Self-template synthesis of CoFe2O4 nanotubes for high-performance lithium storage
Xiao Zhanga,
Yaping Xiea,
Yanfang Sunb,
Qiao Zhanga,
Qiuyu Zhua,
Dan Houa and
Jinxue Guo*a
aKey Laboratory of Sensor Analysis of Tumor Marker (Ministry of Education), Laboratory of Inorganic Synthesis and Applied Chemistry, Qingdao University of Science and Technology, Qingdao 266042, P. R. China. E-mail: gjx1213@126.com; Tel: +86 532 84022681
bCollege of Science and Technology, Agricultural University of Hebei, Cangzhou 061100, P. R. China
First published on 23rd March 2015
Abstract
CoFe2O4 nanotubes are synthesized by thermal annealing the precursor template of core–shell composite nanorods, which are prepared via two-step hydrothermal process. The formation of a core–shell template and the self-template conversion from nanorods into nanotubes are systemically characterized by XRD, SEM, TEM, EDX, and BET techniques. When employed as anode material for lithium-ion batteries, the specific 1D hollow nanostructures can shorten the Li+ transport path, increase the contact between the active materials and electrolyte, and buffer the volume changes during electrochemical cycles. Endowed with these structural benefits, CoFe2O4 nanotubes display excellent electrochemical performance, such as large and stable reversible capacity (988 mA h g−1 at rate of 100 mA g−1 after 100 cycles and 830 mA h g−1 at 1000 mA g−1 after 200 cycles), superior high-rate capability, and durable cycling life (over 500 cycles at high rate of 1000 mA g−1) as well as good capacity retention (94% after 200 cycles at 1000 mA g−1). Moreover, we believe that the present synthesis strategy may offer a universal approach for preparation of other 1D ternary transition metal oxides as high-performance anode materials.
Introduction
Rechargeable lithium-ion batteries (LIBs) are key devices for energy storage and supply due to their high voltage, high energy density, and long cycling life.1 Nevertheless, the increasing demands for high capacity, power, and rate performance have boosted extensive research on exploring novel electrode materials, especially anodes that can satisfy these requirements. Transition metal oxides including Co3O4,2 Fe3O4,3 Fe2O3,4 TiO2,5 MnO2,6 CuO,7 have been considered as the promising alternative anode materials due to their theoretical specific capacities.8 Despite the advantages, the practical application of transition metal oxide anodes is hindered by their intrinsic low conductivity and huge volume variation in lithiation/delithiation process, which will cause rapid capacity fading and poor rate performance.9,10 Nanostructuring is believed to be one of the effective strategies to solve these problems. For instance, one-dimensional (1D) nanostructures such as nanorods,11,12 nanowires,13,14 and nanotubes,15,16 are particularly attractive, because these 1D nanostructures possess shortened Li+ path, large surface area, and facile strain relaxation which can enhance the lithium storage properties. It is reported that, nanotubes usually deliver better electrochemical performance than nanorods, owing to their larger surface area and tubular morphology.16 Therefore, it is desired to develop tubular structured transition metal oxides as high-performance anodes.In the past few years, ternary transition metal oxides (e.g., ZnFe2O4,17–19 NiFe2O4,19 CuFe2O4,20 MnFe2O4,21 CoFe2O4 (ref. 19, 22–32)) have been on the focus as alternative anode materials for LIBs, because the storage capacity and working voltage of ternary transition metal oxides can be tuned by varying the metal content. Among them, CoFe2O4 has attracted special attention due to its high theoretical capacity (916 mA h g−1). Unfortunately, like other transition metal oxides, the application of CoFe2O4 is hindered by the obstacles of poor electronic conductivity and large volume changes during the repeated Li+ insertion/extraction process, which will induce dramatic capacity fading, poor cycling performance and rate capability. With the aim to break the bottleneck, many strategies have been developed, including preparation of hybrid composites composed of CoFe2O4 and carbon materials,24,28,30,32 fabrication of nanosized CoFe2O4 with unique architectures,19,25–27,29 and synthesis of bicomponent hybrids, such as CoO/CoFe2O4 (ref. 23 and 32) and Co3O4/CoFe2O4.31 However, to our best knowledge, synthesis of CoFe2O4 nanotubes for application of high performance anode materials has rarely been reported.
Very recently, we have developed a self-template synthesis strategy to prepare Co nanotubes using core–shell structured nanorods as precursor.33 In this work, for the first time, CoFe2O4 nanotubes are obtained via a similar self-template strategy for LIBs anode materials. Derived from its unique structural features, CoFe2O4 nanotubes deliver outstanding lithium storage performance, including high reversible capacity, good rate capability, and superior cycling stability in addition to long cycling life.
Experiment
The FeOOH nanorods are prepared following the method reported previously.11 FeCl3·6H2O (4.04 g) is dissolved in 30 mL of water. The obtained clear solution is transferred into a 40 mL Teflon-lined stainless steel autoclave and maintained at 160 °C for 6 h. The product is harvested by centrifugation and then washed with water for vacuum drying at 80 °C for 24 h.0.04 g resultant FeOOH, 0.18 g of CoCl2·6H2O, and 0.22 g of urea are dissolved in 30 mL water and then transferred into a 40 mL autoclave. After the autoclave is heated at 100 °C for 12 h, the product is harvested and sintered at 700 °C for 2 h under ambient air. After cooled to room temperature, the final product is collected for characterization and tested as anode materials for LIBs.
The crystallographic information of the as-prepared samples are obtained from powder X-ray powder diffraction (XRD, Philips X'-pert X-ray diffractometer, Cu Kα radiation: λ = 0.154056 nm). Scanning electron microscope (SEM, JEOL JSM-7500F) and transmission electron microscope (TEM, JEOL JEM-2100) are performed to reveal the morphological features. The energy dispersive X-ray spectroscopy (EDX) is obtained on SEM equipped with EDX probe. N2 sorption/desorption isotherms are measured at 77 K on Micromeritics Co. Ltd., Tristar. The specific surface area is determined using the multipoint Braunauer–Emmett–Teller (BET) method.
The lithium storage properties of the sample are performed using the CR2016-type coin cell. The slurry for working electrode is composed of CoFe2O4, acetylene black and polyvinylidene fluoride (PVDF) at the weight ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The slurry is compressed on a copper foil, and then dried in vacuum. The cells are assembled in an argon-filled glove box with the working electrode, metallic lithium, and the Clegard 2300 microporous film. The electrolyte is 1 M LiPF6 in a mixture of ethylene carbonate (EC)/dimethyl carbonate (DMC)/ethyl methyl carbonate (EMC) (1
1. The slurry is compressed on a copper foil, and then dried in vacuum. The cells are assembled in an argon-filled glove box with the working electrode, metallic lithium, and the Clegard 2300 microporous film. The electrolyte is 1 M LiPF6 in a mixture of ethylene carbonate (EC)/dimethyl carbonate (DMC)/ethyl methyl carbonate (EMC) (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in volume). The cells are charged and discharged at various rates between 3 and 0.01 V.
1 in volume). The cells are charged and discharged at various rates between 3 and 0.01 V.
Results and discussion
SEM and TEM techniques are employed to reveal the formation process of CoFe2O4 nanotubes. As shown in the SEM image of FeOOH (Fig. 1a), uniform FeOOH nanorods with diameters of 50–80 nm and lengths of 700–1000 nm are observed. Fig. 1b shows the SEM image of the products obtained after the second hydrothermal process. It is shown that the resultant nanorods show bigger diameters and rough surface in comparison with FeOOH nanorods, suggesting the successful deposition of outer layer on FeOOH nanorods. The TEM images reveal the core–shell structures more intuitively and clearly. As shown in Fig. 1c, core materials of FeOOH nanorods are completely coated with the outer shell materials. The magnified lattice resolved TEM image is shown in Fig. 1d. It is shown that, the FeOOH core is well crystallized and exhibits clear lattice of (200) crystal plane with the d spacing of 0.53 nm, while the crystal lattice of Co-based shell materials can not be identified, suggesting its amorphous phase. In consistent with TEM, powder XRD patterns (Fig. 1e) of the core–shell nanorods further confirm this result. It is clearly shown that, all the irradiation peaks can be well assigned to tetragonal phase FeOOH (JCPDS 34-1266). No obvious irradiation peak that corresponds to the outer shell materials is observed, confirming the amorphous phase of the outer Co-based shell. To reveal the composition of core–shell nanorods, EDX spectrum is collected and shown in Fig. 1f. It can be clearly observed that, Fe and Co elements are present with the atomic ratio of ∼2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, further confirming the formation of composites nanorods.
1, further confirming the formation of composites nanorods.
 | ||
| Fig. 1 (a) SEM image of FeOOH nanorods, (b) SEM and (c and d) TEM images of core–shell nanorods, (e) XRD patterns and (f) EDX spectrum of core–shell nanorods. | ||
After heating-treatment, the XRD pattern (Fig. 2a) of the final product is performed and delivers typical irradiation peaks that can be indexed to cubic CoFe2O4 (JCPDS 22-1086). As shown in the TEM image of Fig. 2b, CoFe2O4 obtained after calcination presents the hollow tubular nanostructures with the wall thickness of 5–15 nm, which is advantageous to electrolyte diffusion and can shorten Li+ transport distance, therefore effectively boosting the lithium storage performance. The SEM image of CoFe2O4 nanotubes is shown in Fig. 2c. It is clearly shown that, uniform CoFe2O4 nanotubes are obtained at a high yield, except for trace amounts of nanoparticle. In comparison with the precursor nanorods, the size of the resultant CoFe2O4 nanotubes shows no obvious difference. However, the obtained nanotubes show smoother surface than the nanorods, indicating the successful transformation. BET gas-sorption measurements of CoFe2O4 nanotubes are performed. As shown in Fig. 2d, the N2 sorption/desorption isotherms deliver type IV curves, showing the typical mesoporous features. The BET surface area is determined to be 22.3 m2 g−1 and the pore volume is 0.11 cm3 g−1. From the insert of Fig. 2d, the corresponding Barrentt–Joyner–Halenda (BJH) pore-size distribution shows that the main size of pores is ∼18 nm. It is believed that the mesoporous nature can shorten ion transfer path and supply void space to accommodate the volume variation, thus boosting the lithium storage performance of electrode materials.
 | ||
| Fig. 2 (a) XRD pattern, (b) TEM image, (c) SEM image, and (d) N2 sorption/desorption isotherms of CoFe2O4 nanotubes. The insert in (d) presents the corresponding pore size distribution. | ||
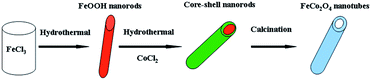
Concluded from the above results, the formation process of CoFe2O4 nanotubes can be assigned to a self-templated approach, as described in Scheme 1. FeOOH nanorods are firstly prepared via a hydrothermal method. Then another hydrothermal process is adopted to coat FeOOH nanorods with Co-based shell to form the 1D core–shell nanorods. The core–shell nanorods are then subjected to calcination and transferred into CoFe2O4 nanotubes based on Kirkendall effect. In this annealing step, FeOOH firstly react with materials in the contacted shell to form a thin layer composed of new phase of Co–Fe oxides. With further heating treatment, core materials continuously evacuate to react with shell materials and deposit on prior formed Co–Fe oxides layer.34,35 Through this evolution process, the hollow 1D nanostructures are eventually formed from the core–shell nanorods.
Fig. 3a shows the charge–discharge voltage profiles of CoFe2O4 nanotubes for its first three cycles at a current density of 100 mA g−1. Clearly, the voltage profiles show the characteristics curves of CoFe2O4 anode.19,22 In the first discharge curve, the sloped region between 1.5 and 0.85 V is assigned to the lithium insertion into CoFe2O4 to form LixCoFe2O4.22 The long plateau observed at about 0.8 V corresponds to the conversion reaction (CoFe2O4 + 8Li+ + 8e− ↔ Co + 2Fe + 4Li2O), accompanied by the decomposition of electrolyte to form a solid electrolyte interphase (SEI) layer. As for the charge profile, a broad plateau region in the range of 1.2–2.2 V is detected, corresponding to the oxidation of metallic Fe and Co.
The cycling performance of CoFe2O4 nanotubes at 100 mA g−1 are shown in Fig. 3b. CoFe2O4 anode delivers an initial discharge and charge capacities of 1417 and 1036 mA h g−1, respectively, with a Coulombic efficiency of 73%. The irreversible capacity loss in the first cycle is usually attributed to the formation of SEI film.31,32 From Fig. 3b, the Coulombic efficiency increases to 92% at the second cycle and keeps over 97% after the 5th cycle, indicating the highly reversible conversion reaction. The large and stable lithium storage properties can be clearly observed in it. After the initial several training cycles, the charge capacity of CoFe2O4 nanotubes maintains stable at approximately 960 mA h g−1 throughout 100 cycles. And a high charge capacity of 988 mA h g−1 can be retained at the 100th cycle, delivering excellent capacity retention of 95% compared with the 1st cycle (1036 mA h g−1). It is worth noting that, the presented values are higher than the theoretical capacity of CoFe2O4. It is not unusual for CoFe2O4 anode materials and has also been observed in the previously reported hollow CoFe2O4 nanospheres.25 The contribution on the extra capacity can be attributed to the specific hollow tubular textures and high surface area, which could serve as a “sponge” to supply more interfacial bonding as extra active sites for lithium insertion.11,36 The lithium storage at high charge–discharge rate of 1 A g−1 is also performed to highlight the good high-rate capability and superior cycling stability of CoFe2O4 nanotubes. As shown in Fig. 3c, the electrochemical training process is also observed at the initial several cycles. It is clearly shown that, CoFe2O4 nanotubes deliver high capacity and excellent cycling stability even at high rate of 1 A g−1 for 500 cycles. The high charge capacities of 830, 780, and 680 mA h g−1 are retained at the 200th, 400th, and 500th cycles, respectively, showing the corresponding capacity retention of 94%, 89%, and 77% compared with the 1st cycle (880 mA h g−1).
To estimate the potential applications of CoFe2O4 nanotubes anode, the rate test with the current density between 100 and 5000 mA g−1 is conducted. In Fig. 3d, the good rate capability for CoFe2O4 nanotubes can be observed. The reversible capacities in the 3rd cycle are 1007, 896, 802, 731, and 654 mA h g−1 at rates of 100, 500, 1000, 2000, and 5000 mA g−1, respectively. Moreover, when the test rate turns back to 100 mA g−1, high capacity of 943 mA h g−1 can be recovered, indicating the good capacity retention.
The comparison of the lithium storage performance between the present CoFe2O4 nanotubes and previously reported CoFe2O4-based anode materials is shown in Fig. 4. It is clearly shown that, the CoFe2O4 nanotubes deliver excellent electrochemical performance both at the low charge–discharge rate and high rate. At low rate (<200 mA g−1), after a longer charge–discharge test, CoFe2O4 nanotubes exhibit higher reversible capacity than that of the reported CoFe2O4/G,22 CoO/CoFe2O4,23 CoFe2O4@C,24 3D macroporous CoFe2O4,28 CoFe2O4/G,30 and Co3O4/CoFe2O4.31 Wang et al. have prepared hollow CoFe2O4 nanospheres as anode materials, which show the highest capacity of 1185 mA h g−1 after 50 cycles at 90 mA g−1.25 Unfortunately, the cycling test is shorter and more importantly, the cycling stability of the hollow CoFe2O4 nanospheres at high rate is lack. At the high-rate region of Fig. 4, the present sample exhibits larger and more stable lithium storage performance than ref. 19, 26, 29 and 30. The CoO/CoFe2O4-NG composite anode materials prepared by Zhao and coauthors deliver higher capacity than this work, but our present sample extends the cycling life to an amazing long life over 500 cycles at high rate, which almost reaches the demand of practical application. To our knowledge, the present CoFe2O4 nanotubes show the longest cycling life at high charge–discharge rate, high specific capacity, and superior cycling stability as well as good high-rate performance.
 | ||
| Fig. 4 The comparison of the electrochemical performance of CoFe2O4-based anode materials between the present work and the previous literatures. | ||
Based on the electrochemical performance shown above, CoFe2O4 nanotubes display extraordinarily excellent cycling stability and high-rate performance as well as high capacity. The outstanding performances should be ascribed to the specific features of nanotubes. The tubular feature is favorable for electrolyte diffusion and can shorten the Li+ and electron transport path, thus enhancing the fast lithium insertion/extraction. The large surface to volume ratio is convenient for the contact between the active materials and electrolyte, improving the utilization of active materials. The 1D architecture and hollow structure with additional void can effectively buffer the repeated volume changes of the tubular wall, inducing prolonged cycling stability and life.
Conclusions
In summary, CoFe2O4 nanotubes are firstly synthesized via a self-template method using core–shell nanorods as precursor. The 1D hollow nanostructures enable CoFe2O4 as the promising integrated anodes for LIBs. As a result of the desirable electrode architecture, the present CoFe2O4 nanotubes can deliver high capacity of 988 mA h g−1 at 100 mA g−1 with excellent capacity retention over 100 cycles. It also shows high capacity of 680 mA h g−1 at 1000 mA g−1 with the outstanding durable cycling life over 500 cycles, making it a promising candidate as advanced anode materials for LIBs. And this synthesis strategy is expected to be employed for the synthesis of other 1D ternary transition metal oxides for applications in energy storage, catalysis, and sensors.Acknowledgements
We thank for the financial support from National Natural Science Foundation of China (21003079), Shandong Provincial Natural Science Foundation, China (ZR2014JL015, ZR2014EMM004).Notes and references
- J.-M. Tarascon and M. Armand, Nature, 2001, 414, 359–367 CrossRef CAS PubMed.
- J. Guo, L. Chen, X. Zhang, B. Jiang and L. Ma, Electrochim. Acta, 2014, 129, 410–415 CrossRef CAS PubMed.
- G. Zhou, D. W. Wang, F. Li, L. Zhang, N. Li, Z. S. Wu, L. Wen, G. Q. Lu and H. M. Cheng, Chem. Mater., 2010, 22, 5306–5313 CrossRef CAS.
- J. Zhang, Y. Sun, Y. Yao, T. Huang and A. Yu, J. Power Sources, 2013, 222, 59–65 CrossRef CAS PubMed.
- Z. Y. Guo, X. L. Dong, D. D. Zhou, Y. J. Du, Y. G. Wang and Y. Y. Xia, RSC Adv., 2013, 3, 3352–3358 RSC.
- A. L. M. Reddy, M. M. Shaijumon, S. R. Gowda and P. M. Ajayan, Nano Lett., 2009, 9, 1002–1006 CrossRef CAS PubMed.
- S. Ko, J. I. Lee, H. S. Yang, S. Park and U. Jeong, Adv. Mater., 2012, 24, 4451–4456 CrossRef CAS PubMed.
- P. Poizot, S. Laruelle, S. Grugeon, L. Dupont and J.-M. Tarascon, Nature, 2000, 407, 497–499 Search PubMed.
- P. G. Bruce, B. Scrosati and J.-M. Tarascon, Angew. Chem., Int. Ed., 2008, 47, 2930–2946 CrossRef CAS PubMed.
- M. Ebner, F. Marone, M. Stampanoni and V. Wood, Science, 2013, 342, 716–720 CrossRef CAS PubMed.
- X. Zhang, H. Chen, Y. Xie and J. Guo, J. Mater. Chem. A, 2014, 2, 3912–3918 CAS.
- N. Jayaprakash, W. D. Jones, S. S. Moganty and L. A. Archer, J. Power Sources, 2012, 200, 53–58 CrossRef CAS PubMed.
- H. Wu, M. Xu, Y. C. Wang and G. F. Zheng, Nano Res., 2013, 6, 167–173 CrossRef CAS.
- Y. G. Li, B. Tan and Y. Y. Wu, Nano Lett., 2008, 8, 265–270 CrossRef CAS PubMed.
- X. W. Lou, D. Deng, J. Y. Lee, J. Feng and L. A. Archer, Adv. Mater., 2008, 20, 258–262 CrossRef CAS.
- L. Chen, H. Xu, L. Li, F. Wu, J. Yang and Y. Qian, J. Power Sources, 2014, 245, 429–435 CrossRef CAS PubMed.
- D. Bresser, E. Paillard, R. Kloepsch, S. Krueger, M. Fiedler, R. Schmitz, D. Baither, M. Winter and S. Passerini, Adv. Energy Mater., 2013, 3, 513–523 CrossRef CAS.
- P. F. Teh, Y. Sharma, S. S. Pramana and M. Srinivasan, J. Mater. Chem., 2011, 21, 14999–15008 RSC.
- N. Wang, H. Xu, L. Chen, X. Gu, J. Yang and Y. Qian, J. Power Sources, 2014, 247, 163–169 CrossRef CAS PubMed.
- Y. Sharma, N. Sharma, G. V. Subba Rao and B. V. R. Chowdari, J. Power Sources, 2007, 173, 495–501 CrossRef CAS PubMed.
- Y. Xiao, J. Zai, L. Tao, B. Li, Q. Han, C. Yu and X. Qian, Phys. Chem. Chem. Phys., 2013, 15, 3939–3945 RSC.
- Y. Zhao, J. Li, Y. Ding and L. Guan, J. Mater. Chem., 2011, 21, 19101–19105 RSC.
- M. Li, Y.-X. Yin, C. Li, F. Zhang, L.-J. Wan, S. Xu and D. G. Evans, Chem. Commun., 2012, 48, 410–412 RSC.
- Z. Ding, B. Yao, J. Feng and J. Zhang, J. Solid State Electrochem., 2014, 18, 19–27 CrossRef CAS PubMed.
- Y. Wang, D. Su, A. Ung, J.-H. Ahn and G. Wang, Nanotechnology, 2012, 23, 055402 CrossRef CAS PubMed.
- Y.-Q. Chu, Z.-W. Fu and Q.-Z. Qin, Electrochim. Acta, 2004, 49, 4915–4921 CrossRef CAS PubMed.
- Z. H. Li, T. P. Zhao, X. Y. Zhan, D. S. Gao, Q. Z. Xiao and G. T. Lei, Electrochim. Acta, 2010, 55, 4594–4598 CrossRef CAS PubMed.
- Y. Wang, J. Park, B. Sun, H. Ahn and G. Wang, Chem.–Asian J., 2012, 7, 1940–1946 CrossRef CAS PubMed.
- P. Lavela and J. L. Tirado, J. Power Sources, 2007, 172, 379–387 CrossRef CAS PubMed.
- S. Liu, J. Xie, C. Fang, G. Cao, T. Zhu and X. Zhao, J. Mater. Chem., 2012, 22, 19738–19743 RSC.
- A. K. Rai, J. Gim, T. V. Thi, D. Ahn, S. J. Cho and J. Kim, J. Phys. Chem. C, 2014, 118, 11234–11243 CAS.
- C. Zhao, C. Yu, S. Liu, J. Yang, X. Fan and J. Qiu, Part. Part. Syst. Charact., 2015, 32, 91–97 CrossRef CAS.
- J. Guo, X. Zhang, T. Zhang, T. Zhou, X. Zhang and Z. Quan, Mater. Lett., 2015, 141, 288–290 CrossRef CAS PubMed.
- L. Yu, L. Zhang, H. B. Wu and X. W. Lou, Angew. Chem., Int. Ed., 2014, 53, 3711–3714 CrossRef CAS PubMed.
- X. W. Lou, D. Deng, J. Y. Lee, J. Feng and L. A. Archer, Adv. Mater., 2008, 20, 258–262 CrossRef CAS.
- J. Wang, N. Yang, H. Tang, Z. Dong, Q. Jin, M. Yang, D. Kisailus, H. Zhao, Z. Tang and D. Wang, Angew. Chem., Int. Ed., 2013, 52, 6417–6420 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |