indicates that the energy gap of ICT band decreases, upon binding metal ions to the electron withdrawing moieties.46 Likewise, we assume that the red shift of 1–Cu2+ complex at 430 nm was induced by binding of Cu2+ to the electron withdrawing groups such as the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N group. In addition, the color change could be explained by LMCT mechanism. The band with the molar extinction coefficient in the thousands, 6.3 × 103 M−1 cm−1 at 525 nm, is too large to be Cu-based d–d transitions and thus must be ligand-based transitions.22,47 Therefore, the change of ICT and LMCT bands might be responsible for the dramatic color change from pale yellow to orange.
N group. In addition, the color change could be explained by LMCT mechanism. The band with the molar extinction coefficient in the thousands, 6.3 × 103 M−1 cm−1 at 525 nm, is too large to be Cu-based d–d transitions and thus must be ligand-based transitions.22,47 Therefore, the change of ICT and LMCT bands might be responsible for the dramatic color change from pale yellow to orange.
The binding properties of 1 with Cu2+ were further studied by UV-vis titration experiments. On the treatment with Cu2+ to the solution of 1, the absorption band at 410 nm significantly decreased, and a new band at 525 nm gradually reached maximum at 1.4 equiv. of Cu2+ (Fig. 2).
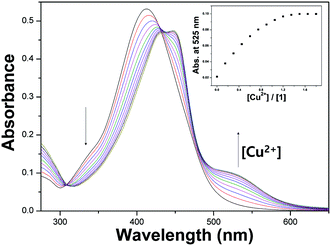 | ||
| Fig. 2 UV-vis titration of 1 (15 μM) with Cu2+ (0–1.4 equiv.). Inset: absorption titration profile of 1 with Cu2+ at 525 nm. | ||
The Job plot revealed a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric ratio between the 1 and Cu2+ (Fig. S1†), which was confirmed by ESI-mass spectrometry analysis.
1 stoichiometric ratio between the 1 and Cu2+ (Fig. S1†), which was confirmed by ESI-mass spectrometry analysis.
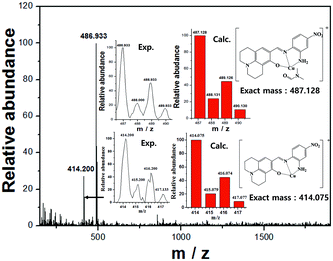
The positive ion mass spectrum indicated the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding mode between 1 and Cu2+ [m/z 486.933; calcd, 487.128] (Fig. 3).
1 binding mode between 1 and Cu2+ [m/z 486.933; calcd, 487.128] (Fig. 3).
Based on Job plot and ESI-mass spectrometry analysis, we propose that the oxygen and the two nitrogen atoms of 1 might bind to Cu2+ as shown in Scheme 2.
Based on the UV-vis titration, the binding ability (K = 6.9 × 104 M−1) of 1 for Cu2+ was determined from Benesi–Hildebrand equation (Fig. S2†).48 This value is within those (104 to 105 M−1) previously reported for Cu2+-binding cheomsensors.49,50 The detection limit (DL) was calculated to be 0.37 μM, using the equation DL = 3σ/m, where σ is the relative standard deviation and m is the slope of the calibration graph.51 This value is far below the WHO acceptable limit (31.5 μM) in drinking water (Fig. S3†).52,53
To study further the ability of 1 for Cu2+ detection, competitive measurements were carried out in the presence of various metal ions. As shown in Fig. 4, the presence of other background metal ions showed no or a little change of absorbance.
To examine the reversibility of 1 toward Cu2+ in DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0), ethylenediaminetetraacetic acid (EDTA, 1.4 equiv.) was added to the mixed solution of 1 and Cu2+ (Fig. S4†). The solution color changed from pale yellow to orange (the original color of 1). Upon addition of Cu2+ into the mixture solution again, the absorbance at 525 nm appeared accompanied by a color change. These results indicate that 1 could be recyclable easily through treatment with a proper reagent such as EDTA. Such reversibility and regeneration might be important for the fabrication of devices to sense the Cu2+ in aqueous environmental solution.
5, v/v, 10 mM, bis-tris, pH 7.0), ethylenediaminetetraacetic acid (EDTA, 1.4 equiv.) was added to the mixed solution of 1 and Cu2+ (Fig. S4†). The solution color changed from pale yellow to orange (the original color of 1). Upon addition of Cu2+ into the mixture solution again, the absorbance at 525 nm appeared accompanied by a color change. These results indicate that 1 could be recyclable easily through treatment with a proper reagent such as EDTA. Such reversibility and regeneration might be important for the fabrication of devices to sense the Cu2+ in aqueous environmental solution.
We studied the effect of pH on the absorption response of 1 to Cu2+ ion in a series of solutions with pH values ranging from 2 to 12 (Fig. S5†). The color of the 1–Cu2+ complex remained in the orange region between pH 7 and 12, while its color changed to the pale yellow between pH 2 and 6. The absorbance increase between pH 6 and 7 indicated that the hydroxyl proton of phenol group in 1–Cu2+ complex began to deprotonate. These results showed that Cu2+ could be clearly detected by naked eye using 1 over the environmentally relevant pH range of 7.0–12.0.
For the practical application of 1, test kits were prepared by immersing filter papers in a DMF solution of 1 and then dried in oven. These test strips were used to detect Cu2+ among various cations. As shown Fig. 5, when the test kits coated with 1 were added to other cation solutions (10 μM), an obvious color change was observed only with Cu2+ in DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0). Hence, the test kits coated with 1 could be conveniently used to detect Cu2+ down to 10 μM much below the WHO guideline (31.5 μM). Importantly, this is the first example that 1 could detect the lowest concentration of copper by using the test strip in aqueous solution, to the best of our knowledge.
5, v/v, 10 mM, bis-tris, pH 7.0). Hence, the test kits coated with 1 could be conveniently used to detect Cu2+ down to 10 μM much below the WHO guideline (31.5 μM). Importantly, this is the first example that 1 could detect the lowest concentration of copper by using the test strip in aqueous solution, to the best of our knowledge.
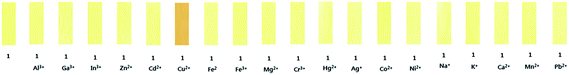 | ||
| Fig. 5 Photographs of the test kits coated with 1 (1 mM) in the presence of various metal ions (10 μM). | ||
We also constructed a calibration curve for the determination of Cu2+ by 1 (Fig. S6†). Receptor 1 exhibited a good linear relationship between the UV-vis absorbance of 1 and Cu2+ concentration (7.5–21.0 μM) with a correlation coefficient of R2 = 0.9895 (n = 3), which means that 1 is suitable for quantitative detection of Cu2+. In order to examine the applicability of the chemosensor 1 in environmental samples, the chemosensor was applied to the determination of Cu2+ in water samples. We prepared artificial polluted water samples by adding various metal ions known as being involved in industrial processes into deionized water. The results were summarized in Table S1,† which exhibited satisfactory recovery and R.S.D. values for the water samples.
Absorption spectroscopic studies of 1–Cu2+ complex toward Cys and His
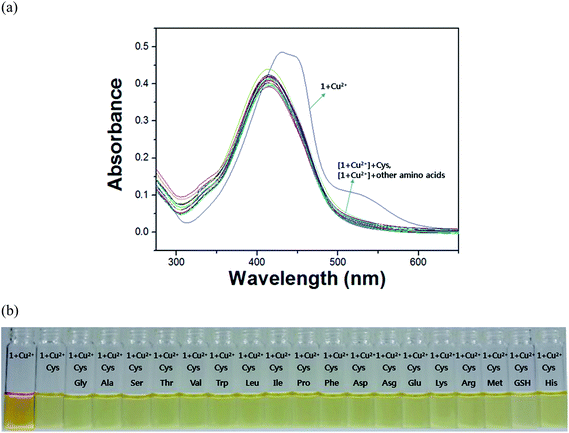
Because it has been reported that the thiol-containing amino acid and peptide strongly bind to Cu2+ ions,55–58 we examined the selectivity of 1–Cu2+ complex to Cys using copper–sulfur affinity. The absorbance spectral study of 1–Cu2+ complex with 19 different amino acids and peptide such as Gly, Ala, Ser, Thr, Cys, Val, Leu, Ile, Met, Pro, Phe, Trp, Asp, Glu, Asn, Gln, His, Lys, Arg and glutathione (GSH) was carried out in DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0). Upon the addition of 7.5 equiv. of each amino acid or GSH, 1–Cu2+ complex showed little or no spectra changes in absorption peaks in the presence of Gly, Ala, Ser, Thr, Val, Leu, Ile, Met, Pro, Phe, Trp, Asp, Glu, Asn, Gln, His, Lys, Arg and GSH as shown in Fig. S7a.† In contrast, the addition of Cys and His to 1–Cu2+ complex showed significant spectral changes. Consistent with the changes of UV-vis spectra, the addition of Cys and His to 1–Cu2+ complex instantly color changes from orange to pale yellow, respectively (Fig. S7b†). This result indicates that 1–Cu2+ could be used as a “naked-eye” sensor for Cys and His in aqueous media, respectively, although His and Cys were indistinguishable with the method proposed herein.
5, v/v, 10 mM, bis-tris, pH 7.0). Upon the addition of 7.5 equiv. of each amino acid or GSH, 1–Cu2+ complex showed little or no spectra changes in absorption peaks in the presence of Gly, Ala, Ser, Thr, Val, Leu, Ile, Met, Pro, Phe, Trp, Asp, Glu, Asn, Gln, His, Lys, Arg and GSH as shown in Fig. S7a.† In contrast, the addition of Cys and His to 1–Cu2+ complex showed significant spectral changes. Consistent with the changes of UV-vis spectra, the addition of Cys and His to 1–Cu2+ complex instantly color changes from orange to pale yellow, respectively (Fig. S7b†). This result indicates that 1–Cu2+ could be used as a “naked-eye” sensor for Cys and His in aqueous media, respectively, although His and Cys were indistinguishable with the method proposed herein.
Importantly, this is the first example of the sequential colorimetric detection of Cys and His by using copper complex as a receptor, to the best of our knowledge (Table 1).27,40–43,55–59
| Sensors | Detection limit (μM) | Interference | Percent of water in solution | Method of detection | Analyte | Reference |
|---|---|---|---|---|---|---|
| a None means that there is no interference among the common amino acids and GSH. | ||||||
 |
No data | No data | 50% | Fluorescence | Cys, Hcy | 27 |
 |
3.1 | Nonea | 99.5% | Fluorescence | His | 40 |
 |
26 (His) | Nonea | 80% | Fluorescence | His | 41 |
 |
None | Nonea | 10% | Fluorescence | His | 42 |
 |
1.6 | No data | 100% | Fluorescence | His | 43 |
 |
No data | Arg, Lys | 50% | Fluorescence | Cys, Hcy, GSH | 55 |
 |
9 | No data | 99.9% | Fluorescence | Cys | 56 |
 |
No data | No data | 70% | Fluorescence | Cys | 57 |
 |
0.065 | Nonea | 100% | Fluorescence | Cys | 58 |
 |
0.04 | GSH | 100% | Fluorescence | Cys | 59 |
 |
Cys: 3.6, His: 9 | Nonea | 83% | Naked eye | Cys, His | This work |
The binding properties of 1–Cu2+ complex with both Cys and His were further studied by UV-vis titration experiments. On the treatment with Cys to the solution of 1–Cu2+, the absorption band at 525 nm significantly decreased, and the band gradually reached minimum at 6 equiv. of Cys (Fig. S8a†). Only one clear isosbestic point at 313 nm indicates the evident reaction of 1–Cu2+ complex with Cys. The titration of 1–Cu2+ with His also blue-shifted with an isosbestic point at 309 nm, when the concentration of His reached to 7.5 equiv. (Fig. S8b†).
The binding modes between 1–Cu2+ complex and the two amino acids, Cys and His, were revealed by using Job plot analysis. The Job plots for the 1–Cu2+ complex with Cys (Fig. S9a†) and His (Fig. S9b†) exhibited a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry, respectively.
1 stoichiometry, respectively.
The interactions of 1–Cu2+ complex with Cys or His were further analyzed by ESI-mass spectrometry analysis. The negative ion mass spectrum of 1–Cu2+ complex upon addition of 1 equiv. of Cys indicated the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding between Cu2+ and Cys [(Cys − H+)− + Cu2+ + 2NO3−]− [m/z 306.933; calcd, 306.917] (Fig. S10a†). For His, the negative ion mass spectrum showed that a peak at m/z = 450.207 was assignable to [(His − H+)− + Cu2+ + 2NO3− + H2O + DMF [calcd, 450.041] (Fig. S10b†). Based on the Job plot and ESI-mass spectrometry analysis, we propose that the 1–Cu2+ complex might undergo the demetallation by Cys and His, respectively (Scheme 3).
1 binding between Cu2+ and Cys [(Cys − H+)− + Cu2+ + 2NO3−]− [m/z 306.933; calcd, 306.917] (Fig. S10a†). For His, the negative ion mass spectrum showed that a peak at m/z = 450.207 was assignable to [(His − H+)− + Cu2+ + 2NO3− + H2O + DMF [calcd, 450.041] (Fig. S10b†). Based on the Job plot and ESI-mass spectrometry analysis, we propose that the 1–Cu2+ complex might undergo the demetallation by Cys and His, respectively (Scheme 3).
From the results of UV-vis titration, the dissociation constants of the 1–Cu2+ for Cys and His were determined as 4.7 × 103 M−1 and 2.4 × 104 M−1, respectively, on the basis of Benesi–Hildebrand equation (Fig. S11†). For practical application, the detection limit was also an important parameter. Thus, the detection limits of 1–Cu2+ for the analysis of Cys and His were determined to be 3.6 μM and 9.0 μM using of the basis 3σ/m, respectively (Fig. S12†).
To further study the ability of 1–Cu2+ for Cys and His detection, inhibition tests were performed with 19 various amino acids and peptide such as Gly, Ala, Ser, Thr, Val, Leu, Ile, Met, Pro, Phe, Trp, Asp, Glu, Asn, Gln, His, Lys, Arg and GSH. When 1–Cu2+ was treated with 6 equiv. of Cys in the presence of the same concentration of other amino acids (Fig. 6), there was no inhibition for the detection of Cys by 1–Cu2+ in DMF–buffer solution (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v, 10 mM, bis-tris, pH 7.0).54 These results demonstrate that 1–Cu2+ could be an excellent chromogenic sensor with high selectivity for Cys over competing amino acids. Similarly, we conducted the preferential selectivity of 1–Cu2+ as a colorimetric sensor for the detection of His in the presence of various amino acids. His was also not inhibited by the competing other amino acids as shown in Fig. S13.†
5, v/v, 10 mM, bis-tris, pH 7.0).54 These results demonstrate that 1–Cu2+ could be an excellent chromogenic sensor with high selectivity for Cys over competing amino acids. Similarly, we conducted the preferential selectivity of 1–Cu2+ as a colorimetric sensor for the detection of His in the presence of various amino acids. His was also not inhibited by the competing other amino acids as shown in Fig. S13.†
To verify its potential usage in environment, we investigated the effect of pH on the absorption responses of 1–Cu2+ complex to Cys and His in a series of solutions with pH values ranging from 2 to 12, respectively (Fig. S14†). The colors of the 1–Cu2+ complex with both Cys and His certainly remained in the orange region between pH 7 and 12, whereas its color changed to the pale yellow between pH 2 and 6. Therefore, these results indicate that both Cys and His could be definitely detected by naked eye using 1–Cu2+ complex over the environmentally relevant pH range of 7–12.
Conclusions
We have developed a new colorimetric sensor 1 for the sequential detection of copper ion and amino acids (cysteine and histidine). 1 induced an obvious color change from pale yellow to orange in the presence of Cu2+ ion, and the bathochromic shift was explained by the change of ICT band. The receptor enabled analysis of Cu2+ ions with a sensitivity limit of 0.37 μM, which is far below the WHO acceptable limit (31.5 μM) in drinking water. Besides, 1 could be used as a practical, visible colorimetric test strip for quantifying Cu2+ (10 μM) in aqueous environment. Moreover, chemosensing ensemble 1–Cu2+ complex was used as a colorimetric sensor for cysteine and histidine, although His and Cys were indistinguishable with the method proposed herein. Importantly, this is the first example of the sequential colorimetric chemosensor for Cys and His by using copper complex as a receptor. This sequential sensing system was simple in design, fast in operation, and more promising and convenient than other methods. Thus, these results may contribute to the development of a novel type of the colorimetric sequential recognition of copper ion and amino acids.Acknowledgements
Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2014R1A2A1A11051794 and 2012008875) are gratefully acknowledged.References
- Y. K. Jang, U. C. Nam, H. L. Kwon, I. H. Hwang and C. Kim, Dyes Pigm., 2013, 99, 6–13 CrossRef CAS PubMed
.
- H. G. Lee, K. B. Kim, G. J. Park, Y. J. Na, H. Y. Jo, S. A. Lee and C. Kim, Inorg. Chem. Commun., 2014, 39, 61–65 CrossRef CAS PubMed
.
- S. Yin, V. Leen, S. V. Snick, N. Boens and W. Dehaen, Chem. Commun., 2010, 46, 6329–6331 RSC
.
- H. S. Jung, P. S. Kwon, J. W. Lee, J. I. Kim, C. S. Hong, J. W. Kim, S. Yan, J. Y. Lee, J. H. Lee, T. Joo and J. S. Kim, J. Am. Chem. Soc., 2009, 131, 2008–2012 CrossRef CAS PubMed
.
- M. H. Kim, J. H. Noh, S. Kim, S. Ahn and S. K. Chang, Dyes Pigm., 2009, 82, 341–346 CrossRef CAS PubMed
.
- X. Huang, Z. Guo, W. Zhu, Y. Xie and H. Tian, Chem. Commun., 2008, 5143–5145 RSC
.
- N. Aksuner, E. Henden, I. Yilmaz and A. Cukurovali, Dyes Pigm., 2009, 83, 211–217 CrossRef CAS PubMed
.
- H. Mu, R. Gong, Q. Ma, Y. Sun and E. Fu, Tetrahedron Lett., 2007, 48, 5525–5529 CrossRef CAS PubMed
.
- H. Kim, Y. J. Na, E. J. Song, K. B. Kim, J. M. Bae and C. Kim, RSC Adv., 2014, 4, 22463–22469 RSC
.
- L. Zeng, E. W. Miller, A. Pralle, E. Y. Isacoff and C. J. Chang, J. Am. Chem. Soc., 2006, 128, 10–11 CrossRef CAS PubMed
.
- L. Wang, W. Qin, X. Tang, W. Dou and W. J. Liu, J. Phys. Chem. A, 2011, 115, 1609–1616 CrossRef CAS PubMed
.
- J. Zhao, S. Ji, Y. Chen, H. Guo and P. Yang, Phys. Chem. Chem. Phys., 2012, 14, 8803–8817 RSC
.
- Y. J. Na, Y. W. Choi, J. Y. Yun and C. Kim, Spectrochim. Acta, Part A, 2015, 136, 1649–1657 CrossRef CAS PubMed
.
- H. J. Kim, S. H. Kim, J. H. Kim, L. N. Anh, J. H. Lee, C. Lee and J. S. Kim, Tetrahedron Lett., 2009, 50, 2782–2786 CrossRef CAS PubMed
.
- S. Banthia and A. Samanta, New J. Chem., 2005, 29, 1007–1010 RSC
.
- K. B. Kim, H. Kim, E. J. Song and C. Kim, Dalton Trans., 2013, 16569–16577 RSC
.
- J. Y. Noh, G. J. Park, Y. J. Na, H. Y. Jo, S. A. Lee and C. Kim, Dalton Trans., 2013, 5652–5656 Search PubMed
.
- Y. J. Zhang, X. P. He, M. Hu, Z. Li, X. X. Shi and G. R. Chen, Dyes Pigm., 2011, 88, 391–395 CrossRef CAS PubMed
.
- D. Maity, A. K. Manna, D. Karthigeyan, T. K. Kundu, S. K. Pati and T. Govindaraju, Chem.–Eur. J., 2011, 17, 11152–11161 CrossRef CAS PubMed
.
- J. S. Wu, P. F. Wang, X. H. Zhang and S. K. Wu, Spectrochim. Acta, Part A, 2006, 65, 749–752 CrossRef PubMed
.
- N. Kumari, N. Dey, S. Jha and S. Bhattacharya, ACS Appl. Mater. Interfaces, 2013, 5, 2438–2445 CAS
.
- H. Y. Jo, G. J. Park, Y. J. Na, Y. W. Choi, G. R. You and C. Kim, Dyes Pigm., 2014, 109, 127–134 CrossRef CAS PubMed
.
- Y. Tang, Y. Qu, Z. Song, X. He, J. Xie, J. Hua and G. Chen, Org. Biomol. Chem., 2012, 10, 555–560 CAS
.
- D. Shi, B. Zhang, Y. Yang, C. Guan, X. He, Y. Li, G. Chen and K. Chen, Analyst, 2013, 138, 2808–2811 RSC
.
-
(a) R. Janaky, V. Varga, A. Hermann, P. Saransaari and S. S. Oja, Neurochem. Res., 2000, 25, 1397–1405 CrossRef CAS
; (b) X. F. Wang and M. S. Cynader, J. Neurosci., 2001, 21, 3322–3331 CAS
.
- N. Saravanan, D. Senthil and P. Varalakshmi, Br. J. Urol., 1996, 78, 22–24 CAS
.
- Q. Li, Y. Guo and S. Shao, Sens. Actuators, B, 2012, 171–172, 872–877 CrossRef CAS PubMed
.
- X. Zeng, X. Zhang, B. Zhu, H. Jia and Y. Li, Dyes Pigm., 2012, 94, 10–15 CrossRef CAS PubMed
.
- G. Chwatko and E. Bald, Talanta, 2000, 52, 509–515 CrossRef CAS
.
- Y. V. Tcherkas and A. D. Denisenko, J. Chromatogr. A, 2001, 913, 309–313 CrossRef CAS
.
- O. Nekrassova, N. S. Lawrence and R. G. Compton, Talanta, 2003, 60, 1085–1095 CrossRef CAS
.
- Y. Zhou and J. Yoon, Chem. Soc. Rev., 2012, 41, 52–67 RSC
.
- G. N. Chen, X. P. Wu, J. P. Duan and H. Q. Chen, Talanta, 1999, 49, 319–330 CrossRef
.
- M. Watanabe, M. E. Suliman, A. R. Qureshi, E. Garcin-Lopez, P. Barany, O. Heimburger, P. Stenvinkel and B. Lindholm, Am. J. Clin. Nutr., 2008, 87, 1860–1866 CAS
.
- J. F. Folmer-Andersen, M. Kitamura and E. V. Anslyn, J. Am. Chem. Soc., 2006, 128, 5652–5653 CrossRef CAS PubMed
.
- D. Leung, J. F. Folmer-Andersen, V. M. Lynch and E. V. Anslyn, J. Am. Chem. Soc., 2008, 130, 12318–12327 CrossRef CAS PubMed
.
- H. Aït-Haddou, S. L. Wiskur, V. M. Lynch and E. V. Anslyn, J. Am. Chem. Soc., 2001, 123, 11296–11297 CrossRef
.
- S.-H. Li, C.-W. Yu and J.-G. Xu, Chem. Commun., 2005, 450–452 RSC
.
- J. F. Folmer-Andersen, V. M. Lynch and E. V. Anslyn, J. Am. Chem. Soc., 2005, 127, 7986–7987 CrossRef CAS PubMed
.
- J. Hou, K. Li, K. Yu, M. Wu and X. Yu, Org. Biomol. Chem., 2013, 11, 717–720 CAS
.
- Q. You, A. Lee, W. Chan and X. K. Leung, Chem. Commun., 2014, 50, 6207–6210 RSC
.
- G. Wei, F. Meng, Y. Wang, Y. Cheng and C. Zhu, Macromol. Rapid Commun., 2014, 35, 2077–2081 CrossRef CAS PubMed
.
- X. Wang, Q. Miao, T. Song, Q. Yuan and J. G. G. Liang, Analyst, 2014, 139, 3360–3364 RSC
.
- S.-J. Yang, X. Z. Tian and I. Shin, Org. Lett., 2009, 11, 3438–3441 CrossRef CAS PubMed
.
- P. Kaur, D. Sareen and K. Singh, Talanta, 2011, 83, 1695–1700 CrossRef CAS PubMed
.
- G. J. Park, I. H. Hwang, E. J. Song, H. Kim and C. Kim, Tetrahedron, 2014, 70, 2822–2828 CrossRef CAS PubMed
.
- E. J. Song, J. Kang, G. R. You, G. J. Park, Y. Kim, S.-J. Kim, C. Kim and R. G. Harrison, Dalton Trans., 2013, 15514–15520 RSC
.
- H. A. Benesi and J. H. Hildebrand, J. Am. Chem. Soc., 1949, 171, 2703–2707 CrossRef
.
- Y. Xiang, A. Tong, P. Jin and Y. Ju, Org. Lett., 2006, 8, 2863–2866 CrossRef CAS PubMed
.
- S. Wu, K. Du and Y. Sung, Dalton Trans., 2010, 4363–4368 RSC
.
- J. Luo, J. Zhang, J. Lin, J. Wang and P. Yang, Microchim. Acta, 2015, 182, 201–208 CrossRef CAS
.
- Y. Shiraishi, S. Sumiya and T. Hirai, Chem. Commun., 2011, 47, 4953–4955 RSC
.
- WHO, Guidelines for Drinking Water Quality, WHO guidelines values for chemicals that are of health significance in drinking water, Geneva, 3rd edn, 2008 Search PubMed
.
- S. Xue, S. Ding, Q. Zhai, H. Zhang and G. Feng, Biosens. Bioelectron., 2015, 68, 316–321 CrossRef CAS PubMed
.
- R. Peng, L. Lin, X. Wu, X. Liu and X. Feng, J. Org. Chem., 2013, 78, 11602–11605 CrossRef CAS PubMed
.
- H. Wang, G. Zhou and X. Chen, Sens. Actuators, B, 2013, 176, 698–703 CrossRef CAS PubMed
.
- O. G. Tsay, K. M. Lee and D. G. Churchill, New J. Chem., 2012, 36, 1949–1952 RSC
.
- W. Liao, F. Wu, Y. Wu and X. Wang, Microchim. Acta, 2008, 162, 147–152 CrossRef CAS
.
- L. Chang, T. Wu and F. Chen, Microchim. Acta, 2012, 177, 295–300 CrossRef CAS
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra00544b |
| This journal is © The Royal Society of Chemistry 2015 |