Green sample clean-up based on magnetic multiwalled carbon nanotubes for the determination of lamivudine by high performance liquid chromatography
Lingling Wang,
Xu Xu,
Zhenzhen Zhang,
Danfeng Zhang,
Xueyan Liu and
Lei Zhang*
College of Chemistry, Liaoning University, 66 Chongshan Middle Road, Shenyang, Liaoning 110036, People's Republic of China. E-mail: zhanglei63@126.com; Fax: +86 24 62202380; Tel: +86 24 62207809
First published on 12th February 2015
Abstract
A simple, green and efficient method for the determination of trace lamivudine (LMV) based on magnetic solid-phase extraction (MSPE) coupled with high performance liquid chromatography (HPLC) was developed. Magnetic multiwalled carbon nanotubes (MMWCNTs) were prepared by assembling Ni nanoparticles onto multiwall carbon nanotubes (Ni/MWCNTs). Due to their excellent adsorption capability, the Ni/MWCNTs were used as adsorbents for magnetic solid-phase extraction (MSPE). Various parameters affecting the extraction efficiency such as the amount of adsorbent, the pH, ionic strength and desorption conditions were optimized. The extracted LMV was desorbed using ionic liquid (IL) “Green solvents” as substitutes of volatile organic solvents. Under optimized conditions, a limit of detection (LOD) of 0.14 ng mL−1 and enrichment factor (EF) of 292 were obtained. The calibration curves showed good linearity (r2 = 0.9978–0.9993) over the concentration ranges of 0.5–500 ng mL−1. The proposed method was applied to the analysis of river water and human urine samples, and the recoveries of analytes obtained were in the range of 97.6–103.0 and 93.4–102.2%, respectively. The relative standard deviations (RSD) were less than 5.93% (n = 5). The high efficiency of the method ensures its successful application in extraction of trace LMV from large volumes of environmental water and biological fluid samples.
1. Introduction
Hepatitis B virus (HBV) and human immune deficiency virus (HIV) remain important public health problems in the world. About 35.3 million adults and children worldwide are estimated to be infected with HIV.1 HIV infection causes a progressive decline in the number and function of CD4+ T cells, leaving the host vulnerable to opportunistic infections.2,3 If untreated, eventually most HIV-infected individuals develop acquired immunodeficiency syndrome (AIDS) and die.4 HBV is highly prevalent in the United States, with approximately 1.2 million chronic carriers. HBV is the ninth leading cause of death worldwide, and there are more than 350 million carriers.5 Hepatitis B may develop into a chronic disease in up to 10 percent of newly-infected people each year. If left untreated, the risk of developing cirrhosis (scarring) and liver cancer becomes higher.Lamivudine (LMV) is chemically known as (2R-cis)-4-amino-1-[2-(hydroxy methyl)-1,3-oxathiolan-5-yl]-2(1H)-pyrimidinone. LMV is an analogue of cytidine. It can inhibit both types (1 and 2) of HIV reverse transcriptase and also the reverse transcriptase of HBV. LMV, the first oral antiviral agent approved for the treatment of chronic Hepatitis B, has been the most commonly prescribed drug for initial therapy of Hepatitis B.
In the literature, HPLC and HPLC-MS methods have been reported for analysis of LMV and its metabolites in biological fluids.6–12 Compared with HPLC, HPLC-MS is a very sensitive, accurate and efficient technique for identification and quantification of LMV. However, LC-MS instrument is expensive and not available in many laboratories. HPLC instrument is widely used due to lower cost and greater robustness in bioavailability studies. But HPLC assay suffered from lack of sensitivity and specificity. LMV in human urine and environmental water samples are at trace level, so a preconcentration procedure is necessary before HPLC analysis.
To date, a variety of methods have been developed for the separation and preconcentration of the target compounds from various samples.13–15 Solid phase extraction (SPE) is one of the most popular techniques due to its high enrichment performance, low solvent-cost and simple to operate. In recent years, a new mode of SPE, based on the use of magnetic or magnetically modified adsorbents termed as magnetic solid-phase extraction (MSPE), has been developed.16–19 In comparison to traditional SPE, MSPE has several advantages. The separation process in MSPE can be performed directly in crude samples containing suspended solid materials by applying an external magnet without the need of additional centrifugation or filtration, and thus enhance extraction efficiency. Various magnetic materials have been applied in the extraction of multifarious compounds, including pharmaceuticals, pesticides and polycyclic aromatic compounds in biological or environmental samples.20–31 Considering the structural feature of the LMV, magnetic multiwalled carbon nanotubes (MMWCNTs) are considered to be extremely superior adsorbent due to their high specific surface area and large micropore volume. Moreover, the main goal of sustainable chemistry is to decrease the use of hazardous or generation material in chemical processes. In recent years, ionic liquids (ILs) have attracted great attention due to their unique chemical and physical properties such as nonvolatility that preserves the environment, excellent solvation qualities, non-flammable and high thermal stability.32,33 Herein ILs are used as “green” eluent for replacing the traditional toxic solvents. As far as we know, studies on the application of Ni/MWCNTs and ILs synergistic microextraction for LMV determination have not been reported.
In this work, magnetic Ni/MWCNTs hybrid materials were fabricated for the extraction of LMV, and the adsorption performance of Ni/MWCNTs was evaluated. “Green solvents” – ILs as environmentally friendly substitutes for volatile organic eluent solvents were selected to desorb LMV. When the extraction was completed, the Ni/MWCNTs were rapidly separated from the sample matrix by an external magnetic field, and finally the eluted LMV with ILs was analysed by HPLC without further enrichment. Ni/MWCNTs were applied to extract trace level LMV from human urine samples. Satisfactory results were obtained with high recoveries, low LOD, good precision and fine accuracy.
2. Materials and methods
2.1 Materials and reagents
MWCNTs (95% purity) were purchased from Chengdu Organic Chemicals Co. Ltd. (Chengdu, China). The standard of LMV was obtained from Northeast Pharmaceutical Science & Technology Development Co. Ltd. (Shenyang, China). 1-Ethyl-3-methylimidazolium tetrafluoroborate ([C2mim][BF4]), was purchased from Chengjie Chemical Co., Ltd. (Shanghai, China). Chromatographic grade methanol (MeOH) was purchased from Fisher Corporation (Pittsburgh, PA, USA). HPLC-grade deionised water was obtained from a MilliQ water purification system (MilliQ Water; Molsheim, France). Analytical grade sodium phosphate and sodium chloride were purchased from Beijing Chemical Co. (Beijing, China). Deionized water used throughout experiments was purified using a Sartorius Arium 611 system (Sartorius, Göttingen, Germany).2.2 Standard solution and samples preparation
The stock solution containing LMV was prepared by dissolving appropriate amount of LMV in deionized water and stored at 4 °C under dark conditions. The working solutions were obtained daily by appropriately diluting the stock solution with deionized water.Blank urine samples were collected from healthy volunteers and stored at −18 °C. Before use, the urine sample was thawed at room temperature and centrifuged at 4000 rpm for 5 min. 10 mL blank and spiked urine samples were diluted at the ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5 with deionized water for further analyses.
1.5 with deionized water for further analyses.
River water samples were taken from Hunhe river (Shenyang, China). The spiked river water samples were prepared by spiking working solutions into 150 mL of river water for further analyses.
2.3 Magnetic Ni/MWCNTs preparation
The preparation of Ni/MWCNTs was illustrated in Fig. 1. The appropriate amount of purified CNTs was suspended in ethylene glycol (EG) of 0.05 mol L−1 NiCl2 by ultrasonic dispersion. Ten minutes later, appropriate amounts of NaOH, Na2CO3 and N2H4·H2O were added in sequence. The solution was placed in the microwave chemical reactor and heated at 700 W and 2450 MHz for 1.5 min, and the suspended particles were precipitated by placing a magnet under the container. Then the precipitation Ni/MWCNTs was washed with distilled water, and dried at 60 °C in vacuum for 5 h.2.4 Characterization of the Ni/MWCNTs
The morphology of the Ni/MWCNTs was observed by a transmission electron microscope (TEM, TecnaiG 200, FIE, USA). The BET surface area and pore distribution were analyzed using N2 adsorption/desorption isotherms on a Tristar 3020 volumetric adsorption analyzers manufactured by Micromeritics (Norcross, GA). The crystal structure of the Ni/MWCNTs was investigated by the Powder X-ray diffraction (XRD, Siemens D5000 Diffractometer, Germany). The magnetic property was characterized by vibrating sample magnetometry (VSM, Lakeshore-7407) at room temperature.2.5 Batch adsorption experiment
Batch adsorption experiments were conducted using 50 mL flasks containing 20.0 mg Ni/MWCNTs and 25 mL of 5.0 μg mL−1 LMV solution. After stirring at a constant rate at 293 K for 20.0 min, the solid/liquid phases were separated by an external magnet. Then the concentration of LMV in suspensions was determined by HPLC. The adsorption percentage (Ads.%) was calculated based on the following equation:
 | (1) |
2.6 MSPE procedure
20.0 mg Ni/MWCNTs were added into 25 mL of the spiked and blank human urine sample solutions (or 150 mL of the spiked water samples), and the extraction was performed at a stirring rate of 180 rpm for 20 min. After extraction, the Ni/MWCNTs were separated rapidly from the solution by an external magnet. The target analytes were ultrasonically eluted from the Ni/MWCNTs with 0.5 mL of ILs/0.03 mol L−1 Na3PO4 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v
3, v![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v) for 2 min. The eluent was separated from the Ni/MWCNTs by a magnet in 30 s. And eluent, as analytical solution, filtered through a 0.45 μm PTFE filter membrane for HPLC analysis.
v) for 2 min. The eluent was separated from the Ni/MWCNTs by a magnet in 30 s. And eluent, as analytical solution, filtered through a 0.45 μm PTFE filter membrane for HPLC analysis.
2.7 HPLC analysis
An Agilent 1100 HPLC (Palo Alto, CA, USA) equipped with an automatic sampler and diode array detector was used for the chromatographic analysis. The analysis of LMV was performed on a SB-C18 (150 mm × 4.6 mm, 5 μm) column (Agilent, Palo Alto, CA, USA). The mobile phase was a mixture of 20 mmol L−1 phosphate buffer (pH 3.5)–MeOH (92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8, v/v). The flow rate and column temperature were set at 1.0 mL min−1 and 30 °C, respectively. The detection wavelength was set at 270 nm. The injection volume of analytical solution was 20 μL.
8, v/v). The flow rate and column temperature were set at 1.0 mL min−1 and 30 °C, respectively. The detection wavelength was set at 270 nm. The injection volume of analytical solution was 20 μL.
3. Results and discussion
3.1 Characteristics of Ni/MWCNTs
The morphology of MWCNTs and Ni/MWCNTs was investigated by TEM. From Fig. 2(a), it is observed that Ni nanoparticles were deposited on the surface of MWCNTs, and the average size of the Ni nanoparticles was about 20 nm. | ||
| Fig. 2 (a) TEM image of Ni/MWCNTs; (b) XRD of Ni/MWCNTs; (c) magnetic hysteresis curves for Ni/MWCNTs; (d) N2 adsorption–desorption isotherms measured of Ni/MWCNTs. | ||
The XRD patterns of MWCNTs and Ni/MWCNTs are shown in Fig. 2(b). The diffraction peaks at 2θ = 26.2° and 44.2° were assigned to (002) and (101) plane of MWCNTs. As can be seen from Fig. 2(b), Ni/MWCNTs hybrids were a mixture of two phases: Ni and MWCNTs. Three peaks corresponding to the structure of Ni existed in the XRD pattern of the magnetic composites. The peaks could be assigned to (111), (200), (222) plane of fcc structure Ni. The two peaks corresponding to the structure of MWCNTs also existed in the XRD pattern of the Ni/MWCNTs. The results revealed that the Ni/MWCNTs adsorbents were composed of Ni and MWCNTs.
The magnetization properties of Ni/MWCNTs was investigated at room temperature by measuring magnetization curves (Fig. 2(c)). The saturation magnetization Ms of Ni/MWCNTs was 9.371 emu g−1 (magnetic field = ±10 kOe). The loop of Ni/MWCNTs exhibited very low coercive field and remanence values, which indicated that the Ni/MWCNTs was very close to the behaviour of superparamagnet at room temperature. The simple magnetic separation experiment confirmed that Ni/MWCNTs can be used as a magnetic adsorbent to remove LMV from aqueous solutions. Compared with conventional adsorbents in SPE, the magnet adsorbent Ni/MWCNTs can be reused and the phase separation can be conveniently realized by an external magnetic field.
BET surface area and Barrett–Joyner–Halenda (BJH) pore-size distribution curves (inset) of the adsorbent were showed in Fig. 2(d). The specific surface areas of MWCNTs and Ni/MWCNTs were determined to be 343.603 and 444.393 m2 g−1, respectively. The larger specific surface areas of Ni/MWCNTs were attributed to the smaller size of Ni particles in the presence of MWCNTs. The average pore diameter of sorbents was determined to be 3.020 nm for MWCNTs and 3.011 nm for Ni/MWCNTs. The effective surface area and active sites of adsorbent increased, which would enhance the adsorption quantity of LMV.
3.2 Optimization of extraction conditions
In order to select the optimum extraction conditions, 25 mL 50.0 ng mL−1 LMV solution was applied to test the adsorption behavior at different conditions. The experimental parameters, including the amount of adsorbent, the pH and ionic strength of sample solution and elution conditions, were investigated.The addition of salt has both positive and negative impacts on the extraction process. On the one hand, the occurring of saltingout effect can decrease the solubility of organic analytes, which is conductive to the improvement of extraction efficiency; on the other hand, the addition of salt can also increase the viscosity of the solution, which is against the adsorption of LMV by Ni/MWCNTs due to the reduction of extraction capability and diffusion coefficient. In this study, LMV adsorption data under different ionic strengths (0.02, 0.05, 0.10 and 0.20 mol L−1 NaCl) were analyzed. The results showed that the addition of NaCl had no significant effect on recoveries of the analytes in the concentration range investigated. Hence, the LMV solution without adjusting ionic strength was adopted.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 1
3, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 4
1, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v), and ILs/0.03 mol L−1 Na3PO4 (1
1 v/v), and ILs/0.03 mol L−1 Na3PO4 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, 1
9, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 2
4, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) were investigated. The results showed in Fig. 3, the eluent of ILs/0.03 mol L−1 Na3PO4 (2
3, v/v) were investigated. The results showed in Fig. 3, the eluent of ILs/0.03 mol L−1 Na3PO4 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) gave the highest recovery for the target analytes. Therefore, the eluent containing ILs/0.03 mol L−1 Na3PO4 (2
3, v/v) gave the highest recovery for the target analytes. Therefore, the eluent containing ILs/0.03 mol L−1 Na3PO4 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) was selected as the desorption solvent for the subsequent studies.
3, v/v) was selected as the desorption solvent for the subsequent studies.
3.3 Adsorption kinetics and adsorption diffusion mechanism
The influence of contact time on the adsorption of LMV was presented in the Fig. 4. The LMV adsorption reaction was found to be fast, the adsorption process could reach equilibrium within 20 min. To understand the adsorption mechanism, the pseudo-first-order and pseudo-second-order models were used to investigate the adsorption kinetics of adsorbent. The parameters of kinetics models were extracted from the experimental data of this study, and the results were presented in Table 1. Compared to that of the pseudo-first-order kinetic model, the correlation coefficient of the pseudo-second-order model was higher (R2 ≥ 0.999). Therefore, the pseudo-second-order kinetic model was feasible to describe the adsorption process of LMV on the Ni/MWCNTs. | ||
| Fig. 4 The kinetics curve of LMV adsorption on Ni/MWCNTs at different temperatures. (Ni/MWCNTs, 20.0 mg; LMV concentration, 5.0 μg mL−1; sample solution volume, 25 mL; extraction time, 20 min). | ||
| T (K) | Pseudo-first-order model | Pseudo-second-order model | |||||
|---|---|---|---|---|---|---|---|
| k1 (min−1) | q1 (mg g−1) | r1 | k2 (g mg−1 min−1) | q2 (mg g−1) | h2 (mg g−1 min−1) | r2 | |
| 318 | 0.244 | 0.867 | 0.930 | 0.882 | 5.952 | 31.246 | 0.999 |
| 298 | 0.193 | 1.270 | 0.957 | 0.508 | 6.211 | 19.597 | 1.000 |
| 278 | 0.228 | 1.702 | 0.931 | 0.389 | 6.289 | 15.386 | 1.000 |
In the adsorption process, only external mass transfer and intra-particle diffusion play important role in rate determination. In order to evaluate the relative importance of the two processes, time-course LMV adsorption data were processed using the intra-particle diffusion and the external mass transfer models. Weber–Morris intra-particle diffusion model is represented as follows:
| qt = Kdt1/2 + I | (2) |
| T (K) | Initial linear portion | Second linear portion | Third linear portion | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Kd1 (mg g−1 min−1/2) | I1 | r1 | Kd2 (mg g−1 min−1/2) | I2 | r2 | Kd3 (mg g−1 min−1/2) | I3 | r3 | |
| 318 | 0.597 | 4.406 | 0.977 | 0.218 | 5.177 | 0.930 | 0.014 | 5.817 | 0.988 |
| 298 | 0.921 | 3.866 | 0.930 | 0.304 | 5.508 | 0.989 | 0.050 | 5.865 | 0.962 |
| 278 | 1.595 | 2.627 | 0.934 | 0.348 | 5.016 | 0.909 | 0.040 | 5.980 | 0.958 |
The external mass transfer model is represented by eqn (3):
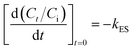 | (3) |
It is amply clear that both intra-particle and external mass transfer processes played important roles in adsorption of LMV. However, it was unclear as to which one exerted a greater influence on the rate of LMV adsorption. This point was resolved using the Boyd plot. The Boyd plot is obtained by plotting Bt versus time t. The Bt is expressed by following equation:
| Bt = −ln(1 − F) − 0.4977 | (4) |
3.4 Adsorption capacity and adsorption thermodynamic
The equilibrium adsorption amount of LMV on Ni/MWCNTs as a function of the equilibrium concentration of LMV was depicted in Fig. 6. An increased adsorption was observed for LMV until saturation was attained. In order to investigate the adsorption isotherm of LMV on Ni/MWCNTs, the Langmuir and Freundlich isotherm models were used.Langmuir equation:
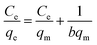 | (5) |
Freundlich equation is given by:
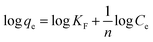 | (6) |
As shown in Table 3, the adsorption data was well correlated by Langmuir equation under the studied concentration range. The maximum adsorption capacity of LMV on Ni/MWCNTs was 129.20, 136.80, 140.06 mg g−1 at 318, 298 and 278 K, respectively.
| T/K | Langmuir model | Freundlich model | |||
|---|---|---|---|---|---|
| qm/(mg g−1) | r2 | kF (mg−1 g−1) | n | r2 | |
| 318 | 129.20 | 1.000 | 14.224 | 2.503 | 0.956 |
| 298 | 136.80 | 1.000 | 19.346 | 2.782 | 0.936 |
| 278 | 140.06 | 1.000 | 20.890 | 2.833 | 0.933 |
3.5 Interference studies
Human urine and river water samples contains minimal organic matters and considerable amounts of inorganic ions. These substances may have a potentially competitive adsorption effect on LMV in human urine samples, so it is necessary to investigate the effect of various ions on the extraction of LMV. The LMV solutions (50.0 ng mL−1) consists of Na+, K+, NH4+, Mg2+, Ca2+, H2PO4−, HCO3−, SO42−, PO43−, CO32− and ascorbic acid, urea were applied in the studies, respectively. The tolerance ratio of each substance was taken as the largest amount yielding an error in the determination of the analyte not exceeding 5% (Table 4).| Interference | Interference to LMV ratio (w/w) |
|---|---|
| Na+, K+, NH4+, Mg2+, Ca2+ | 1000 |
| H2PO4−, HCO3−, SO42− | 1000 |
| PO43−, CO32− | 300 |
| Ascorbic acid | 1000 |
| Urea | 700 |
3.6 Reusability of Ni/MWCNTs
To investigate the reusability of Ni/MWCNTs, the adsorbent was reused for extraction of LMV after desorption of the analytes from Ni/MWCNTs. The results indicated that above 90% recovery for the target analytes was obtained even after a 10-cycle run of Ni/MWCNTs for the adsorption and desorption of LMV. It can be seen that Ni/MWCNTs nanocomposites have the advantage of reuse stability, which can significantly reduce the cost in sample preparation.3.7 Analytical performances and applications of the method for LMV
Under the optimum experimental conditions, different quality parameters were evaluated to assess the method performance. Several important parameters including dynamic linear ranges (DLRs), regression equations, correlation coefficient (r2), enrichment factors (EFs), extraction recoveries (ER%), precision, the limit of detection (LOD) and limit of quantification (LOQ) were determined to validate the method. The working curves were constructed by plotting the peak areas measured versus the concentrations of analytes in the samples. All the experiments were performed in triplicate.The LODs and LOQs were calculated by the following equations:
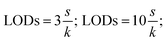 | (7) |
| Sample | DLR | Regression equation | LOD | LOQ | r2 | EF | ERd (%) | RSD (%) (n = 5) intra-day | RSD (%) (n = 5) inter-day |
|---|---|---|---|---|---|---|---|---|---|
| a All concentrations are based on ng mL−1.b Sample volume was 150 mL.c Sample volume was 25 mL.d Extraction recoveries were obtained at 50.0 ng mL−1 level of the analytes. | |||||||||
| Waterb | 0.5–500.0 | y = 17.244C + 45.594 | 0.14 | 0.46 | 0.9982 | 292 | 97.3 | 2.46 | 3.75 |
| Waterc | 5.0–500.0 | y = 2.8871C + 15.353 | 0.78 | 2.60 | 0.9993 | 50 | 99.3 | 1.62 | 2.83 |
| Urinec | 5.0–500.0 | y = 2.6973C + 24.457 | 0.97 | 3.23 | 0.9978 | 48 | 96.8 | 3.85 | 4.36 |
In order to evaluate the applicability of the present method, human urine and river water samples were analyzed. It can be seen (Fig. 7) that no significant interference peaks were found at the retention positions of LMV. To test the accuracy of the proposed method, recoveries of method were investigated by determinating LMV in the spiked human urine and river water samples. The analytical results were showed in Table 6. The recoveries of the spiked samples were in the range from 93.4% to 103.0%. The relative standard deviations varied between 2.80% and 5.93% (n = 5). It can be considered that the current method provides acceptable recoveries and precision for the determination of LMV in real samples.
| Sample | Spiked (ng mL−1) | Found (ng mL−1) | RRa (%) | RSD (%) (n = 5) |
|---|---|---|---|---|
| a Relative recovery.b ND = not detected. | ||||
| Human urine | 0 | NDb | — | — |
| 10.0 | 10.22 ± 0.16 | 102.2 | 5.93 | |
| 50.0 | 49.23 ± 0.47 | 98.5 | 3.53 | |
| 250.0 | 233.51 ± 1.31 | 93.4 | 2.80 | |
| River water | 0 | ND | — | — |
| 1.0 | 1.03 ± 0.82 | 103.0 | 4.85 | |
| 50.0 | 50.12 ± 0.53 | 100.2 | 3.12 | |
| 250.0 | 243.93 ± 1.85 | 97.6 | 3.42 | |
3.8 Comparison with other methods
Table 7 provides a comparison of the proposed method with other analytical methods previously reported in literature for the determination of LMV in biological fluids and environmental water. As can be seen, the LOD of this work was lower than the reported method, showing the high sensitivity. Moreover, the separation of adsorbent from sample solutions was simple and fast owing to the magnetic property of Ni/MWCNTs. It can be concluded that Ni/MWCNTs can be an excellent adsorbent for the extraction and enrichment of LMV from real samples.| Sample matrix | Extraction technique | Detection method | Linearity (ng mL−1) | LOD/LOQ (ng mL−1) | Recovery (%) | Ref. |
|---|---|---|---|---|---|---|
| a LLE: liquid–liquid extraction.b SPE: solid-phase extraction. | ||||||
| Rat plasma | LLEa | HPLC-UV | 100–50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
—/100 | 72–79 | 6 |
| Human serum | LLE | HPLC-UV | 5–2500 | 2/5 | 88–98 | 13 |
| Human plasma | SPEb cartridges | HPLC-MS/MS | 10–4000 | —/10 | 56.5–67.5 | 11 |
| Human plasma | SPE columns | HPLC-UV | 15–5000 | —/15 | 97.7–103.1 | 12 |
| Human urine | SPE-MIPs | HPLC-UV | 60–700 | 7.95/24.05 | 82.5–90.8 | 14 |
| Human urine | SPE-Ni/MWCNTs | HPLC-DAD | 5–500 | 0.97/3.23 | 93.4–102.2 | |
| River water | 0.5–500 | 0.14/0.46 | 97.6–103.0 | This work | ||
4. Conclusions
In this research, magnetic Ni/MWCNTs were synthesized and applied to enrich trace level LMV from large volumes of urine samples in a short extraction time. Compared with other extraction modes, the obtained Ni/MWCNTs adsorbents exhibit excellent extraction ability to LMV due to the large surface area and synergistic effect between the MWCNTs and the Ni nanoparticles. Ni/MWCNTs adsorbents can be easily isolated from sample solution with an external magnetic field. On this basis, a method of MSPE-HPLC-DAD has been developed for the analysis of LMV. This proposed method also has advantages of simplicity, ease of operation, and environmental benignity. Low detection limit and satisfactory recoveries were obtained, indicating that the proposed method using Ni/MWCNTs as SPE adsorbent is considerable potential in pre-concentrating and detecting trace LMV in environmental water and biological fluid samples.Acknowledgements
This project was supported by the National Nature Science Foundation of China (NSFC51178212), Liaoning Provincial Department of Education Innovation Team Projects (LT2012001), Shenyang Science and Technology Plan Project (F13-289-1-00), the Foundation of 211 Project for Innovative Talent Training, Liaoning University, and the Foundation for Young Scholars of Liaoning University (no. 2012LDQN06). The authors also thank their colleagues and other students who participated in this study.References
- S. K. Dey, N. Zahan, S. Afrose, T. Islam, M. Shajahan, S. Saha, S. A. Mahmud, A. A. Talukder, M. Mizuguchi and H. Ushijima, HIV & AIDS Rev., 2014, 13, 33–39 Search PubMed.
- A. G. Dalgleish, P. C. L. Beverley, P. R. Clapham, D. H. Crawford, M. F. Greaves and R. A. Weiss, Nature, 1984, 312, 763–767 CrossRef CAS.
- D. Klatzmann, F. Barre-Sinoussi, M. Nugeyre, C. Danquet, E. Vilmer, C. Griscelli, F. Brun-Veziret, C. Rouzioux, J. Gluckman and J. Chermann, et al., Science, 1984, 225, 59–63 CAS.
- J. P. Christensen, R. D. Cardin, K. C. Branum and P. C. Doherty, Proc. Natl. Acad. Sci. U. S. A., 1999, 96, 5135–5140 CrossRef CAS.
- W. M. Lee, N. Engl. J. Med., 1997, 337, 1733–1745 CrossRef CAS PubMed.
- Y. Alnouti, C. A. White and M. G. Bartlett, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2004, 803, 279–284 CrossRef CAS PubMed.
- B. Fan and J. T. Stewart, J. Pharm. Biomed. Anal., 2002, 28, 903–908 CrossRef CAS.
- K. B. Kenney, S. A. Wring, R. M. Carr, G. N. Wells and J. A. Dunn, J. Pharm. Biomed. Anal., 2000, 22, 967–983 CrossRef CAS.
- J. J. Zheng, S. T. Wu and T. A. Emm, J. Chromatogr. B: Biomed. Sci. Appl., 2001, 761, 195–201 CrossRef CAS.
- X.-J. Zhou and J.-P. Sommadossi, J. Chromatogr. B: Biomed. Sci. Appl., 1997, 691, 417–424 CrossRef CAS.
- R. K. Valluru, P. B. Reddy B, K. Sumanth S, P. Kumar V and N. B. Kilaru, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2013, 931, 117–126 CrossRef CAS PubMed.
- C. P. W. G. M. Verweij-van Wissen, R. E. Aarnoutse and D. M. Burger, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2005, 816, 121–129 CrossRef CAS PubMed.
- G. Bahrami, S. Mirzaeei, A. Kiani and B. Mohammadi, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2005, 823, 213–217 CrossRef CAS PubMed.
- M. Shekarchi, M. Pourfarzib, B. Akbari-Adergani, A. Mehramizi, M. Javanbakht and R. Dinarvand, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2013, 931, 50–55 CrossRef CAS PubMed.
- X. Zhao, Y. Shi, T. Wang, Y. Cai and G. Jiang, J. Chromatogr. A, 2008, 1188, 140–147 CrossRef CAS PubMed.
- Y. Xiong, Y. Xie, S. Chen and Z. Li, Chem.–Eur. J., 2003, 9, 4991–4996 CrossRef CAS PubMed.
- H. Niu, Y. Cai, Y. Shi, F. Wei, S. Mou and G. Jiang, J. Chromatogr. A, 2007, 1172, 113–120 CrossRef CAS PubMed.
- S. Zhang, H. Niu, Y. Cai and Y. Shi, Anal. Chim. Acta, 2010, 665, 167–175 CrossRef CAS PubMed.
- L. Sun, C. Zhang, L. Chen, J. Liu, H. Jin, H. Xu and L. Ding, Anal. Chim. Acta, 2009, 638, 162–168 CrossRef CAS PubMed.
- Y. Hou, J. Yu and S. Gao, J. Mater. Chem., 2003, 13, 1983–1987 RSC.
- Y. Lu, Y. Yin, B. T. Mayers and Y. Xia, Nano Lett., 2002, 2, 183–186 CrossRef CAS.
- L. Zhou, L. Ji, P.-C. Ma, Y. Shao, H. Zhang, W. Gao and Y. Li, J. Hazard. Mater., 2014, 265, 104–114 CrossRef CAS PubMed.
- D. Yang, F. Yang, J. Hu, J. Long, C. Wang, D. Fu and Q. Ni, Chem. Commun., 2009, 4447–4449, 10.1039/b908012k.
- F. Bianchi, V. Chiesi, F. Casoli, P. Luches, L. Nasi, M. Careri and A. Mangia, J. Chromatogr. A, 2012, 1231, 8–15 CrossRef CAS PubMed.
- N. Rastkari and R. Ahmadkhaniha, J. Chromatogr. A, 2013, 1286, 22–28 CrossRef CAS PubMed.
- H.-F. Zhang and Y.-P. Shi, Anal. Chim. Acta, 2012, 724, 54–60 CrossRef CAS PubMed.
- Y. Xu, J. Ding, H. Chen, Q. Zhao, J. Hou, J. Yan, H. Wang, L. Ding and N. Ren, Food Chem., 2013, 140, 83–90 CrossRef CAS PubMed.
- A. V. Herrera-Herrera, J. Hernández-Borges, M. M. Afonso, J. A. Palenzuela and M. Á. Rodríguez-Delgado, Talanta, 2013, 116, 695–703 CrossRef CAS PubMed.
- B. Chen, S. Wang, Q. Zhang and Y. Huang, Analyst, 2012, 137, 1232–1240 RSC.
- T. Madrakian, M. Ahmadi, A. Afkhami and M. Soleimani, Analyst, 2013, 138, 4542–4549 RSC.
- F. Pena-Pereira, R. M. B. O. Duarte, T. Trindade and A. C. Duarte, J. Chromatogr. A, 2013, 1299, 25–32 CrossRef CAS PubMed.
- J. L. Anderson, J. Ding, T. Welton and D. W. Armstrong, J. Am. Chem. Soc., 2002, 124, 14247–14254 CrossRef CAS PubMed.
- E. G. Yanes, S. R. Gratz, M. J. Baldwin, S. E. Robison and A. M. Stalcup, Anal. Chem., 2001, 73, 3838–3844 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |





